Abstract
Background:
Cartilage lesions with concomitant subchondral plate damage are challenging pathologies for foot and ankle surgeons. Recent clinical reports suggest that recombinant human bone morphogenetic protein-2 (rhBMP-2) can restore structural integrity of the subchondral plate and promote osseous healing, potentially improving outcomes for cartilage repair procedures. This study seeks to evaluate the complication rate and patient-reported outcomes of rhBMP-2-augmented repair for ankle osteochondral lesions with subchondral involvement.
Methods:
Patients who received surgical repair with rhBMP-2, extracellular matrix (ECM), and bone marrow aspirate concentrate (BMAC) for the treatment of an ankle osteochondral defect with an associated subchondral damage between January 2023 and September 2024 were retrospectively chart reviewed. Twenty-one patients who fit the criteria were included. All patients completed Patient-Reported Outcomes Measurement Information System (PROMIS) surveys preoperatively and at minimum 1 year postoperatively. Clinical outcomes, postoperative complications, and revision surgery were recorded.
Results:
The average time from surgery to postoperative survey follow-up was 17.9 (range, 12.0-27.3) months. Average cartilage lesion size was 88.4 (range, 36.0-120) mm2. Significant pre- to postoperative improvement in Physical Function, Pain Interference, and Pain Intensity were observed among the patient cohort. Physical Function score increased by an average of 10.4 points (P < .05). Pain Interference improved by an average of 10.4 points (P < .05). Pain Intensity decreased by an average of 8.6 points (P < .05). Global Physical Health increased by an average of 8.4 points (P < .05). Global Mental Health and Depression did not change significantly. Observed changes exceeded commonly cited PROMIS minimal clinically important differences for lower-extremity populations. The complication rate was low, with only 2 patients (9.5%) reporting persistent pain requiring a return to the operating room for revision surgery.
Conclusion:
Our results suggest that this procedure is an effective treatment with a significant improvement in clinical outcomes and a low complication rate.
Level of Evidence:
Level IV, case series.

Keywords
Introduction
Articular cartilage defects can be limited to the superficial layer of cartilage or can extend deeper, affecting the underlying subchondral bone of the ankle. Certain defects, such as ankle osteochondral lesions (OCLs), may in fact start in the subchondral bone, only secondarily affecting the overlying cartilage. Other joint pathologies involving the subchondral bone include osteonecrosis and osteochondral fractures. Full-thickness ankle cartilage defects are seen in a range of 18% to 33% of OCL cases.1,11 This subset of cases is associated with poorer outcomes and a higher rate of progression to new cartilage damage as well as osteoarthritis if left untreated.14,18,30
Bone grafting is a reasonable treatment option for repairing a subchondral bone defect after debridement of a cyst that avoids obvious donor site injuries. Recombinant human bone morphogenetic protein-2, rhBMP-2 (Medtronic) is a widely used osteoinductive adjuvant that has shown promise in promoting subchondral bone healing in full-thickness cartilage defects.22,27 However, the repair of the cartilage lesion still remains a concern. Previous studies have indicated the use of biological adjuncts such as extracellular matrix (ECM) (Arthrex) and bone marrow aspirate concentrate (BMAC) as a viable option to improve clinical and radiographic outcomes in the repair of ankle osteochondral lesions.12,13 Considering the potential of rhBMP-2 in cartilage repair and regeneration, we applied a combination of rhBMP-2, ECM, and BMAC for treatment of full-thickness cartilage lesions with subchondral bone damage.
The presented study evaluated pre- to postoperative clinical outcomes and complicate rate of ankle osteochondral lesions with subchondral bone involvement treated with rhBMP-2, ECM, and BMAC. We hypothesized that patients treated with rhBMP-2, ECM, and BMAC will demonstrate a significant improvement in patient-reported outcomes following surgical repair. To the authors’ knowledge, this study was the first collection of human clinical outcomes after this intervention.
