Abstract
Background:
TAA has gained wide acceptance and is a commonly performed surgery. In spite of this rise in popularity, both deep and superficial postoperative infections remain a significant issue following procedures performed through an anterior approach. Our study sought to identify whether a relationship exists between the thickness of the anterior ankle soft tissues and infection rates after TAA.
Methods:
We performed a retrospective cohort study of 293 patients who underwent primary TAA from 2017 to 2023 at a single institution. All patients had a preoperative computed tomography (CT) scan that was used for patient-specific instrumentation (PSI). All measurements of the anterior soft tissue envelope were obtained from CT scans. All procedures were done through an anterior approach. Patient demographics including body mass index (BMI) and diabetic status were recorded. A logistic regression analysis was performed to identify significant risk factors for postoperative infection after TAA.
Results:
Increasing anterior ankle soft tissue thickness was found to be significantly associated with postoperative infection after TAA (OR 1.31, 95% CI 1.17, 1.46, P < .001). This was also true of diabetic status (OR 4.9, 95% CI 1.70, 14.4, P = .003). When stratifying for deep and superficial infections, the relationship of ankle soft tissue thickness and infection remained significant for superficial infection (OR 1.26, 95% CI 1.13, 1.40, P = .013), but not for deep infection (P = .056). The mean age of our cohort was 68.2 years at the time of surgery (SD 8.6 years). Median follow-up was 16 months. Half (50.2%) of the patients were female; 9.6% were diabetic. Mean BMI was 30.3 (SD 5.3). Average anterior ankle soft tissue thickness was 16.5 mm (SD 4.0 mm).
Conclusion:
Our study demonstrates that a thicker anterior ankle soft tissue envelope was found to be significantly associated with superficial infection risk after TAA. This risk was independent of patient BMI, nor did BMI correlate with ankle soft tissue thickness (R2 = 0.03). Our study may help inform perioperative care and counseling after this increasingly common procedure.
Level of Evidence:
Level III, retrospective cohort study.
This is a visual representation of the abstract.
Introduction
Total ankle arthroplasty (TAA) has become an increasingly used treatment for end-stage ankle arthritis, offering pain relief and restoration of joint motion in appropriately selected patients.1,2 Despite improvements in implant design, surgical technique, and perioperative protocols, complication rates after TAA remain higher than those reported for total hip or knee arthroplasty.3-5 Among these, wound complications and periprosthetic infection remain particularly concerning because of the limited soft tissue coverage of the ankle and its relatively tenuous vascular supply.5-7
The quality and integrity of the soft tissue envelope have long been recognized as critical determinants of postoperative outcomes following ankle surgery.8,9 Prior studies have shown that compromised soft tissue conditions, such as scarring, edema, or thin coverage available for usage during closure, may increase the risk of wound healing problems and infection after TAA.10-12 However, objective quantification of local soft tissue characteristics, particularly anterior soft tissue thickness, has been limited. Although systemic factors such as diabetes mellitus and obesity are well-established risk factors for infection following arthroplasty,13-15 it remains unclear whether local anatomic factors, such as anterior ankle soft tissue thickness, independently contribute to postoperative infection risk.
The purpose of this study was to evaluate whether anterior ankle soft tissue thickness, measured on preoperative computed tomography (CT) scans, was associated with postoperative infection following TAA performed through an anterior approach. We hypothesized that increased anterior soft tissue thickness would be an independent risk factor for infection after TAA, even after adjusting for patient comorbidities such as diabetes and body mass index (BMI).
