Abstract
Objective
(1) Characterize the distribution of M1 and M2 macrophages in vestibular schwannomas by hearing status. (2) Develop assays to assess monocyte migration and macrophage polarization in cocultures with vestibular schwannoma cells.
Study Design
Basic and translational science.
Setting
Tertiary care center.
Methods
A retrospective chart review of 30 patients with vestibular schwannoma (VS) was performed. Patients were stratified into serviceable and unserviceable hearing groups. Immunohistochemistry for CD80+ M1 and CD163+ M2 macrophages was conducted. Primary VS cultures (n = 4) were developed and cocultured with monocytes. Immunohistochemistry for macrophage markers was performed to assess monocyte migration and macrophage polarization.
Results
Although tumors associated with unserviceable hearing had higher levels of CD80 and CD163 than those with serviceable hearing, the relationship was only significant with CD163 (P = .0161). However, CD163 level did not remain a significant predictor variable associated with unserviceable hearing on multivariate analysis when adjusted for other variables. In vitro assays show that VS cells induced monocyte migration and polarization toward CD80+ M1 or CD163+ M2 macrophage phenotypes, with qualitative differences in CD163+ macrophage morphologies between serviceable and unserviceable hearing groups.
Conclusion
Vestibular schwannomas express varying degrees of CD80+ M1 and CD163+ M2 macrophages. We present evidence that higher expression of CD163+ may contribute to poorer hearing outcomes in patients with VS. We also describe in vitro assays in a proof-of-concept investigation that VS cells can initiate monocyte migration and macrophage polarization. Future investigations are warranted to explore the relationships between tumor, macrophages, secreted cytokines, and hearing outcomes in patients with VS.
Vestibular schwannomas (VSs) are benign intracranial tumors arising from Schwann cells of cochleovestibular nerves. They can cause hearing loss (HL), imbalance, and life-threatening intracranial complications. HL occurs in over 80% of cases and can affect quality of life.1-4
Historically, VS-associated HL was thought to arise from direct tumor compression of cochleovestibular nerves and therefore related to tumor size. However, several studies revealed this relationship is not straightforward, most likely multifactorial, and affected by the tumor microenvironment (TME).5-9 In efforts to find new therapies to preserve hearing in patients with VS, the VS TME has become a growing area of research.
Tumor-associated macrophages (TAMs) are immune cells found in the TME. TAMs occur when monocytes infiltrate tumor and polarize to different macrophage phenotypes, depending on the cytokines and chemokines existing in the TME. These macrophages are divided into 2 broad phenotypic categories: M1 and M2 macrophages.10,11
In the presence of certain cytokines, such as lipopolysaccharide and interferon-γ, monocytes polarize into M1 macrophages that express CD80 (a surface glycoprotein) and secrete proinflammatory cytokines such as tumor necrosis factor–α that can damage cochlear hair cells and cause HL.12-16 M1 macrophages are considered tumoricidal due to production of other toxic intermediates.17,18 In the presence of other factors, such as interleukin (IL)–4 and IL-13, monocytes polarize into M2 macrophages that express different surface glycoproteins, such as CD163. M2 macrophages secrete anti-inflammatory cytokines, including IL-10 and transforming growth factor–β; promote angiogenesis; and facilitate tumor growth.18-22
In this study, we described distributions of CD80+ M1 and CD163+ M2 macrophages within VS tumors and how TAM polarization may contribute to HL. We also describe in vitro assays exploring monocyte migration and polarization in the presence of VS cells as a proof of concept that can be further expanded to study the TME effect on clinical outcomes in the future.
Methods
This study was approved by the University of Miami Institutional Review Board (Protocol #20150637).
Patient Consent and Tumor Harvesting
Patients undergoing VS surgery at University of Miami/Jackson Memorial Hospital between April 2018 and September 2020 were consented for tumor harvesting. Tumor chunks were collected from the operating room and placed in chilled Dulbecco’s modified Eagle’s medium (DMEM). Tissues were processed for cell culture and immunohistochemistry and banked.
Retrospective Chart Review and Radiographic Tumor Measurements
Retrospective chart reviews were performed to collect demographic, presenting, audiometric, radiographic, and surgical information on 30 consecutive patients who were consented for tumor harvesting. Patients were stratified into 2 groups based on hearing status: (1) serviceable hearing (SH), defined as American Academy of Otolaryngology–Head and Neck Surgery (AAO) hearing grade of A or B, and (2) unserviceable hearing (UH) defined as AAO hearing grade of C or D.
Tumor volumes were calculated using preoperative magnetic resonance imaging (MRI) of the brain with and without gadolinium using 3D Slicer software (version 4.10.2; Brigham and Women’s Hospital). In brief, enhancing tumor was manually outlined on axial T1-weighted images with contrast. Tumor outlines were confirmed using coronal and sagittal images and tumor volume was measured in cm3.