Methods
This was a single-center retrospective study conducted from the institutional review board–approved Foot and Ankle Registry data, and the protocol was approved by the steering committee at the investigators’ institution. Registry data were retrospectively reviewed to identify patients who received osteochondral lesion repair with rhBMP-2, ECM, and BMAC between January 2023 and September 2024 by the principal investigator.
Inclusion criteria were patients aged 18 years or older who underwent surgery for a primary diagnosis of osteochondral lesion with subchondral plate disruption on the talus and who completed Patient-Reported Outcomes Measurement Information System (PROMIS) surveys both preoperatively and at a minimum of 1 year postoperatively. Patients were excluded if they did not receive rhBMP-2, ECM, or BMAC together; had histories of previous ipsilateral forefoot or ankle surgery; presented with an ankle fracture, rheumatoid arthritis, or gout; had concomitant procedures (6 patients); had a calcaneal or navicular OCL (2 patients); or had insufficient baseline or follow-up functional outcome scores (16 patients). Forty-five patients who received the combination treatment were initially identified from the registry; after applying inclusion criteria, 21 patients were included in the study cohort.
Preoperative magnetic resonance imaging (MRI) radiographs were available for all patients and were obtained to confirm subchondral damage and collect cartilage demographic data. Lesion location and surface area were noted for each patient. Retrospective chart review was performed to collect patient age, sex, BMI, race, and ethnicity. Operative and postoperative clinic notes were reviewed for all performed procedures, clinical outcomes, and complications including persistent pain, stiffness, wound healing issues, and return to the operating room for revision surgery.
Survey Outcomes
Patient-reported outcomes were assessed using PROMIS scores, which has been validated in various foot and ankle surgeries. PROMIS is a computerized adaptive test (CAT) used to assess functional outcomes in the following domains: Physical Function, Pain Interference, Pain Intensity, Global Physical Health, Global Mental Health, and Depression. Scores have a standardized mean of 50, the reference population average, with an SD (T score) of 10. Higher scores indicate greater physical function, pain interference, pain intensity, global health, and depression. All patients received PROMIS surveys preoperatively and at 1, 2, 5, and 10 years postoperatively, if applicable, through the foot and ankle registry at our institution. For the purposes of this study, patients’ preoperative and most recent postoperative PROMIS surveys, collected at a minimum of 1 year postoperatively, were analyzed.
Surgical Technique
The patient was placed in the supine position. The technique to collect BMAC has been described before in the literature.12,13 Approximately 60 mL of bone marrow was aspirated from the anterior superior iliac crest to produce 3 mL of BMAC. Next, the patient was placed in noninvasive ankle distraction. Standard anteromedial and anterolateral portals were established to allow for diagnostic arthroscopy. The area was debrided, and any scar tissue, osteophytes, and loose fragments were removed. All fluid was removed using an epidural spinal needle and vacuum suction, and the remainder of the arthroscopy was performed dry.
Attention was then turned to the talus. After the size of the defect and extent of the cystic changes in the talus were assessed, the mixture of rhBMP-2, ECM, and BMAC was placed in the base of the defect to fill the subchondral plate and bony layer (Figure 1). A fibrin sealant was then placed over the mixture to allow for additional fixation and left to dry. Patients were placed in a splint and remained nonweightbearing for 2 weeks. At 2 weeks postoperatively, they were transitioned to a boot and began active and passive range-of-motion ankle exercises as well as hip- and knee-strengthening exercises. Nonweightbearing status completed at 6 weeks postoperatively, at which point patients were allowed to begin partial weightbearing with a boot and crutches. At approximately 8 weeks postoperatively, patients advanced to full weightbearing in a sneaker.
