Abstract
Background:
Haglund syndrome is characterized by heel pain associated with posterosuperior calcaneal exostosis, insertional Achilles tendinopathy, and retrocalcaneal bursitis. When conservative treatments fail, surgical intervention is required. This study aims to evaluate the effectiveness of a combined surgical approach, integrating percutaneous resection of the calcaneal exostosis and proximal medial gastrocnemius release (PMGR) using the Barouk technique in treating Haglund syndrome. The goal is to assess whether this approach offers superior clinical outcomes compared to percutaneous resection alone.
Methods:
We prospectively enrolled 224 patients undergoing combined percutaneous resection and PMGR, divided into group A (n = 106; with Achilles–plantar complex contracture) and group B (n = 118; without contracture) based on passive dorsiflexion testing. Outcomes were compared to 2 historical retrospective control groups treated with resection only: group 1 (n = 124; with contracture) and group 2 (n = 135; without contracture). All prospective patients received the combined treatment regardless of contracture status. Patients with body mass index >30 were excluded. The Foot Function Index (FFI) and Victorian Institute of Sport Assessment–Achilles (VISA-A) Questionnaire scores were collected at baseline and 3, 6, and 12 months.
Results:
Groups A and B improved a mean 30 ± 5 FFI points and 40 ± 7 VISA-A points at 12 months (both P < .001). When compared to historical controls who underwent resection alone, the combined treatment groups showed statistically superior outcomes at all follow-up intervals (P < .05). However, these comparisons are limited by the nonconcurrent, unmatched study design.
Conclusion:
This mixed prospective-retrospective study suggests potential benefits of adding Achilles–plantar complex lengthening to percutaneous calcaneal resection. However, the nonrandomized design, historical controls, and lack of patient/outcome assessor masking significantly limit causal inference. Although the combined approach showed statistically superior outcomes compared with historical controls, these findings should be considered hypothesis-generating pending validation in randomized controlled trials.
Level of Evidence:
Level III, retrospective-comparative cohort.
Introduction
The Achilles–plantar complex includes the Achilles tendon and plantar fascia. The Achilles tendon, the largest and strongest tendon in the body, arises from the gastrocnemius and soleus muscles, with occasional input from the plantaris (present in ~92%-94%). 16 It is ~15 cm long, flattening before inserting into the calcaneus, with some fibers extending into the plantar fascia.9,10 The plantar fascia, originating from the medial calcaneal tubercle and extending to the toes, supports the medial arch and gait biomechanics. 24 Studies suggest anatomical and functional interconnectivity between the 2 structures. Haglund syndrome is an orthopaedic condition characterized by heel pain, typically caused by a triad of posterosuperior calcaneal exostosis, insertional Achilles tendinopathy, and retrocalcaneal bursitis.
Although consensus on precise definitions remains lacking (Haglund deformity, Haglund syndrome, and Haglund disease), we will hereafter adopt the term “Haglund syndrome,” as it most closely aligns with the definition used in our study. 17 Haglund syndrome is an orthopaedic condition that presents with heel pain, particularly at the Achilles tendon insertion. This syndrome is identified by a characteristic triad: posterosuperior calcaneal exostosis, insertional Achilles tendinopathy, and retrocalcaneal bursitis, leading to significant discomfort and impact on mobility and quality of life.18,20
Radiographic diagnosis of Haglund deformity was performed by evaluating the following parameters for each patient: Fowler-Philip angle (FPA), Heneghan-Pavlov parallel pitch lines (PPL), Haglund deformity height, bump height, and the bump-to-calcaneus ratio. In addition, to better assess the presence of retrocalcaneal bursitis and Achilles tendon calcifications, all patients underwent both ultrasonographic and magnetic resonance imaging (MRI) prior to surgery.
The pathophysiology of Haglund syndrome involves mechanical irritation and inflammation caused by the bony prominence against footwear, leading to chronic pain and bursitis.
