Abstract
Background:
Plantar fasciitis is the most common cause of plantar heel pain and the use of custom foot orthosis is a well-supported treatment modality. Traditional custom orthoses are fabricated using foam impressions and manual techniques, but these methods are time-intensive and susceptible to variability in fit and quality. The HP Arize platform applies high-resolution 3D laser scanning with algorithm-driven modeling to streamline fabrication of custom foot orthoses. Although early reports suggest comparable comfort and durability to traditional devices, clinical data evaluating patient-reported outcomes remain limited.
Methods:
This prospective pilot, randomized study assigned 34 adults diagnosed with plantar fasciitis to receive either traditionally fabricated custom orthoses (n = 18) or HP Arize 3D-printed orthoses (n = 16). Primary analyses used a modified intention‑to‑treat cohort (n = 30) comprising participants who received an insert and completed baseline assessments. Patient-Reported Outcomes Measurement Information System (PROMIS) physical function, pain interference, and mobility scores were collected at baseline, 4 weeks, and 8 weeks. Secondary measures included patient satisfaction, frequency of orthotic use, and adverse effects. Statistical comparisons were performed using Fisher exact test and Student t test or Wilcoxon signed-rank test, with significance set at P < .05.
Results:
Baseline demographic and clinical characteristics did not differ between groups. At 4 weeks, satisfaction with customization, fit, and comfort was high across both groups (median 4.0-5.0). Traditional orthosis users reported greater daily use (P = .04) but also higher rates of rubbing/discomfort (P = .047). By 8 weeks, side effects were not statistically significantly different. Exploratory comparisons suggested higher mean PROMIS physical function (49.0 vs 41.9, Δ 7.1; P = .008) and mobility scores (46.9 vs 41.4, Δ 5.5; P = .02) at 8 weeks in the traditional group, whereas pain interference scores did not differ.
Conclusion:
Both traditionally fabricated and HP Arize 3D-printed custom orthoses were associated with high satisfaction and functional improvement. Although traditional devices showed greater early compliance and superior 8-week functional outcomes, the HP Arize system demonstrated fewer adverse effects and offers a scalable, efficient alternative. As an exploratory pilot study, these findings are preliminary and intended to inform feasibility and effect‑size estimation for future trials.
Level of Evidence:
Level IV, pilot prospective comparative study
Type of Study:
Quasi-experimental pre-post study.

Introduction
Plantar fasciitis is the leading cause of plantar heel pain in adults and one of the most common foot and ankle conditions encountered in orthopaedic outpatient clinics. It accounts for a significant proportion of visits to foot and ankle specialists, with upward of 1 million patients seeking treatment annually in the United States.1,2
Patients with this condition suffer from significant impairments in activities of daily living and overall well‑being. 3 Previous research has noted that contributing factors can include occupations requiring prolonged standing, repetitive weightbearing activities, running, limited ankle dorsiflexion, obesity, and pes planus.4 -6 Conservative treatment remains the mainstay of management and may include nonsteroidal anti-inflammatory drugs, physical therapy, stretching programs, night splints, plantar fascia massage, steroids and/or platelet-rich plasma injections, and/or orthotic inserts. 7
Custom foot orthoses are frequently prescribed for plantar fasciitis. These devices are designed to reduce strain on the plantar fascia by supporting the medial longitudinal arch and cushioning the heel. Their use has been supported in recent clinical guidelines for both short- and long-term improvement of patient symptoms, with the caveat that they should not be used as an isolated treatment for short-term symptom relief; rather, they are to be considered one adjunct among other conservative options.8,9 Although the patient-specific structural support of custom orthotics makes them an attractive option for both patients and practitioners, their fabrication presents notable manufacturing challenges. Traditionally, custom orthoses are fabricated using foam box impressions and manual techniques, which are labor-intensive and prone to variability in fit and quality. A systematic review emphasized that making impressions with foam boxes and plaster models requires specialized infrastructure, technical skill, and significant time, suggesting limitations in efficiency and reproducibility. 10
In response, 3D printing technology has emerged as a modern alternative. High-resolution foot scanning, combined with algorithm-driven modeling, 11 enables the rapid creation of patient-specific orthoses. Early data indicate that 3D scanning and additive manufacturing (AM) can be faster than traditional casting methods,12,13 with one study boasting a mere 10-hour print-to-processing workflow for their 3D-printed ankle-foot orthoses. 14 Additionally, narrative reviews indicate that 3D-printed orthoses, including those for the upper limb, lower limb, and spine, are durable, comfortable, and comparable to (or superior) to conventional devices in terms of biomechanical outcomes.13,15,16 However, comparative clinical evidence remains limited, underscoring the need for rigorous trials. Pilot data are needed to confirm the feasibility of recruitment, retention, and electronic PROMIS capture in this population and to estimate effect sizes for future sample size calculations.