Methods
Data Collection
We constructed a patient cohort using radiographic studies and demographic information from patients who underwent total ankle arthroplasty at our institution from 2017 to 2023. Patients required at least six months of follow-up. Inclusion criteria included age greater than 18 years. Patients were treated by a single surgeon (P.F.). All obtained preoperative CT scans in order to design their patient-specific instrumentation (ie, personalized tibial and talar cutting guides) from which anterior ankle soft tissue thickness was measured using the technique shown in Figure 1. In addition to radiographic measurements, we collected routine demographic information on our patients including age, sex, height, weight, BMI, diabetic status. Revision total ankle arthroplasty cases were excluded, as were conversions from patients previously treated with ankle arthrodesis. Patients treated with concurrent supramalleolar osteotomies were also excluded. Although 15 patients were excluded for insufficient follow-up, their baseline demographics were not significantly different from the final cohort, suggesting minimal selection bias. In terms of surgical history, we recorded whether patients had a previous operatively treated ankle fracture (AO 43 and AO 44B/C injuries treated with internal fixation). This was chosen because of the close association of ankle arthritis to trauma, and the fact that operatively treated ankle fractures could be determined not only retrospectively through clinic intake notes, but also through office radiographs and CT, making it a reliable indicator of previous surgeries in a way that ligamentous repair or nonoperatively treated ankle fracture patterns would not be.

Computed tomography–based method of estimating ankle soft tissue thickness. Scan is brought to the widest point of the tibial plafond. A transmalleolar line is drawn. An orthogonal line is created at the point of maximal ankle soft tissue thickness, and it is measured.
Our end point of interest was postoperative infection after total ankle arthroplasty. We used both a composite end point that included both superficial and deep infection, as well as divided our analysis to look specifically at superficial infection and deep. Superficial infection was determined retroactively from patient clinic notes. It was determined based on clinical description of cellulitic change treated with antibiotics or superficial wound dehiscence treated with local wound care (usually wet to dry dressing changes) and antibiotics. Deep infection was defined either through positive aspiration culture or synovial white blood cell count >3000 cells/μL), or draining sinus tract. We included both superficial and deep infections to increase ability to detect trends in what is a relatively rare end point.
Data Analysis
Demographic information was reported with percentages for binomial variables, and medians with IQRs for continuous variables when appropriate.
Statistical significance for demographic differences was calculated using an alpha value of .05. Cohort demographic differences were determined using t test for quantitative variables and χ2 tests for qualitative variables, respectively. Shapiro-Wilk testing was used to assess for normal distribution of data before using t tests for quantitative variables. For our logistic regression, our plan was to run our regression for diabetic status, anterior soft tissue thickness, and patient BMI. BMI was included to determine whether anterior soft tissue thickness was an independent risk factor for postoperative infection, or whether it was operating as a surrogate for general patient body habitus. Bonferroni correction and a modified alpha value of 0.017 was used for our regression to account for 3 independent variables. All data analysis was performed using Python, version 3.13.7.
Procedure Details
All procedures were performed through an anterior approach to the ankle joint. Meticulous care was taken not to violate the tibialis anterior tendon sheath. The attending surgeon (P.F.) is a fellowship-trained Foot and Ankle surgeon with more than 30 years’ experience and performs approximately 100 to 115 total ankle replacements per year. All patients were treated with the Infinity Total Ankle System implant (Stryker).
At the end of the procedure, a 0.05% chlorhexidine solution is used for final lavage, followed by normal saline solution. The retinaculum is closed with 2-0 Vicryl suture. The subcutaneous layer is closed with 3-0 undyed Vicryl. The skin is closed with 4-0 nylon.
Ethical Considerations
This study was approved by our hospital’s institutional review board (IRB no. 2022-148). No informed consent was required for this project.
Results
A total of 308 patients met inclusion criteria for this study, of which 293 patients had the required duration of follow-up. Mean age was 68.2 years of age (SD = 8.6 years). Half (50.2%) of our cohort was female. The mean BMI of our patients was 30.3. In addition, 9.6% of our cohort was diabetic. Recent hemoglobin A1c (within 3 months of total ankle replacement) was not available for the vast majority of patients, and was not reported nor included in analysis. The mean preoperative anterior ankle soft tissue thickness in this cohort was 16.5 mm (SD 4.0 mm). This value was not significantly different between male and female patients and was independent of patient weight (mean 88.6 kg, SD 21.1) or BMI (mean 30.3, SD 5.3). Median patient follow-up was 16 months. Full demographic and outcome information for this cohort is contained in Table 1.
Patient Cohort Demographics (N = 293).
