Abstract
Background:
Primary implant stability is important for successful outcomes after uncemented total ankle replacement (TAR). However, the influence of patient-specific bone density on TAR performance is poorly understood, especially for implants that rely on press-fit for stable primary fixation. Our goal was to evaluate how bone density influences implant-bone interfacial micromotions in 3 press-fit tibial component designs by sampling from a TAR preoperative planning database using finite element analysis (FEA).
Methods:
FEA was conducted in 4 TAR patients with relatively low-density (n = 2, lowest 10% of a sample including 58 patients) and average-density (n = 2, midrange of sample) bone as assessed from deidentified patient CT scans. Three tibial implant designs were evaluated: a bone-sparing resurfacing implant, a cortex-sparing anterior approach monoblock stemmed implant, and a distal-reaming modular stemmed implant. Implants were inserted into tibia geometries obtained from the CT scans. Press-fit implantation was modeled first, followed by loadings from the stance phase of gait, and the associated micromotions were computed from the FEA output.
Results:
In general, patients with average-density bone had FEA predicted lower micromotions than patients with low-density bone. FEA suggests that implant fixation features had less influence on micromotions in patients with average-density bone, with peak micromotions ranging from 2 to 23 µm (3.1 ± 1.3 µm average micromotion). For patients with low-density bone, interfacial regions are predicted to experience micromotions exceeding the bony ingrowth threshold of 50 µm only for the resurfacing implant.
Conclusion:
We investigated the influence of bone density on implant-bone micromotions with varying primary fixation features using FEA. The model predicts that micromotions are less in average-density bone, regardless of implant fixation features. However, both stemmed devices showed lower micromotions in less-dense bone, albeit with the corresponding clinical trade-off of requiring more tibial bone removal.
Clinical Relevance:
The results presented here implicate the complementary role that local bone density plays in the primary fixation stability of uncemented TAR.
Keywords
Clinical Relevance
We investigated the influence of bone density on implant-bone micromotions with varying primary fixation features. Micromotions were lower in average-density bone, regardless of fixation features. However, stemmed devices showed lower micromotions in less-dense bone at the expense of more tibial bone removal. Our results underscore the important role that local bone density plays in the primary fixation stability of uncemented TAR, a key factor in long-term implant survivorship.
Introduction
Primary implant stability following surgical implantation is important for successful outcomes after uncemented total ankle replacement (TAR). Uncemented TAR fixation relies on adequate osseointegration for long-term survival of the implant. Unfavorable primary stability may lead to inadequate secondary osseointegration at the implant-bone interface, resulting in inferior interfacial mechanics longer-term and subsequent failure of the implant due to aseptic loosening.5,7,23 The relatively low longer-term survivorship of TAR compared with cementless total knee arthroplasty1,18,19,22 suggests a need for better understanding of the interplay between interfacial mechanics and implant stability in TAR.
Fixation features play a key role in initial implant stability.17,24 Commercial TAR tibial components are available with a wide range of fixation features designed to aid primary fixation stability. These fixation features include geometric designs with varying shapes and sizes (eg, pegs or stem), surface coatings (eg, plasma-sprayed or 3D-printed), and implantation method (eg, cemented or interference press-fit). Each fixation feature is designed to provide stability during functional load transfer, while taking the density of surrounding bone into consideration. Consequently, the interaction between fixation features and surrounding bone density should be appropriately accounted for when evaluating the stability performance of TAR implants.
Bone density, along with TAR implant design, is a key contributing factor to primary implant stability. 3 Resurfacing implants are designed to achieve stable fixation while maximally preserving bone, and they depend on interference press fit in sufficiently dense local bone to provide initial stability. Stemmed implants necessitate more bone resection, but their correspondingly larger fixation features present a greater interfacial area that is likely to reduce the dependence upon local bone density for primary fixation stability. Unfortunately, there is little guidance available regarding objective decision making in the selection of a given implant design for a patient based on their apparent bone density. The stability benefits of interference press-fit may be diminished in low-density bone particularly for implants with lower-profile fixation features, thereby influencing implant-bone interfacial mechanics.
Finite element analysis (FEA) is commonly used to evaluate TAR implant-bone mechanics because of its efficiency in simulating the complex interactions between implant geometry, surface behavior, bone density, and loading conditions. FEA studies have shown that tibial component geometry, surface characteristics (approximated using global frictional properties), and tibial bone density all influence the performance of TAR implants.14,15,20,24,27,28 However, some of these findings were based on simulation of purely compressive loads,14,15,24,27 and parametric variation of tibial bone density acquired from healthy volunteers. 15 More importantly, these studies did not simulate press-fit implantation, which is contrary to their typical use, thereby limiting clinical applicability of the findings.
