Abstract
Introduction
Fibroblast growth factor receptor (FGFR) initiates a signaling cascade that is critical for cell proliferation, differentiation, and survival. 1 FGFR and its numerous downstream pathways have become a target for the development of new chemotherapeutic agents. 2 The integrity of the retinal pigment epithelium (RPE), however, relies heavily on FGFR activation and stimulation of the mitogen-activated protein kinase (MAPK) pathway through the mitogen-activated protein kinase kinase (MEK) enzyme. 3 Inhibitors of MEK, such as trametinib, have been well known to induce reversible retinopathy. 3 More recently, erdafitinib, a pan-inhibitor of FGFR, was approved by the US Food and Drug Administration and has been associated with a reversible retinopathy similar to MEK inhibitors. 4
Combination therapy of both FGFR inhibitors and MEK inhibitors has been proposed as a solution to overcome drug-resistant cancers. 5 To our knowledge, the potential combined effect of MEK and FGFR inhibitors, an experimental combination therapy, on retinal toxicity has not been previously reported.
Case Report
A 67-year-old white male physician with a history of prostate and pancreatic cancer presented for an eye examination while on an experimental chemotherapy regimen, including trametinib, a MEK inhibitor, and erdafitinib, an FGFR inhibitor. The patient had previous surgical resection of his pancreatic cancer and was subsequently treated with FOLFIRINOX (5-fluorouracil with leucovorin, irinotecan, and oxaliplatin). Gene expression profile testing of tumor tissue identified a mutation in KRAS.