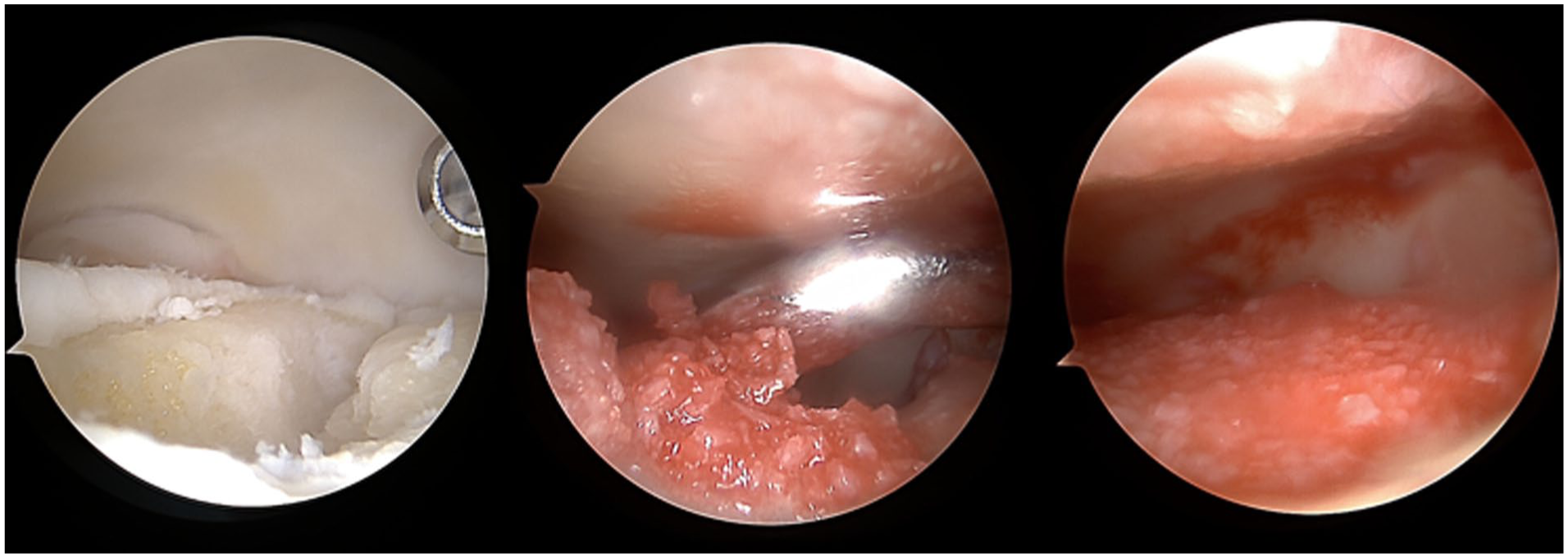
Intraoperative images of osteochondral lesion treated with rhBMP-2, ECM, and BMAC. BMAC, bone marrow aspirate concentrate; ECM, extracellular matrix; rhBMP-2, recombinant human bone morphogenetic protein-2.
Statistical Analysis
Descriptive statistics were reported as means and SDs for continuous variables and counts and percentages for categorical variables. All patients treated with this combination therapy from our registry were considered. Paired t tests were used to compare preoperative and postoperative PROMIS scores after normality was assessed and confirmed using the Shapiro-Wilk test. Listwise deletion was conducted for patients with missing data. Statistical significance was determined with an alpha of .05. Analysis was conducted on R: A Language and Environment for Statistical Computing (R Core Team 2021).
Results
Patient Demographics
Chart review was performed to collect demographic information. Patient demographics are listed in Table 1. The average time from index procedure to last clinical follow-up visit was 6.4 (range, 2.3-13.1) months. The average age of the patient cohort was 42.0 (range, 24.2-70.8) years. Thirteen men and 8 women were included. The average BMI was 27.5 (range, 21.3-45.9). Radiographic analysis of preoperative MRIs demonstrated an average cartilage lesion surface area of 88.4 (range, 36.0-120) mm2. Fourteen patients presented with osteochondral dissecans, and 7 patients presented with an acute osteochondral lesion. Seventeen patients had 1 lesion, 3 patients had 2 lesions, and 1 patient had 3 lesions. Of the 26 lesions, there were 12 medial talar defects, 5 lateral talar defects, 1 central talar defect, and 8 distal tibial plafond defects (Table 1).
