Abstract
Background:
Globally, rising rates of ankle arthritis have largely driven increases in total ankle arthroplasty (TAA) and revision TAA (rTAA). INVISION (Wright Medical/Stryker) is a novel modular implant system designed for addressing rTAA-associated challenges; however, clinical outcomes remain limited. Therefore, the purpose of this systematic review is to synthesize the available INVISION rTAA literature to evaluate implant survivorship and complication rates.
Methods:
A PROSPERO-preregistered query of PubMed/MEDLINE, Embase, Cochrane, CINAHL, and Web of Science was performed on August 18, 2025, to identify INVISION-using rTAA studies. Quality was assessed using the Methodological Index for Non-Randomized Studies (MINORS). Extractions included demographics and operative outcomes. Statistics included frequency-weighted means (FWMs) and meta-analyses.
Results:
Five retrospective studies were included (mean MINORS = 10.2 ± 0.8; n = 100; FWM age = 64.0 ± 8.4 years; follow-up = 2.8 ± 1.2 years; 56% male; body mass index = 30.5 ± 5.5 kg/m2; time-to-revision = 4.5 ± 2.5 years). Tibial-talar implant permutations were INVISION-INVISION (25%), INVISION-INBONEII (19%), and INBONEII-INVISION (56%). Patients underwent an average of 1.8 additional procedures to rTAA: frequently medial malleolus fixation (21.8%) and hardware removal (17.3%). Pooled implant survivorship was 88%. Complication rates varied (0%-39.3%), and the all-cause rTAA pooled reoperation rate was 21%, driven primarily by aseptic loosening (38.1%) and infection (28.6%). The pooled re-revision rate was 11%. Meta-analyses revealed no significant differences in either all-cause reoperation or re-revision risk when comparing INVISION-INVISION to any other implant permutations.
Conclusion:
INVISION rTAA demonstrated early survivorship and complication rates that should be interpreted cautiously given retrospective study designs, high heterogeneity, “very low” GRADE outcome certainty, and limited follow-up duration. Aseptic loosening and infection remain the predominant drivers of reoperation and re-revision. No significant differences in these risks were detected across implant permutations. Future longitudinal, comparative studies are needed.
Introduction
Total ankle arthroplasty (TAA) utilization has grown substantially in recent years, attributable to rising global incidences of post-traumatic ankle arthritis.1 -5 Correspondingly, revision TAA (rTAA) procedures have also increased considerably as a reflection of primary TAA failures, with similar growth in procedure volume anticipated.5 -8 Indications for rTAA are variable, but prosthesis subsidence and aseptic loosening are among the most common. 9 Compared with primary TAA, rTAA procedures are also often considered more technically demanding and are associated with considerable reoperation rates, frequently because of peri-implant complications.7,10,11 These challenges have prompted the development of newer-generation implant systems with unique, ameliorative features.10,12,13 One such system demonstrating increasing utilization is the INVISION modular implant (Wright Medical/Stryker).13,14
Initially approved in the United States following FDA 510(k) clearance in May 2016 (K153008), INVISION was poised as the first rTAA-specific implant system, with indications spanning various forms of arthritis to failed prior surgeries. 15 Distinguishing features of this fourth-generation system include the interchangeability of its tibial and talar components with those of other widely used implants, such as Infinity and INBONE (both: Wright Medical Technology), alongside offerings of multiple plate-thickness selections, versatility when facing compromised bone stock, and a broad anatomic footprint for improved stability. 14 Although several smaller studies have evaluated the performance of INVISION in traditionally challenging rTAA scenarios and reported mixed clinical outcomes, the available evidence remains uncollated and relatively underpowered in isolation. Therefore, this systematic review aims to evaluate the clinical outcomes and complications in patients undergoing rTAA using the INVISION modular implant system.