We have recently shown that the effects of tibial component geometry and surface characteristics on implant-bone interfacial mechanics are less important when interference press fit is simulated. 13 However, the influence of bone density from patients indicated for TAR on implant-bone micromotions is poorly understood, especially for implants that rely on press fit for stable primary fixation. Therefore, the goal of this study was to computationally evaluate how patient-specific bone density influences implant-bone interfacial mechanics in 3 TAR tibial component implant fixation designs (resurfacing, monoblock stemmed, and modular stemmed) under the same implantation conditions when press fit and subsequent gait loading is simulated.
Methods
To evaluate the influence of patient-specific bone density, 4 patients with end-stage ankle arthritis were selected from a TAR surgical planning database that included anonymized clinical CT scans from 58 patients. The patient CT scans were selected after screening to exclude hardware artifacts and for consistency of acquisition parameters (120 kVp, 100-150 mA, in-plane pixel size 0.484-0.625 mm, slice thickness 1-1.25 mm). Based on average Hounsfield unit (HU) values measured at the distal tibia implantation region from CT, the selected patients corresponded to roughly 50% (average density; patients 1 and 2) and 10% (low density; patients 3 and 4) of the range of HU values (Figure 1). The influence of bone density was evaluated for 3 tibial component implant fixation features; a bone-sparing resurfacing implant with pegs, a cortex-sparing anterior approach monoblock stemmed implant, and a distal-reaming modular stemmed implant (Figure 2, top row). The implant fixation features were chosen to generically represent a range of available tibial implant fixation feature sizes. Patients were selected also for having similar tibia sizes (based on the same baseplate size surgically planned for these TAR cases) such that the same baseplate tray size and design could be used for all implant representations to minimize confounding effects.

Histogram showing the distribution of averaged patient-specific HU values measured at the distal tibia region, 15 mm from the resection height, for 58 patients in a TAR surgical planning database. Overlaid symbols indicate individual patient values. Patients for this study were selected from 10% (low) and 50% (average) of the distribution, and their values are indicated by the red square symbols. HU, Hounsfield unit; TAR, total ankle replacement.
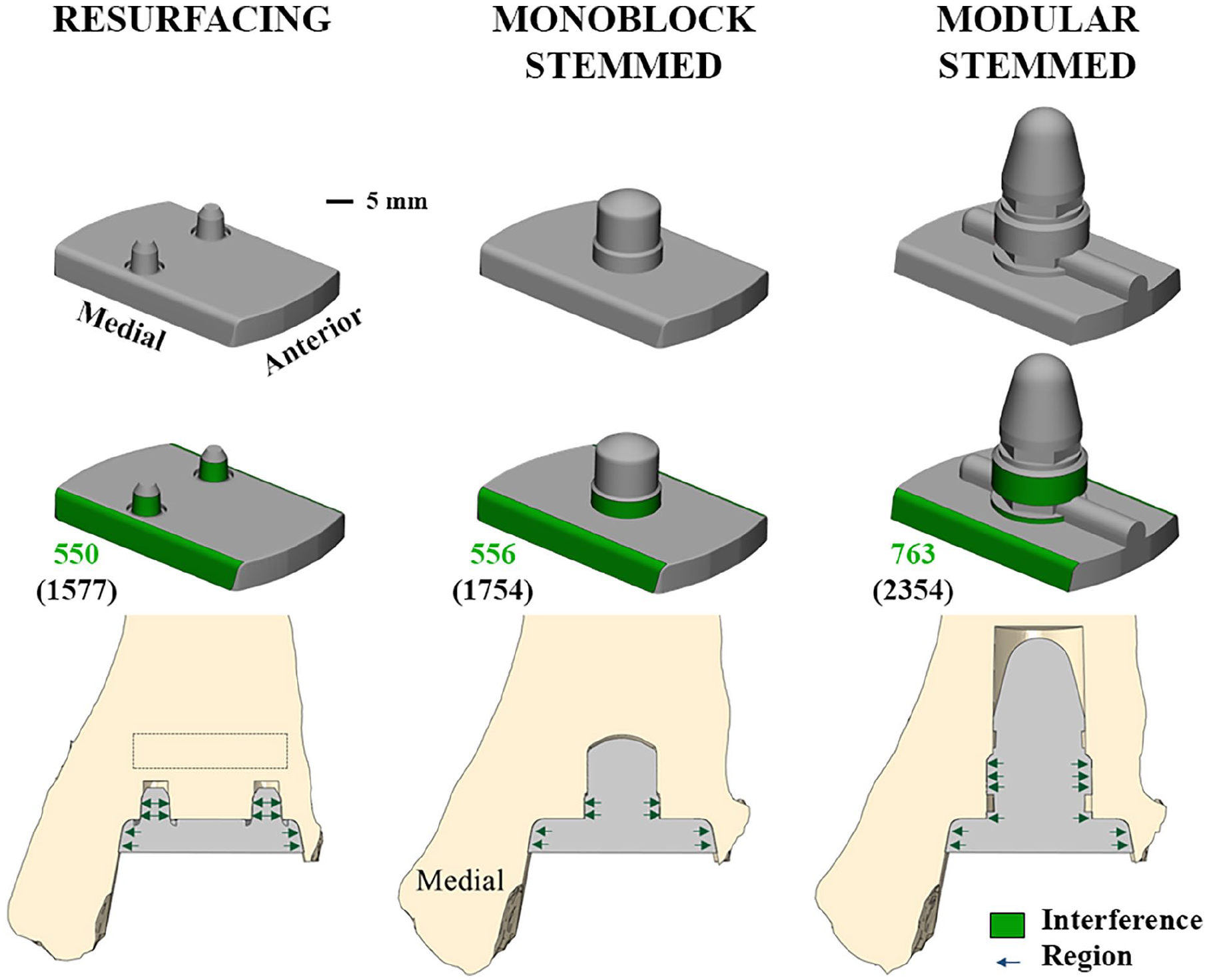