For a subset of patients, these conservative measures fail to provide lasting relief, necessitating surgical intervention.
Several surgical techniques have been developed for the treatment of Haglund syndrome, each with its own approach and considerations. Traditional "open" surgical procedures can be performed with either a lateral approach or transverse approach, with or without detachment of the Achilles tendon, or a medial approach involving an Achilles tendon split. These methods, although effective, are often associated with significant recovery times and risk of complications.1,4,7,12
Endoscopic techniques, such as the one developed by Van Dijk, offer a less invasive alternative, focusing on the removal of the bony prominence and debridement of the bursa via small incisions. 19
Similarly, percutaneous procedures have gained popularity for their minimal invasiveness and quicker recovery periods. Among these, the ultrasound-guided technique described by Madarevic et al 11 and the lateral percutaneous approach under fluoroscopic guidance introduced by Sergio et al, 21 represent significant advancements in the surgical treatment of Haglund syndrome.
Haglund syndrome is often associated with a contracture of the Achilles–plantar complex; currently, there are no studies in the literature that evaluate the impact of surgical treatment addressing this contracture alongside the treatment for Haglund syndrome. In more severe cases of Achilles–plantar complex contracture, a true equinus deformity of the foot may develop, biomechanically resulting from the shortening of both the gastrocnemius and soleus muscles.
The focus of our study is on the innovative combination of a minimally invasive percutaneous technique for the resection of the calcaneal exostosis as described by Sergio et al, 21 paired with the proximal medial gastrocnemius release (PMGR), as originally described by Barouk. 2
The combined approach, integrating both percutaneous resection and PMGR, aims to address the bony exostosis and soft tissue contracture of Haglund syndrome, based on the hypothesis that both posterior impingement and tension contribute synergistically to symptom persistence. By evaluating the efficacy of this combined surgical intervention, our study seeks to contribute to the ongoing development of treatment modalities for Haglund syndrome, offering new insights and options for both patients and practitioners.
Materials and Methods
Study Design and Patient Selection
A power analysis was conducted using G*Power (version 3.1), assuming a large effect size (d = 0.8). To detect this effect using a 2-tailed paired samples t test, with an alpha level of .05 and a statistical power of 0.95, a minimum sample size of 19 participants was required. The sample used in the present study substantially exceeded this threshold.
This study is a prospective cohort study with a retrospectively collected control group. The prospective phase evaluated the efficacy of a novel surgical intervention for Haglund syndrome, a debilitating condition characterized by persistent heel pain that significantly impairs quality of life. We enrolled a cohort of 224 patients (120 males, 104 females) with an average age of 38 ± 7.4 years (range 26-49 years). Eligibility for inclusion required a clinical and radiologic diagnosis (in addition to standard radiographic evaluation, all patients underwent both ultrasonography and MRI, which confirmed the presence of retrocalcaneal bursitis and Achilles tendinopathy) of Haglund syndrome, with symptoms persisting for more than 3 months despite conservative treatment efforts. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the local ethics committee.
Prior to considering surgical intervention, all participants underwent an array of conservative treatments, including physical therapy, nonsteroidal antiinflammatory drugs, and footwear modifications.21,25 These measures, however, did not yield long-term relief, necessitating exploration of surgical options to address the chronic heel pain associated with Haglund syndrome.
Furthermore, the retrospective analysis was conducted with patients in our database who underwent surgery for Haglund syndrome in the years before the start of this study involving only percutaneous resection of the posterior-superior angle of the calcaneus.
Inclusion and Exclusion Criteria
Inclusion criteria mandated a history of persistent pain refractory to conservative measures and confirmed diagnosis of Haglund syndrome. We excluded individuals with a body mass index greater than 30, those who had undergone previous orthopaedic surgeries on the lower limbs, patients with rheumatic or neurologic disorders, and individuals with recent trauma or injuries to the lower limb within 6 months prior to the surgical intervention and people who have had at least 1 fragility fracture due to osteoporosis. 15 These criteria ensured a homogenous study population and integrity of the outcome data.