Immunohistochemistry for CD80 and CD163 on VS Tumors
Tumor chunks (n = 30) were fixed in 4% paraformaldehyde overnight, instilled in 30% sucrose, and embedded in optimal cutting temperature (OCT) medium, snap frozen, and sectioned at 5 µm (Leica CM 1860). Tissue sections were permeabilized and blocked prior to antigen retrieval with citrate buffer. Sections were incubated with CD80 (1:500, #8679; ProSci) or CD163 (1:500, #163M; Cell Marque) primary antibodies at 4°C overnight, Alexa Fluor 594–conjugated secondary antibody (Life Technologies) for 2 hours at room temperature, and 4′6-diamidino-2-phenylindole (DAPI) nuclear stain for 10 minutes. Antifade mounting medium was added before cover-slipping. Confocal images were obtained for 3 different areas (Zeiss LSM 700 Microscope, 40× oil immersion lens). Serum-treated sections were used as negative controls. After the minimum and maximum threshold values were set to eliminate nonspecific staining and artifact on ImageJ software (National Institutes of Health), expression levels for CD80 and CD163 were determined as percent area per region of interest on high-powered field. Median values were compared between samples.
Cocultures of VS and Monocytes
VS tumor chunks (n = 4) were enzymatically dissociated and cultured in Schwann media (Sciencell), as previously described. 23 Human CD14+ monocytes (PromoCell) were cultured in mononuclear cell medium (PromoCell), according to the manufacturer’s protocol. Subsequently, VS cells and monocytes were cocultured in either (1) 24-well dishes with transwell inserts to assess monocyte migration and polarization or (2) directly in 16-well cultures slides to assess monocyte polarization.
For migration assays, primary VS cells were plated on 24-well dishes at 50,000 cells/well in Schwann medium and incubated at 37°C and 5% CO2 for 24 hours. Subsequently, monocytes were added to each transwell insert at a density of 0 and 50,000 monocytes/insert. For direct cocultures, VS cells were plated at 5000 cells/well on 16-well culture slides in Schwann medium at 37°C and 5% CO2 for 24 hours. Monocytes were then added at 5000 monocytes/well. Progressive media changes (30%-50%) were performed every 24 to 72 hours for 11 days prior to being fixed with 4% paraformaldehyde. Immunohistochemistry was performed using the following primary antibodies: CD80 (1:100 for transwell cocultures; ThermoFisher, PA5-85913), CD80 (1:100 for direct cocultures; ProSci, 8679), CD163 (1:100, OriGene, TA506383), and S100 (1:500, Abcam, ab868). Confocal images were obtained with a 20× lens for transwell cocultures and a 40× oil immersion lens for direct cocultures (Leica SP5 Inverted Microscope). Qualitative observations were described.
Statistical Analysis
With results from the initial 15 specimens, a sample size calculation was performed, and 30 specimens were necessary to determine a difference in macrophage expression between hearing cohorts (power = 0.8; α = 0.05). Differences in clinical data and CD80 and CD163 levels were compared between SH and UH groups using t test or Fisher exact tests with Bonferroni correction for multiple comparisons. Spearman rank correlation was used to assess relationships between tumor volume and macrophage markers. Univariate and multivariate logistic regression analysis with generalized linear modeling was also performed to determine the impact of clinically relevant predictor variables on preoperative UH. Variables included age at time of surgery, prior treatment (surgery, radiation, and/or chemotherapy), tumor volume, and CD80 and CD163 expression levels. Odds ratios, 95% CIs, and P values were determined for univariate and multivariate analyses. Significance was set at a P value less than .05. Statistical analysis was performed using R (version 4.1.1; The R Foundation).
Results
Clinical Data by Hearing Status
Clinical data for patients with SH and UH are displayed in Table 1 . The average age was 53.3 years (range, 25-77 years). Fifty percent were male, 83% were white, and 36.7% were Hispanic. There were no significant differences in age, sex, or ethnicity between SH and UH groups. However, there was a significant difference in race distribution between both groups (P = .0306) that did not remain significant after Bonferroni correction (P = .6426).
Demographic, Clinical, Audiometric, Radiographic, and Surgical Information.
