Abstract
Background:
It is unknown how the increased range of motion and improved gait mechanics provided by total ankle arthroplasty (TAA) affects the progression of knee arthritis compared with ankle arthrodesis (AA). We hypothesized that patients treated with TAA would have a lower incidence of postoperative knee arthritis and total knee arthroplasty (TKA) compared with AA.
Methods:
We retrospectively reviewed a matched cohort of 3751 AAs and 3751 TAAs performed between January 1, 2007, and December 31, 2021, using a commercial claims database. Patients with a history of preoperative knee arthritis or TKA were excluded. Patients were matched 1:1 for gender, age at time of ankle surgery, indication for ankle surgery, year of ankle surgery, and geographical region. Univariate and multivariate regression analyses were performed. Significance was set at P < .05.
Results:
Demographics were similar between AA and TAA. AA (absolute risk [AR] = 17.54%, odds ratio [OR] = 1.24, CI = 1.07-1.44), earlier years of ankle surgery, and older age increased the risk of postoperative knee arthritis compared with TAA (AR = 10.10%, absolute risk reduction [ARR] = 7.44%, CI = 5.89%-8.99%). Posttraumatic ankle arthritis, inpatient surgery, and surgery in the West region were protective against developing postoperative knee arthritis. AA (AR = 4.19%, OR = 2.37, CI = 1.65-3.39), earlier year of ankle surgery, males, and other indication for ankle surgery increased the risk of requiring a postoperative TKA compared with TAA (AR = 1.36%, ARR = 2.83%, CI = 2.09%-3.57%). Posttraumatic ankle arthritis and inpatient surgery were protective factors against requiring a TKA.
Conclusion:
There is a significantly higher incidence of patients who underwent TKA after AA compared with TAA, although the inability to confirm laterality in 38% of cases dilutes the hypothesis of a specific biomechanical association in the study. We recommend that surgeons carefully weigh the risks and benefits of TAA and AA with this in mind, especially in patients with minimal knee arthritis, as a potential strategy to mitigate risk of requiring TKA in the future.
Level of Evidence:
Level III, retrospective comparative study.

Keywords
Introduction
Both ankle arthrodesis (AA) and total ankle arthroplasty (TAA) are valid therapeutic options for addressing end-stage ankle arthritis, offering substantial and enduring enhancements in pain alleviation and functional restoration. 1 TAA provides the advantage of maintaining ankle joint range of motion, thereby enhancing the biomechanics of the arthritic ankle. 2
A critical consideration in the choice between these interventions is the potential for downstream effects on adjacent joints. Previous investigations have underscored a possible association between AA and subsequent subtalar joint osteoarthritis necessitating subtalar joint arthrodesis.2,3 Moreover, modified gait dynamics resulting from ankle fusion could impose additional stresses on the knee joint, potentially accelerating degeneration and predisposing patients to knee osteoarthritis. In contrast, TAA serves to retain a more natural motion within the ankle joint, possibly mitigating knee deterioration.
Gait studies have demonstrated that AA can lead to compensatory increases in hip and knee motion and elevated heel-strike forces, suggesting a broader impact on lower limb biomechanics beyond the ankle alone.4,5 Increased plantar pressures and anterior shifts in center-of-pressure distribution have also been reported following AA, potentially contributing to altered knee joint loading patterns.6,7 Conversely, TAA patients retain more physiologic gait symmetry and hindfoot mobility, which may protect adjacent joints such as the knee.8,9
We hypothesize that patients treated with TAA would have a lower incidence of postoperative knee arthritis and progression to TKA compared with AA. We evaluated whether AA poses a greater risk of progression to TKA when compared with TAA. Such insights are crucial for informed decision making by patients and orthopaedic surgeons when choosing the most suitable intervention for end-stage ankle arthritis. Additionally, this analysis holds implications for long-term patient management, aiming to minimize downstream impacts on adjacent joints.