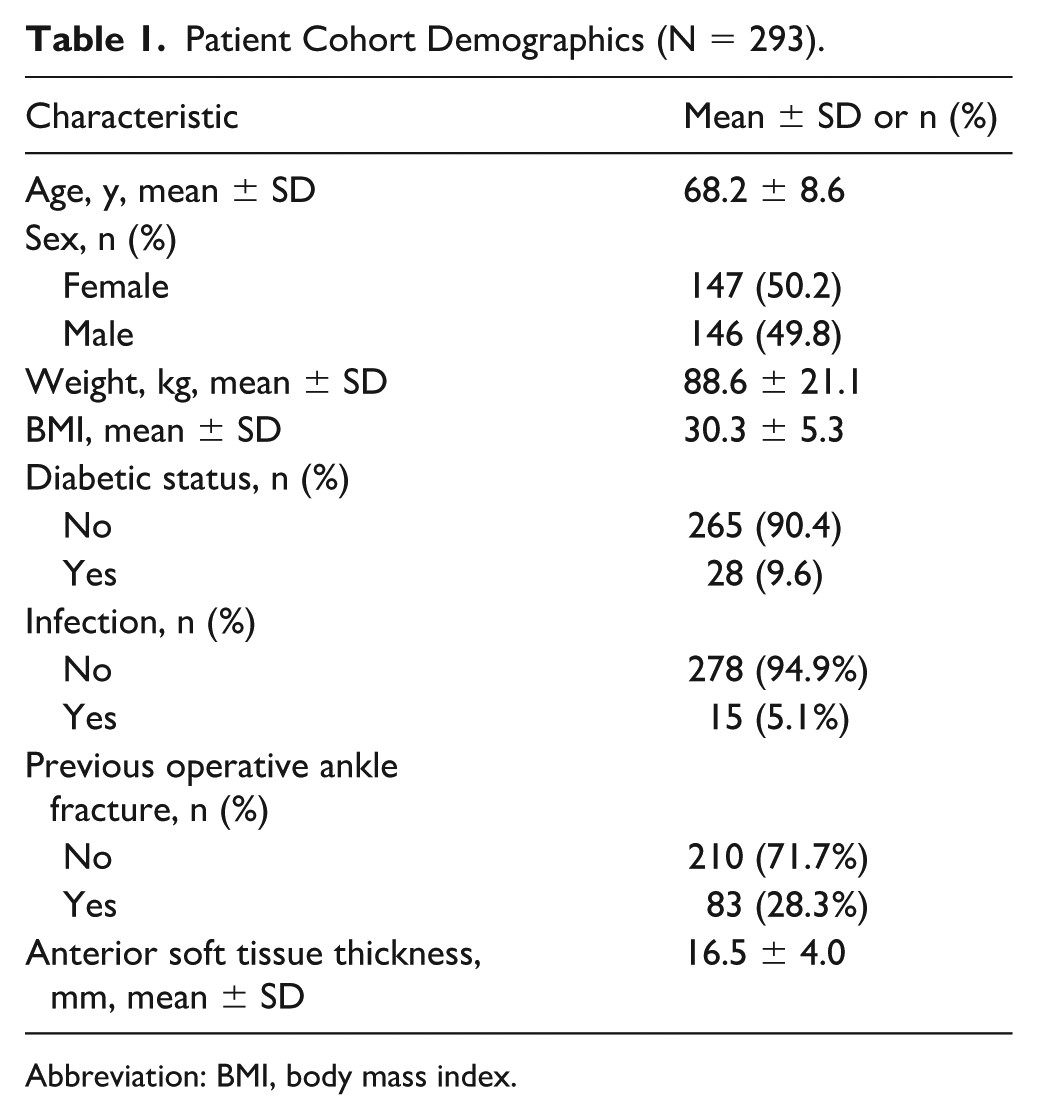
Abbreviation: BMI, body mass index.