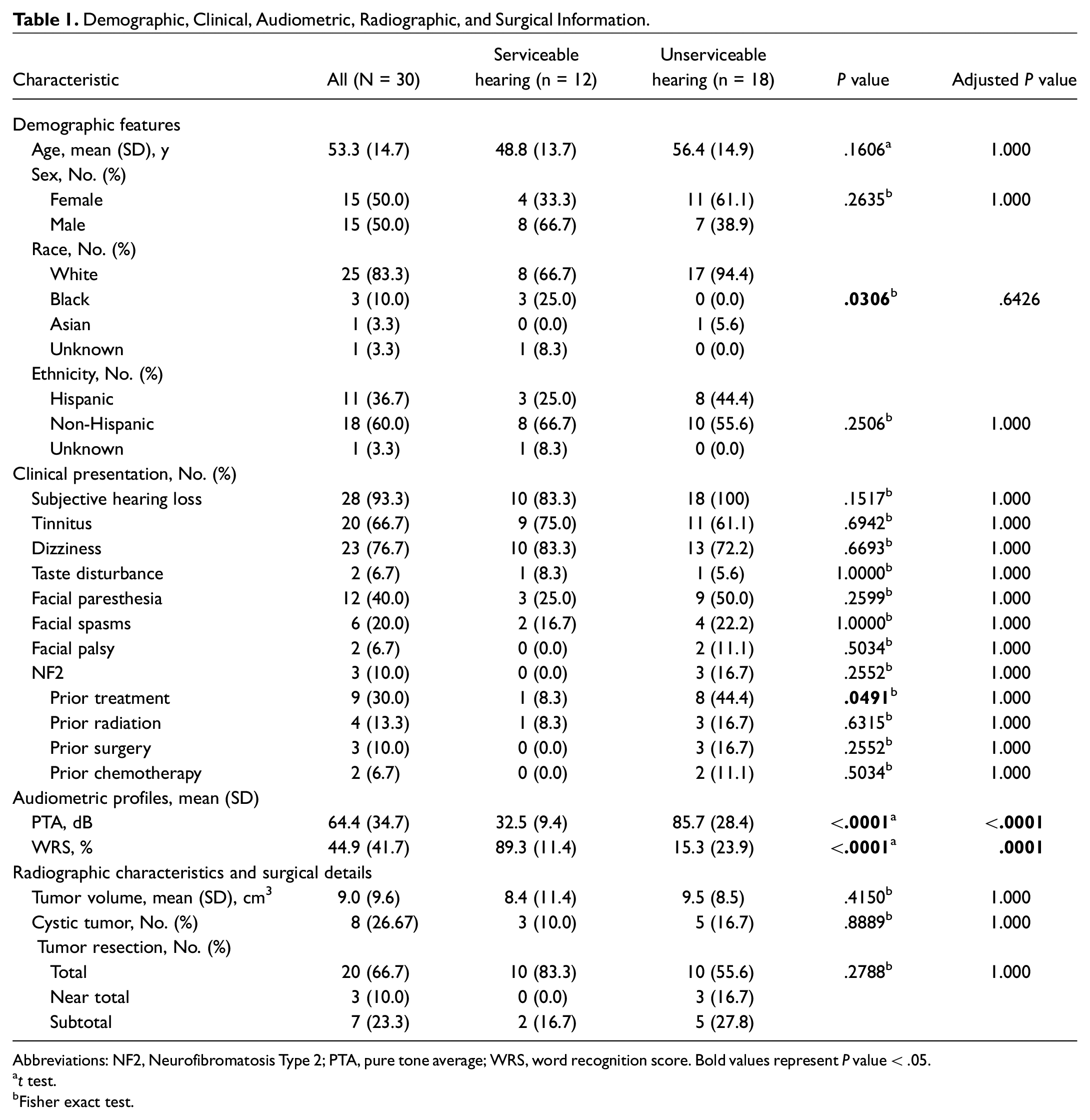
Abbreviations: NF2, Neurofibromatosis Type 2; PTA, pure tone average; WRS, word recognition score. Bold values represent P value < .05.
t test.
Fisher exact test.
Most patients presented with subjective HL, tinnitus, and/or dizziness. Fewer patients presented with taste disturbance (6.7%), facial paresthesia (40%), facial spasm (20%), and facial palsy (6.7%). There were no significant differences in the distribution of preoperative symptoms between groups. One patient (8.4%) in the SH group received radiation. In the UH group, 3 (16.7%) received radiation, 3 (16.7%) underwent prior surgery, and 2 (11.1%) received bevacizumab. Of these 8 UH patients with prior treatment, 1 patient who received radiation and the 2 patients treated with bevacizumab had Neurofibromatosis Type 2 (NF2). Although the proportion of patients with NF2 and prior VS treatment was higher in the UH group, there were no significant differences between hearing groups.
Because the SH and UH groups were defined by pure tone average (PTA) and word recognition score (WRS), it was not unexpected to find that the UH group had higher pure-tone averages (P < .0001) and WRS (P < .0001) than the SH group ( Figure 1A , B ). The tumor volumes for both cohorts were similar (P = .4150) with means of 8.4 cm3 and 9.5 cm3, respectively ( Figure 1C ). The proportion of cystic tumors was also comparable (P = .8889). In addition, all 30 tumors had tumor components located in the internal auditory canal and cerebellopontine angles.