Patient Cohort Demographics (N = 21).

Abbreviations: BMI, body mass index; F, female; M, male; PROMIS, Patient-Reported Outcomes Measurement Information System.
PROMIS Outcomes
Twenty-one patients had both preoperative and minimum 1-year postoperative PROMIS scores. The average time from the index procedure to postoperative survey completion was 17.9 (range, 12.0-27.3) months. The patient cohort demonstrated significant pre- to postoperative improvement in physical function, pain interference, pain intensity, and global physical health. However, no differences were observed in global mental health and depression (Table 2).
Pre- to Postoperative PROMIS Outcomes

Abbreviation: PROMIS, Patient-Reported Outcomes Measurement Information System.aBoldface indicates significance (P < .05).
Complications
Two patients reported persistent pain that required a return to the operating room. One patient presented as a revision case that had already received ankle arthroscopy with attempted cartilage repair for the treatment of both talar and tibial plafond osteochondral dissecans and osteoarthritis. Five months after their procedure with rhBMP-2, ECM, and BMAC, the patient received another arthroscopy debridement with anterior osteophyte decompression, OCD microfracture of lateral tibial plafond lesion with autograft bone grafting of calcaneus, and a superficial peroneal nerve decompression. At 16 months postoperative to the second procedure, new radiographs indicated mild degenerative changes in the ankle joint as well as a small anterior osteophyte from the distal tibia. Physical therapy was recommended for this patient, who had some persistent symptoms.
The second patient who initially presented with osteochondral dissecans lesion measuring 12 mm × 16 mm × 8 mm demonstrated ankle pain and persistent edema 16 months postoperatively with no improvement with multiple hyaluronan injections (Figure 2). At 2 years postoperatively, the patient received a total ankle arthroplasty as well as a lateral ligament reconstruction. The patient responded well to the procedure and has reported an improvement in symptoms.

Sixty-nine-year-old female patient. (A) Preoperative MRI shows large lesion on talar dome (12 mm × 16 mm × 8 mm). (B) Fifteen-month postoperative MRI shows healing of the subchondral plate and cartilage (fat-suppressed MRI). (C) Preoperative MRI. (D) Twenty-one-month postoperative MRI shows significant edema status of the joint. The patient was indicated for a total ankle repair 2 years after initial arthroscopic rhBMP-2, ECM, and BMAC treatment. BMAC, bone marrow aspirate concentrate; ECM, extracellular matrix; MRI, magnetic resonance imaging; rhBMP-2, recombinant human bone morphogenetic protein-2.
Discussion
This study demonstrated significant pre- to postoperative clinical improvements after an average of 17 months of follow-up for the treatment of ankle osteochondral lesions with subchondral involvement using rhBMP-2, ECM, and BMAC. In addition, our patient cohort identified 2 of 21 patients (9.5%) with persistent postoperative pain who required reoperation. We observed a low complication rate associated with the use of rhBMP-2. However, more data are required to corroborate the use of rhBMP-2 for ankle subchondral lesions. Drakos et al 13 reported the use of bone graft with ECM-BMAC in several osteochondral lesions with subchondral involvement, but an insufficient number of cases prevented the authors from greater statistical analysis in their study. This study serves as an investigative follow-up for this particular subset of injuries.