Methods
Study Creation and Initial Search
PubMed/MEDLINE, Embase, Cochrane, CINAHL, and Web of Science were queried through August 18, 2025, for studies reporting INVISION-rTAA clinical outcomes. The PubMed/MEDLINE search strategy was: (1) [(“total ankle replacement”[tiab] OR “total ankle arthroplasty”[tiab] OR “total ankle replacements”[tiab] OR “total ankle arthroplasties” [tiab] OR arthroplasty, ankle replacement[MeSH Terms]) AND (invision[tw])]; (2) [(Invision[tw])]; (3) strategy 1 OR 2. Additionally, a grey literature search was also performed on August 18, 2025, during which the first 5 pages of Google Scholar were examined. All database and grey literature search strategies are provided in Supplemental Table 1. In accordance with standardized practices, this study was performed under the guidelines of the most recent Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), and preregistered in the PROSPERO registry for systematic reviews and meta-analyses (CRD420251118668). 16
Inclusion and Exclusion Criteria
To meet full inclusion criteria, studies must have reported surgical details (such as primary TAA implant characteristics or additional operations), clinical scores in the form of patient-reported outcome measures (PROMs) assessing endpoints like patient function or pain, complications, reoperations, and/or other relevant clinical details in accordance with predefined, preregistered outcome domains. Studies were excluded if they were primarily nonhuman (cadaver, animal, etc.) research, had only non-rTAA results, reported non-INVISION-only implant findings, were technical reports, case studies, reviews, surveys, letters to the editor, lacking full-text access, and/or were not published in English.
Article Screening Process
Rayyan, a public website used for systematic reviews, was used for screening. 17 First, one author independently performed manual deduplication, followed then by title and abstract screening (S.Y.). Then, a second author independently performed the same screening and confirmed findings, blinded to the initial author’s screening decisions (B.G.). Any resultant conflicts here were resolved by the present study’s first author.
Data Extraction
Two authors independently completed data extraction and cross-checked accuracy (S.Y. and B.G.). Variables retrieved included author information, follow-up, body mass index (BMI), complications, and clinical scores/PROMs, among other predefined, preregistered endpoints. Any resultant conflicts here were resolved by the present study’s first author.
Article Quality Grading
To assess the quality of included studies, the Methodological Index for Non-Randomized Studies (MINORS) scale was used. Within MINORS, all studies are first classified as either “noncomparative” or “comparative”. 18 If deemed noncomparative in design, articles are graded out of 16 points, on a 12-item spectrum, and with each item rated from 0 to 2 points. If deemed comparative, articles are graded out of 24 points, in similar fashion to above. Based on the summative score, studies were considered “high quality” if generating 16 or 24 points, “moderate quality” if generating 10-15 or 15-23 points, or “low quality” if generating less than 10 or less than 15 points for noncomparative and comparative studies, respectively. 19
Certainty Assessment
The Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) system was used to assess outcome certainty. Through GRADE, outcomes can be labeled as having “very low,” “low,” “moderate,” or “high” certainty based on several factors. 20 Within this assessment model, it is important to note that all outcomes from observational studies inherently begin at “low” certainty and proceed to either increase or decrease, largely because of natural limitations stemming from nonrandomization and associated imprecision.
Statistical Analysis
The IBM Statistical Package for the Social Sciences (SPSS) version 30.0 (IBM Corporation) and Microsoft Excel for Microsoft 365 Apps for Enterprise, version 2505 (Microsoft Corporation) were used for statistics. Frequency-weighted means (FWMs), standard deviations (SDs), and percentages (%) were employed for the collation of data. It should be noted that pooling was only performed when outcomes were deemed to have sufficiently comparable definitions, such as if identical PROM instruments were used across studies, to avoid potential misrepresentations. 21 Furthermore, if an outcome of interest was not reported or reported in an undistinguishable format in a respective study, it was excluded from pooling and/or additional statistical transformations due to insufficient data granularity. Where quantitative transformations may not have been appropriate, a qualitative/narrative approach was substituted. In instances where 2 or more studies demonstrated appropriate homogeneity and comparability, random effect meta-analyses were performed using risk ratios (RRs) with 95% confidence intervals (CIs) for binary variables. 22 In analyses involving multiple subgroup comparisons, resultant P values were Bonferroni-corrected to control for family-wise error (α = 0.0125). During such meta-analyses, if a study reported at least one zero-event, a treatment-arm continuity correction was applied, whereby a small sample-size-weighted adjustment was added only to the affected study arm to enable setting-appropriate, literature-backed meta-analytic practices.23,24 Of note, studies with complete separation (zeros in opposing arms) were excluded from such meta-analytic calculations because of undefined variance. For estimating the transformation of ranges to SDs, an extrapolated approach to the standards described by Hozo et al 25 was adopted. Similarly, for converting medians and interquartile ranges (IQRs) to means and SDs, an extrapolated approach to the standards described by Wan et al 26 was adopted. Lastly, when marked temporal heterogeneity was noted, such as follow-up reported in months instead of years, all value reporting types were converted to the value reporting type that followed the majority trend, such as months being converted to years in the aforementioned example.