Generalized representations of 3 TAR tibial component fixation features (top row), showing fixation and sidewall regions where interference press-fit was modeled (green in middle row), and implanted position shown through the anterior-distal tibia cross section (bottom row). The press-fit areas (green values) and implant-bone interfacial areas (values in parentheses) in square millimeters are also indicated. Rectangle shows the distal tibia region of HU evaluation. HU, Hounsfield unit; TAR, total ankle replacement.
FEA was performed according to previously described methodology.4,13,20 Patient tibial geometries and bone densities were acquired from the clinical CT scan data. Tibial geometries were segmented using Mimics (Materialise), with only the distal 14 cm of the tibia modeled for computational efficiency. 20
The resurfacing implant was virtually inserted into the distal tibia according to standard clinical practice. Implants with a stemmed fixation feature are typically inserted more proximally to interface with cortical bone, but to remove confounding effects of implant placement, 14 both stemmed implants in this study were aligned to match the implanted location and orientation of the resurfacing baseplate tray (Figure 2, middle row). However, the amount of resected bone for implant insertion was different for all implants and followed manufacturer guidelines (Figure 2, bottom row). To model press-fit in all implants, a 100-μm interference was generated along the implant fixation features and sidewalls mimicking clinical practice (Figure 2, middle and bottom rows). We only modeled the TAR tibial component, because our goal was to evaluate implant-bone interfacial mechanics in the tibia, as tibial components are observed to undergo earlier revision than talar components, clinically. 8
A total of 12 FE models were analyzed (4 patients × 3 tibial component implant fixation features). All geometries were meshed using second-order tetrahedral elements in 3-matic (Materialise). Characteristic element edge lengths (2 mm for implant; varying 2 mm distal to 3 mm proximal for bone) were determined from a convergence study. 13 A more refined mesh size was used for the implant-bone interface (0.5 mm for implant; 1 mm for bone). Titanium alloy linear elastic material properties were assigned to implant geometries, while each bone element was assigned distinct isotropic, 26 nonlinear (elastic-plastic),2,6,16 CT HU-specific 21 material properties (Figure 3). Incorporating the nonlinear material behavior of bone on an element-by-element basis allowed for patient-specific modeling of permanent bone deformation associated with interference press-fit, as well as loading-associated deformations during the analysis. Implant-bone interfacial contact was modeled with a coefficient of friction of 0.6.13,20

Distribution of tibia elastic modulus at the bone-resurfacing implant interface for each patient reflecting the variation in local bone density across patients. Patients 1 and 2 had substantially higher density bone / greater modulus than patients 3 and 4.