Materials and Methods
After approval from our institutional review board, a retrospective cohort study was conducted using the MarketScan Commercial Claims Database (Merative, Ann Arbor, Michigan). This database utilizes employer-sourced insurance claims data but is limited in the clinical data it provides as it is not linked to patient electronic health records. A total of 26 628 primary AA and 11 339 primary TAA claims were identified between January 1, 2007, and December 31, 2021, based on the Current Procedural Terminology (CPT), International Classification of Diseases, Ninth (ICD-9) and Tenth Revision (ICD-10), codes listed in Supplementary Table S2. Indication for ankle surgery, including ankle osteoarthritis, posttraumatic ankle arthritis, rheumatoid arthritis, or another indication for ankle surgery was determined based on the ICD-9 and ICD-10 codes listed in Supplementary Table S2. We were unable to confirm the ipsilaterality of ankle surgery and future knee arthritis or TKA in 38% of cases because of ICD-9 codes not providing information about laterality. Claims were excluded based on the criteria described in Figures 1 and 2, leaving 10 221 AAs and 3883 TAAs in the final study population. Of note, we excluded all patients with prior diagnosis of knee arthritis and diagnosis of knee arthritis or TKA within 30 days of ankle surgery to ensure our analysis only considers postoperative onset of arthritis.
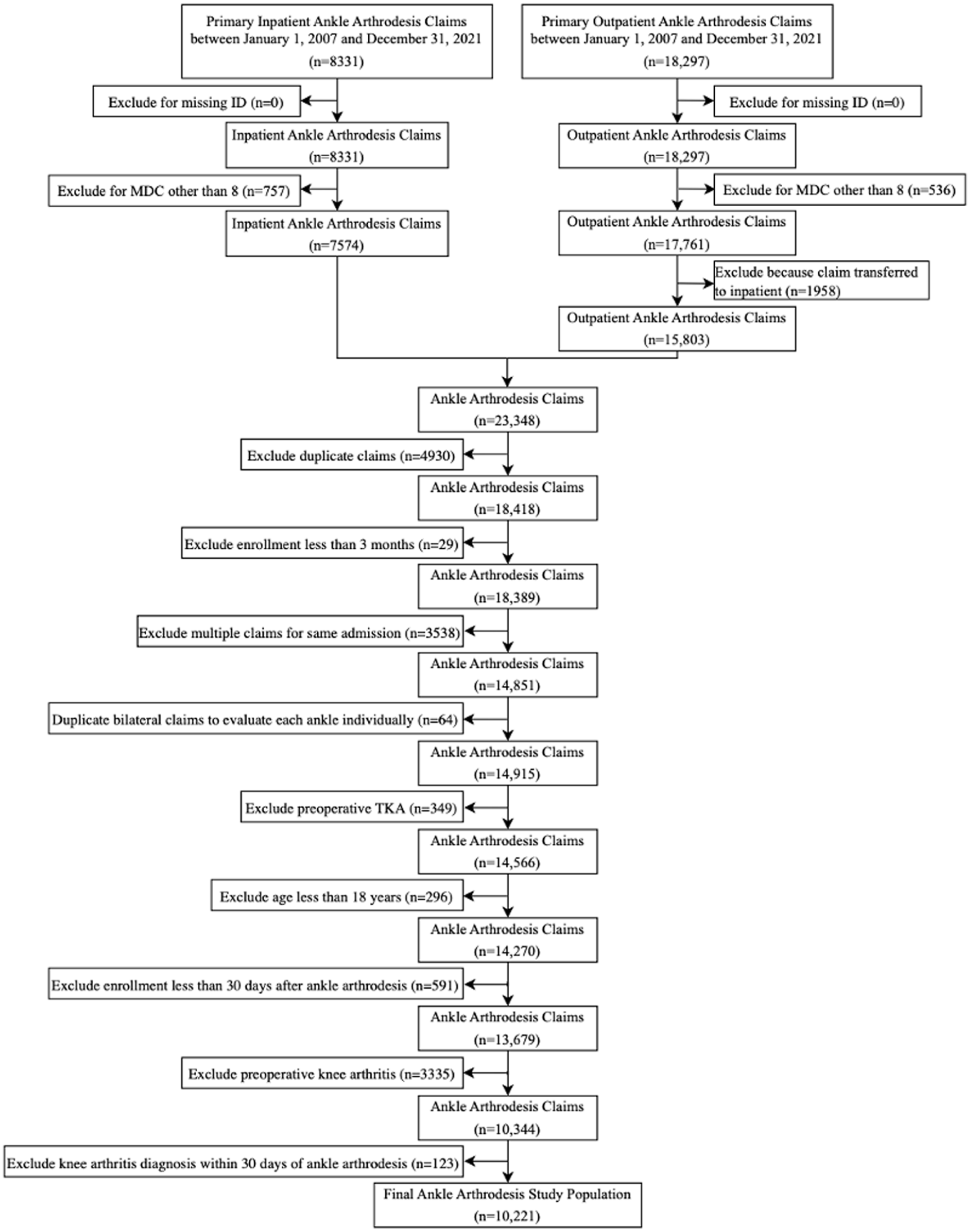
Flowchart demonstrating study design for ankle arthrodesis patients. ID, identification number; MDC, major diagnostic category.