Results
Search Results
The initial query retrieved 294 articles, of which 221 remained after deduplication. Title and abstract screening yielded 9 articles, 4 of which met inclusion criteria. One additional study was identified through grey literature search, resulting in 5 total included studies (Figure 1).14,27 -30
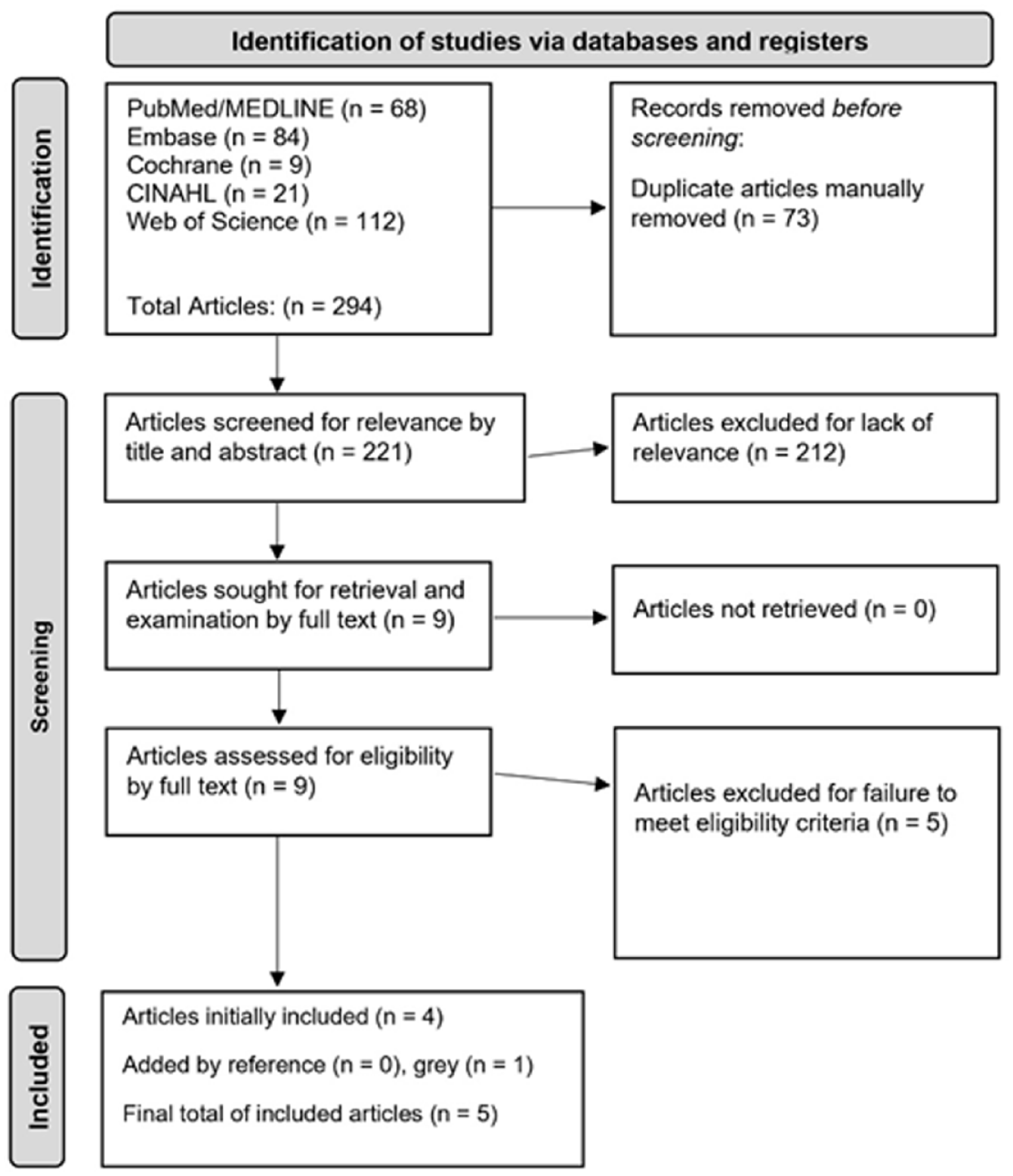
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram, outlining this study’s search algorithm progression.
