Abstract

Iron chelation plays a central role in the treatment of hemosiderosis. Historically, the chelator of choice was deferoxamine. While effective, this medication is administered subcutaneously via lengthy transfusions and exhibits a robust side effect profile, including sensorineural hearing loss (SNHL) in 25% to 30% of patients. 1 Because of this, orally administered chelators deferiprone and deferasirox (DFX) have been developed. To date, there are limited data regarding the otologic side effect profile for these new-generation iron chelators. The most convincing data are derived from clinical trials of DFX in which hearing loss was reported in up to 1.1% of patients.2,3 Subsequent studies have found rates of mild hearing loss ranging up to 45% in patients who have beta thalassemia treated with DFX.4,5 Descriptions of patients with greater than mild DFX-associated hearing loss or those requiring cochlear implantation are not present in the current literature.
Case Report
This study was deemed exempt from review by the Department of Research Programs at Walter Reed National Military Medical Center.
Our patient is a man in his 60s with a history of myelodysplastic syndrome and nontransfusional iron overload manifesting with elevated ferritin levels despite standard treatment. A routine audiogram predating treatment to monitor known hearing loss revealed mild SNHL bilaterally with right and left speech discrimination scores (SDSs) of 100% and 92%, respectively ( Table 1 ). The patient was prescribed long-term DFX (1000 mg, daily) from 2010 to 2012. In 2012, he reported sudden left hearing loss. An audiogram confirmed severe left SNHL with a 70-dB pure tone average and SDS of 12%; the right side was relatively unchanged. Discontinuation of DFX and high-dose oral steroids were unsuccessful at restoring hearing. The patient was fitted with a hearing aid with minimal benefit.
Patient’s Audiometric Data Over Time.
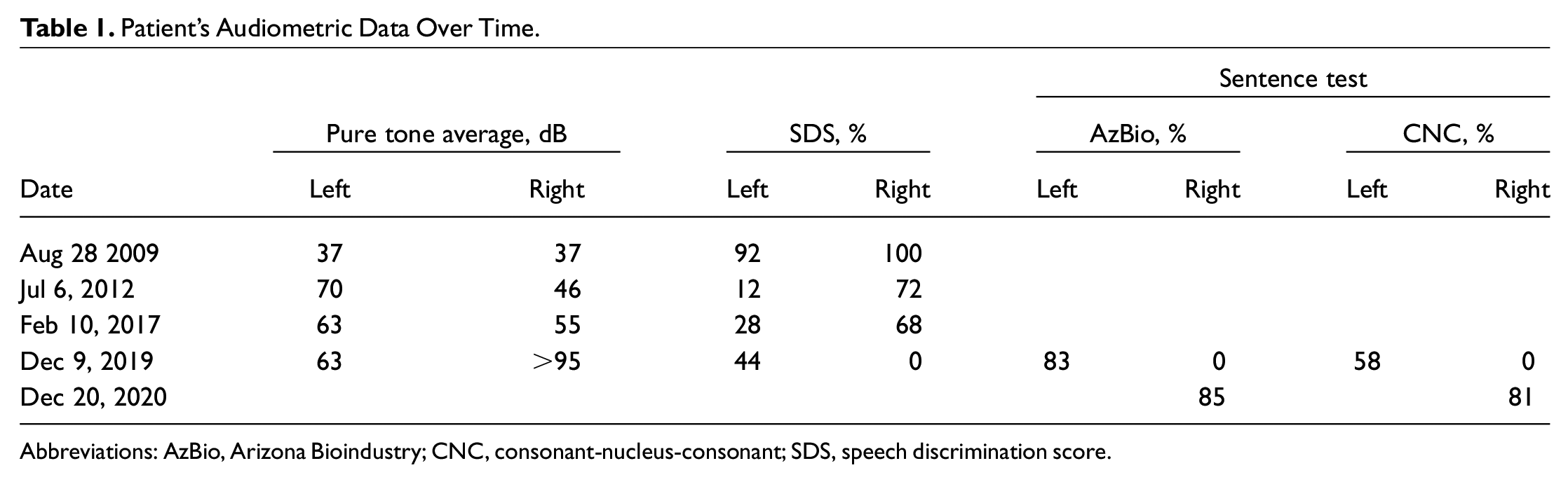
Abbreviations: AzBio, Arizona Bioindustry; CNC, consonant-nucleus-consonant; SDS, speech discrimination score.
By 2015, elevated ferritin levels necessitated DFX treatment at 500 mg daily. Within 18 months, it was discontinued secondary to worsening right SNHL, decreasing from 37- to 55-dB pure tone average and diminished SDS to 68%. This time, the left ear was unchanged. Again, steroids were unsuccessful.
Finally, DFX (250 mg) was restarted in 2018, but again acute right SNHL occurred. At this time, hearing loss had progressed to profound right-sided deafness. A cochlear implant evaluation confirmed 0% and 83% on the AzBio sentence test (Arizona Bioindusty) and 0% and 58% CNC sentence test (consonant-nucleus-consonant) in quiet on the right and left, respectively. Magnetic resonance imaging confirmed normal anatomy and no retrocochlear pathology ( Figure 1 ). Due to the lack of benefit from hearing aids and profound deafness, the decision was made to perform right-sided cochlear implantation in 2020. The patient was implanted with a precurved perimodiolar electrode uneventfully. Six months postoperatively, the patient had improved AzBio and CNC scores of 85% and 81%, respectively.