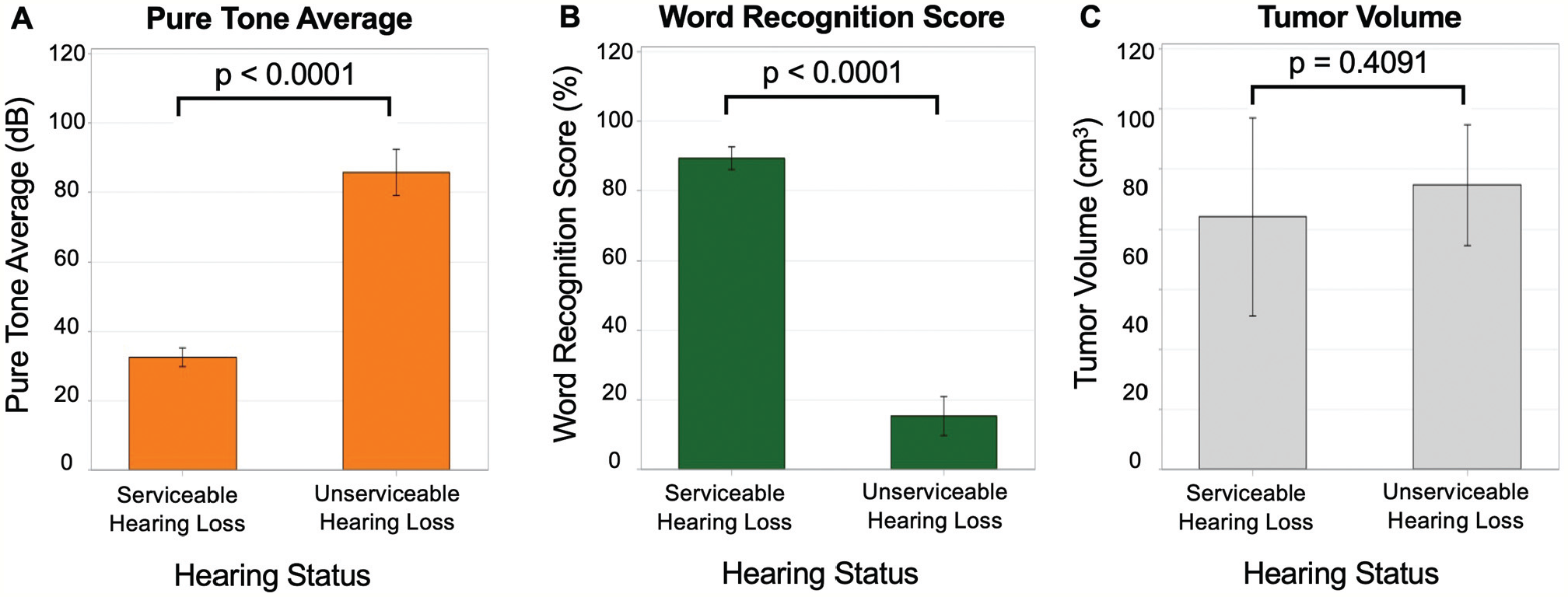
Audiometry and tumor volume. (A, B) Although patients with serviceable hearing had better pure-tone averages and word recognition scores than those with unserviceable hearing, (C) tumor volumes were similar.
Furthermore, most patients had gross total resection of tumor (66.7%). The UH group had higher percentages with near-total and subtotal resections than the SH group, but the difference was not significant (P = .2788).
CD80 and CD163 by Hearing Status and Tumor Volume
Immunohistochemistry for CD80 and CD163 was performed to determine expression levels of M1 and M2 macrophages, respectively. We quantified percent area, and results were stratified by hearing status ( Figure 2A , B ; bars show mean and error bars represent standard error). For both macrophage markers, UH tumors had higher mean expression levels than SH tumors. Although not statistically significant for CD80 (P = .0692), the increase in CD163 was significantly higher in UH than SH tumors (P = .0161). Figure 2C demonstrates representative confocal images illustrating a UH tumor expressing more CD80 and CD163 than another SH tumor. In addition, we found no significant correlations between tumor volume and CD80 (r = 0.1364, P = .4723) or between tumor volume and CD163 (r = 0.1409, P = .4578).
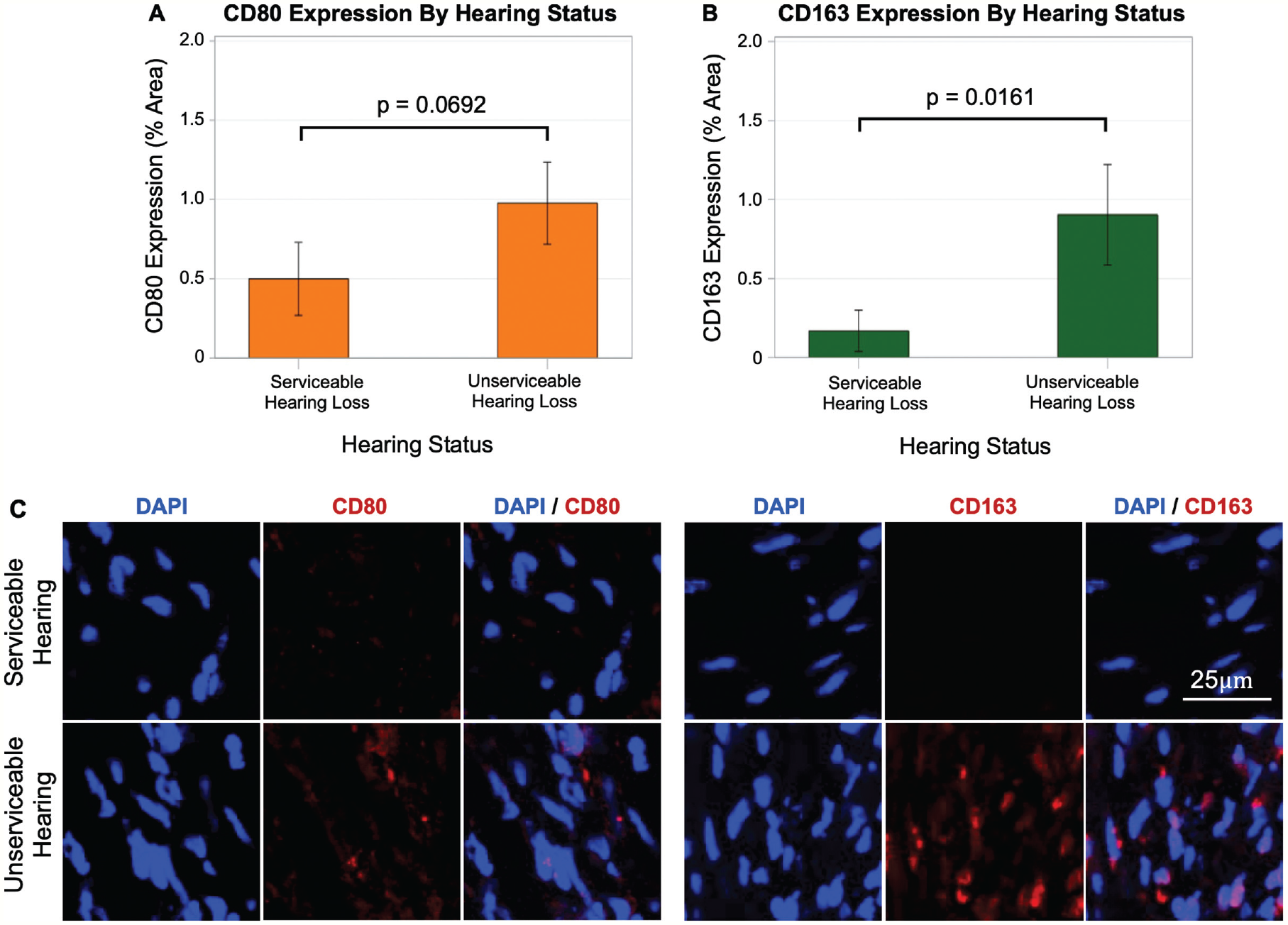
CD80 and CD163 expression. (A, B) Mean expression levels for CD80 and CD163 were higher in patients with unserviceable hearing vs serviceable hearing. (C) Representative confocal images are shown.