Article Quality Results
All 5 included studies were observational, noncomparative, and retrospective in design (mean MINORS = 10.2 ± 0.8). Four studies were of moderate quality, and 1 study was of low quality (Supplemental Table 2). All extracted variables were conservatively downgraded to “very low” outcome certainty under GRADE because of the observational natures, instances of marked heterogeneity, capacities for imprecision, and potential biases.
Patient Demographics and Study Characteristics
A total of 100 patients underwent rTAA with INVISION (FWM age = 64.0 ± 8.4 years; follow-up = 2.8 years [FWM 80% reporting] ± 1.2 years [SD 60% reporting]; 56.0% male [80% reporting]; BMI 30.5 ± 5.5 kg/m2 [40% reporting]) (Supplemental Table 3). Most studies outlined inclusion/exclusion criteria that largely revolved around patients undergoing rTAAs with INVISION and a minimum follow-up requirement. Permutation proportions within this cohort (tibial-talar) were most frequently INBONEII-INVISION (56%), then INVISION-INVISION (25%), and lastly INVISION-INBONEII (19%) (Supplemental Table 3).
Primary Operation Details: Before rTAA
The primary operation before rTAA in this cohort was nearly exclusively TAA (99%), with only 1 case of ankle fusion (1%) (Table 1). Of the studies that delineated primary operation indications, the reasoning was exclusively joint arthritis (40% reporting). Four studies (80% reporting) specified primary operation implants: the most common were Infinity (23.3%), STAR (14.4%), INBONEI (13.3%), and INBONEII (13.3%), further described in Table 1. The FWM time from the primary operation to rTAA was 4.5 ± 2.4 years (80% reporting).
Operative, Indication, and Implant Details of the Primary, Pre-rTAA Operations From Included Studies. a
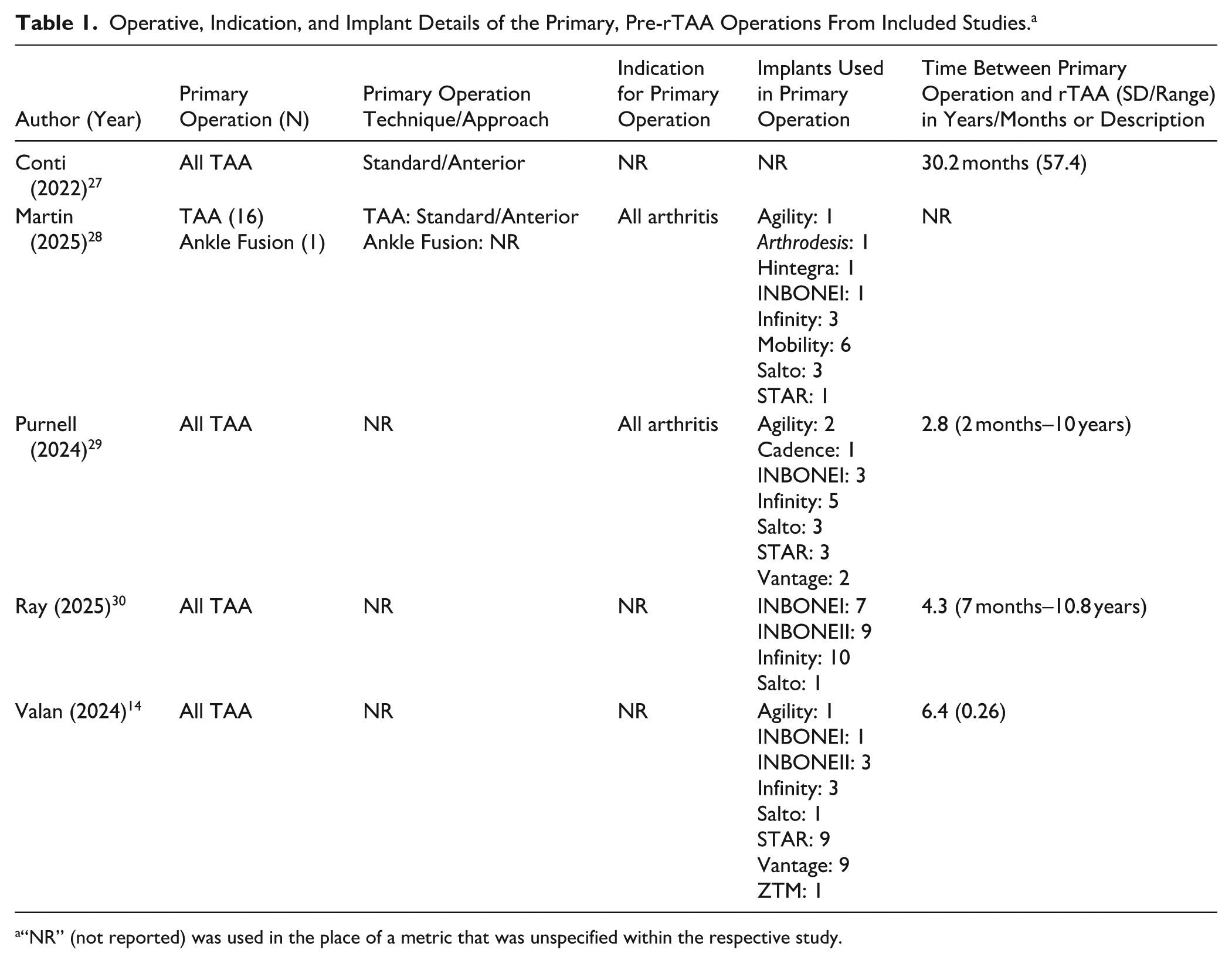
“NR” (not reported) was used in the place of a metric that was unspecified within the respective study.
rTAA Operative and Surgical Details
Indications for rTAA were variable. The most common was implant subsidence (27.8%), followed by aseptic loosening (26.4%), infection (23.6%), and cyst formation (6.9%): further delineated in Table 2 (80% reporting). Across studies that reported additional procedures (60% reporting), there were approximately 1.8 per patient. These were most frequently medial malleolus fixations (21.8%), followed then by bone grafting (18%), hardware removal (17.3%), and gastrocnemius recession (12.8%) (Table 2). Notably, only 7% of patients had radiograph-detectable postoperative cysts.
Operative and Surgical Details Specific to the rTAAs of Included Studies. a

NR (not reported) was used in the place of a metric that was unspecified within the respective study. UD (undistinguishable) was used where data were not adequately granularized for reporting within the respective study.