Flowchart demonstrating study design for ankle arthroplasty patients. ID, identification number; MDC, major diagnostic category.
Patients were matched 1:1 by exact matching on gender, age at surgery, and surgical indication along with Mahalanobis nearest-neighbor matching on year of ankle surgery, type of health care facility, and geographical region. The 3751 AAs and 3751 TAAs that could be matched were included in the analysis. This matching strategy aligns with contemporary cohort study methodologies to mitigate confounding variables and ensure comparable baseline characteristics between treatment groups.10,11
Statistical Analysis
Python (Python Software Foundation) was used for statistical analysis. R (R Core Team) was used for matching and to calculate cumulative incidence of postoperative knee arthritis and TKA. Univariate and multivariate logistic regression were used to evaluate risk factors for progression to postoperative knee arthritis and TKA. Odds ratios (ORs) and 95% CIs were calculated. Independent samples t test was used to compare follow-up. Significance was set at P <.05. Minimum sample size was calculated to be 174 AAs and 174 TAAs. 12 Similar statistical approaches have been employed in large-database studies examining outcomes following ankle arthrodesis and arthroplasty.13,14
Results
Demographics
Demographics were similar between AA and TAA, including age (OR = 1.00, CI = 0.99-1.01), gender (OR = 1.00, CI = 0.91-1.09), type of health care facility where surgery occurred (OR = 1.01, CI = 0.92-1.11), diagnosis of posttraumatic ankle arthritis (OR = 1.00, CI = 0.91-1.10), or another indication for ankle surgery (OR = 1.00, CI = 0.79-1.27) compared with osteoarthritis. Patients who underwent an AA tended to have surgery closer to 2007 (OR = 0.78, CI = 0.77-0.79) and therefore had longer follow-up (P < .001). Specifically, the AA cohort had a mean follow-up of 2.50 years (range = 0.08-14.54 years), whereas TAA patients had a shorter average follow-up duration of 1.89 years (range = 0.08-14.85 years) (Supplementary Table S1). There were regional differences between cohorts, including more TAAs were performed in the South (OR = 0.79, CI = 0.68-0.91) and West (OR = 0.68, CI = 0.58-0.81) compared with the Northeast (Supplementary Table S2).
Risk factors for developing postoperative knee arthritis
At 10 years, the cumulative incidence of knee arthritis was higher for AA than TAA (53.1% vs 41.9%, P < .001; Figure 3). Univariate analysis confirmed increased risk with AA (OR = 1.89, CI = 1.65-2.17; absolute risk [AR] = 17.54%) compared with TAA (AR = 10.10%, absolute risk reduction [ARR] = 7.44%, CI = 5.89%-8.99%). Earlier years of ankle surgery (OR = 0.88, CI = 0.87-0.90) also increased the risk of developing knee arthritis. A diagnosis of posttraumatic ankle arthritis compared with ankle osteoarthritis (OR = 0.77, CI = 0.67-0.88) and having ankle surgery in the West region compared with the Northeast (OR = 0.75, CI = 0.60-0.95) were protective factors against developing knee arthritis. Gender (OR = 1.00, CI = 0.88-1.14), age (OR = 1.00, CI = 1.00-1.01), inpatient ankle surgery (OR = 0.89, CI = 0.77-1.01), another indication for ankle surgery compared with ankle osteoarthritis (OR = 1.34, CI = 0.99-1.83), and the other geographical regions were not risk factors for developing knee arthritis (Table 1).

Cumulative incidence of postoperative knee arthritis. This line graph demonstrates the cumulative incidence of developing postoperative knee arthritis based on if the patient underwent total ankle arthroplasty vs ankle arthrodesis. Patients who underwent an ankle arthrodesis had a higher cumulative incidence of developing postoperative knee arthritis (53.1%) compared with those who underwent a total ankle arthroplasty (41.9%) at 10 years after ankle surgery (P < .001). Of note, cohort numbers decrease at a relatively higher rate after 10 years.