Bone morphogenetic protein 2 (BMP-2) is a member of the transforming growth factor β (TGF-β) superfamily critically involved in osteogenesis. 21 BMP-2 binds to type I and type II serine/threonine kinase receptors on the surface of various cell types, including mesenchymal stem cells, initiating a signaling cascade predominately via the suppressor of mothers against decapentaplegic (SMAD) pathway. 21 This cascade culminates in the transcription of osteogenic genes, driving the differentiation of mesenchymal stem cells into osteoblasts.5,8 The clinical use of BMP was initially limited because of its required isolation from donor bone, which required several kilograms of donor bone to obtain grams of BMP. 25 Advances in molecular biology in the 1980s allowed sequencing of BMPs, with the first version of rhBMP-2 being cloned in 1988. 31 RhBMP-2, the active ingredient in Infuse Bone Graft, has been shown to induce bone formation as an “osteoinductive” autograft replacement in a number of preclinical and clinical investigations and has been cleared for use in interbody spine fusion, open tibial fractures, and oral maxillofacial bone grafting procedures.3,4,6,9,15,16,23,24,29,32,33
The use of BMPs in cartilage regeneration has been reported in several studies.10,22,26 -28 However, there is a scarcity of data for its use in human trials. To the authors’ knowledge, this study represents the first case series observing the effects of rhBMP-2 on human ankle cartilage regeneration. Taniyama et al 27 evaluated the effect of transplantation of porous hydroxyapatite collagen (HAp/Col) impregnated with rhBMP-2 on osteochondral defect repair in a rabbit model and demonstrated significant subchondral bone repair compared with the untreated defect control group. Sellers et al 22 examined repair tissue quality of full-thickness cartilage repairs 1 year postoperatively with rhBMP-2 in rabbit models and reported significant improvement in histologic appearance and composition of the ECM compared to untreated control groups. The specific categories that were improved by treatment with rhBMP-2 included integration at the margin, cellular morphology, architecture within the defect, and formation of a new tidemark. The authors suggested that this improvement may have been due to the acceleration of subchondral bone replacement, which may provide increased physical support to the newly forming articular surface in the treated defects. Tokuhara et al 28 observed dose-dependent effects of Escherichia coli–derived dimeric rhBMP-2 (E-rhBMP-2) for the repair of large osteochondral defects in a rabbit model and reported accelerated rates of bone repair with high doses of E-rhBMP-2. At 24 weeks, the repair of cartilage tissue was significantly improved with E-rhBMP-2 treatment compared to defects treated without E-rhBMP-2.
Although only rhBMP-2 directly addresses subchondral plate damage, outcomes with the combination of rhBMP-2, ECM, and BMAC in our study are comparable to alternative cartilage repair treatments, such as microfracture and ECM-BMAC. Drakos et al 12 reported revision rates of 4.8% for ECM-BMAC and 20.9% for microfracture, which are similar to the 9% revision rate observed in our cohort. The patient-reported functional outcomes after ECM-BMAC and microfracture are also consistent with our findings, with patients reporting significant improvement in physical function and pain at short-term follow-up. 12 The key distinction between the rhBMP-2, ECM, BMAC combination, and other cartilage repair strategies is that the combination therapy repairs both the subchondral plate and overlying cartilage defect, whereas microfracture and ECM-BMAC primarily target surface cartilage.7,12 For this reason, the authors refrained from direct comparison between rhBMP-2 and cartilage-only repair methods.
Cartilage replacement techniques, such as the osteochondral autograft transplant (OAT) procedure have also been used to address OCLs with subchondral damage. In the OAT procedure, damaged subchondral bone and cartilage are replaced with healthy tissue, typically a plug harvested from the patient’s knee. However, OAT is best suited for large or cystic OCLs with subchondral damage, typically measuring greater than 10 mm in diameter. 17 Because our study focused on small to medium-sized lesions (average lesion surface area 88.4 mm2), the authors believe direct comparison between the use of rhBMP-2 and OATs would be inappropriate. Nevertheless, the favorable results of our study suggest that rhBMP-2 could be used for intermediate-sized lesions with subchondral damage as an alternative to OATs.