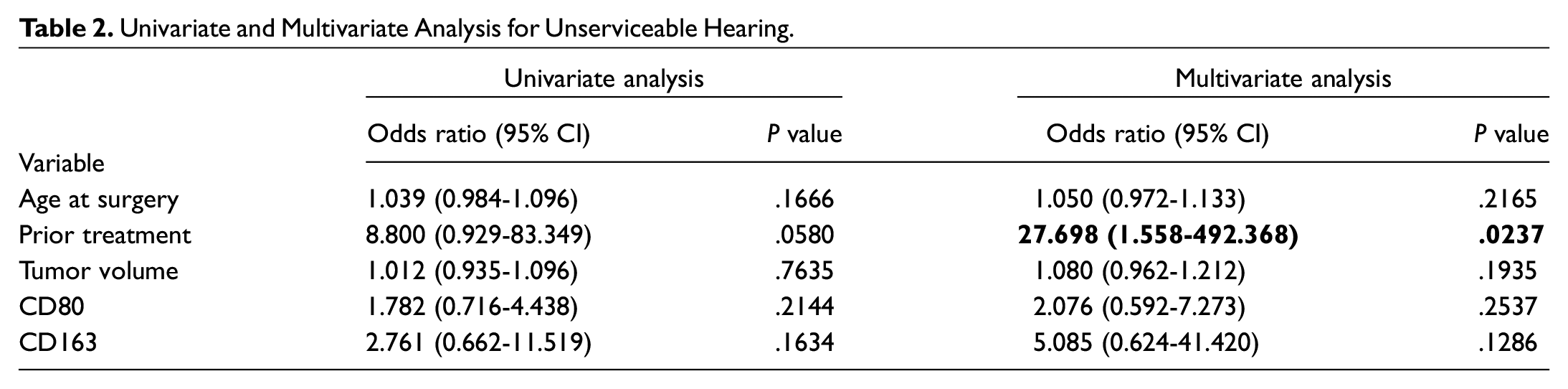
Subsequently, univariate and multivariate analyses were performed to determine the impact of clinically relevant predictor variables on preoperative hearing status ( Table 2 ). On univariate analyses, no variables were independently associated with UH. However, on multivariate analyses, history of prior treatment (surgery, radiation, and/or chemotherapy) was independently associated with UH at time of surgery (P = .0237; adjusted odds ratio, 27.698; 95% CI, 1.558-492.368), when adjusted for all other variables. Although the odds ratios for CD80 and CD163 were 2.076 and 5.085, respectively, they were not significant by 95% CI and P values. When macrophage expression was compared between patients with and without prior treatments, there were no significant differences in CD80 (P = .9459) or CD163 (P = .3451) levels.
Univariate and Multivariate Analysis for Unserviceable Hearing.
Models of Monocyte Migration and Differentiation With Vestibular Schwannoma
To determine if the VS TME can cause monocyte migration and polarization, transwell and direct cocultures assays were performed. Representative confocal images of cocultures on day 11 are shown in Figures 3 and 4 , respectively. The transwell assay revealed that monocytes migrated through the insert and attached to the bottom of the plate, which contained adherent VS cells. Some monocytes remained small and round and expressed both CD80 and CD163 markers. Other monocytes polarized to express either CD80 or CD163 and began developing different macrophage morphologies ( Figure 3 ).