The patient did well for several years, but unfortunately his disease recurred and he was subsequently started on a new chemotherapy regimen of irinotecan, hydroxychloroquine, and trametinib. His cancer burden continued to progress despite this regimen, and erdafitinib was added and the irinotecan discontinued. Two weeks before presentation to the eye clinic, the patient experienced blurred vision in both eyes. He self-discontinued hydroxychloroquine at this time over concern for ocular toxicity. At the time of presentation, the patient had been taking erdafitinib 9 mg daily for 1 month and trametinib 2 mg daily for 4 months.
An ophthalmic examination found a best-corrected visual acuity of 20/70 OD and 20/60 OS. On dilated funduscopic examination, there were multifocal pockets of subretinal fluid (SRF), including a large area of subfoveal fluid in both eyes. Optical coherence tomography imaging showed central SRF with thickened photoreceptors in both eyes (Figure 1). Hypoautofluorescence and hyperautofluorescence were seen corresponding to pockets of SRF (Figure 2). One week after the ophthalmic examination, both trametinib and erdafitinib were discontinued as the patient experienced intolerable systemic side effects including bloody emesis. Unfortunately, the patient was unable to return for follow-up because of his systemic health and he died 2 months later.
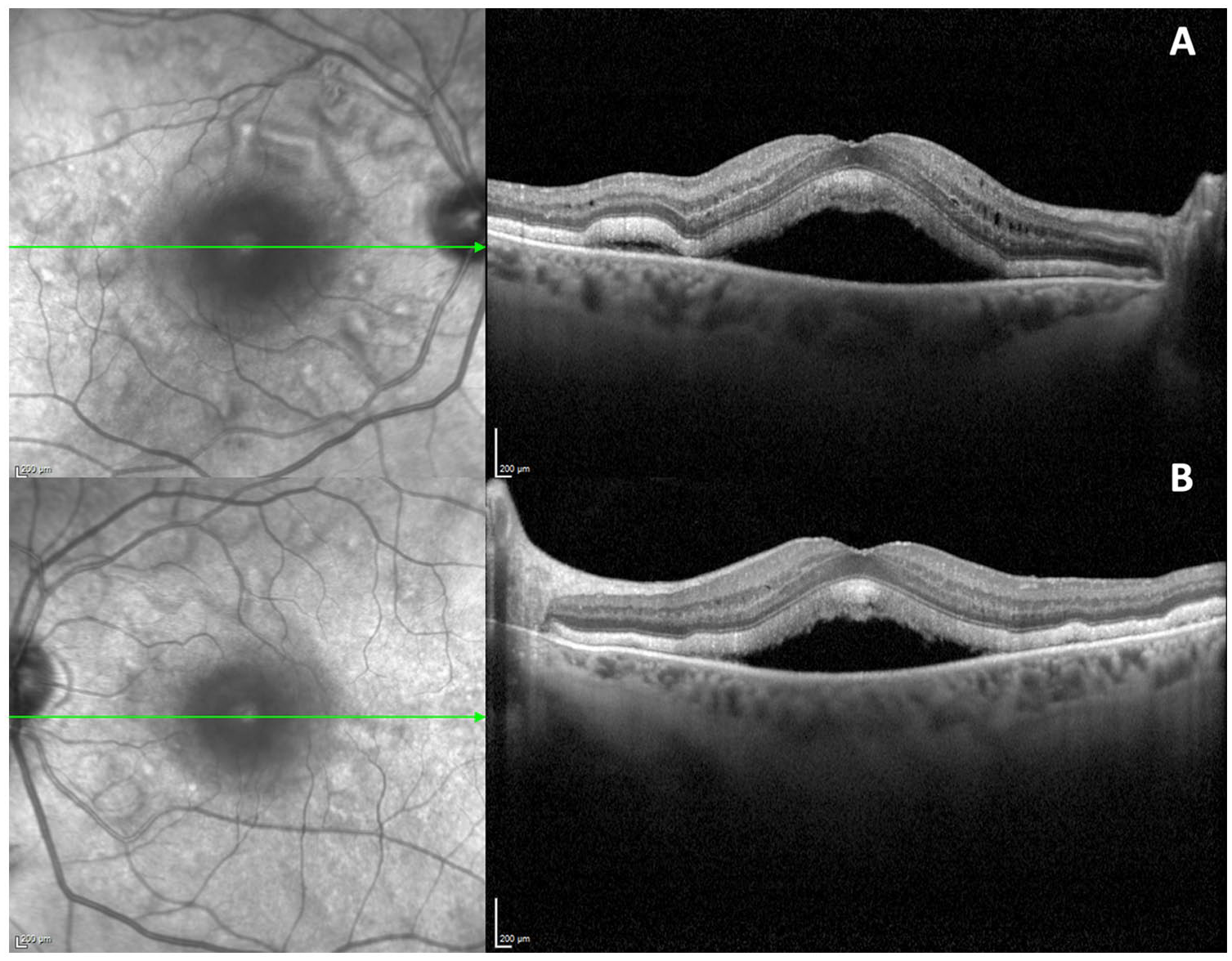
Optical coherence tomography of the right eye and left eye shows multifocal subretinal fluid and a thickened photoreceptor layer.
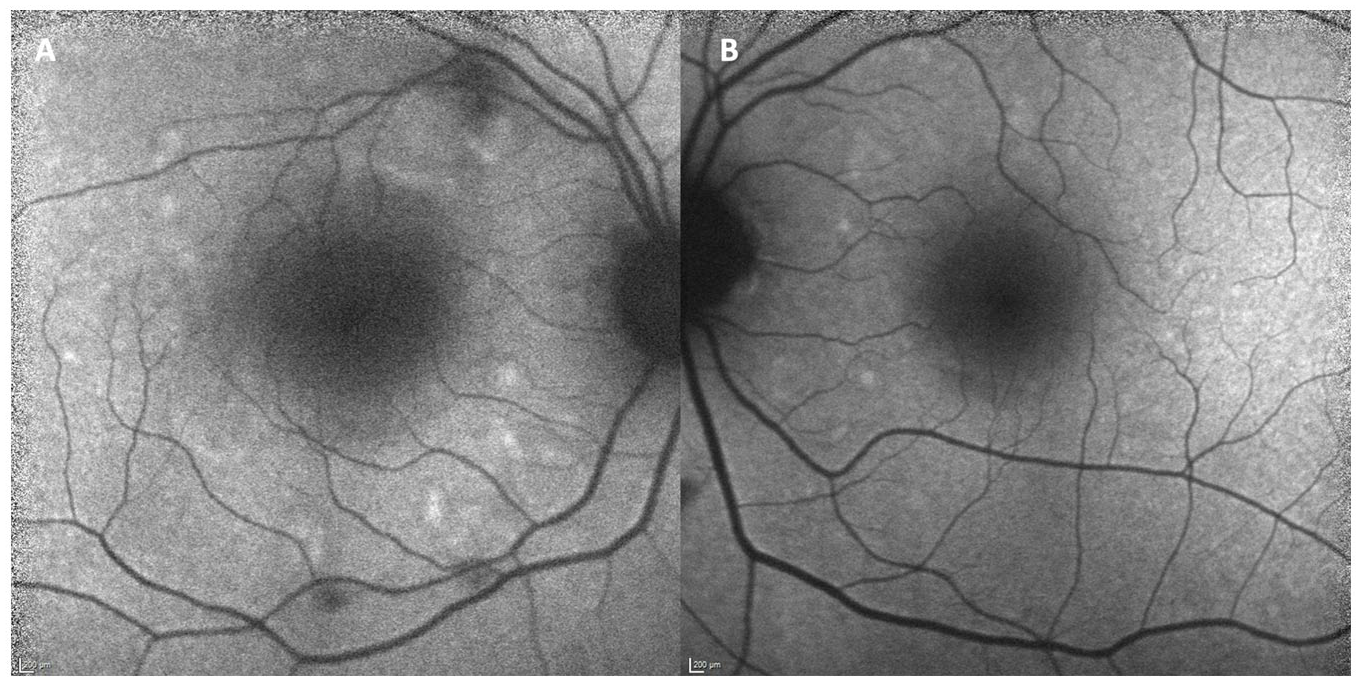
Fundus autofluorescence photographs revealing hyper-and hypo-autofluorescence corresponding to pockets of presumably new and older subretinal fluid, respectively, in the right (A) and left (B) eyes.