Risk Factors for Postoperative Knee Arthritis.
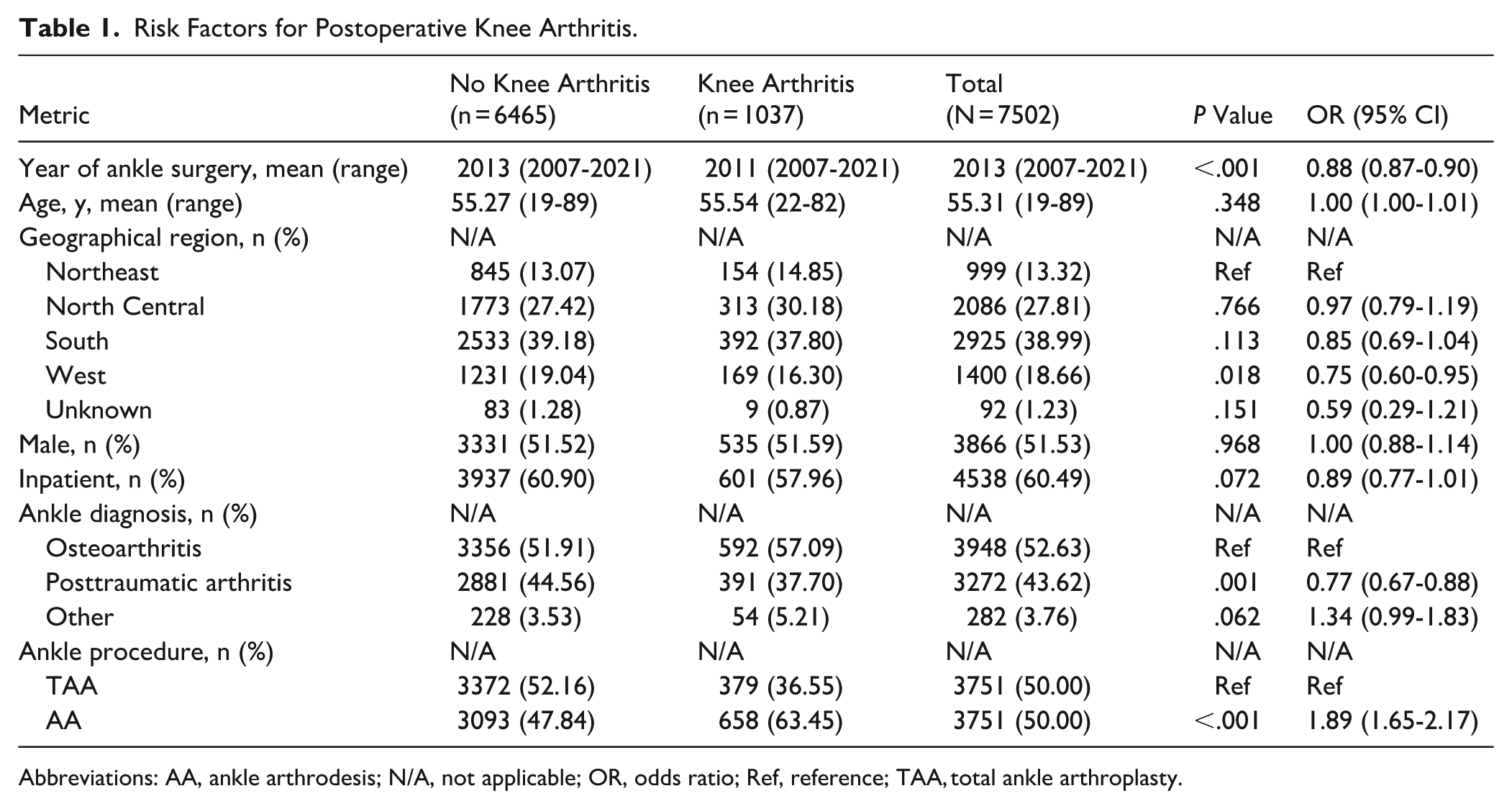
Abbreviations: AA, ankle arthrodesis; N/A, not applicable; OR, odds ratio; Ref, reference; TAA, total ankle arthroplasty.
