Abstract
Introduction
Intravitreal injections are a mainstay of treatment for retinal diseases and the most performed ophthalmic procedure in the world. In 2017, the Centers for Medicare and Medicaid Services reported more than 3 million intravitreal injections were administered in the United States, and that number is growing by 6% annually. 1 Biologics that inhibit vascular endothelial growth factor A (anti-VEGF) are the most common type of pharmaceutical agent given intravitreally. Anti-VEGF agents are used to treat multiple exudative retinal diseases including diabetic macular edema (DME), neovascular age-related macular degeneration (nAMD), and retinal venous occlusive (RVO) disease among others. Another class of pharmaceutical agents injected intravitreally is corticosteroids. 2
Exogenous endophthalmitis is a rare but serious complication of intravitreal injection procedures. A meta-analysis of 43 articles between January 2005 and May 2012 reported the overall rate of endophthalmitis following 350 535 anti-VEGF injections to be 0.056%. 3 Bacterial endophthalmitis following injection may cause permanent vision loss despite appropriate intervention. 4 For that study, rates of endophthalmitis were evaluated in a large urban retina practice relative to 2 variables: prefilled syringes (PFS) and universal masking.
Commonly used anti-VEGF pharmaceutical agents include ranibizumab (Lucentis), bevacizumab (Avastin), and aflibercept (Eylea). In routine clinical practice, single-use plastic syringes of bevacizumab are prepared in sterile fashion by a compounding pharmacy through the repackaging of larger bevacizumab vials into multiple syringes. In comparison, when first introduced commercially in 2006 and 2011 respectively, ranibizumab and aflibercept were provided in single-dose glass vials. The contents of the vial were aspirated into a syringe using a filter needle, and the filter needle was exchanged for a needle that was used to deliver the injection intravitreally. This transfer of the vial contents to a syringe theoretically could allow external contamination of the medication, leading to endophthalmitis. 5
Subsequently, the United States Food and Drug Administration approved ranibizumab as a PFS with a dose of 0.5 mg/0.05 mL in 2016, then approved it as a PFS in 2018 with a dose of 0.3 mg/0.05 mL, and approved aflibercept as a PFS in 2019. The introduction of syringes prefilled with sterile medication may theoretically reduce the risk of contamination and thus decrease the risk of endophthalmitis compared with clinical use of these medications from glass-vial preparation (GVP). A French study involving 1 285 034 injections of ranibizumab 0.5 mg/0.05 mL reported a 40% decreased rate of endophthalmitis with the use of PFS vs conventional GVP, from a rate of 0.021% to 0.013%. 6 In the present study, endophthalmitis rates before and after the introduction of PFS were investigated.
The second variable considered in this work was the impact of universal masking. 7 Streptococcus species, which was isolated in nearly 30% of culture-positive cases, are the second leading causative organisms of infectious endophthalmitis and exist as a normal part of oral flora.3,7 Physician masking has been proposed to potentially decrease the transmission of nasopharyngeal and oral flora to patients and instruments.8,9 Conversely, patient masking may cause increased transmission as air containing nasopharyngeal droplets can leak in the direction of the eye from the superior edge of a poorly fitted mask.10–13
Although the Euretina Board released an expert consensus in 2018 recommending implementation of a “no talking” or physician masking mandate during intravitreal injections, no universal, official procedure protocol exists in the United States. When implemented, the no-talking protocol has been reported to be effective at reducing the rate of infectious and oral pathogen–associated endophthalmitis.11,14
To reduce the transmission of COVID-19, large percentages of ophthalmologists, ancillary staff, and patients began wearing surgical masks consistently in 2020 during the pandemic. In the present study, the incidence of endophthalmitis following universal masking was examined to determine whether there was any change in the rate of endophthalmitis.
Methods
This multicenter, retrospective consecutive case series was approved by the Institutional Review Board of Houston Methodist Hospital in Texas. The medical records of all patients seen at Retina Consultants of Texas (RCTX) in the Greater Houston Area between January 1, 2015, and August 30, 2021, were examined. The total number of intravitreal injections during that time was identified using billing codes, and billing records were used to identify any cases of endophthalmitis seen during that time. Indications for intravitreal injection included nAMD, DME, RVO disease (branch and central), diabetic retinopathy, retinal edema, and chorioretinitis.