PROMs and Clinical Scores
Three studies detailed PROMs/clinical scores (60% reporting). Although entirely heterogenous, when perioperative values were provided across the various PROM instruments in Martin et al 28 and Purnell et al, 29 improvements were tallied and occurred in 85.7% (12 of 14) of these individual PROM domains pre- to postoperatively (40% reporting; Table 3). Among these, outcomes meeting ankle-literature-supported minimal clinically important difference (MCID) thresholds, where available, included Patient Reported Outcome Measurement Information System (PROMIS) pain intensity, PROMIS pain interference, Manchester-Oxford Foot Questionnaire (MOXFQ) walking, MOXFQ pain, MOXFQ social, Ankle Osteoarthritis Scale (AOS) Pain, and AOS Disability.31 -34
PROM and Clinical Score Details of Included Studies. a

NR (not reported) was used in the place of a metric that was unspecified within the respective study. UD (undistinguishable) was used where data was not adequately granularized for reporting within the respective study.
Complications, Reoperations, and Implant Survivorship
Four studies provided distinguishable, author-defined complication enumeration, with rates ranging from 0% to 39.3% (80% reporting). Because of heterogeneity in classification, these granular complications were unable to be pooled or analyzed further (Table 4). In contrast, all included studies reported rates for reoperation and re-revision, with event-specific detailing when above zero percent. The pooled all-cause reoperation rate was 21% (0%-40.7%): a collation represented most frequently by aseptic loosening (38.1%) and infection (28.6%) (100% reporting). Only 2 studies reported amputations, with 1 case in each (2% cohort rate): one occurred in an INVISION-INVISION revision and the other was INBONEII-INVISION, both due to recurrent infections. Of all-cause reoperations, the implant permutation with the greatest reoperation proportion was INBONEII-INVISION at 38.1%, followed then by INVISION-INVISION (33.3%) and lastly INVISION-INBONEII (28.6%) (Table 4). Re-revisions occurred at an 11% rate (0%-44.4%), with infection (45.5%) and aseptic loosening (36.4%) representing the greatest proportions (100% reporting). The INVISION-INVISION and INBONEII-INVISION implant permutations accounted for the greatest proportions of these re-revisions, each at 36.4%, followed then by INVISION-INBONEII (27.3%) (Table 4). No discernible subsequent revisions beyond the reported re-revisions were identified. Pooled implant survivorship, based on reported survivorship and re-revision data within each study’s respective follow-up period (FWM 2.8 years ± 1.2 years), was 88% (55.6%-100%; range 100% survivorship reporting).