Assessment of Achilles-Plantar Complex Contracture
A pivotal aspect of our patient assessment was the evaluation of passive ankle dorsiflexion with the knee extended, with a dorsiflexion angle of less than 10° being considered indicative of Achilles–plantar complex contracture. 14 The findings from this test allowed us to stratify our patient cohort into 2 distinct groups: group A, comprising 106 subjects with identified Achilles-plantar contracture, and group B, consisting of 118 subjects without such contractures. This stratification was not merely a division but a step toward understanding the nuanced responses to our surgical technique based on the preexisting conditions of the Achilles-plantar complex. 22
In the retrospective analysis, patients previously treated with only percutaneous resection were divided into 2 groups based on the presence (group 1 [n = 124]: patients with Haglund syndrome and contracture of the plantar Achilles complex, treated only with percutaneous resection of the posterior-superior angle of the calcaneus) or absence (group 2 [n = 135]: patients with Haglund syndrome and no contracture of the Plantar Achilles complex, treated only with percutaneous resection of the posterior-superior angle of the calcaneus) of contracture of the plantar Achilles complex.
Outcome Measures and Follow-up
Participants were assessed using the Foot Function Index (FFI) and the Victorian Institute of Sport Assessment–Achilles (VISA-A) Questionnaire at baseline and at 3, 6, and 12 months postoperatively.3,6, 8 These instruments measured pain, functionality, and overall well-being, providing a quantitative framework for evaluating the success of the surgical intervention. Baseline FFI scores and VISA-A Questionnaire scores were compared with those recorded at 3, 6, and 12 months postoperatively for all patients. Additional analyses were conducted comparing preoperative scores between groups A and B, group A and group 1, and group B and group 2, and subsequently at 3, 6, and 12 months.
Surgical Technique
All patients underwent a standardized surgical procedure involving percutaneous decompression with lateral access to the Achilles tendon as described by Sergio et al and PMGR. This combined approach aimed at addressing the calcaneal exostosis and soft tissue pathology was anticipated to not only alleviate pain but also improve functionality and correct anatomical alignment.