This pilot study was designed to compare 2 methods of custom orthotic fabrication in patients with plantar fasciitis: traditional molded inserts and 3D-printed inserts produced using the HP Arize system. The primary objective was to evaluate short-term patient-reported outcomes related to physical function, mobility, and comfort. Secondary outcomes included patient satisfaction, frequency of insert use, and the incidence of adverse effects. It was hypothesized that 3D-printed orthoses would yield similar short-term improvements in function, mobility, and comfort compared with traditionally fabricated orthoses, without an increased rate of adverse effects. This study aimed to generate preliminary data to inform the design or potential utility of future larger-scale investigations.
Methods
Study Design
This was a prospective pilot study conducted in outpatient orthopaedic surgery clinics at a single academic health center. The study received institutional review board (IRB) approval prior to participant enrollment (IRB number 1876792). This study was conducted as an exploratory pilot to evaluate feasibility, estimate effect sizes, and inform the design of future adequately powered comparative trials; it was not intended or powered to establish definitive between-group differences.
Participants
Adults aged 18 years and older with a clinical diagnosis of plantar fasciitis (International Classification of Diseases, Tenth Revision [ICD-10] code M72.2) were eligible. Exclusion criteria included prior use of prescription orthotic inserts, need for bracing below the knee, presence of neuropathy or recent fracture, pregnancy, Medicare insurance status, and inability to communicate in English. All participants were required to have access to electronic communication to complete follow-up surveys. Participants were recruited during routine clinic visits by study physicians or advanced practice practitioners. Eligible patients who consented to participate were randomly assigned to either the traditional insert group or the HP Arize insert group using a computer-generated random number. An a priori power analysis was not conducted, as this was a pilot study designed to enroll 30 patients, 15 per arm. A total of 34 patients were enrolled, of whom 4 withdrew before receiving the insert. These 4 individuals were excluded from analysis because of the absence of either baseline or follow-up data.
Interventions
Patients randomized to the traditional insert group were referred to a certified pedorthist, who obtained custom molds using foam impression boxes. Inserts were fabricated off-site and fitted during a return visit. Patients in the HP Arize insert group underwent in-office foot scanning using the HP Arize 3D laser scanner (Figure 1). The scans were reviewed by a physician and submitted electronically to HP for fabrication. Completed inserts (Figure 2) were mailed to the clinic, and patients returned for fitting and counseling. All patients received standardized education regarding insert use, expected wear schedules, and signs of adverse reactions.

(A) Foot scanning device from HP Arize in the clinic. (B) Example of patient setup for foot scanning.

Example of a 3D-printed custom orthotic device (Arize) illustrating 3D-printed insole architecture. Image sourced from the Northeast Foot and Ankle website and reproduced here for noncommercial scholarly and educational purposes under a fair-use rationale. 17
Outcome Measures
Participants completed assessments at baseline and again at 4 and 8 weeks following receipt of the orthotic. Four- and 8-week surveys were administered electronically through REDCap; participants who did not complete their surveys online were contacted via phone to complete survey questions.
Baseline measures included demographic characteristics (age, sex, race, and body mass index), baseline foot pain and function ratings, and history of prior conservative treatments for plantar fasciitis such as medications, physical therapy, night splints, injections, over-the-counter inserts, heel cups, braces, exercises, or massage.
The primary outcomes were changes in Patient-Reported Outcomes Measurement Information System (PROMIS) physical function, pain interference, and mobility scores, assessed at baseline, 4 weeks, and 8 weeks after receipt of the orthotic inserts. These validated domains were selected to evaluate pain-related disability, general physical function, and mobility and were considered the principal measures of treatment effect.
Secondary outcomes included the frequency of orthotic wear at 4 and 8 weeks, the presence of adverse effects such as rubbing, redness, blisters, or soreness, and patient satisfaction with the customization process, ease of delivery, and fit and comfort of the inserts. Satisfaction ratings were quantified on a 5-point Likert scale, where 1 indicated very dissatisfied and 5 indicated very satisfied.