Details Pertaining to the Review Cohort’s Complications, Reoperations, Re-revisions, and Implant Survivorship. a

Abbreviations: INBII, INBONEII; INV, INVISION.
NR (not reported) was used in the place of a metric that was unspecified within the respective study. UD (undistinguishable) was used where data was not adequately granularized for reporting within the respective study.
Subgroupings and Meta-analyses
Surgical outcomes were sub-grouped by tibial-talar implant permutation variations for further analyses where author-defined granular data was available, sufficient, and appropriate. Beginning with all-cause reoperations, there was no significant difference in risk between a collation of any INVISION/INBONEII permutation versus INVISION-INVISION (RR = 1.0, 95% CI = 0.39-2.59; P = 1.0; Bonferroni-adjusted P = 1.0; 80% reporting). Subgrouping further, no significant differences in risk of all-cause reoperations were observed between either INVISION-INBONEII versus INVISION-INVISION (RR = 1.19, 95% CI = 0.48-2.94; P = .7; adjusted P = 1.0; 80% reporting) or INBONEII-INVISION versus INVISION-INVISION (RR = 0.51, 95% CI = 0.17-1.53; P = .23; adjusted P = .92; 60% reporting). Finally, when examining risk of re-revision, no significant difference was observed between any permutation of INVISION/INBONEII versus INVISION-INVISION (RR = 0.47, 95% CI = 0.12-1.81; P = .27; adjusted P = 1.00; 80% reporting) (Figure 2).

Forest plot comparing re-revision risk between INVISION-INVISION (“INV-INV”) (tibial-talar) rTAA and any permutation (“perm.”) of INVISION/INBONEII (“INV/INBII”) tibial and talar rTAA implants.
Discussion
This systematic review evaluated the INVISION rTAA system to determine survivorship and safety, finding early outcome data that are broadly comparable to other rTAA reports in the contemporary literature, although limited by “very low” GRADE certainty. All-cause reoperations and re-revisions, driven primarily by aseptic loosening and infection, remained prevalent, and meta-analytic comparisons revealed no significant differences attributable to implant permutation.
Nevertheless, the 21% all-cause reoperation rate warrants further discourse. Although high, this rate may not be entirely unprecedented within contemporary TAA literature. For example, in a large review by Vale et al 35 of primary TAAs (INBONE, Salto, Agility, etc.), a 17.4% reoperation rate was noted at an approximate 5.5-year pooled follow-up. Similarly, a 23.5% reoperation rate was reported at a mean 5.8-year follow-up by Locke et al 36 examining primary Hintegra TAA. Ashy et al 37 reported lower reoperation rates (approximately 12% each) for INBONEII primary TAAs and rTAAs within 4-year follow-up, though this mixed primary and revision cohort further limits direct comparability. Importantly, all of these comparator rates were measured at substantially longer follow-up periods (approximately 4-6 years) than the present cohort’s FWM of 2.8 years, and several reflect primary rather than revision TAA populations; direct rate comparisons should therefore be interpreted with caution, as INVISION’s reoperation burden may increase with longer observation. As many of these reoperations arose from comparable underlying causes like implant subsidence and wound-related issues, they help to bracket and contextualize our INVISION observations. 35 Persisting reoperation rates, even with modular implant designs, likely reflect multifactorial challenges relating to the technical complexity of rTAAs, especially in settings of compromised bone stock for which INVISION is indicated.14,38 Furthermore, case-specific anatomic factors and implant-specific learning curves may have also been contributory.38,39 Nonetheless, considerations such as longer follow-up, direct comparisons, and broader experience with INVISION are needed for more translational and confirmative analyses.