The decision to perform Achilles–plantar complex lengthening even in patients with a negative Silfverskiöld and no Achilles–plantar complex contracture was made with the goal of reducing tension in the posterior region of the ankle and foot (Figure 1).
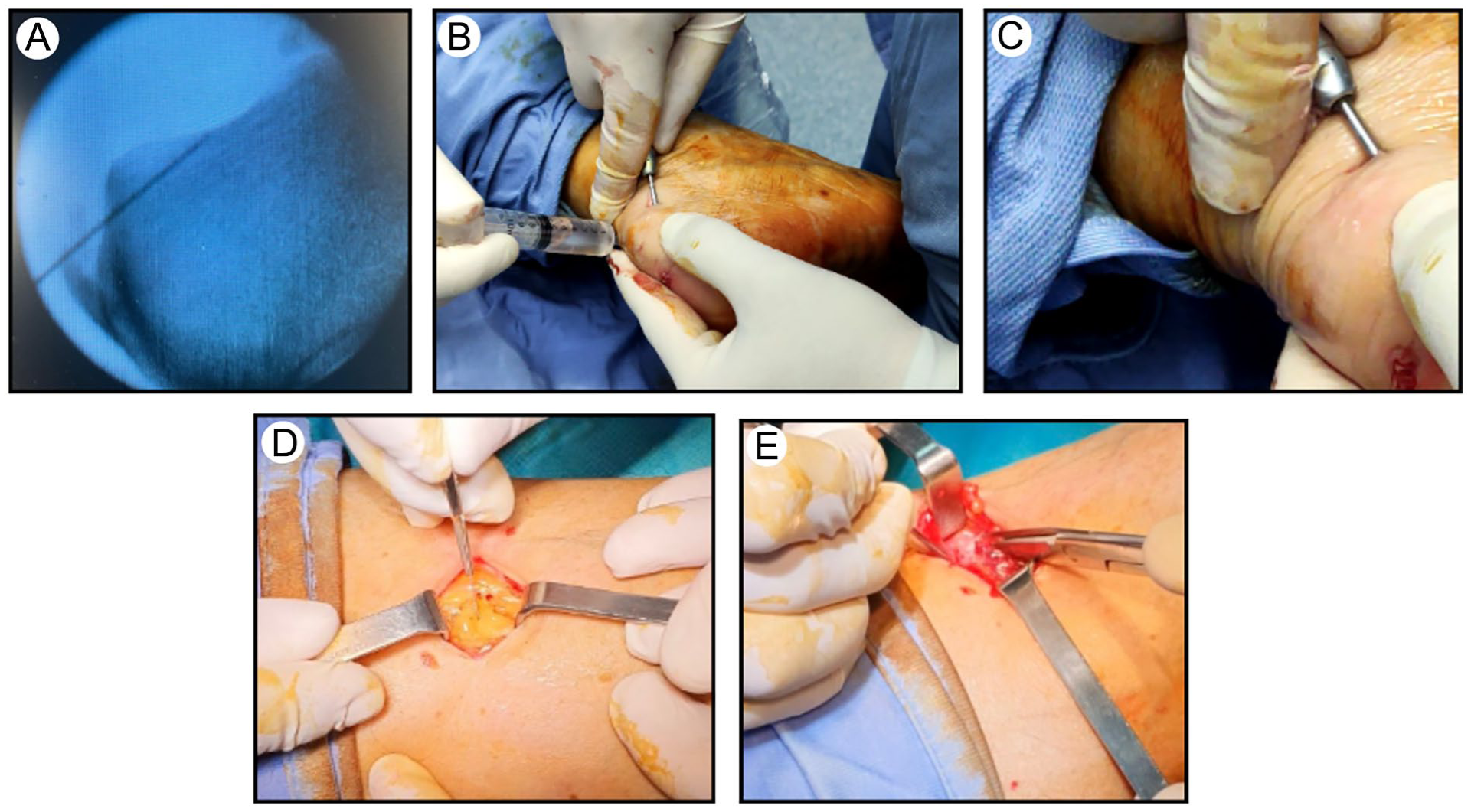
Intraoperative images. (A) Finding the limit of the retrocalcaneal exostosis under fluoroscopy with a needle. (B, C) Minimally invasive decompression. (D) Incision for the release of the medial gastrocnemius. (E) Cutting the fascia of the proximal medial gastrocnemius through a small incision.
Statistical Analysis
The analytical cornerstone of our study was the application of the paired t test to scrutinize the efficacy of the surgical intervention over time. We used the IBM SPSS Statistics software (IBM Corp, Armonk, NY), a leading platform for statistical analysis, to perform the paired t test. This statistical approach was pivotal in comparing the preoperative scores of both the FFI and the VISA-A Questionnaire against the scores obtained at subsequent follow-up intervals: preoperative, 3 months, 6 months, and 1 year postsurgery. The decision to use the paired t test was informed by its proficiency in evaluating the mean differences within our paired observations, thereby allowing us to assess the significance of our surgical intervention in improving patient outcomes over the designated time points.
To compare the outcomes and recovery trajectories between different groups, we used the unpaired t test. 22 This statistical tool was applied to the FFI and VISA-A Questionnaire scores gathered at the preoperative and at the 3, 6, and 12-month postoperative follow-ups.
In our rigorous statistical framework, we established a threshold of P <.05 to denote statistical significance, and an MCID of 6.5 points was identified for the VISA-A Questionnaire. 13
No formal correction for multiple comparisons was applied. Group comparisons were predefined and clinically relevant. Given the exploratory nature of the study, P values are reported without adjustment but interpreted with caution.