The reported side effects of rhBMP-2 use include ectopic bone formation, inflammatory responses, seroma formation, and wound healing issues. 2 None of these side effects were reported in our study cohort. The complications seen in our cohort, which included 2 patients who returned to the operating room for revision surgery, were related to the persistent pain likely associated with the severity of their injury rather than negative side effects associated with rhBMP-2. Our low complication rate with the use of a small dose of rhBMP-2 is consistent with previous studies which reported that rhBMP-2 side effects are dose-dependent. 20 Mendenhall et al 20 evaluated the low-dose impact of rhBMP-2 for anterior cervical fusions and determined it is safe, reporting solid arthrodesis in 96% of patients with an average 15-month follow-up. The authors’ low-dosage rhBMP-2 protocol (0.50 ± 0.09 mg per level) also demonstrated significantly reduced incidence of complications in comparison to higher dosage protocols for ACFs within the literature. A similar small dose of rhBMP-2 was used for our cohort, with rhBMP-2 applied on a scaffold as per manufacturing instructions. 19 In our study, ECM-BMAC served as a scaffold, preventing rhBMP-2 from being assessed in isolation.
This study presented with several limitations. First, this study is limited to a short-term follow-up, so there is still a possibility for future adverse effects. A study with longer follow-up is required to better illustrate the clinical timeline of rhBMP-2 for this procedure. Second, this retrospective case series lacked radiographic and/or cellular ossification analysis to correlate clinical results with. Postoperative MRIs or CT scans were not routinely collected within the patient cohort, so postoperative radiographic analysis was unable to be performed. Future studies should collect postoperative MRIs and cell samples to determine the quality of postoperative repair tissue. Third, the combination of 3 different components (rhBMP-2, ECM, and BMAC) to address cartilage injury in this case series prevented rhBMP-2 from being isolated as the independent variable in this study. And importantly, there was no control group in this study. As noted, the authors believe the unique mechanism of rhBMP-2 in repairing subchondral damage of OCLs makes it difficult to compare to existing cartilage repair techniques. Future well-controlled studies with postoperative imaging are needed to further assess the efficacy of rhBMP-2 in repairing OCLs with subchondral damage (Figure 3) (Figure 4).

Twenty-eight-year-old male patient. (A) Preoperative MRI shows OCL of talar dome with significant cartilage degeneration. (B) Twenty-month postoperative MRI cartilage surface repair with rhBMP-2, ECM, and BMAC. BMAC, bone marrow aspirate concentrate; ECM, extracellular matrix; MRI, magnetic resonance imaging; OCL, osteochondral lesion; rhBMP-2, recombinant human bone morphogenetic protein-2.

Forty-seven-year-old female patient. (A) Preoperative MRI shows tibial plafond lesion. (B) Seven-month postoperative MRI cartilage surface repair after rhBMP-2, ECM, and BMAC. BMAC, bone marrow aspirate concentrate; ECM, extracellular matrix; MRI, magnetic resonance imaging; rhBMP-2, recombinant human bone morphogenetic protein-2.
Conclusion
This study observed improved short-term patient-reported outcomes for the treatment of osteochondral defects with subchondral involvement with rhBMP-2, ECM, and BMAC. The patient cohort reported a significant improvement in pain and physical function with an average of 1.5 years of follow-up and was associated with a low return to OR rate of 9.5%. Although this study presents favorable outcomes for this procedure, more data are required to further investigate the use of bone graft and biologic adjuncts.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114251398507 – Supplemental material for Evaluating Clinical Outcomes of Recombinant Human Bone Morphogenetic Protein-2 in the Treatment of Subchondral Plate Damage Associated With Ankle Osteochondral Lesions
Supplemental material, sj-pdf-1-fao-10.1177_24730114251398507 for Evaluating Clinical Outcomes of Recombinant Human Bone Morphogenetic Protein-2 in the Treatment of Subchondral Plate Damage Associated With Ankle Osteochondral Lesions by David Cho, Cordelia Burn, Jonathan Gamarra, Jayson Stern, Prerana Katiyar and Mark Drakos in Foot & Ankle Orthopaedics
Footnotes
Ethical Considerations
Ethical approval for this study was obtained from the HSS Foot and Ankle Steering Committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mark Drakos, MD, reports general disclosures of Arthrex, Extremity, and Aevumed. Disclosure forms for all authors are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