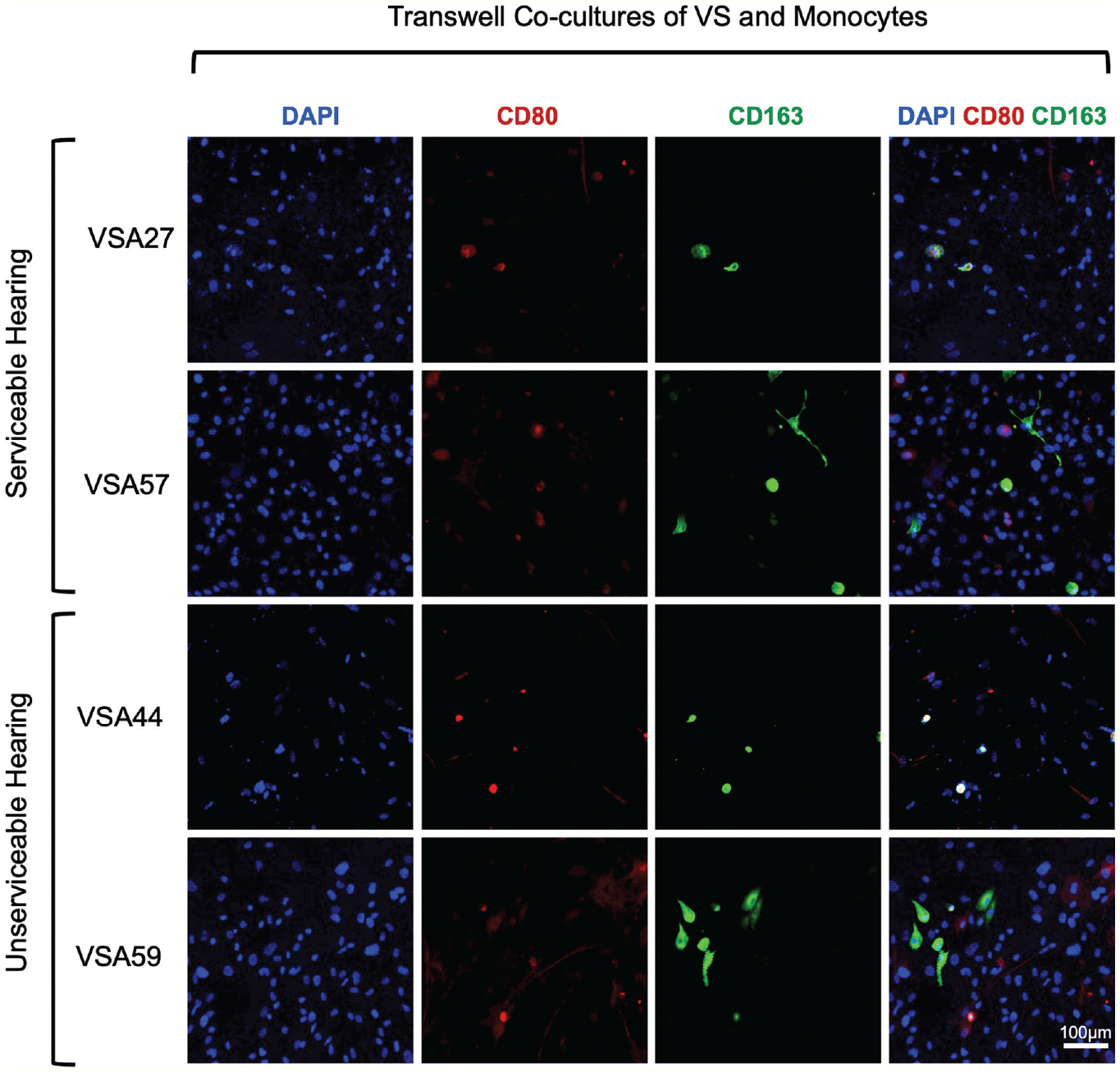
Transwell cocultures. Confocal images show monocytes migrated toward vestibular schwannoma (VS) cells associated with serviceable and unserviceable hearing and polarized to M1-type CD80+, M2-type CD163+, or mixed-phenotype macrophages.