To mimic surgical technique, press fit was simulated by a static displacement of the implant into the tibia along the primary axis of the implant fixation features while the proximal end of the tibia was held fixed.10,13 To facilitate nonlinear solution convergence when modeling press fit with the larger interference regions, the stemmed fixation features required simulation of press fit in 2 stages; initial static displacement to model press fit along the sidewalls, followed by a contact algorithm-based modeling of press fit around the stems, which generates stresses as interference is resolved by adjustment of the bone elements (shrink-fit algorithm in Abaqus). This was deemed appropriate as our goal was to account for the interfacial influence of bone compaction during loading rather than local mechanics related to the actual process of press-fit implantation. The implant was allowed to recoil after press-fit. Then, multiaxial forces and moments from the stance phase of gait 25 were applied to the distal implant surface.13,20 The same load magnitudes (not patient-scaled) were applied to all patient models to eliminate confounding effects.
All analyses were performed in Abaqus 2018 (Dassault Systemes Simulia Corp). Implant stability was evaluated by comparing interfacial micromotions, because small micromotions (below 20-50 µm) are known to facilitate bony ingrowth, whereas excessive micromotions (above 150 µm) result in deleterious fibrous tissue ingrowth.11,12 Micromotion was computed from FEA output as the difference in displacement between implant-bone closest nodal pairs.13,20 Peak and average implant-bone micromotions throughout stance were calculated for general comparison between patients across the implant fixation features. Micromotion patterns plotted over the implant-bone interface were qualitatively compared at 73% stance where the overall peak micromotion occurred.
To better capture the complex micromotion patterns at the implant-bone interface and to quantify the area of the interface experiencing excessive micromotions, we also compared the distributions of micromotions over implant-bone interfacial regions (expressed as % of total interfacial area). Implant interfacial regions were binned according to the magnitude of micromotion and plotted as a cumulative sum of the entire gait cycle.
Results
In general, micromotions were lower in patients with average-density bone (patients 1 and 2) than in patients with low-density bone (patients 3 and 4; Figure 4). Implant fixation features had less of an influence on micromotions in patients with average-density bone, with minimum and maximum peak micromotions throughout stance ranging from 2 to 16 µm for the modular stemmed implant, 3 to 22 µm for the monoblock stemmed implant, and 2 to 23 µm for the resurfacing implant. This implies that similar implant stability was achieved regardless of the implant fixation feature, despite differences in the required amount of resected bone volume (7.3% ± 1.0% [mean ± SD throughout] more with monoblock stemmed implant, and 40.8% ± 4.7% more with modular stemmed implant, both compared to the resurfacing implant). Also, similar trend in micromotions was observed throughout stance in patients with average-density bone, with peaks occurring between 70% and 80% stance (Figure 4).

Peak and average micromotions throughout stance for all 4 patients for each implant fixation feature.
The influence of implant fixation features was more apparent in patients with low-density bone (Figure 4). For these patients, micromotions were lowest with the modular stemmed implant (29 and 32 µm overall peak values for patients 3 and 4, respectively), compared to the monoblock stemmed implant (47 and 50 µm overall peak values) and the resurfacing implant (108 and 67 µm overall peak values). Average micromotions were less than 50 µm in all cases (Figure 4). Micromotion patterns were consistent between patients (Figure 5) and corresponded to the dominant loads acting during the specific instance of stance.

Micromotion patterns at 73% stance for all 4 patients for each implant fixation feature.
The amount of implant-bone interfacial regions experiencing high micromotions progressively decreased with increasing size of the implant fixation feature (Figure 6). In patients with average-density bone, no implant interfacial regions were subject to micromotions greater than 50 µm across all implant fixation features. In patients with low-density bone, interfacial regions of only the resurfacing implant (5% and 2% for patients 3 and 4, respectively) were subject to micromotions greater than 50 µm over the entire gait cycle.

Micromotion frequency distributions for all patients and implant fixation features over the entire stance phase.
Discussion
The influence of bone density on TAR performance is poorly understood, especially for implants that rely on press fit for stable primary fixation. Using FEA, we investigated the influence of bone density on implant-bone micromotions during physiologic loading with varying implant fixation features and clinically relevant interference press-fit implantation. Micromotions were lower with bone compaction in average-density bone, regardless of the implant fixation feature (resurfacing, monoblock stemmed, modular stemmed). However, both stemmed devices demonstrated lower micromotions in less-dense bone, albeit with the clinical trade-off of more tibial bone removal, and in the case of the modular stem, potential disruption to the calcaneus and talus associated with the surgical approach.