Feasibility Outcomes
Because this was a pilot investigation, feasibility outcomes were also evaluated. These included (1) recruitment feasibility (number screened, enrolled, and randomized), (2) retention and follow-up feasibility (completion rates at 4 and 8 weeks), (3) feasibility of PROMIS data capture through REDCap, and (4) feasibility of device fabrication workflows (time from scan/mold to insert receipt). These outcomes were collected to assess study processes and inform the design and operational parameters of a future adequately powered trial.
Data Management and Statistical Analysis
All study data were stored in REDCap, and participants were assigned a study identification number to maintain confidentiality. Descriptive statistics were calculated for baseline characteristics and outcome measures. Between-group comparisons were made using Fisher exact test for categorical variables and either Student t test or Wilcoxon signed-rank test for continuous variables. A P value < .05 was considered statistically significant. Because this was a pilot study, between-group statistical comparisons were conducted solely to estimate preliminary effect sizes and assess variability; P values should not be interpreted as testing for definitive differences between groups. Because 4 randomized participants withdrew before receiving their assigned insert and consequently did not contribute any outcome data, analyses were conducted using a modified intention-to-treat (mITT) approach that encompassed all participants who received an insert and completed at least the baseline assessment assessments. All statistical analyses were performed using SAS Enterprise Guide Version 8.3 (SAS Institute Inc, Cary, NC, USA).
Results
A total of 34 participants were enrolled in the study, including 18 assigned to the traditional insert group and 16 to the HP Arize insert group. Baseline demographics are presented in Table 1. Follow-up data were available for the majority of participants, with 3 missing surveys at 4 weeks and 2 at 8 weeks. Among the 30 participants who received inserts and contributed baseline data, follow-up completion was 90% at 4 weeks and 93% at 8 weeks, supporting the feasibility of retention and PROMIS data capture in this population.
Baseline Demographic, Clinical Characteristics, and Participant-Reported Prior Treatments for Plantar Fasciitis by Insert Group.

P values from χ2 or Fisher test for categorical variables, t test for age and BMI, and Wilcoxon signed-rank test for median baseline pain and function ratings.
Other includes over-the-counter inserts, heel cups, boots, braces, home exercises, and massage.
Traditional and HP Arize insert group participants were similar, and there were no statistically significant differences between groups with respect to sex, race, age, or BMI (all P > .05; Table 1). Similarly, baseline median pain and function scores for both the right and left foot did not differ significantly between groups (Table 1). At baseline, the most reported prior treatments among participants were medication (58.8%), physical therapy (33.3%), and other interventions (66.7%; Table 1). Of the other reported interventions, the highest frequencies were for at-home exercise (9 participants), over-the-counter inserts (8 participants), and heel cups (8 participants). No participants reported having received injections as prior therapies. There were no significant differences between the insert groups in the distributions of prior treatments (Table 1).
Baseline, 4-week, and 8-week PROMIS scores are presented in Table 2. No significant differences were observed between the insert groups at baseline or at the 4-week follow-up. However, at 8 weeks, participants in the traditional insert group had significantly higher mean PROMIS physical function scores (49.0 vs 41.9; P = 0.008) and PROMIS mobility scores (46.9 vs 41.4; P = 0.02) compared with the HP Arize insert group. However, changes over time (difference in 4-week and 8-week PROMIS scores from baseline) were not significantly different by insert group (Table 3).
PROMIS Scores at Baseline, 4 Weeks, and 8 Weeks by Insert Group.

Boldface indicates statistically significant p-values (p < 0.05)
Changes in PROMIS scores and number of patients meeting MCID by insert group.

Abbreviations: MCID, minimal clinically important difference; PF = physical function (PROMIS subscale), PI = pain inference (PROMIS subscale); PROMIS, Patient-Reported Outcome Measures Information System.
MCID cutoff was considered a ±5 or more PROMIS score improvement from baseline.
At both the 4- and 8-week follow-up intervals, similar proportions of patients in the Traditional and HP Arize insert groups achieved a minimal clinically important difference (MCID), defined as a ≥5-point change in PROMIS domains (Table 3). From baseline to 4 weeks, between 33% and 47% of patients in the Traditional group and 27% to 40% of patients in the HP Arize group reached the MCID threshold across physical function, pain interference, and mobility, with no statistically significant differences between groups. When assessed from baseline to 8 weeks, approximately one-third to more than half of patients in each cohort achieved MCID, including 46.7% vs 26.7% for physical function, 60% vs 53.3% for pain interference, and 40% vs 33.3% for mobility, again without significant between-group differences.