All cases of endophthalmitis included in this analysis were diagnosed and treated for clinically suspected endophthalmitis after intravitreal injection at RCTX. Cases of postsurgical endophthalmitis, community-acquired endophthalmitis, bleb-associated endophthalmitis, endogenous endophthalmitis, and infection secondary to trauma or corneal disease were excluded. Endophthalmitis cases were required to be documented within 6 weeks of receiving an injection at RCTX to be included.
All patients with presumed infectious endophthalmitis received intravitreal antibiotic and steroid injections according to local standard of care. Additionally, anterior chamber or vitreous taps were performed for all patients. Microbiological analyses were conducted by the Microbiology Department of Houston Methodist Hospitals. Medical records were reviewed, and relevant clinical data were extracted. Endophthalmitis cases were defined as culture-positive if there was bacterial growth on culture or a positive gram stain from the anterior chamber or vitreous tap.
All intravitreal injections were performed by board-certified physicians within RCTX in office-based settings with minor variations in injection technique. Treatment eyes were prepared with local anesthetic and topical 5%–10% povidone-iodine. Subconjunctival lidocaine, viscous lidocaine hydrocholoride ophthalmic gel, or topical anesthetic drops were used based on physician preference. Lid retraction was performed either manually or with a bladed lid speculum. Injection was performed 3.5 to 4.0 mm from the limbus. The total number of GVP and PFS injections were identified for each medication.
The study included dates prior to the approval of PFS during which only GVP was available. At RCTX, the transition from using primarily GVP to PFS varied according to injection type. PFS of ranibizumab 0.5 mg/0.05 mL, ranibizumab 0.3 mg/0.05 mL, and aflibercept were first used in clinic on February 12, 2017, April 26, 2018, and December 20, 2019, respectively. After PFS was available, both GVP and PFS were used in clinic and began to be included in this study; determining which drug-transport system had been used was done through billing codes.
Bevacizumab was prefilled by a compounding pharmacy and was considered neither GVP nor PFS in this study. Intravitreal triamcinolone acetonide (IVTA) was available as only a GVP injection, and the dexamethasone intravitreal implant was preloaded into a drug-specific injector.
At RCTX, masks were mandated for all patients, physicians, and ancillary staff beginning March 22, 2020; therefore, records between January 1, 2015, and March 21, 2020, were designated as pre-universal masking, and those between March 22, 2020, and August 30, 2021, were designated as universal masking. Intravitreal injections included in this study were anti-VEGF agents (aflibercept [2 mg/0.05 mL], ranibizumab [0.5 mg/0.05 mL and 0.3 mg/0.05 mL], and bevacizumab [1.25 mg/0.05 mL]) and steroid agents (dexamethasone intravitreal implant [0.7 mg; Ozurdex] and IVTA [4 mg/0.1 mL; Kenalog]).
Endophthalmitis rates were compared between treatment groups, including type of injection, method of administration, dosage, and timing of administration with respect to the when universal masking began. A limited number of the endophthalmitis cases described in the present study have been previously reported.5,15 Statistical comparisons between all treatment groups were performed using Fisher exact test. Statistical significance was set at P < .05 (2-tailed test), and all analyses were performed using R version 3.5.2 software (R Foundation).
Additional descriptive statistics were also collected, including treatment indication, phakic status, days to presentation, status of diabetes mellitus type 2, and visual acuity. Best-corrected visual acuity was reported at the date of the causative injection, at presentation of endophthalmitis, and at the 3-month follow-up visit in all cases. As established by previous studies, vision levels of counting fingers, hand motion, light perception, and no light perception were assigned visual acuity values of 1.0/200, 0.5/200, 0.25/200, and 0.125/200 (logMAR equivalent 2.3, 2.6, 2.9, and 3.2, respectively).16,17
Results
Between January 1, 2015, and August 30, 2021, there were 307 349 intravitreal injections performed. A total of 101 cases of postinjection endophthalmitis from 100 patients were identified and treated, yielding a rate of 0.033% or approximately 1 case for every 3030 injections (Table 1). The number of endophthalmitis cases identified and treated per year was: 9 in 2015, 16 in 2016, 16 in 2017, 15 in 2018, 17 in 2019, 15 in 2020, and 13 in 2021. The total rate of endophthalmitis after various types of anti-VEGF injections was 0.031% (n = 93/302 443), or approximately 1 case for every 3252 injections; this was significantly less than the rate after injection of steroid agents, which was 0.163% (n = 8/4906) (relative risk [RR], 0.189; 95% CI, 0.092-0.338; P < .001).