A multivariate analysis showed that AA compared with TAA (OR = 1.24, CI = 1.07-1.44), earlier year of ankle surgery (OR = 0.89, CI = 0.87-0.90), and older age (OR = 1.01, CI = 1.01-1.02) increased the risk of developing knee arthritis. A diagnosis of posttraumatic ankle arthritis compared with ankle osteoarthritis (OR = 0.82, CI = 0.72-0.95), inpatient ankle surgery (OR = 0.84, CI = 0.73-0.96), and having ankle surgery in the West region compared with the Northeast (OR = 0.71, CI = 0.56-0.91) were protective factors against developing knee arthritis. Gender (OR = 0.98, CI = 0.86-1.12), another indication for ankle surgery compared with ankle osteoarthritis (OR = 1.37, CI = 0.99-1.88), and the other geographical regions were not risk factors for developing knee arthritis (Table 2).
Multivariate Analysis for Risk Factors for Postoperative Knee Arthritis.

Abbreviations: AA, ankle arthrodesis; N/A, not applicable; OR, odds ratio; Ref, reference.
Risk Factors for Progression to Postoperative TKA
AA had a higher cumulative incidence of undergoing a TKA (36.3%) compared with TAA (21.7%) at 10 years after ankle surgery (P < .001; Figure 4). A univariate analysis showed that AA (OR = 3.17, CI = 2.30-4.36; AR = 4.19%) compared with TAA (AR = 1.36%, ARR = 2.83%, CI = 2.09%-3.57%), earlier year of ankle surgery (OR = 0.87, CI = 0.84-0.91), males (OR = 1.59, CI = 1.19-2.11), and another indication for ankle surgery other than osteoarthritis (OR = 3.42, CI = 2.24-5.22) increased the risk of requiring a TKA. A diagnosis of posttraumatic ankle arthritis compared with ankle osteoarthritis (OR = 0.47, CI = 0.34-0.66) and inpatient ankle surgery (OR = 0.54, CI = 0.41-0.71) were protective factors against requiring a TKA. Age (OR = 1.00, CI = 0.98-1.02) and geographical region were not risk factors for requiring a TKA (Table 3).

Cumulative incidence of postoperative total knee arthroplasty. This line graph demonstrates the cumulative incidence of requiring a postoperative total knee arthroplasty based on if the patient underwent total ankle arthroplasty vs ankle arthrodesis. Patients who underwent an ankle arthrodesis had a higher cumulative incidence of developing postoperative knee arthritis (36.3%) compared with those who underwent a total ankle arthroplasty (21.7%) at 10 years after ankle surgery (P < .001). Of note, cohort numbers decrease at a relatively higher rate after 10 years.
Risk Factors for Postoperative TKA.

Abbreviations: AA, ankle arthrodesis; N/A, not applicable; OR, odds ratio; Ref, reference; TAA, total ankle arthroplasty; TKA, total knee arthroplasty.
A multivariate analysis showed that AA compared with TAA (OR = 2.37, CI = 1.65-3.39), earlier year of ankle surgery (OR = 0.91, CI = 0.88-0.96), males (OR = 1.63, CI = 1.22-2.19), and another indication for ankle surgery other than osteoarthritis (OR = 3.77, CI = 2.41-5.89) increased the risk of requiring a TKA. A diagnosis of posttraumatic ankle arthritis compared with ankle osteoarthritis (OR = 0.51, CI = 0.37-0.71) and inpatient ankle surgery (OR = 0.50, CI = 0.37-0.66) were protective factors against requiring a TKA. Age (OR = 1.00, CI = 0.99-1.02) and geographical region were not risk factors for requiring a TKA (Table 4).
Multivariate Analysis for Risk Factors for Postoperative TKA.

Abbreviations: AA, ankle arthrodesis; N/A, not applicable; OR, odds ratio; Ref, reference.
Discussion
The management of end-stage ankle arthritis, whether through AA or TAA, is a complex decision that requires careful consideration of the potential implications on adjacent joints. A primary concern was the risk of progression to TKA following ankle surgery. We sought to critically review the findings of our analysis and examine implications of ankle surgery on the knee joint, bearing in mind that 38% of the cohort had unknown laterality, and this inclusion of potential contralateral disease dilutes the hypothesis of a specific biomechanical association in the study.
The data indicated that patients treated with AA were 3.17 times more likely to undergo subsequent TKA compared with those who were treated with TAA. We also demonstrate a 2.83% risk reduction for undergoing TKA and a 7.44% risk reduction for developing knee arthritis when TAA is chosen over AA. These findings present an opportunity for surgeons to opt for treatment that decreases the chances of developing knee arthritis, and to a lesser extent, decreases the likelihood of requiring another major surgical intervention. Importantly, preventing the potential development of knee arthritis and progression to TKA improves patient quality of life, reduces health care costs, and eliminates risks associated with further surgery.