At 4 weeks post–insert receipt, participants in both the traditional and HP Arize insert groups reported high levels of satisfaction. Median Likert scale ratings indicated strong satisfaction with the overall customization process (5.0 in the traditional group and 4.5 in the HP Arize group) and with the ease of the process from customization to delivery (5.0 in both groups). Participants also reported satisfaction with the overall fit and feel of the custom inserts, with a median score of 4.0 in both groups. At 8 weeks, median responses for fit and feel remained unchanged at 4.0 for both insert groups.
At 4 weeks, study participants in the traditional insert group reported a significantly higher frequency of wearing their inserts every day or on most days than those in the HP Arize insert group (P = .04). However, those in the traditional insert group also reported a significantly higher frequency of rubbing or discomfort (P = .047) and other side effects (P = .02; Table 2). Only 2 participants in the HP Arize insert group reported any side effects at either time point. By 8 weeks post–insert receipt, these differences in side effects were no longer statistically significant (Table 4).
Insert Wear Frequency and Reported Side Effects at 4 and 8 Weeks by Insert Group.
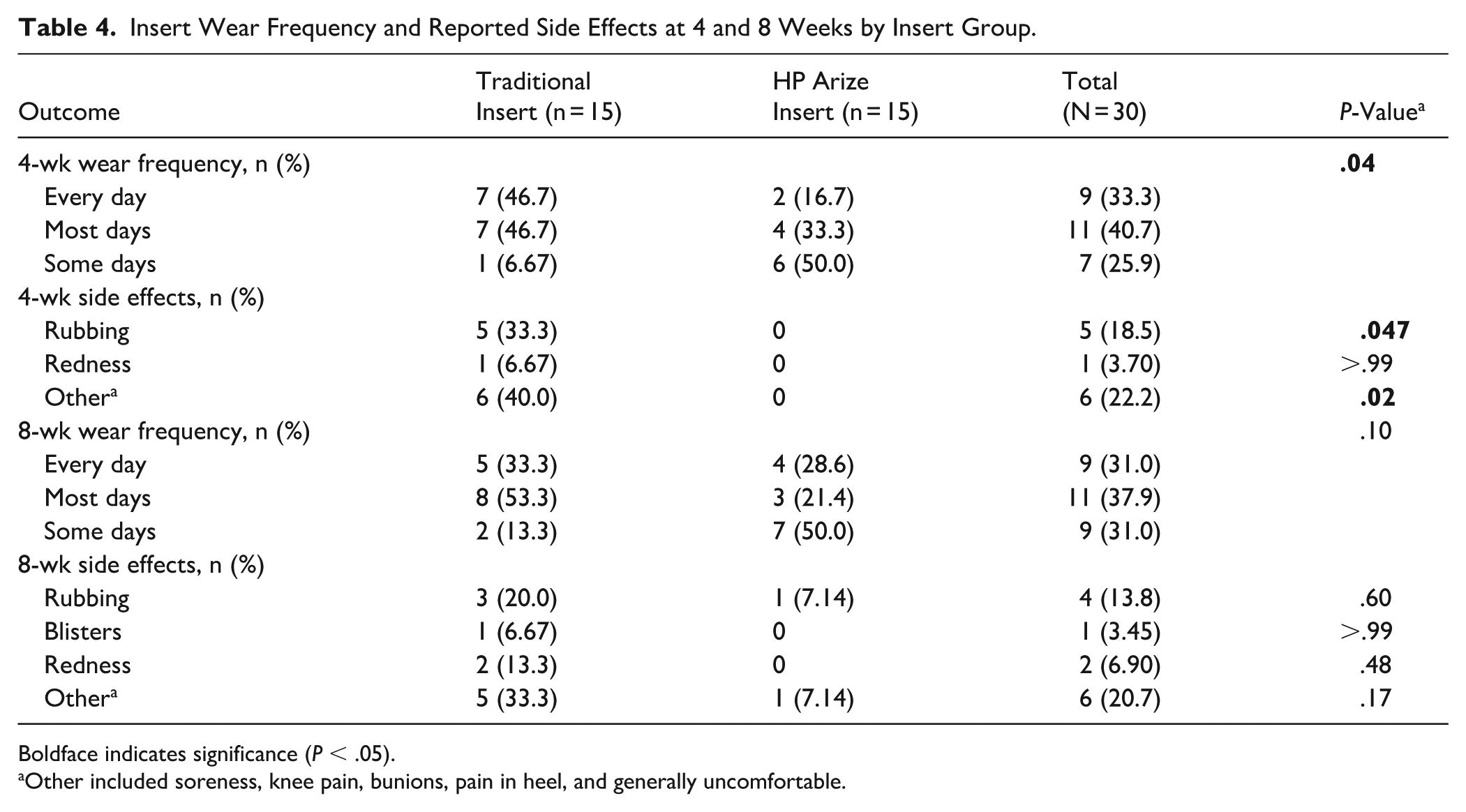
Boldface indicates significance (P < .05).