In terms of infection risk, 5.1% of patients who underwent total ankle arthroplasty had an identified infectious or wound healing complication, totaling 15 patients. Of these, 13 experienced a superficial wound infection or dehiscence. These patients were treated with oral antibiotics and in the case of wound dehiscence, wet to dry dressing changes. All went on to uneventful healing and resolution of infection. There were 2 patients who had a deep infection (0.68% of the total cohort). Both patients with deep infections were late infections (>6 weeks) that were treated in a 2-stage fashion with explant and spacer placement with later conversion. More granular information on these 2 deep infections is included in Appendix A. In our logistic regression, increasing anterior ankle soft tissue thickness was found to be significantly associated with postoperative infection after TAA (OR 1.31, 95% CI 1.17, 1.46, P < .001). This was also true of diabetic status (OR 4.9, 95% CI 1.70, 14.4, P = .003). When stratifying for deep and superficial infections, the relationship of ankle soft tissue thickness and infection remained significant for superficial infection (OR 1.26, 95% CI 1.13, 1.40, P < .001) but not for deep infection (OR 2.1, 95% CI 0.98, 82.3, P = .056). This relationship was independent of patient BMI. Full results of the logistic regression are included in Table 2.
Multivariable Logistic Regression for Patient Infection Risk.

Abbreviations: BMI, body mass index; DM, diabetes mellitus.
Combined refers to combined end point of infection including both superficial and deep cases.
When comparing the groups without and with infections, anterior ankle soft tissue thickness was found to be greater in the group with infections (mean of 20.4 mm vs 16.1 mm, P < .001). This is also true when narrowing the analysis to superficial wound infections only (mean of 20.1 mm for patients with superficial infections vs 16.2 mm for those without superficial infections, P = .01), consistent with the fact that most infections were superficial. Increased ankle soft tissue thickness was also associated with having a previous ankle fracture open reduction and internal fixation (16.2 mm for those without previous operative ankle fractures vs 17.3 mm for those with previous operative ankle fractures, P = .03). Figure 2 shows the direct correlation between anterior ankle soft tissue thickness and patient BMI was also calculated. The R2 value for this relationship was 0.03 (P = .003), indicating a negligible relationship.

Relationship between BMI and Anterior Ankle Soft Tissue Thickness.
Discussion
In this study of 293 patients that underwent primary total ankle arthroplasty, we found that increased anterior ankle soft tissue thickness was significantly and independently associated with postoperative superficial infection. This relationship persisted even after adjusting for established systemic risk factors such as diabetes mellitus and BMI. These findings suggest that the local soft tissue envelope surrounding the ankle plays a clinically relevant role in postoperative wound healing and infection risk.
We found that increased soft tissue thickness correlated with superficial infection. This may appear counterintuitive, as thin soft tissue envelopes are classically considered higher risk. However, thicker soft tissue likely reflects greater subcutaneous adiposity, which has been associated with poor perfusion, delayed wound healing, and increased bacterial burden in other anatomic regions.16,17 Moreover, excessive soft tissue bulk may increase wound tension during closure or create dead space that fosters infection. These mechanisms align with prior studies linking subcutaneous fat thickness to surgical site infections after total knee and hip arthroplasty.18-20
Our study came to similar conclusions as the work by Wu et al. 21 Their group found an association between increased preoperative ankle soft tissue thickness and the need for eventual revision surgery. They also found that this risk was independent of patient BMI, implying that it is specifically the ankle soft tissue envelope that serves as a risk factor for complication. Our work differs from theirs in a number of ways. Our end point of combined infection risk was distinct from theirs, which only included all-cause revision. They also utilized preoperative radiographs, rather than CT scans. Because radiographic assessment of the ankle is highly dependent on foot and ankle positioning, our study sought to use preoperative CT scans as a more reliable marker of anterior ankle thickness. 22 Their work also looked at both soft tissue thickness at the level of the distal tibia as well as the talus, whereas our standardized system specifically homed in tibial soft tissue thickness at the level of the plafond.
Our work also parallels the study by Vahedi et al 23 that looked at anterior knee soft tissue thickness and its association with infection and total knee arthroplasty (TKA). They found that greater anterior knee soft tissue thickness was an independent risk factor for prosthetic joint infection. They found a similar association with medial knee soft tissue thickness. This association was maintained even when accounting for patient BMI. They posited that distribution of body fat may be as important or more than total body fat when it comes to infection. This conclusion is in line with our findings, in which there was an association with soft tissue infection for ankle soft tissue thickness, but not for BMI. This may be because the distribution of soft tissue, that is, about the ankle carries a higher impact on infection risk that global fat distribution. It also raises questions on whether anterior ankle soft tissue thickness is a modifiable risk factor, as it appears independent of patient habitus. Therefore, although this study may help inform how surgeons counsel their patients on risk of TAA, it may not be something that can easily translate into improved patient outcomes.
