Abstract
Research Type:
Level 4 – Case series
Introduction/Purpose:
Total talus replacement (TTR) was originally developed in Asia using stainless steel or ceramic material for the treatment of talar avascular necrosis (AVN). Over the past 15 years, advancements in materials, technology, and 3D printing have allowed for its combination with a distal tibial prosthesis i.e., total ankle-total talus replacement (TATTR). The indications for TTR have expanded beyond talar AVN to include various conditions that lead to talar bone loss, with or without ankle arthritis. However, there is a paucity of data on the clinical outcomes of isolated TTR and TATTR, particularly in North America. This study evaluates the clinical and functional outcomes of both procedures performed in one North American center.
Methods:
In this IRB-approved retrospective study, we identified and reviewed the records of all patients who underwent TTR, or TATTR, with at least 6 months of follow-up in one North American medical center.
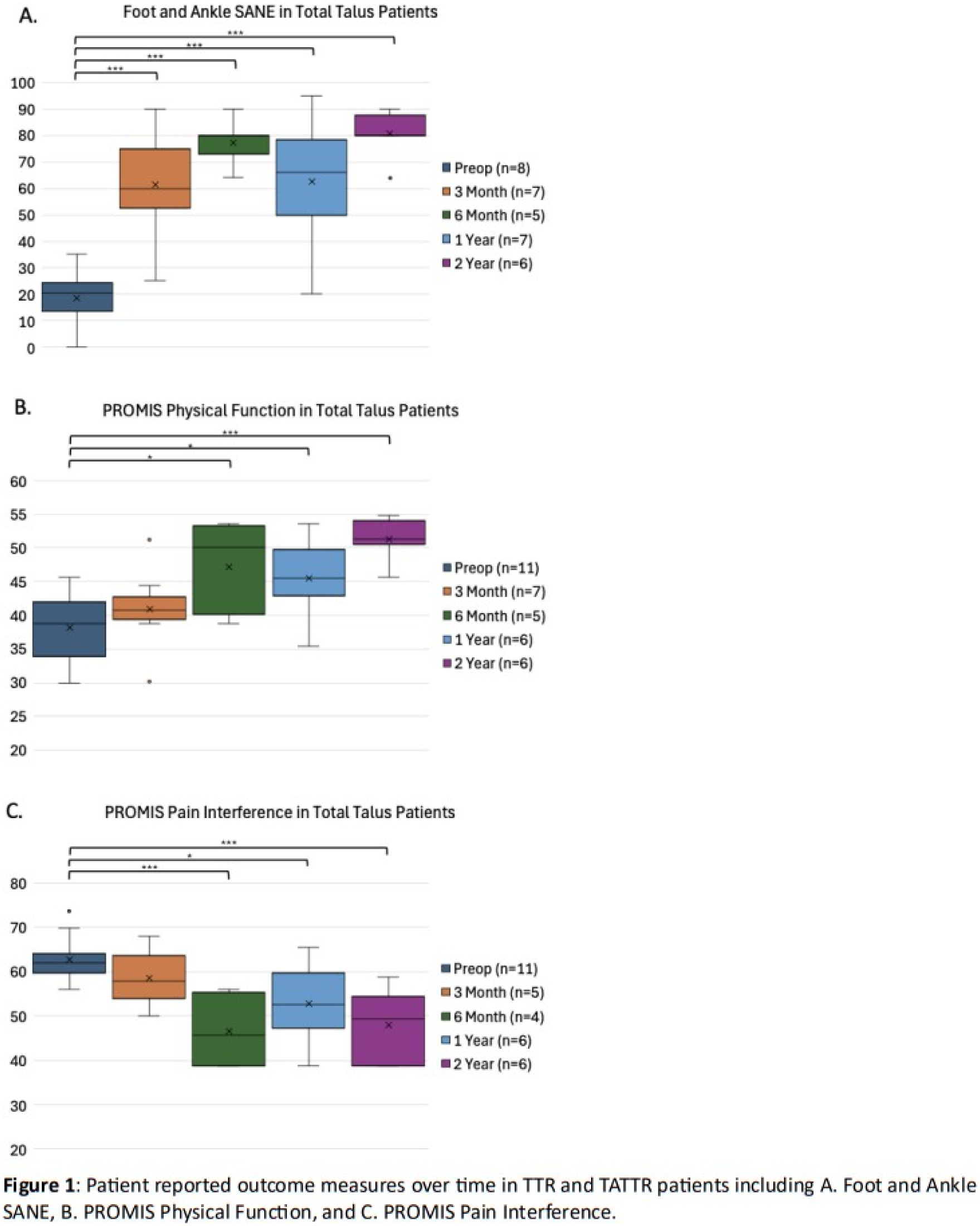
Demographics, associated surgeries, and peri-operative details, including complications, were recorded. Patient-reported outcome measures, including Foot and Ankle Single Assessment Numeric Evaluation (SANE), PROMIS physical function, and PROMIS pain interference, were collected preoperatively and at postoperative follow-up visits (3, 6, 12, and 24 months). Statistical analyses were conducted using SAS v9.04.
Results:
A total of 15 patients (mean age: 48.8 years, range: 27–86 years) were included. The etiologies for talar bone loss included AVN (primary or posttraumatic) in 10 patients, revision of failed TTR in 1 patient, and revision of a failed total ankle replacement in 4 patients. No intra-operative complications or implant failures were observed. Post-operative complications occurred in 5 of 15 patients, including one minor wound infection, one minor nerve injury, one case of worsening peroneal tendon pain requiring subsequent surgery, and two cases with decreased range of motion necessitating additional procedures. The mean clinical follow-up time was 23.3 +/- 15.3 months (range: 6.6-58.4 months). All patient-reported outcome measures showed statistically significant improvement by 6 months postoperatively (Figure 1).
Conclusion:
This study demonstrates good clinical and patient-reported outcomes following both isolated TTR and TATTR for patients with significant talus destruction, with no early failures. To our knowledge, this is the first report from a North American Center detailing outcomes of both isolated TTR and TATTR using titanium and cobalt chromium prostheses. Further follow-up is necessary to assess implant longevity and long-term functional improvements following these two procedures. Additionally, we are actively investigating the functional and activity-related benefits of these procedures compared to pantalar fusion.