Results
Our study demonstrated statistically significant improvements in both the FFI and VISA-A Questionnaire scores at 3, 6, and 12 months postoperatively compared with preoperative values, for both group A (patients with Achilles–plantar complex contracture) and group B (patients without Achilles–plantar complex contracture) (Table 1).
FFI and VISA-A Scores (All 224 Patients: Group A + Group B).

Abbreviations: FFI, Foot Function Index; Preop., preoperative; VISA-A, Victorian Institute of Sport Assessment–Achilles.
When examining the baseline characteristics between the 2 groups, preoperative scores for both FFI and VISA-A were found to be comparable. This similarity suggests that the initial conditions of the 2 patient groups were statistically indistinguishable, providing a balanced foundation for evaluating the effects of the surgical intervention.
Furthermore, the comparative analysis conducted between group A and group B at the follow-up intervals of 3, 6, and 12 months postsurgery revealed no statistically significant differences in the recovery trajectories or outcomes between the groups (Table 2).
FFI and VISA-A Scores: Group A vs Group B.
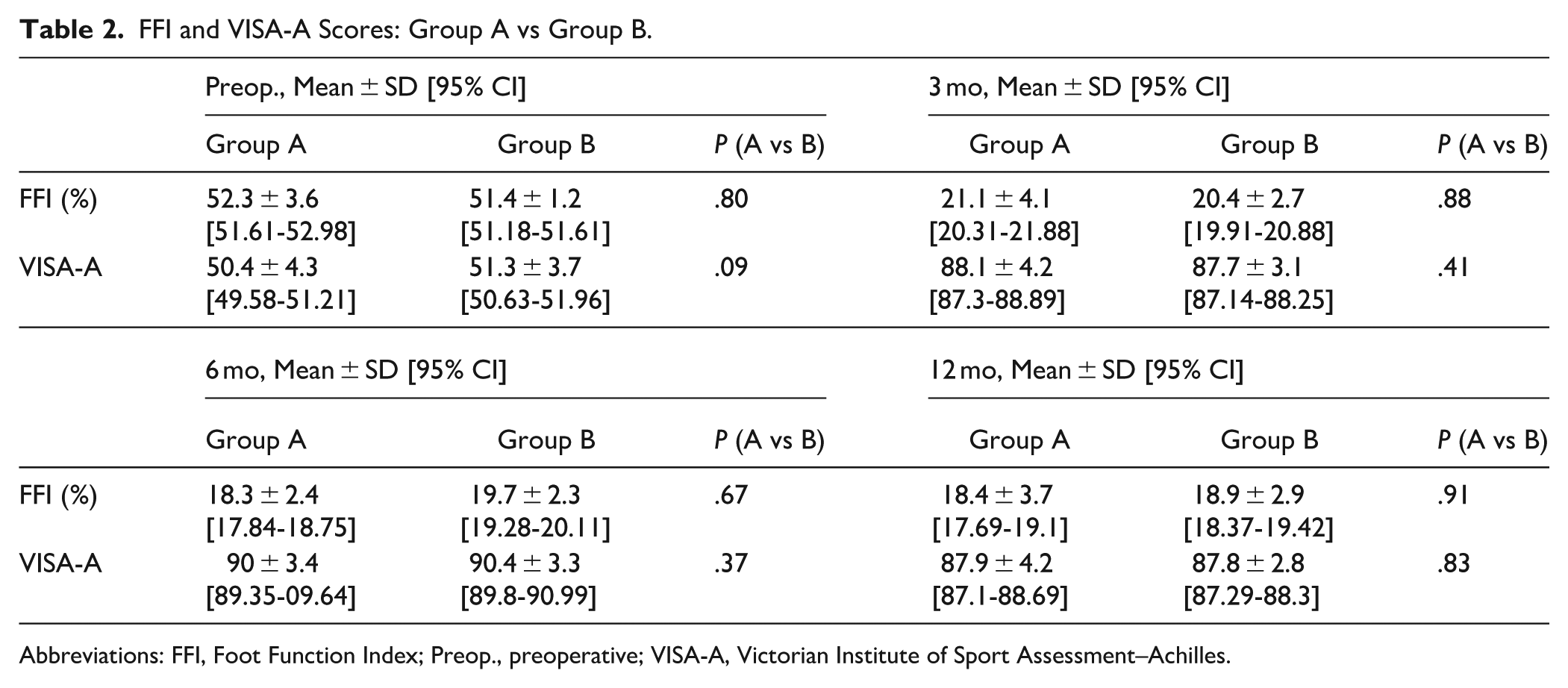
Abbreviations: FFI, Foot Function Index; Preop., preoperative; VISA-A, Victorian Institute of Sport Assessment–Achilles.