Our work has limitations. One is the exclusion of patients who had revision ankle arthroplasty or conversion from arthrodesis to arthroplasty. Our goal was to select for patients without postoperative anterior scar in order to better evaluate the effect of ankle thickness on infection risk. However, that choice does limit generalizability. We also only looked at infection as an end point. It is possible and even plausible that increasing ankle soft tissue has effects beyond infection, for instance, in aseptic component loosening or osteolysis. However, because wound complications and infection are common, we wanted to focus on that specific factor and its relationship to the ankle soft tissue environment and improve our ability to discern a meaningful difference in patient outcomes. We also had insufficient information regarding preoperative (within 3 months) hemoglobin A1c values for our diabetic patients. Therefore, although we were able to identify diabetes as a factor associated with infection risk, we are unable to comment on how the control or lack thereof of a patient’s diabetes influences that risk. Another weakness is that we used a fairly basic risk factor, soft tissue thickness, that encompasses soft tissue in many forms. Fat, scar, tendon, etc are all contained within that risk factor and are not differentiated. Unfortunately, the capacity of CT scans to differentiate between these different soft tissue subtypes is limited. MRI is the gold standard for this type of granular information on soft tissue subtypes. A more complete study would integrate both preoperative CT as well as MRI in determining risk factors for infection after TAA. Our study was a pragmatic foray into the idea of soft tissue thickness as a risk factor, driven by the fact that CT was universally available for our cohort of patients given our use of patient-specific instrumentation. A preoperative MRI for patients undergoing TAA is rare, and most likely would require a prospective cohort whose design would be built on more basic studies such as ours. Finally, we only had 2 events of deep infection in our cohort. It is likely that the effect of soft tissue thickness is significant beyond superficial infection, but because deep infection is a rare end point, our current cohort is unable to sufficiently investigate this relationship.
Conclusion
The ankle soft tissue environment is emerging as an important consideration for patient outcomes after total ankle arthroplasty. Our study demonstrates a significant association between increasing ankle soft tissue thickness and superficial infection risk following anterior approach primary TAA. Future studies will be required to fully understand the relationship between these factors and whether this information can be used to optimize patient outcomes after this increasingly common procedure.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114261420160 – Supplemental material for Association of Preoperative Anterior Ankle Soft Tissue Thickness and Infection After Primary Total Ankle Arthroplasty: A Retrospective Cohort Study
Supplemental material, sj-pdf-1-fao-10.1177_24730114261420160 for Association of Preoperative Anterior Ankle Soft Tissue Thickness and Infection After Primary Total Ankle Arthroplasty: A Retrospective Cohort Study by Vivek Nair, Dylan Moran, Matthew Cederman, Robert Dean, Paul Fortin and Zein El-Zein in Foot & Ankle Orthopaedics
Footnotes
Appendix
Summary Information on Patients With Deep Infections.

| Patient | Age | Sex | Height, cm | Weight, kg | BMI | DM | Soft Tissue Thickness, mm | Past Operative Ankle Fracture | Causative Organism |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 69 | M | 191.1 | 118 | 32.3 | No | 27.9 | Yes | Staphylococcus aureus (MSSA) |
| 2 | 70 | F | 157.5 | 76 | 27.9 | Yes | 20.7 | Yes | S aureus (MSSA) |
Abbreviation: DM, diabetes mellitus; MSSA, methicillin-sensitive Staphylococcus aureus.
Ethical Considerations
Ethical approval for this study was obtained through institutional review board approval at Oakland University William Beaumont hospital (IRB 2022-148).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Paul Fortin, MD, reports disclosures relevant to manuscript of Paragon 28 consultant and royalties. Disclosure forms for all authors are available online.
Data Availability Statement
Data for this project is derived from non-publicly available patient health information compiled in a secure database for research purposes. De-identified cohort data available on request from corresponding author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
