Abstract
Research Type:
Level 3 - Retrospective cohort study, Case-control study, Meta-analysis of Level 3 studies
Introduction/Purpose:
Subtle Lisfranc joint injuries represents a diagnostic challenge. While weightbearing radiographs remain widely used, weightbearing CT (WBCT) is emerging as a promising alternative. Recent WBCT Lisfranc studies rely on manual measurements, cadaveric specimens, or contralateral foot comparisons using non-normalized metrics such as joint space distance. However, these methods fail to account for patient size and varying injury complexity. A shift toward normalized 3D metrics within the injured foot itself, such as joint coverage percentage, eliminates size-dependent variability by standardizing measurements across patients. Therefore, this study aimed to develop a normalized 3D measurement capable of detecting Lisfranc joint injuries, integrating machine learning to assess its robustness. We hypothesized that injury could be detected with an area under the curve (AUC) greater than 0.90.
Methods:
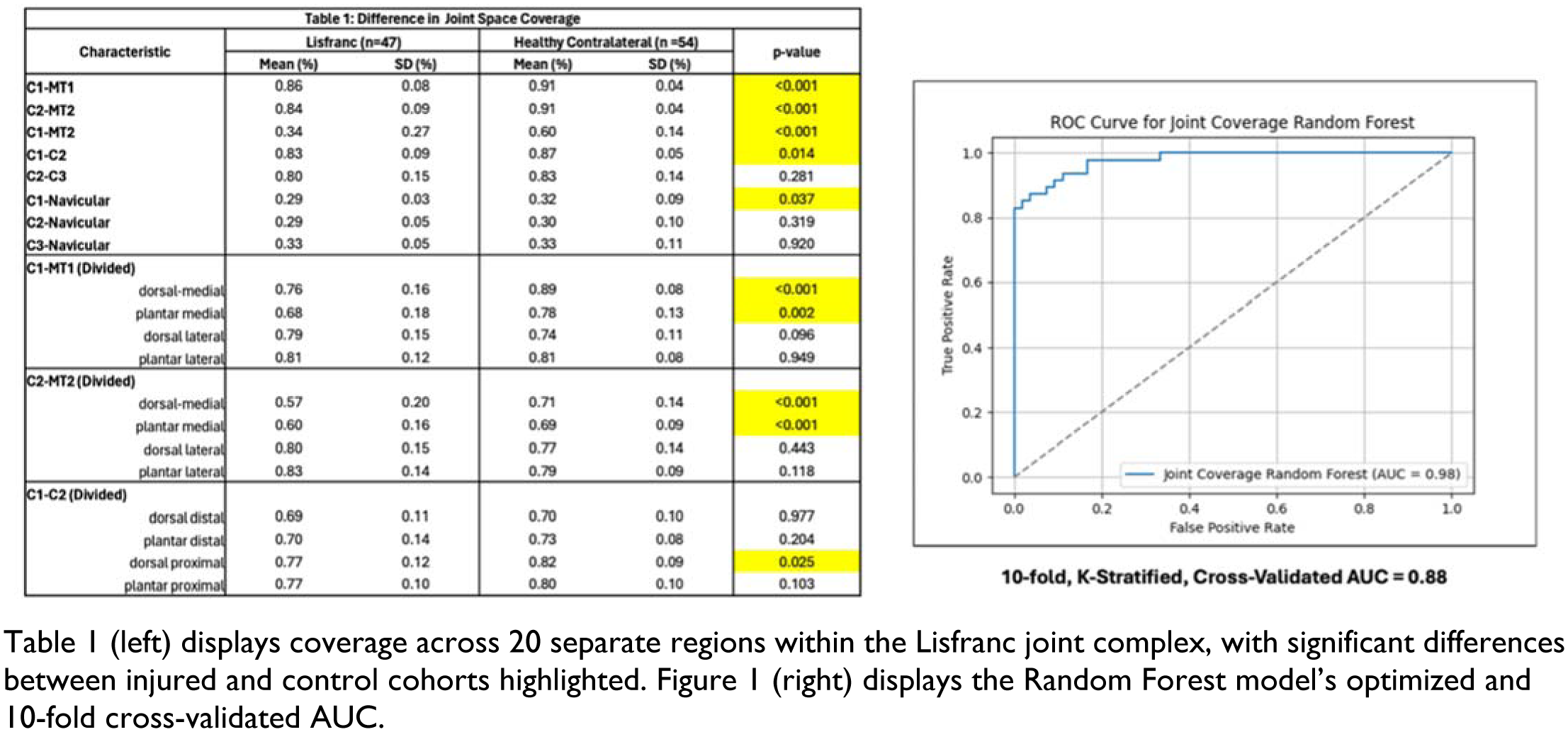
This retrospective study included 47 patients with Lisfranc injuries who underwent WBCT for further stability assessment. Of these, 27 had bilateral scans (27 injured, 27 contralateral feet), and 20 had unilateral scans (20 injured feet). An additional 27 matched controls provided 27 healthy feet, totaling 54 control feet. To perform distance mapping, a foot and ankle surgeon manually selected opposing surfaces of eight Lisfranc complex regions, including seven articulating (M1-C1, M2-C2, C1-C2, C2-C3, and each naviculocuneiform joint) and one non-articulating (C1-M2). The M1-C1, M2-C2, and C1-C2 regions were divided into four subregions. Coverage was assessed in MATLAB by calculating the percentage of distances < 4mm, as reduced coverage indicates subluxation. A random forest model was trained on all coverage metrics to optimize injury detection, using percent impact on decision, decision tree count, depth, and leaf size. Model performance was evaluated via AUC ROC analysis with 10-fold cross-validation to estimate diagnostic accuracy.
Results:
A significant decrease in coverage was evident for 5/8 primary regions (C1-M1, C2-M2, C1-M2, and C1-C2, C1-Nav.) and certain medial aspects of the divided C1-M1 and C2-M2 subregions (Table 1). The optimized random forest solely analyzed the C1-M1, C2-M2, C1-M2, C1-M1 (dorsal medial), and C2-M2 (dorsal and plantar medial), as these were the only metrics over the percent impact on decision of 5%. Upon testing across numerous percent thresholds, 5% removed joints that were correlated or redundant in determining injury. The optimized random forest parameters included 700 decision trees with a depth of 6 questions, 2 samples/split, and 4 samples/leaf. The optimized random forest model’s AUC equaled 0.98 (Figure 1). Finally, the cross-validated AUC equaled 0.88, demonstrating little overfitting from our relatively small sample size.
Conclusion:
In conclusion, this diagnostic algorithm demonstrated promising results, representing the first study to utilize normalized metrics to assess possible Lisfranc injury. While there is utility in assessment of joint space width, normalized metrics such as coverage allow for injury assessment without the need for contralateral comparison. A strength of this study was the optimization of our model, as we allowed the model to learn from our dataset without providing any assumptions, such as expected decreases in coverage in certain regions. Future testing is needed to determine the validity of this measurement and diagnostic algorithm on an entirely unsupervised dataset.