Total Injections per Year and Endophthalmitis Rate by Medication Type.
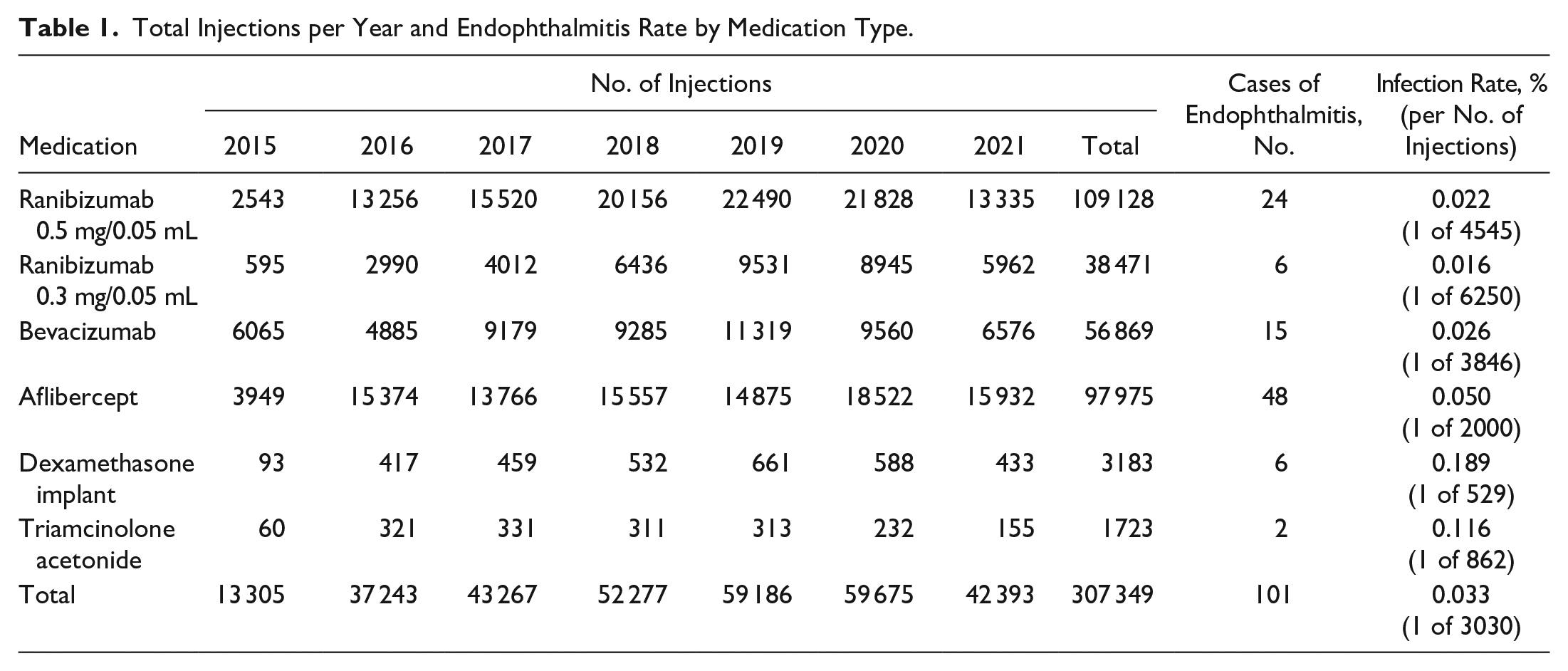
The treatment indication for intravitreal injection was nAMD in 54.46%, DME in 23.76%, diabetic retinopathy in 2.97%, central RVO in 4.95%, branch RVO in 8.91%, retinal edema in 2.97%, and chorioretinitis in 1.98% of the affected patient population. The mean time to presentation was 5.5 days. Sixty-six (66.0%) of patients were women, and 37 patients (37.0%) had type 2 diabetes mellitus. There were 55 right eyes (54.46%), and 26 were phakic (25.74%).