Conclusions
To our knowledge, this is the first case of a bilateral symptomatic multifocal serous retinopathy in a patient recently on a combination of an MEK inhibitor (trametinib) and an FGFR inhibitor (erdafitinib). This was part of an experimental “last-resort” protocol performed outside a clinical trial by an oncologist and the patient, who was also a physician, in the setting of an aggressive cancer resistant to multiple previous regimens.
Downstream signaling pathways activated by FGFRs include phosphatidylinositol 3-kinase (PI3K), inositol-1,4,5-trisphosphate, and diacylglycerol and the RAS–RAF–MEK–extracellular signal-regulated kinase system. 1 This large network controls cell metabolism, immunity, survival, and angiogenesis. Mutations in this pathway are a known trigger for the development of cancer, leading to recent drug discovery of pan-inhibitors of FGFR receptors as well as individual downstream targets such as MEK. In particular, the unique structural features of MEK have made it an ideal target for small molecule inhibitors as a means of anticancer treatment. 6
A reversible serous retinopathy resulting from MEK inhibitors has been well established in the literature. Classically, patients develop multifocal pockets of SRF without evidence of focal RPE defects or leakage as expected in central serous chorioretinopathy. In one clinical trial, 90% of patients taking an MEK inhibitor developed SRF, with a large portion remaining asymptomatic with few long-term ocular sequalae. 7 A similar retinopathy has been more recently described among inhibitors of the upstream target, FGFR.8,9
The majority of previous reports largely involved patients who had routine asymptomatic screening after initiation of treatment; in most cases the treatment is continued despite the presence of mild retinal dysfunction, which often resolves over time.8,10 In our case, the patient became profoundly symptomatic with large areas of subfoveal fluid bilaterally as well as multifocal pockets of SRF. Alekseev et al 11 reported a patient who developed similar OCT findings after treatment of pemigatinib, another small molecule inhibitor of FGFR. Other reports of retinopathy associated with erdafitinib have demonstrated more focalized areas of subfoveal fluid, with hyperreflective subretinal material that resolved after discontinuation of treatment. 4 In our case, the patient developed symptoms shortly after starting erdafitinib, suggesting FGFR inhibition as the culprit of more symptomatic retinopathy. However, it is possible the patient may have had asymptomatic pockets of SRF from trametinib before initiation of erdafitinib that notably worsened on combination therapy.
The development of SRF associated with hypoautofluorescence and hyperautofluorescence implicated involvement of the RPE pump. In vivo and in vitro studies have shown that the maintenance, repair, and protection of the RPE rely heavily on MAPK signaling. 3 MEK depends on stimulation by FGFR, and this common pathway explains the similarities in MEK-associated and FGFR-associated retinopathy. However, FGFR also independently triggers the PI3K signaling pathway, which has been shown to be critical in the antioxidant protection of the RPE. 12 Combination treatment of erdafitinib and trametinib may have an additive effect on RPE dysfunction as a result of increasing stress on the FGFR–MEK signaling pathway. It is also plausible that FGFR inhibition creates additional oxidative stress on the RPE through channels separate from the MEK pathways.
In most patients on MEK or FGFR inhibitors, screening visits before and within 1 month after the initiation of treatment are recommended. As the retinopathy on a single drug alone is usually self-limited and not vision threatening, these life-saving therapies are continued. However, future studies are necessary to assess the risk of combination therapy of MEK and FGFR inhibitors to guide ophthalmologic surveillance for retinopathy in these patients, which may be more severe.
Footnotes
Ethical Approval
This case report was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information was performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
The consent to publish was not required in this case as neither the images nor text reveal the identity of the patient.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Finn is on the advisory boards of Allergan, EyePoint, Iveric Bio, and Alimera and is a consultant to Genentech.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