Both FFI and VISA-A Questionnaire scores showed statistically significant improvements at 3 months, 6 months, and 1 year postoperatively compared to preoperative values in both groups.
The retrospective analysis, comparing group A with group 1 (Table 3) and group B with group 2 (Table 4), revealed that preoperative values of FFI and VISA-A Questionnaire scores were also comparable across all 4 groups, indicating a similar baseline condition. However, at the follow-up intervals of 3, 6, and 12 months, groups A and B showed significantly better outcomes in terms of FFI and VISA-A Questionnaire scores compared with groups 1 and 2, respectively.
FFI and VISA-A Scores: Group A vs Group 1.

Abbreviations: FFI, Foot Function Index; Preop., preoperative; VISA-A, Victorian Institute of Sport Assessment–Achilles.
FFI and VISA-A Scores: Group B vs Group 2.
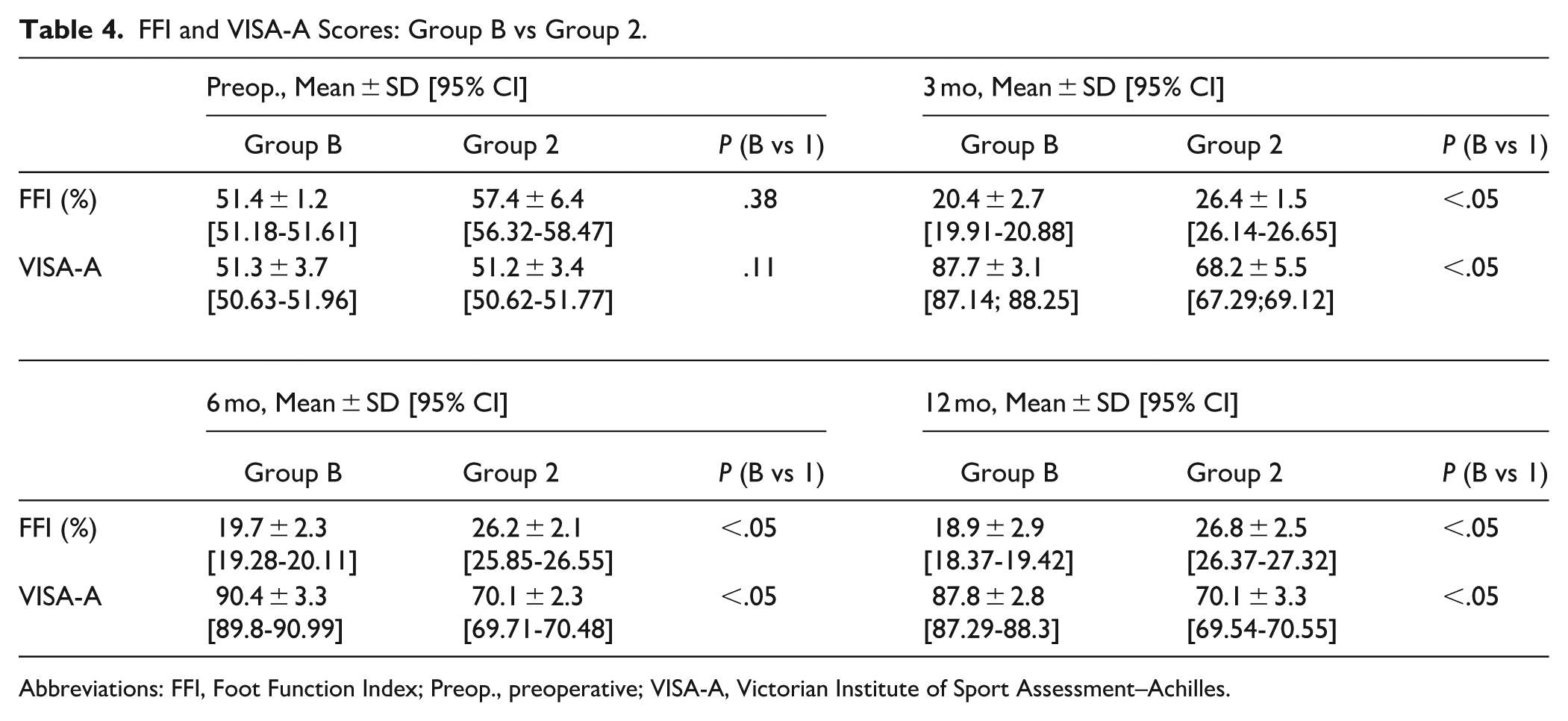
Abbreviations: FFI, Foot Function Index; Preop., preoperative; VISA-A, Victorian Institute of Sport Assessment–Achilles.
Regarding complications, the overall rate was low, with only 2% (5 patients) of cases affected. The reported complications included postoperative hematoma in the gastrocnemius region and superficial skin infections. Importantly, all cases were managed conservatively and resolved completely without the need for surgical revision.
Notably, no cases of overlengthening were observed, and there were no instances of sural nerve injury or wound healing delay. The percutaneous nature of the calcaneal resection and the minimal invasiveness of the PMGR procedure contribute to a favorable safety profile (Figure 2).

Radiographic features of Haglund syndrome (A) before treatment and (B) immediately after surgery.
Discussion