Implant fixation features were found to minimally influence implant-bone interfacial micromotions in patients with average-density bone. To date, most FEA studies investigating stability performance of TAR implants have only simulated tibial component implantation with line-to-line interfacial contact between the implant and bone. These studies found that keel fixation features tended to demonstrate lower micromotions (25-100 µm peak micromotions) compared with peg fixation features (40-235 µm peak micromotions), whereas micromotions were found to be largest for stemmed fixation features (60-1125 µm peak micromotions).15,20,24,27,28 In the clinic, uncemented TAR relies on press-fit implantation for stable primary fixation, where stresses generated from elastic recoil after press-fit implantation contribute to provide implant stability during subsequent loading. From prior 13 and current findings, the stability produced by press-fit implantation in average bone density appears to be sufficient to restrict micromotions independent from the implant fixation features. This is clinically meaningful because stable primary fixation could potentially be achieved with less bone resection, preserving bone in case of later complications or future revision surgery.
Implant fixation features appeared to play more of a role in patients with low-density bone. One prior FEA study reported increasing micromotions with decreasing bone density when evaluating keel and stem fixation features under uniaxial loading. 15 However, bone density was parametrically varied in a healthy tibia geometry, and bone compaction was not simulated. Consequently, the variations in micromotions were proportional to the factor of density change and showed a somewhat linear relationship corresponding to the linear material model. 15 Interestingly, the larger-profile stemmed fixation feature has been shown to perform poorly in simulations without press-fit implantation as mentioned above. However, with press-fit implantation, the monoblock and modular stemmed fixation features contributed to implant stability in relation to feature size in the patients with low-density bone. With the impact of elastic recoil potentially diminishing with bone compaction in low-density bone, implant stability appears to be supplemented by implant fixation feature size (Figure 2). This effect may be more pronounced when evaluating TAR performance in patients with osteoporotic bone, which is a topic for future investigation.
Patients with low-density bone had higher micromotions. Micromotion trends also showed more variability (Figure 4; patient 4), likely due to the interaction between local bone density around the fixation features and late stance kinetics, which are dominated by high internal rotation and inversion moments and low axial compressive force, 25 thereby influencing the ability to resist subsequent eccentric micromotions. Similarly, the high anteromedial micromotion patterns observed during the instance of overall peak micromotion (Figure 5) corresponded to the dominant dorsiflexion and internal rotation moments that occur at 73% stance. 25 However, average micromotions were all within the reported 50-µm threshold for bony ingrowth.11,12 These results are of course specific to the patients in our study, who were selected from the available cohort based on distal tibia HU measurements. Peak micromotions were, however, above the bony ingrowth threshold for the resurfacing fixation feature. What is presently unknown and requires further investigation is whether a single peak micromotion value or interfacial regions subject to micromotions above the threshold over time contribute to failed osseointegration. Furthermore, the relationship between the amount of interfacial surface area available for bony ingrowth and successful osseointegration is also unclear.
This is important because a recent clinical study found that tibial components with larger-profile stemmed fixation features lowered tibial failure likelihood by 95%, although patient bone density variation was not considered. 9 The resurfacing and stemmed fixation features performed similarly in patients with average bone density in our study, but the findings of the recent clinical study suggest micromotions within the osseointegration threshold may be but one mechanical factor among others such as available bony ingrowth surface, that play a role in successful TAR outcomes. A prospective evaluation of the interaction between mechanical, clinical, and patient factors is needed to improve our understanding of implant stability performance. Nevertheless, our data may be able to provide relevant feedback to surgeons on the selection choice of implant resurfacing vs stemmed fixation feature for the studied tray geometry based on patient-specific CT HU assessment.
Other fixation features, such as surface coating, are also implemented to aid primary fixation stability of TAR tibial components. To reduce model complexity, microstructural surface characteristics are typically approximated using global frictional properties.13,15,20 Varying coefficients of friction from 0.1 to 0.5 has been shown to result in ~15-µm decrease in average micromotions when line-to-line implantation was simulated. 15 With press-fit implantation, however, we previously showed the influence of surface characteristics (titanium plasma spray vs 3D-printed porous coating), as approximated by friction coefficient, to be minimal, with ~1 µm decrease in average micromotions (resurfacing fixation feature) with increasing friction coefficient. 13 Hence, we did not evaluate the influence of varying friction coefficient in this study. We also showed that changing interference press-fit magnitudes from 50 to 200 µm only marginally affected micromotions (~1 µm change), 13 so we chose the 100 µm intermediate interference magnitude for this study.