Likewise, the 11% pooled re-revision rate was also literature-comparable. In the 999-patient rTAA review by Sun et al, 40 a 9.9% pooled re-revision rate was observed within 5-year follow-up. Furthermore, Jennison et al 41 reviewed rTAAs (INBONEI, Salto, etc.) and determined a 14.4% pooled re-revision rate, indicating that the present study’s 11% re-revision rate at 2.8 years falls within a range seen in comparable rTAA literature, although again the shorter follow-up period limits direct comparisons. The present study’s pooled implant survivorship of 88% at a FWM follow-up of 2.8 ± 1.2 years also showed contextualizable promise as a relatively early measure of durability. Ashy et al 37 reported an implant survivorship of 93.9% at a mean 37.3-month follow-up. Similarly, Hintermann et al 42 examined 116 rTAAs and reported survivorships of 81%, 74%, and 70% at 5, 10, and 15 years, respectively. Furthermore, summated survivorships within 6 years have been reported to range from 70% to 98% in generalized TAA literature. 43 Within such context, the re-revision and survivorship outcomes observed for INVISION appear broadly consistent with early published rTAA data, although firm conclusions await longer-term, controlled study.
In examining the factors driving reoperations and re-revisions, aseptic loosening and infection constituted the two most common. Such findings have literature-backed precedent, both as frequent causes and risk factors for initial and secondary rTAAs.40,42,43 For example, in Locke et al, 36 aseptic loosening occurred in 14% of patients and constituted 100% of revisions, whereas a separate 7.8% of patients developed infections. Furthermore, aseptic loosening and infection were significantly associated with TAA revisions and reoperations in a multi-implant study by Sanchez-Correa et al. 44 Although not directly assessed in comparative fashion within the present review, INVISION does not appear to narratively confer added protection against these complications, highlighting the broader need for more investigation.
The meta-analyses demonstrated no statistically significant differences in reoperation or re-revision risk between implant permutations, a finding that is hypothesis-generating but should be interpreted cautiously given the small study sample sizes and “very low” GRADE certainty of the underlying evidence. This study builds upon prior literature supporting INVISION as a unique option among revision-specific interchangeable TAA systems, particularly in its capacity to address instability and provide augmentation in settings of substantial bone resection. 43 Although literature specifically examining INVISION’s modularity in this manner is limited, Sun et al 40 found no significant differences in re-revision rates attributable to the INVISION tibial or talar components individually. This example is consistent with the present study’s findings; however, both analyses are limited by small sample sizes and low statistical power, such that the absence of a significant difference should not be interpreted as equivalence for safety assurance. Such a finding may be especially meaningful in scenarios where retained prostheses or limited implant availability pose constraints to decision making.
This review’s findings and analyses warrant caution due to inherent limitations. First, the collated studies were largely retrospective and of moderate MINORS quality. This, coupled with the outcome certainty grading of “very low” and high statistical heterogeneity, emphasizes potential risks of biases that ultimately limit the strengths of conclusions made. Conversely, the narrow study base of relatively newer publications underscores INVISON’s novelty, underrepresentation, and growing interest compared with other implant systems. For such reasons, this review was restricted by smaller sample sizes and specific statistical inferences, meriting nuanced, power-related alertness when contextualizing findings. Additionally, marked heterogeneity in data reporting was observed for certain measures; this was particularly apparent when examining patient comorbidities, PROMs, and author-reported significance assessments. Only one study detailed a postoperative rehabilitation protocol, whereas only one other study reported radiographic outcomes such as alignments and/or deformities. Such investigative variables are commonplace in TAA literature; however, the current INVISION-focused evidence base provides insufficient exploration of these measures for meaningful synthesis beyond what was performed.37,45 Furthermore, there was also widespread ambiguity surrounding tibial/talar implant selection (INVISION vs INBONEII) for rTAA, which likely varied by surgeon judgment and case-specific factors in included studies, and thus may have influenced outcomes. Finally, a pooled complication rate could not be calculated, primarily because of heterogeneity in author-defined complication criteria. Variance in how complications are classified, such as differentiating minor versus major or intraoperative versus postoperative, has been a challenge noted in prior orthopaedic literature.30,46 Although this limitation was transparently addressed, it must remain a point of vigilance during appraisal. Even so, to the authors’ best knowledge, this study is the first, most extensive, and largest analysis of clinical outcomes following INVISION-based rTAA. Future high-quality studies with longer follow-up are needed to further corroborate the aforementioned findings and directly evaluate INVISION against other implant systems, for example, with concerted efforts to address the above-described limitations: one such method being greater standardization and homogeneity among outcome measures like PROMs.