All patients underwent diagnostic anterior chamber paracentesis or vitreous tap at clinical presentation. Cultures were submitted for all 101 cases of endophthalmitis, of which 37 (36.63%) were culture positive and 64 (63.37%) were culture negative. Overall mean visual acuity at the time of the causative injection was logMAR 0.62 (approximately 20/84), and mean logMAR visual acuity at endophthalmitis presentation was 2.01 (approximately 20/2064). Mean visual acuity at 3-month follow-up was logMAR 1.06 (approximately 20/229). Twenty-three patients (22.77%) subsequently underwent pars plana vitrectomy.
Endophthalmitis Rates: GVP vs PFS
Compared with GVP, use of PFS was associated with a lower rate of endophthalmitis (Table 2). In the GVP group, a total of 102 866 injections were administered and 54 cases of endophthalmitis occurred (0.052%; 1 in 1923 injections). In the PFS group, a total of 142 708 injections were administered, and 24 cases of endophthalmitis occurred (0.017%; 1 in 5882 injections).
Comparison of Endophthalmitis Rates Between GVP and PFS by Medication Type.
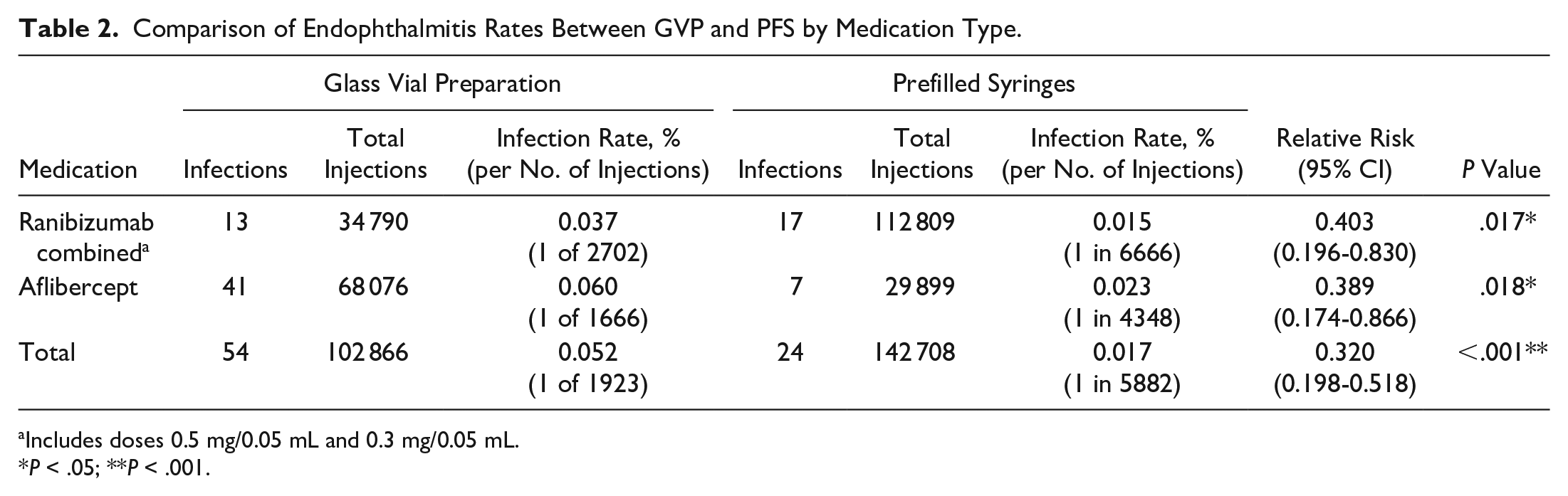
Includes doses 0.5 mg/0.05 mL and 0.3 mg/0.05 mL.
p < .05; **p < .001.