Other included soreness, knee pain, bunions, pain in heel, and generally uncomfortable.
Discussion
In this pilot, randomized study comparing traditional, custom-made inserts with HP Arize 3D-printed inserts for plantar fasciitis, both were associated with generally favorable short‑term patient‑reported outcomes and high satisfaction over 8 weeks. Despite differences in manufacturing and fitting workflows (wherein traditional inserts required 2 separate in-person visits and HP Arize inserts required follow-up for in-person fitting post-scan) participants in both groups reported similarly high satisfaction with the customization and delivery process. These pilot findings suggest that digital scanning and 3D printing can produce patient-specific orthoses that are generally acceptable to users, although lower early wear frequency in the HP Arize group limits conclusions about relative usability. These comparative observations should be interpreted cautiously, as this study was exploratory and not powered to detect definitive between-group differences.
A notable finding was the difference in short-term compliance between groups. At 4 weeks, 94% of participants in the traditional insert group reported wearing their inserts every or most days, compared with only 50% in the HP Arize group. Although this difference diminished slightly by 8 weeks (with compliance in the traditional group declining to 86% and remaining unchanged in the HP Arize group), it raises important considerations regarding early adherence to orthotic use. It is unclear if the difference in the fabrication process, visit with an orthotist or that experience, had an impact on the use of the orthotic or if there were other, as yet unidentified, factors that could have affected this rate. The low rate of reported side effects in the HP Arize insert group suggests that factors beyond discomfort, such as personal preference, perception of benefit, or other unmeasured influences, may have contributed to the lower compliance. Future qualitative or mixed-methods studies could help identify these drivers.
Because this was a pilot feasibility study, between-group comparisons are intended to offer preliminary estimates of effect size rather than to establish the clinical superiority of either orthotic device type. Although PROMIS scores for physical function, mobility, and pain were similar across groups at baseline and 4 weeks, participants in the traditional insert group showed significantly greater improvement in mean function and mobility by 8 weeks. This pattern may reflect the higher initial compliance observed in that group. However, attributing improvements solely to the orthoses is challenging, as nearly all participants received additional interventions during their initial visit (eg, medications, therapeutic exercises, or corticosteroid injections). As such, the observed improvements likely reflect the combined effect of multimodal management rather than the inserts alone. This was observed by Pfeffer et al, who examined patient-reported outcomes in a group of individuals treated with stretching only as compared to stretching with inserts; participants who combined stretching with inserts reported significantly less pain on the Foot Function Index pain subscale. 18
In our cohort, the proportion of patients achieving an MCID, defined as a ≥5-point change in PROMIS domains, was comparable between insert types. Across all time points, roughly one-quarter to one-half of patients achieved MCID in physical function, mobility, or pain interference, with no statistically significant differences between traditional and HP Arize inserts. These findings suggest that both orthoses may facilitate modest short-term improvements in patient-reported outcomes, but that clinically meaningful gains are not consistently attributable to insert type alone. The lack of difference further underscores the influence of concurrent multimodal interventions and highlights the need for adequately powered prospective studies to isolate the effect of orthotic design on functional recovery.
Emerging evidence suggests that customized 3D-printed foot orthoses may offer superior outcomes compared with conventional devices for plantar fasciitis and plantar heel pain. In a randomized controlled trial, Xu et al found that customized 3D-printed ankle-foot orthoses provided greater comfort and significantly reduced peak plantar pressure at the mid- and lateral heel compared with standard prefabricated ankle-foot orthoses over 8 weeks, indicating more effective redistribution of load away from symptomatic regions. 19 Comparative studies further report that 3D-printed orthoses perform at least as well as traditional devices in reducing ankle plantarflexion moment and power, but demonstrate clear advantages in limiting arch height drop, a parameter linked to arch stability, in unilateral plantar fasciopathy. 20 Finite element and gait analyses of 3D-printed heel cups also show improved heel pad support, reduced pressure on the plantar fascia and calcaneus, and pain relief within 4 weeks of use.13,15 Taken together, these findings highlight the growing body of evidence supporting 3D-printed orthoses as a potentially superior option for optimizing plantar biomechanics, reducing focal pressure, and enhancing patient comfort compared with conventional orthotic interventions.