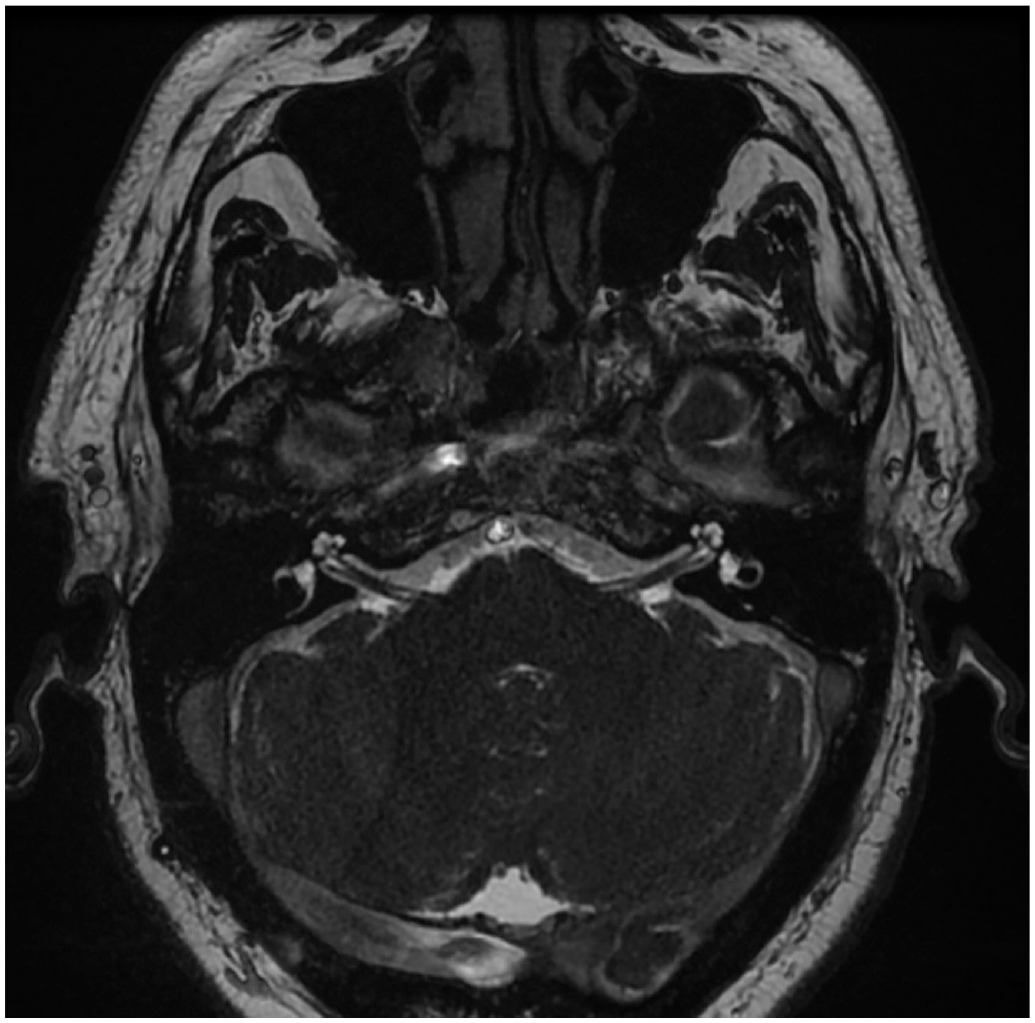
Preoperative high-resolution T2 magnetic resonance image of the internal auditory canals demonstrating normal cochlear anatomy and internal auditory canals bilaterally.
Discussion
Hearing loss is a well-known side effect of first-generation iron chelators, but the incidence of hearing loss from second-generation medications such as DFX is not well established. The existing literature largely fails to consider preexisting hearing loss, its magnitude, or the need for hearing restoration.
The prospective trials’ data on hearing loss are limited to subjective reports without objective audiometric data.2,3 The 2 largest cross-sectional studies report a rate of DFX ototoxicity between 0% and 45%; however, both are limited to patients with beta thalassemia treated for transfusion-associated hemosiderosis, and neither provided audiometric data.4,5 Derin et al reported that no patient demonstrated hearing loss above grade 2 on the National Cancer Institute’s Common Terminology Criteria for Adverse Events, indicating no threshold for intervention. Khan et al described all patients as having a mild SNHL.
The case presented here offers the first longitudinal look at the hearing status of a patient treated with DFX. The case as reported offers a valuable timeline of events with comprehensive audiometric data spanning the patient’s clinical course. This case is also the first to describe profound DFX treatment-related SNHL and subsequent successful cochlear implant rehabilitation. This patient did well with a cochlear implant achieving typical postimplantation speech scores. Importantly, there was no evidence for cochlear lumen obstruction or abnormal anatomy.
Although it is impossible to confirm causality between hearing loss and DFX or fully exclude idiopathic sudden SNHL, the temporal relationship between the use of DFX and the patient’s symptoms provides strong evidence for a possible link. Future prospective research is needed to fully elucidate the rate and degree at which second-generation iron chelation therapy affects hearing.
Author Contributions
Disclosures
Footnotes
The contents of this publication are the sole responsibility of the authors and do not necessarily reflect the view, opinions, or policies of the Uniformed Services University of the Health Sciences; the Department of Defense; or the Department of the Army, Navy, or Air Force. Mention of trade names, commercial products, or organizations does not imply endorsement by the US government.