Endophthalmitis Rates: Before and After Universal Masking
There was no significant difference in endophthalmitis rates when comparing the pre-universal masking and universal masking cohorts (RR, 0.953; 95% CI, 0.616-1.473; P = .91) (Table 3). In the pre-universal masking period, a total of 219 131 injections were administered, and 73 cases of endophthalmitis were reported (0.033%; 1 of 3030 injections) (Table 3). Twenty-nine (39.73%) of these cases were culture positive and 44 (60.27%) of the cases were culture negative. During the time of universal masking, a total of 88 218 injections were administered, and 28 cases of endophthalmitis were reported (0.032%; 1 of 3150 injections).
Comparison of Endophthalmitis Rates Before and After Universal Masking by Medication Type.
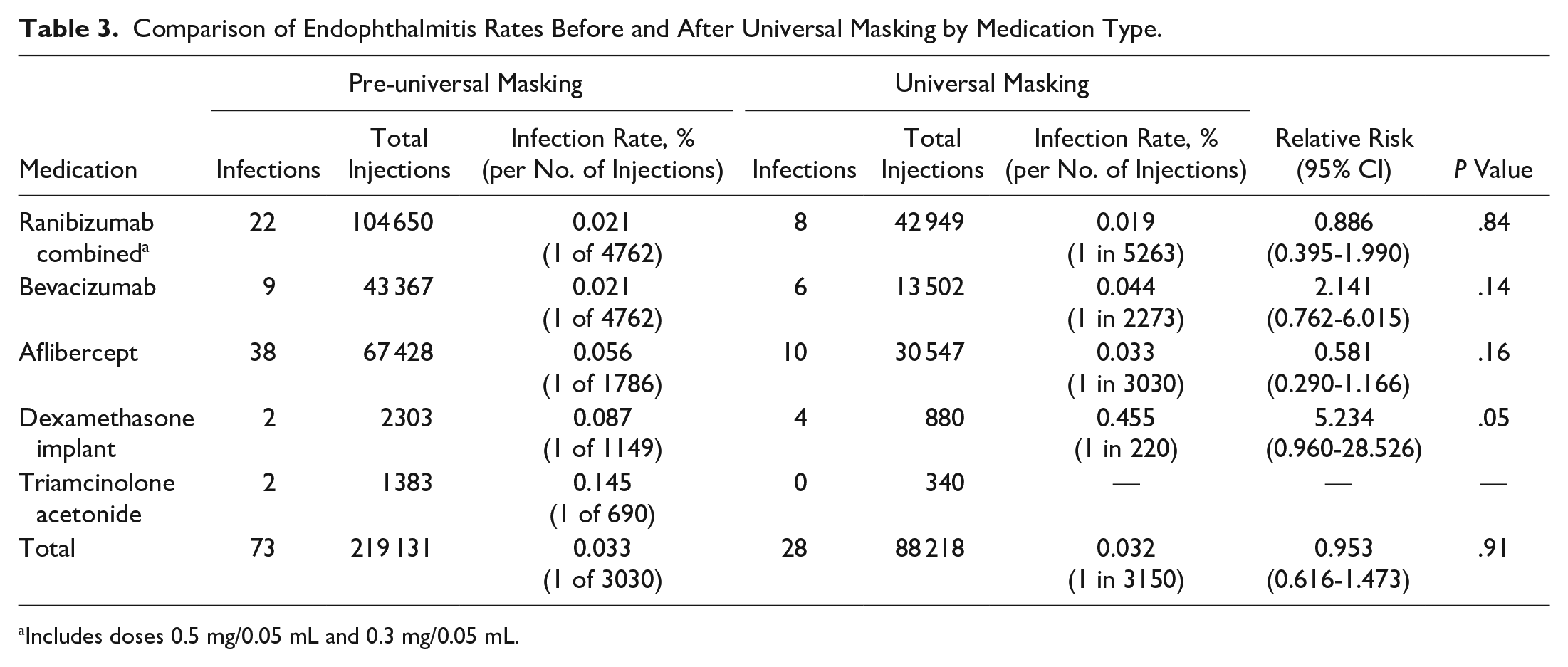
Includes doses 0.5 mg/0.05 mL and 0.3 mg/0.05 mL.