To our knowledge, this is the first clinical pilot study to evaluate patient outcomes associated with the HP Arize 3D-printed orthotic platform. By prospectively comparing HP Arize with traditional custom inserts using validated PROMIS measures and patient-reported metrics, our study offers preliminary evidence of the acceptability and potential effectiveness of digitally manufactured orthoses in a clinical environment. Feasibility metrics in this pilot study were encouraging. Recruitment reached the prespecified target, and follow-up completion exceeded 90% at both time points, supporting the practicality of electronic PROMIS capture and adherence monitoring in this patient population. These findings suggest that a larger, adequately powered comparative trial is feasible. These findings contribute to a growing body of research on the clinical utility of 3D-printed foot orthoses and help inform future design, implementation, and adoption strategies.
However, there are important limitations to consider. Given the pilot study's relatively small sample size, the analyses were not powered to detect small or moderate between-group differences, and nonsignificant P values should not be interpreted as evidence of equivalence or lack of effect. The limited sample also reduces generalizability. Because participants were aware of their insert type, the study is susceptible to expectation bias. Perceptions favoring or questioning new technology could have influenced satisfaction ratings and PROMIS responses independent of true device performance. Nearly all participants also received adjunctive treatments during their initial visit, making it difficult to isolate the effects of the inserts themselves. Additionally, follow-up data at the 4-week point were missing for 3 participants, and at 8 weeks for 2 participants, which may introduce attrition bias. Lastly, the insert use frequency (Table 4) was self-reported, and objective measures of wear-time were not collected.
The findings indicate that both traditional and 3D-printed custom orthotics are deemed acceptable by patients and may result in functional improvements over time. The digital workflow associated with the HP Arize system may provide benefits in scalability and manufacturing efficiency, especially within clinical environments seeking to optimize orthotic delivery. Although workflow efficiency is often cited as a benefit of 3D-printed orthoses, this study did not assess efficiency metrics such as scanning time, fabrication duration, costs, or impact on clinical workflow. More research is necessary to examine these factors. Nonetheless, lower early compliance with the HP Arize insert emphasizes the importance of patient education and follow-up, particularly when introducing newer technologies. Clinicians should consider individual patient preferences, expectations, and potential barriers to adherence when recommending orthotic interventions.
Conclusion
In this pilot study, both custom-made and 3D-printed custom foot orthoses were generally well tolerated and showed favorable short-term patient-reported outcomes over 8 weeks in patients with plantar fasciitis. Traditional inserts demonstrated higher early self-reported use and greater improvements in PROMIS physical function and mobility scores at 8 weeks but were also associated with higher rates of discomfort. This pilot study found no signal of harm and indicated potential clinical benefits of 3D-printed custom inserts compared with traditional orthotics for the treatment of plantar fasciitis. These findings should be viewed as preliminary and warrant confirmation in a fully powered clinical trial.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114251413246 – Supplemental material for Pilot, Randomized Comparison of Traditionally Fabricated Custom Foot Orthoses vs HP Arize 3D-Printed Orthoses for Plantar Fasciitis
Supplemental material, sj-pdf-1-fao-10.1177_24730114251413246 for Pilot, Randomized Comparison of Traditionally Fabricated Custom Foot Orthoses vs HP Arize 3D-Printed Orthoses for Plantar Fasciitis by J. Benjamin Jackson, Kaleb Eaton, Harley T. Davis, Emily McKinney and Tyler A. Gonzalez in Foot & Ankle Orthopaedics
Footnotes
ORCID iDs
Ethical Considerations
The study received institutional review board (IRB) approval prior to participant enrollment (IRB number 1876792) on April 12, 2022.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors’ institution received a research grant from HP Inc. that covered the costs associated with IRB submission fees, research staff time, and orthotic fabrication for the HP and standard groups. The authors received no direct funding. HP Inc had no role in study design, data collection, analysis, interpretation, or manuscript preparation.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J. Benjamin Jackson III, MD, MBA, reports disclosures relevant to manuscript of Vilex – consultant. Tyler A. Gonzalez, MD, MBA, reports disclosures relevant to manuscript of Vilex, Treace Medical Concepts Inc., Enovis, ConMed, SBM – consultant; Vilex, Treace Medical Concepts Inc, Enovis, SBM – royalties. Disclosure forms for all authors are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