Conclusion
This study examined early clinical outcomes following rTAA with the INVISION modular implant system, finding notable rates of reoperation and re-revision driven predominantly by aseptic loosening and infection, highlighting a continued need for focused mitigation strategies. Meta-analysis detected no significant differences in reoperation or re-revision risk across the available INVISION tibial-talar implant permutations, though this should be interpreted cautiously given small sample sizes and limited statistical power. Overall, conclusions are constrained by retrospective study designs, “very low” GRADE outcome certainty, marked heterogeneity, and short follow-up duration; future high-quality longitudinal and comparative studies are required before firm conclusions about INVISION’s efficacy can be made.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114261435959 – Supplemental material for Revision Total Ankle Arthroplasty Using the INVISION Modular Implant System: A Systematic Review and Meta-Analysis
Supplemental material, sj-pdf-1-fao-10.1177_24730114261435959 for Revision Total Ankle Arthroplasty Using the INVISION Modular Implant System: A Systematic Review and Meta-Analysis by Shahabeddin Yazdanpanah, Braeden R. Gooch, Sashrik Sribhashyam, John W. Cyrus, Benjamin P. Cassidy, Samuel B. Adams, Albert T. Anastasio and Conor N. O’Neill in Foot & Ankle Orthopaedics
Footnotes
Appendix
Study, Demographic, and Cohort Details Table for Included Studies.

| Author (Year) | Inclusion Criteria | Exclusion Criteria | Patients (Number) | Modular Implant Permutations, Organized as Tibial-Talar Component | Average Age or Median (SD/Range/IQR) in Years | Average Follow-up (SD/Range) in Years or Description | Male Number (%) | BMI (SD/Range) | Patient Comorbidities |
|---|---|---|---|---|---|---|---|---|---|
| Conti (2022) 27 | Two-stage rTAA with removal of all implants and placement of antibiotic spacer in patients with chronic periprosthetic joint infections of greater than 4 weeks following primary TAA | Less than 2 years of postoperative clinical score reporting | 9 | INV-INV: 4 INV-INBII: 4 INBII-INV: 1 |
58.9 (11.0) | Minimum 2 years | NR | NR | NR |
| Martin (2025) 28 | All patients who received ankle arthroplasty using INVISION | NR | 17 | INV-INV: 5 INV-INBII: 3 INBII-INV: 9 |
67.9 (56-80) | 40.5 months (7-78) | 14 (82.4) | NR | NR |
| Purnell (2024) 29 | Adult patients who underwent rTAA with INVISION from 2018-2021 as well as a minimum of 2 years of follow-up | NR | 19 | INV-INV: 5 INV-INBII: 5 INBII-INV: 9 |
62 (40-82) | 3.5 (2-5.9) | 10 (52.6) | 30 (23-36) | NR |
| Ray (2025) 30 | All patients who underwent rTAA with INVISION | Less than 2 years of postoperative follow-up and unwilling to return to clinic | 27 | INV-INV: 11 INV-INBII: 7 INBII-INV: 9 |
61.4 (8.1) | 3.6 (NR) | 17 (63) | NR | Hypertension: 16 Hyperlipidemia: 15 Type 2 Diabetes: 6 Coronary Artery Disease: 6 Mental Health Disorder: 6 Rheumatoid Arthritis: 4 Anemia: 3 Hypothyroidism: 3 Fibromyalgia: 3 Gastroesophageal Reflux Disease: 2 Chronic Obstructive Pulmonary Disease: 2 Tobacco Use History: 2 Congestive Heart Failure: 2 Obstructive Sleep Apnea: 1 Chronic Kidney Disease: 1 Peripheral Artery Disease: 1 Atrial Fibrillation: 1 Cervical Myelopathy: 1 Venous Thromboembolism History: 2 Cancer History: 1 Irritable Bowel Syndrome: 1 Asthma: 1 |
| Valan (2024) 14 | All patients who underwent rTAA with INVISION | NR | 28 | INBII-INV: 28 | 67.0 (7.2) | 1.3 (0.9) | 10 (35.7) | 30.8 (6.6) | Tobacco Use History: 13 Rheumatoid Arthritis: 4 Mental Health Disorder: 17 |
Abbreviations: INBII, INBONEII; INV, INVISION; rTAA, revision total ankle arthroplasty; TAA, total ankle arthroplasty.
NR (not reported) was used in the place of a metric that was unspecified within the respective study.
ORCID iDs
Ethical Considerations
Not applicable.
Consent for Publication
Informed consent was not required to gather the data to publish this paper.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Disclosure forms for all authors are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