Of the universal-masking cases of endophthalmitis, 9 (32.14%) were culture positive and 19 (67.86%) were culture negative. Similarly, there was no difference in culture positivity rates between these periods (RR, 0.809; 95% CI, 0.441-1.486; P = .65). The mean time to presentation in the pre-universal and universal masking groups was 5 and 6.9 days, respectively.
Conclusions
Within the current retrospective analysis involving 307 349 intravitreal injections performed over a nearly 8-year interval, a 0.033% rate of endophthalmitis after intravitreal injections was observed. Previous studies have reported similar rates of endophthalmitis following intravitreal injection, ranging from 0.034% to 0.056%.3,5,18,19
In the current study, the rate of endophthalmitis after corticosteroid injection was 0.163% compared with 0.031% after anti-VEGF agents. This finding is consistent with previous studies that identified higher rates of infection among patients receiving corticosteroid injections. 20 This increased rate of endophthalmitis is likely multifactorial and may be influenced by the immunosuppressive effects of corticosteroids that can result in a decreased bacterial load required to cause disease.20,21 Additionally, the needle used for corticosteroid injection is typically larger than that used to deliver anti-VEGF injections, potentially allowing more bacteria to enter the larger wound tract.20,22,23
The rate of endophthalmitis was significantly decreased with the use of PFS vs GVP (odds ratio, 0.320; 95% CI, 0.198-0.518; P < .001). Several preparation steps involved in the transfer of GVP medications from the storage vial to injection syringe are eliminated with the use of PFS, likely contributing to the decreased risk for endophthalmitis.24–26 Specifically, as the transfer of GVP from single-use vials to syringes typically occurs in a nonsterile environment, there may be an increased risk of contamination with skin flora and aerosolized oral bacteria during preparation.
As some medications given by intravitreal injection are unavailable in PFS, it is important to transfer medication under optimal hygienic conditions to minimize risk of contamination.27,28 This process typically includes use of an alcohol wipe to clean the vial top before withdrawing the medication into a sterile syringe through a filter needle.29,30 Regardless of whether the medication is GVP or PFS, it is imperative to follow standard precautionary measures when performing intravitreal injection procedures. Preparing the ocular surface with an antiseptic such as povidone-iodine, minimizing oral flora exposure, and avoiding needle contact with lashes or lids have all been hypothesized to reduce endophthalmitis risk.31–33
Data related to the impact of masking on the risk of endophthalmitis have been mixed among physicians and patients. In 2018, before the COVID-19 pandemic, a survey of retina specialists found that 32.9% of the 399 respondents, 317 (79.5%) of whom practice in the United States, regularly wore surgical masks while administering intravitreal injections. 12 Some studies have suggested that masking can affect the rate of endophthalmitis. For example, studies simulating intravitreal injection procedures have proposed that physician masking may decrease the risk of bacterial contamination.8,9 One study involving 505 968 injections across 12 institutions reported that the rate of culture-positive endophthalmitis cases decreased with universal masking. 11 This may be a result of decreased oral flora contamination with mask usage.