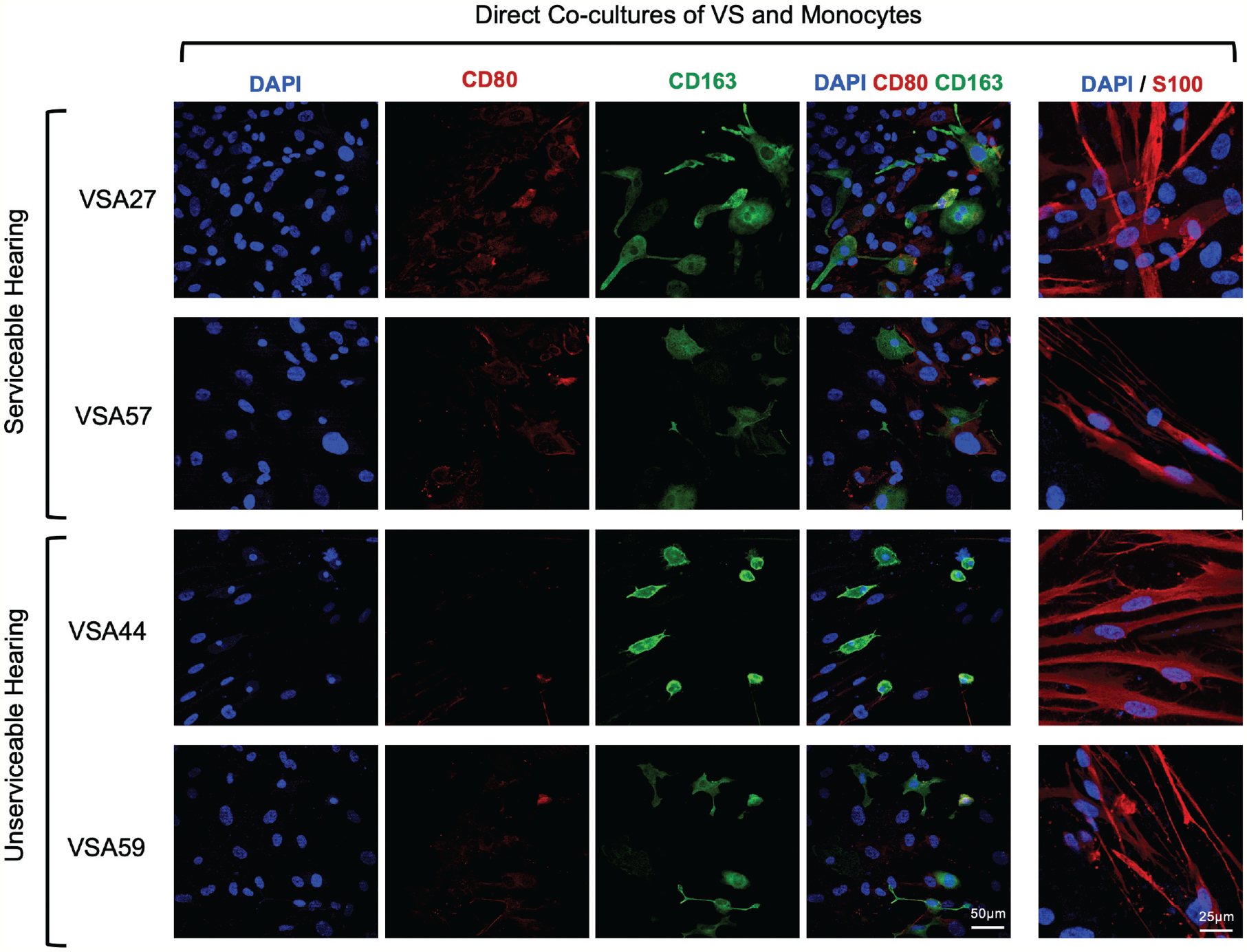
Direct cocultures. Regardless of hearing status, confocal images demonstrate monocytes polarize predominantly toward M2-type CD163+ macrophages, rather than M1-type CD80+ macrophages, when cultured with vestibular schwannoma (VS) cells.
In direct coculture, monocytes differentiated into macrophages, expressing CD80 and/or CD163 ( Figure 4 ). Most macrophages had either more intense CD80+ or CD163+ staining. We observed more CD163+ M2 macrophages than CD80+ M1 macrophages overall. The morphologies of M2 macrophages were heterogenous, and some cells had pseudopodia or cytoplasmic projections. Upon close inspection, M2 macrophages from the SH group appeared as (1) giant cells occasionally with multiple nuclei, (2) large amoeboid-shaped cells with pseudopodia, and (3) spindle-shaped cells. M2 macrophages from the UH group were smaller in size and either (1) round or spindle-shaped with fine cytoplasmic projections or (2) amoeboid shaped with pseudopodia.
Discussion
The TME is a dynamic and complex entity, consisting of tumor cells, infiltrating and resident immune cells, stromal cells and blood vessels, secreted factors, and extracellular matrix components. Although the composition varies with each tumor, immune cells can infiltrate the TME and take on several roles that promote and/or resist inflammation and tumor proliferation. 24 Although the mechanisms have not been fully elucidated, several studies have found associations between TAMs, tumor growth, and HL in VS.5,22,25-28
In a retrospective investigation of 46 patients with VS with subtotal resections, Graffeo et al 25 found that higher macrophage density (defined as the ratio of CD68+ macrophages to S100+ cells) was significantly associated with tumor progression postoperatively. However, it was unknown whether M1 and/or M2 macrophages were implicated, as CD68 is a nonspecific pan-macrophage marker. 26 Although there is some evidence that M1 TAMs may be associated with VS tumor progression, 27 several investigations have found a strong relationship between CD163+ M2 macrophages and greater tumor growth rates and/or larger-sized tumors.21,28,29 Because of differences in how tumor growth was measured across studies, it remains unclear from these investigations whether macrophage infiltration is actually protective and slows the dynamic volumetric tumor growth rate. 28
One study investigated the macrophage expression and HL in patients with VS. In a retrospective investigation of 11 VSs associated with poor hearing and 11 VSs associated with good hearing, Sagers et al 5 demonstrated a consistent but nonsignificant trend toward increased positivity for CD68+ macrophages in the poor hearing group. Similarly, in our 30 patients, we found mean expression levels for CD80 and CD163 to be higher in UH tumors ( Figure 2 ), but only the relationship between higher CD163 and UH was significant (P = .0161). In addition, the mean difference in CD163 between UH and SH groups was more pronounced than with CD80. Unfortunately, the CD163 level did not remain significant as an independent predictor variable associated with UH on multivariate analysis, when controlling for confounding factors such as tumor volume. This may be related, in part, to the sample size and/or the method of quantification (eg, percent area, rather than cell density). However, this trend toward increased positivity for CD80+ and CD163+ macrophages is consistent with Sagers et al, 5 suggesting that TAM is likely one factor of several TME components that contributes to HL. Further investigations are needed to measure impacts of other TME components and delve deeper into different M2 macrophage subtypes.