TAA, with its preservation of ankle joint motion, is inherently less likely to exert adverse effects on the knee joint. Although both AA and TAA may significantly alter gait, Barton et al 8 noted the preservation of natural ankle motion in TAA that may lead to reduced magnitude of biomechanical changes. Similarly, the TARVA randomized controlled trial demonstrated that TAA recipients achieved significantly greater improvements in walking and standing scores than AA patients. 15
The absence of ankle motion after AA can alter gait mechanics and transmit abnormal forces up the kinetic chain, potentially overloading and accelerating degenerative changes in the knee joint. A systematic review by Deleu et al 16 demonstrated that while both AA and TAA induce modification in gait biomechanics, TAA showed improved lower limb kinematics, including increased range of motion (ROM), ankle power, and improved knee and hip motion. However, AA was associated with greater ankle moment and compensatory hip motion without improved knee ROM.4 -8,11,17
Biomechanical differences between AA and TAA have been well documented, prompting several studies evaluating their impact on gait. 16 Brodsky et al 18 conducted a prospective study of 50 patients with advanced ankle arthritis treated with unilateral TAA using the STAR prosthesis. Three-dimensional gait analysis showed significant improvements in ankle power and ROM, as well as knee and hip ROM compared with preoperative values. Doets et al 19 compared the gait characteristics of a patient who had undergone mobile-bearing TAA with those of age-matched and sex-matched healthy controls. Their findings revealed that, following a successful TAA, individuals could achieve a nearly normal gait pattern in terms of knee, ankle, and tarsal joint kinematics, despite a slight reduction in ankle dorsiflexion. Additionally, in the surgical group, EMG activity patterns were largely normal, except for higher activity in the gastrocnemius during early stance and the tibialis anterior muscle during late stance. In a study by Buck and colleagues, gait analysis, including knee function, was conducted on patients who had undergone AA. Their findings revealed that AA indeed leads to changes in gait. However, it was determined that positioning AA in a neutral flexion, with slight (zero to 5 degrees) valgus angulation and approximately 5 to 10 degrees of external rotation, allows for the most compensatory motion at the foot and places the least strain on the knee. 20 In their study comparing gait analysis in patients with ankle osteoarthritis both before and after AA, Brodsky and his coauthors discovered improvements in various temporal-spatial, kinematic, and kinetic measures. These improvements encompassed step length, velocity, ankle moment, hip power, and ROM. These compensatory adjustments resulted in a decrease in the total time of support on the unaffected limb. The authors concluded that postoperative compensatory movements in adjacent joints, even if they do not lead to a complete return to a normal gait, effectively substituted for the lost motion at the ankle. 21
Recent randomized trials reinforce these trends: although both AA and TAA improve pain and function, TAA tends to support more efficient gait and reduce stress on adjacent joints. 9 Similarly, long-term registry data indicated fewer secondary surgeries after TAA, highlighting its potential protective effect on the knee and subtalar joints. 22
It is important to note that we were unable to confirm the laterality of subsequent knee arthritis and TKA in 38% of the cohort. This inclusion of potential contralateral disease does dilute the specific biomechanical argument of this study; however, we still believe the insights generated by this analysis are valuable. In future studies, it will be crucial to confirm the ipsilaterality of AA/TAA and subsequent degenerative knee disease to verify our hypothesis.
The observed likelihood of requiring TKA following AA compared with TAA suggests that factors extending beyond ankle joint motion may contribute to knee joint degeneration. We noted that advanced age, primary osteoarthritis, and earlier ankle surgeries were associated with a heightened risk of proceeding to TKA. Moreover, variables such as surgical technique, the specific design of the TAA prosthetic implant, and postoperative rehabilitation may also be linked to the need for TKA. In a prospective multicenter cohort study conducted by Paterson and colleagues, they found a significant association between foot and ankle pathology and an elevated risk of developing knee osteoarthritis within the following 4 years. They proposed that the mechanisms leading to knee osteoarthritis might be related to alterations in limb biomechanics resulting from ankle dysfunction. Furthermore, ankle malalignment, particularly excessive pronation, may predispose individuals to increased forces on the medial knee compartment. Finally, genetic predispositions such as changes in central pain processing may contribute to development of the lower limb polyarthritis. 23
Patients facing end-stage ankle arthritis, in consultation with their orthopaedic surgeons, must carefully weigh the risks and benefits of AA and TAA. Although TAA may offer the advantage of preserved ankle motion and potentially reduced risk of subsequent knee joint degeneration, it is essential to consider the individual patient’s unique anatomy and comorbidities and the expertise of the surgeon. Moreover, our findings underscore the importance of long-term follow-up and vigilance in patients who undergo AA. Orthopaedic surgeons should be attentive to changes in gait mechanics, knee joint symptoms, and subtalar joint function in patients with a history of AA, as these may indicate a need for early intervention to mitigate further joint degeneration.