The observation of similarly low preoperative VISA-A Questionnaire scores in both groups, with and without Achilles–plantar complex contracture, may reflect a common degree of functional impairment in patients affected by Haglund syndrome. Although this does not necessarily imply a uniform structural involvement of the Achilles–plantar complex, it highlights the significant clinical burden of the condition across different patient profiles. The consistent postoperative improvement in VISA-A Questionnaire scores, regardless of preoperative contracture status, confirms the benefit of adding Achilles–plantar complex release to percutaneous calcaneal resection, even in patients without clinical contracture.
Moreover, the dual improvement in FFI and VISA-A reflects not only pain relief but also enhanced biomechanical performance and function.
This reinforces the idea that the combined surgical strategy targets both the source of impingement and the mechanical contributors to dysfunction. The results of our retrospective analysis demonstrate a clear superiority in postoperative outcomes for groups A and B, who underwent Achilles–plantar complex release, compared with groups 1 and 2.
The significant improvements observed in groups A and B may also suggest that the release of the Achilles–plantar complex not only reduces tension in this structure but could also alleviate pressure at the calcaneal level. This decompression may also help reduce retrocalcaneal friction and recurrence risk, contributing to improved comfort and faster return to activity.
In addition to the clinical outcomes, the adoption of a minimally invasive percutaneous technique for the resection of the calcaneal exostosis offers several procedural advantages, including reduced operative times, lower risk of complications, lower risk of infection, and a diminished recovery period.5,6,23
However, it is paramount to acknowledge that the success of this technique is heavily reliant on the surgeon’s proficiency and experience. The utilization of fluoroscopic guidance during the procedure is indispensable, ensuring the precision of the surgical intervention while minimizing the risks associated with this approach. This highlights the importance of a skilled surgical team and the appropriate use of technology in optimizing patient outcomes.