Some key limitations of our study deserve mention. Data were evaluated from a relatively low number of patients. However, the patients analyzed represent a clinical population indicated for TAR due to end-state ankle arthritis, and a subset was purposefully selected to span a portion of the bone density variation normally observed in the clinic. Also, with simulated interference press-fit implantation, we previously found minimal variation in micromotions throughout stance in patients with HU values in the 400-HU range. 4 Given that the highest frequency of HU values fell in the 400-HU range, we focused our current comparisons to patients from this group and from the lowest end of the spectrum to capture the worst-case influence. While we would expect to see marginal differences in micromotions evaluating patients from the higher range of HU values compared to the 400-HU range, establishing a trend in micromotions with bone density variation was beyond the current scope and is a potential topic for future investigation.
Further limitations relate to inability to capture all the vagaries of local interference fit from the clinical setting. The depth and geometry of resection plays a role in bone support for TAR, 3 and implant placement is design dependent. For example, stemmed fixation features are designed to interface with cortical bone with a greater depth of resection of the tibia. However, to eliminate confounding effects, we aligned the tray of the stemmed fixation feature implants to the resection height of the resurfacing implant tray. Consequently, our estimates of resected bone volume were conservative, and more bone would likely be resected during surgery. Also, the relative change in micromotions has been shown to decrease with progressively more proximal placement of stemmed implants with line-to-line implantation. 14 Therefore, although the stemmed implant placement in our study did not ideally imitate clinical practice, interpretation of our results related to the relative influence of bone density remains valid, but the dependence of absolute micromotion values of stemmed fixation feature implants on resection parameters with press-fit implantation requires further investigation. Another limitation is that it is not possible with the tools currently used in surgery to perform TAR with a pure 100-µm press fit on the baseplate of the tibial component. More likely, press fit is lost as bone is removed to ensure that the baseplate is fully apposed against bone.
Lastly, our FEA methodology has not been directly validated with experimental data, and a direct comparison to literature was not possible because of the scarcity of TAR FEA simulating press-fit implantation. However, to verify our modeling approach, we previously performed TAR FEA simulations with line-to-line implantation and showed that our computed micromotions matched well with the range of values reported in the literature (~60-200 μm13 vs ~50-1100 μm peak values 20 ).
In conclusion, we investigated the influence of bone density on implant-bone micromotions with varying primary fixation features. Our results suggest that local bone density plays an important complementary role in the primary fixation stability of TAR tibial components. However, the correlation between primary fixation stability of TAR tibial components in varying density bone and long-term clinical outcome requires further clinical evidence. Better understanding how local bone density, implant fixation features, and implantation method act together to provide implant stability in TAR should help improve implant performance.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114241308577 – Supplemental material for Finite Element Analysis of the Influence that Bone Density and Implant Fixation Features have on Total Ankle Tibial Component Stability
Supplemental material, sj-pdf-1-fao-10.1177_24730114241308577 for Finite Element Analysis of the Influence that Bone Density and Implant Fixation Features have on Total Ankle Tibial Component Stability by Joshua E. Johnson, Gabriel Clarke, Cesar de Cesar Netto, Bopha Chrea and Donald D. Anderson in Foot & Ankle Orthopaedics
Footnotes
Acknowledgements
We thank Donald Hume and Mathew Anderle from Paragon 28 for their scientific feedback.
Ethical Approval
Ethical approval was not sought for the present study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Cesar de Cesar Netto, MD, PhD, reports general disclosures: unrelated research funding from Paragon 28; royalty payments from Artelon, Medartis, Nextremity Solutions, Paragon 28, and Zimmer Biomet; consulting fees from Exactech, Medartis, Nextremity Solutions, Paragon 28, Ossio, Stryker, and Zimmer Biomet; payment for lectures and presentations from Medartis, Nextremity Solutions, Paragon 28, and Zimmer Biomet; and meeting travel support from CurveBeam, Medartis, Paragon 28, and Zimmer Biomet. Bopha Chrea, MD, reports general disclosures of consulting fees from Novastep. Disclosure forms for all authors are available online.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was provided by a grant from Paragon 28 to the University of Iowa. Paragon 28 also provided deidentified patient CT scans and implant geometries.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