We also demonstrated that prior VS treatment with surgery, radiation, or chemotherapy was a significant independent predictor of UH. This association may relate to selection bias, as previously treated patients with good hearing may not have been selected for salvage surgery and thus not represented in the patient cohort. Alternatively, prior treatment may have altered the local TME, causing local inflammation and promoting more monocyte migration and macrophage polarization. But on further analyses, there were no differences in CD80 and CD163 expression in tumors of patients with and without prior treatments. Further investigations are warranted to fully elucidate the relationship between prior treatment and HL.
Furthermore, we did not find significant correlations between preoperative tumor volume and CD80 or CD163 levels, which is contradictory to other investigations that have found links between macrophages and tumor size and/or progression.21,27-29 This discrepancy may reflect several factors, including differences in patient selection for surgery and proportions of small to large tumors. While we analyzed 30 consecutive patients, some researchers analyzed fast- and slow-growing tumors, excluding tumors where growth rate was not assessed.21,27-29 We did not assess tumor growth rates because only a third of patients underwent initial observation. The discrepancy may also reflect methodologic differences in tissue processing, macrophage subtypes evaluated, antibodies used, staining protocols, and target quantification.21,27-29 We used fixed tissue embedded in OCT medium and analyzed percent area of expression, while others used paraffin-embedded tissue and quantified staining intensity, number of positive cells, percent area, or a combination.
In the interest of developing an in vitro assay to study VS TME on macrophage polarization and hearing, we cocultured VS cells and monocytes. In transwell experiments, monocytes migrated through insert filters toward the bipolar, S100+ VS cells adherent to the culture dish ( Figure 3 ). In our direct coculture experiments, monocytes polarized to CD80+ and CD163+ macrophages, with CD163+ M2 macrophages as the predominant type ( Figure 4 ). Some monocytes costained for CD80 and CD163, consistent with macrophage polarization occurring across a spectrum, with CD80 and CD163 representing phenotypic extremes of M1 and M2 macrophages, respectively.18,30-32
Although there were no obvious differences in macrophage densities, we observed subtle differences in CD163+ M2 macrophage morphologies between hearing groups ( Figure 4 ). The SH group had larger M2 macrophages—many amoeboid shaped with pseudopodia and some with multiple nuclei. In contrast, the UH group demonstrated small M2 macrophages that were amoeboid, round, or spindle-like. In the literature, M1 macrophages have been described as small, spindle-like, or amoeboid, with delicate cytoplasmic extensions, while M2 macrophages were distinctly spindle-like, amoeboid, or round or resembled large, multinucleated giant cells.26,33,34 There is some evidence that macrophage morphology can affect profiles of secreted cytokines. 35 In addition, M2 macrophages can be divided into phenotypic subtypes (M2a, M2b, M2c, and M2d) associated with distinct cell markers, each with their own polarization triggers, cytokines, and roles, including wound healing, infection response, phagocytosis of apoptotic cells, angiogenesis, and tumor progression.17,36-40 Our findings suggest that the VS TME may cause monocytes to polarize to M2 macrophages of different sizes and morphologies; however, this raises questions whether macrophage morphologies are different phenotypically in the cytokines they secrete and whether that leads to HL or other clinical manifestations. Future investigations elucidating these relationships can lead to new therapies for HL and tumor progression in patients with VS.
Study limitations include a small cohort of patients with varying clinical presentations and tumor characteristics, inability to measure tumor growth rate in most patients, and inadequate specimens to formally analyze cytokine expression. CD80 and CD163 expression was quantified objectively using ImageJ software; however, the reviewer was not blinded to hearing status, potentially adding a source of bias. Although we have developed in vitro assays as a proof of concept that VS can drive monocyte migration and differentiation, further experiments with a larger sample size will be needed to perform quantitative evaluations of macrophage polarization, morphology and subtyping, cytokine profiling, and effect on auditory hair cell survival. Although in vitro experiments do not have direct clinical implications, they may help researchers understand the TME, investigate cytokines involved in tumor progression and HL, provide a framework for applying precision medicine in future treatment algorithms, and provide an avenue for testing new therapies for patients with VS.
Conclusion
VS tumors express varying degrees of CD80+ M1 and CD163+ M2 TAMs. We found higher CD163+ expression in patients with VS with UH, but this did not remain significant on multivariate analysis. Although these findings suggest M2 macrophages may contribute partly to HL in patients with VS, TAMs may be one of several TME components responsible for HL. We also describe in vitro assays in a proof-of-concept investigation that VS cells can initiate monocyte migration and polarization toward M1 and M2 macrophage phenotypes. Future investigations are warranted to investigate the relationships between individual VS, macrophage phenotypes, subtypes, and morphologies and secreted cytokines as they relate to hearing and tumor progression. By elucidating these relationships, we will be able to develop and test new therapies for HL and tumor progression in patients with VS.