The comprehensive improvement seen in our study, particularly highlighted by the comparison between patients who underwent only percutaneous resection of the posterior-superior angle of the calcaneus and those in groups A and B who also underwent Achilles–plantar complex release, is well supported by both VISA-A and FFI scores. This emphasizes the multifaceted benefits of combining PMGR with a minimally invasive percutaneous approach to address both the bony and soft tissue components of Haglund syndrome. This surgical strategy not only targets the mechanical impingement but also facilitates the functional rehabilitation of the Achilles tendon, offering a holistic solution to the condition. By effectively addressing both the structural and functional aspects, our approach helps in optimizing recovery and enhancing the overall quality of life for patients. The integration of these surgical techniques appears to be superior in managing the complexities of Haglund syndrome, providing a robust framework for future therapeutic strategies.
Therefore, our findings advocate for the integration of soft tissue management with minimally invasive techniques as a standard in the surgical treatment of Haglund syndrome, underscoring the necessity of skilled surgical execution and the judicious use of fluoroscopic guidance to achieve the best outcomes.
Moreover, our study included patients without Achilles–plantar complex contracture (group B) and a retrospective comparison including groups 1 and 2, who underwent isolated resection of the calcaneal prominence with or without contracture. The outcomes across these subgroups consistently demonstrated better clinical results when PMGR was added to the calcaneal resection, irrespective of the presence or absence of contracture. These findings support the hypothesis that PMGR may be beneficial beyond its traditional indication, offering a biomechanical advantage even in patients with preserved dorsiflexion.
This study has some limitations. Primarily, the assessment of outcomes was based solely on clinical scoring systems (FFI and VISA-A), without the support of objective instrumental evaluations such as imaging studies or biomechanical analyses. Moreover, the retrospective component may be affected by incomplete or inconsistent data collection and lacks the standardized data monitoring present in the prospective phase. Additionally, differences in clinical practice or data availability over time may have introduced selection bias or confounding factors, potentially affecting comparability between groups. Although these clinical tools are widely validated, future research would benefit from integrating instrumental assessments to provide a more comprehensive evaluation of functional recovery and anatomical changes following surgery.
Conclusions
This study suggests that combining minimally invasive percutaneous calcaneal resection with Achilles–plantar complex release is safe, with low complication rates (2%) and significant functional improvements at 12-month follow-up. However, the use of historical rather than concurrent controls introduces significant bias that limits our ability to establish treatment superiority. While outcomes appeared better than historical controls treated with resection alone, this study design cannot determine whether the combined approach is truly superior due to potential confounding factors. These preliminary findings are hypothesis-generating and warrant validation in randomized controlled trials before the technique can be recommended as standard care. The study’s primary value lies in demonstrating safety and feasibility of the combined technique.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114251372601 – Supplemental material for Preliminary Anatomical and Surgical Assessment of Combined Percutaneous Resection and Proximal Medial Gastrocnemius Release vs Historical Controls in Treating Haglund syndrome: A Mixed Prospective-Retrospective Study
Supplemental material, sj-pdf-1-fao-10.1177_24730114251372601 for Preliminary Anatomical and Surgical Assessment of Combined Percutaneous Resection and Proximal Medial Gastrocnemius Release vs Historical Controls in Treating Haglund syndrome: A Mixed Prospective-Retrospective Study by Alessandro Cattolico, Fabrizio Sergio, Alessia Boemio, Ottorino Catani, Massimo Noviello, Ciro Ivan De Girolamo, Luigi Bagella, Mario Boccino, Angela Lucariello, Luigi Aurelio Nasto, Enrico Pola and Fabio Zanchini in Foot & Ankle Orthopaedics
Footnotes
Ethical Approval
Ethical approval for this study was obtained from the ethical committee of the University of Campania “Luigi Vanvitelli” (20200031574a) and all participants of the study signed a written informed consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Disclosure forms for all authors are available online.
Data Availability Statement
The data supporting the conclusion of this article will be made available by the authors, without undue reservation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