Limitations
Although our study provides important insights into the comparative risks of TKA following ankle surgery, several limitations must be acknowledged to provide a comprehensive perspective on the findings. This study relied on a commercial claim database, which has inherent limitations. The data are subject to coding errors, omissions, and variations in coding practices across health care providers. These databases primarily capture billing information, which may lack detailed clinical nuances and comprehensive patient histories. With regard to our study, delayed or absent coding could misclassify existing knee arthritis as a “new” problem and thus overestimate cases of postoperative knee disease and TKA. Additionally, we were unable to confirm laterality in 38% of the cases used in the study. Our decision to presume that the claim was referring to the ipsilateral leg may also overestimate the prevalence of postoperative knee arthritis and TKA.
The choice between AA and TAA is influenced by various factors, including patient preferences/comorbidities, surgeon expertise, and the specific characteristics of ankle pathology. These factors may introduce selection bias into the study, making it challenging to establish a cause-and-effect relationship between the type of ankle surgery and the risk of subsequent TKA. Additionally, we were unable to follow any patients once they had unenrolled from the database, which may underestimate the prevalence of postoperative knee arthritis and TKA. The duration of follow-up may not capture the long-term consequences of ankle surgery adequately. Knee joint degeneration, a gradual process, may manifest after the study period, leading to an underestimation of the risk associated with AA or TAA. Furthermore, patients undergoing AA had a longer mean follow-up compared with TAA patients. Although our methodology used a matched cohort, an important limitation is that the reported increased risk of postoperative knee arthritis and TKA in this study after AA may be an artifact of the significantly longer follow-up time in the arthrodesis group. Additionally, 38% of the cohort had unknown laterality, and this inclusion of potential contralateral disease does dilute the hypothesis of a specific biomechanical association in the study.
The database did not account for variations in surgical techniques, implant designs, or postoperative rehabilitation protocols. These factors can influence outcomes, including their impact on the knee joint, but were beyond the scope of this study. The findings of this study may have limited generalizability to populations outside those covered by commercial insurance, potentially excluding uninsured or publicly insured individuals.
Considering these limitations, our results should be interpreted with caution. Further research is warranted to better understand the mechanisms underlying the increased risk of TKA following AA. Prospective studies with larger sample sizes, longer follow-up periods, and detailed clinical data, are necessary to provide more robust data to validate our findings. Additionally, investigations into the specific factors contributing to knee joint degeneration following ankle surgery, such as biomechanical changes, patient-specific variables, and postoperative rehabilitation can offer valuable insights for optimizing patient outcomes.
Conclusion
We found a significantly higher incidence of patients who underwent TKA after AA compared with TAA. We recommend that surgeons carefully weigh the risks and benefits of TAA and AA with this in mind, especially in patients with minimal knee arthritis, as a potential strategy to mitigate risk of requiring TKA in the future, recognizing that future studies confirming ipsilaterality are necessary to fully verify this hypothesized biomechanical association.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114261435963 – Supplemental material for Ankle Arthrodesis Associated With Risk of Progression to Total Knee Arthroplasty Compared to Total Ankle Arthroplasty
Supplemental material, sj-pdf-1-fao-10.1177_24730114261435963 for Ankle Arthrodesis Associated With Risk of Progression to Total Knee Arthroplasty Compared to Total Ankle Arthroplasty by Gloria Coden, Colin Wood, Andrzej Brzezinski, Matthew Beckles, Shannon Gray, Riley Smith and Kurt Hofmann in Foot & Ankle Orthopaedics
Footnotes
Appendix
Medical Billing Codes.