Other analyses, including the present study, have suggested that rates of endophthalmitis are not impacted by face mask use in both physicians and patients.11,15,34 One study examined the impact of patient masking on endophthalmitis rates; physicians were masked during both the pre-COVID and COVID time periods. The study involved 53 927 injections administered from February 2019 to February 2021 and reported that in the pre-COVID period, during which patients were not masked, the rate of endophthalmitis following the administration of 34 277 injections was 0.020%. In the COVID period, during which patients were masked, the rate of endophthalmitis following the administration of 19 650 injections was 0.036% (P = .40). 35 Another study examining the effect of physician masking on rates of endophthalmitis shared similar findings; the study included 483 222 injections and reported an endophthalmitis rate of 0.0371% without face mask use and 0.0298% with face mask use (P = .53). 36
One caveat related to patient masking to consider clinically is the superior portion of the mask; specifically, securing the superior portion closed with adhesive tape has been proposed to decrease the risk of endophthalmitis by reducing oral bacterial dispersion toward the eyes.10,13,37 A prospective study at Wills Eye Hospital reported taping the superior portion of patient masks significantly reduced small particle transmission when the patient was talking but not when the patient was silent. 37
Another prospective report measuring colony-forming units during simulated intravitreal injections found significantly less growth in silent groups wearing tight-fitting surgical masks with tape compared with those without tape. 13 Interestingly, the study also included a no–face mask group and found no significant difference in growth between this group and any of the masked groups. 13 An analysis of 18 602 injections performed with the superior portion of patients’ masks taped reported no significant difference in the rate of endophthalmitis compared with the group that did not take this additional precautionary measure 11 ; a significant effect may not have been detected because the study was underpowered, or taping may not be sufficiently preventing the spread of bacterial particles that cause endophthalmitis. Overall, given the mixed results from published studies, it remains unclear whether masking, and/or the details of precisely how best to mask, have an impact on overall rates of endophthalmitis following intravitreal injection.
Limitations of the current study are inherent to its retrospective design, which is susceptible to inconsistent data entry and may not include all cases of endophthalmitis. Additionally, procedures were performed by several physicians who used varied injection techniques.
There may have been retina specialists who wore masks and also required patients to wear a mask during injection procedures as a precautionary measure during the pre-universal masking period. The universal masking group included patients and physicians wearing varying types of masks, including cloth, surgical, and N95, with and without tape.
The incidence of endophthalmitis after intravitreal injection remains low. As injection procedures are common and increasing, it is important to regularly evaluate and reevaluate interventions that may modify the risk of endophthalmitis. Related to this and consistent with previous reports, the use of PFS compared with GVP did appear to confer a decreased risk of endophthalmitis; as new pharmacotherapies become available, ideally a transition to PFS would occur as efficiently as possible.38,39 In comparison, within the current analysis, universal masking was found to neither increase nor decrease the rate of endophthalmitis among patients receiving intravitreal injections.
Footnotes
Authors’ Note
Presented virtually at the American Academy of Ophthalmology, October 2022.
Ethical Approval
This retrospective study was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information was performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
Not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CCW declares the following conflicts of interest:
Consulting fees/honoraria for ongoing services provided as a Consultant for: 4DMT, AbbVie, Adverum, Aerie, AGTC, Alcon, Annexon, Apellis, Arrowhead, Bausch + Lomb, Boehringer Ingelheim, Cholgene, Clearside, Curacle, EyePoint, Foresite, Frontera, Genentech, IVERIC Bio, Janssen, Kato, Kiora, Kodiak, Kriya, Merck, Nanoscope, NGM, Notal Vision, Novartis, Ocular Therapeutix, OcuTerra, ONL, Opthea, Oxular, Palatin, PerceiveBio, Perfuse, Ray, RecensMedical, Regeneron, RegenXBio, Resonance, Roche, SciNeuro, Stealth, Surrozen, Suzhou Raymon, THEA, TissueGen, Valo
Grants paid to my institution for ongoing research support as a Principal Investigator for trials sponsored by: 4DMT, Adverum, AffaMed, Alexion, Alimera, Alkahest, Allgenesis, Amgen, Annexin, Annexon, Apellis, Asclepix, Bayer, Boehringer Ingelheim, Chengdu Kanghong, Clearside, Curacle, EyePoint, Gemini, Genentech, GlaxoSmithKline, Gyroscope, IONIS, iRENIX, IVERIC bio, Kodiak, LMRI, Nanoscope, Neurotech, NGM, Novartis, Ocular Therapeutix, Ocuphire, OcuTerra, Ophthotech, Opthea, Oxurion, Oxular, Oyster Point, PerceiveBio, Regeneron, RegenXBio, Roche, SamChunDang Pharm, Sandoz, UNITY, Verily, Xbrane
Stock Options (not owner) from ongoing relationships as a Consultant from Private For-Profit Entities: ONL, PolyPhotonix, RecensMedical, TissueGen, Visgenx, Vitranu
The remaining authors declare that there are no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