| ICD-9 codes | ICD-10 codes | CPT codes | |
|---|---|---|---|
| Ankle arthrodesis | 81.11 | 0SGF03Z, 0SGF04Z, 0SGF05Z, 0SGF07Z, 0SGF0JZ, 0SGF0KZ, 0SGF33Z, 0SGF34Z, 0SGF35Z, 0SGF37Z, 0SGF3JZ, 0SGF3KZ, 0SGF43Z, 0SGF44Z, 0SGF45Z, 0SGF47Z, 0SGF4JZ, 0SGF4KZ, 0SGG03Z, 0SGG04Z, 0SGG05Z, 0SGG07Z, 0SGG0JZ, 0SGG0KZ, 0SGG33Z, 0SGG34Z, 0SGG35Z, 0SGG37Z, 0SGG3JZ, 0SGG3KZ, 0SGG43Z, 0SGG44Z, 0SGG45Z, 0SGG47Z, 0SGG4JZ, 0SGG4KZ | 27870, 29899 |
| Total ankle arthroplasty | 81.56 | 0SRF07Z, 0SRF0J9, 0SRF0JA, 0SRF0JZ, 0SRF0KZ, 0SRG07Z, 0SRG0J9, 0SRG0JA, 0SRG0JZ, 0SRG0KZ | 27702 |
| Ankle osteoarthritis | 715.17, 715.37, 715.97, 716.57, 716.67, 716.97 | M13.171, M13.172, M13.179, M19.071, M19.072, M19.079 | N/A |
| Ankle posttraumatic arthritis | 715.27, 716.17 | M12.571, M12.572, M12.579, M19.171, M19.172, M19.179, M19.271, M19.272, M19.279 | N/A |
| Rheumatoid arthritis | 714 | M05.60, M05.69, M05.70, M05.79, M05.80, M05.89, M05.9, M06.00, M06.09, M06.80, M06.89, M069 |
N/A |
| Ankle rheumatoid arthritis | N/A | M05.071, M05.072, M05.079, M05.171, M05.172, M05.179, M05.271, M05.272, M05.279, M05. 371, M05.372, M05.379, M05.471, M05.472, M05.479, M05.571, M05.572, M05.579, M05.671, M05.672, M05.679, M05.771, M05.772, M05.779, M05.871, M05.872, M05.879, M06.071, M06. 072, M06.079, M06.871, M06.872, M06.879, M08.071, M08.072, M08.079, M08.271, M08.272, M08.279, M08.471, M08.472, M08.479, M08.871, M08.872, M08.879, M08.971, M08.972, M08. 979 |
N/A |
| Total knee arthroplasty | 81.54 | 0SRC069, 0SRC06A, 0SRC06Z, 0SRC0J9, 0SRC0JA, 0SRC0JZ, 0SRD069, 0SRD06A, 0SRD06Z, 0SRD0J9, 0SRD0JA, 0SRD0JZ |
27445, 27447 |
| Knee osteoarthritis | 715.16, 715.36, 715.96, 716.56, 716.66, 716.96 | M13.161, M13.162, M13.169, M17.0, M17.10, M17.11, M17.12, M17.9 | N/A |
| Knee posttraumatic arthritis | 715.26, 716.16 | M12.561, M12.562, M12.569, M17.2, M17.30, M17.31, M17.32, M17.4, M17.5 | N/A |
| Knee rheumatoid arthritis | N/A | M05.061, M05.062, M05.069, M05.161, M05.162, M05.169, M05.261, M05.262, M05.269, M05.361, M05.362, M05.369, M05.461, M05.462, M05.469, M05.561, M05.562, M05.569, M05.661, M05.662, M05.669, M05.761, M05.762, M05.769, M05.861, M05.862, M05.869, M06.061, M06.062, M06.069, M06.861, M06.862, M06.869, M08.061, M08.062, M08.069, M08.261, M08.262, M08.269, M08.461, M08.462, M08.469, M08.861, M08.862, M08.869, M08.961, M08.962, M08.969 | N/A |
Abbreviations: CPT, Current Procedural Terminology; ICD-9, International Classification of Diseases, Ninth Edition; ICD-10, International Classification of Diseases, Tenth Edition.
Author Note
Work performed at: New England Baptist Hospital Department of Orthopaedic Surgery, Boston, MA, USA.
Ethical Considerations
This study was approved by the institutional review board of the New England Baptist Hospital
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Disclosure forms for all authors are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
