Abstract
Background:
Particulated autograft cartilage implantation is a surgical technique that has been previously described for the repair of osteochondral lesions of the talus (OLT). It uses cartilage fragments harvested from the OLT that are minced into 1-2-mm3 fragments and then immediately reimplanted back into the chondral defect and sealed with fibrin glue during a single-stage surgery. The purpose of this study was to characterize the suitability of these minced cartilage fragments as immediate autograft for the treatment of OLTs.
Methods:
Thirty-one patients undergoing primary arthroscopic surgery for their OLT consented to have their loose or damaged cartilage fragments removed and analyzed in the laboratory. Harvested specimens were minced into 1- to 2-mm3 fragments and cell count, cell density, and cell viability were determined. In addition, physical characteristics of the OLT lesion were recorded intraoperatively and analyzed including size, location, Outerbridge chondromalacia grade of the surrounding cartilage, density of underlying bone, and whether the surgeon thought the OLT was primarily hyaline or fibrocartilage.
Results:
An average of 419 000 cells was able to be obtained from the harvested OLT fragments. The cells were 71.2% viable after mincing. Specimens from younger patients and from lesions with worse chondromalacia adjacent to the OLT had significantly higher cell numbers. Those from lateral lesions and with worse neighboring chondromalacia had a significantly higher cell density. None of the remaining physical OLT characteristics studied seemed to significantly affect cell number or viability.
Conclusion:
A large number of viable cells are available for immediate autografting by removing the loose or damaged cartilage from an OLT and mincing it into 1- to 2-mm3 fragments. These can be reimplanted into the chondral defect in a single-stage surgery. Future clinical studies are needed to determine if the addition of these live autologous cells either alone or in conjunction with other techniques significantly improves the quality of the repair tissue and clinical outcomes.
Level of Evidence:
Level IV, case series.
Introduction
Multiple techniques have been described to biologically augment the surgical repair of osteochondral lesions of the talus (OLT). These include procedures where viable chondrocytes are implanted into the site of the cartilage defect. One of these uses particulated juvenile allograft cartilage to deliver live differentiated chondrocytes to the repair site; however, this procedure is costly and the tissue has a very short shelf life.1,5,6,8,13,14,16,21,22,24,25 Another strategy is matrix-induced autologous chondrocyte implantation (MACI) that uses autogenous chondrocytes harvested arthroscopically in a first-stage procedure. The cells are amplified in vitro in a lab and then reimplanted into the patient’s OLT within a collagen matrix scaffold during a second-stage surgery. The disadvantages of this technique include high cost and the need for 2 separate surgical procedures.4,9,10,17,19 MACI is also not currently FDA approved for use in the ankle joint.
Lu et al 20 described the concept of using minced autologous cartilage tissue fragments taken from the injured knee as an immediate source of chondrocytes to augment repair of chondral defects during a single-stage procedure. A recent clinical study applying this same strategy to osteochondral lesions in the ankle found significant improvements in patient-reported outcomes, Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) scores, as well as International Cartilage Repair Society (ICRS) scores on second-look arthroscopy at minimum 1-year follow-up. 26
The purpose of this study was to determine if minced cartilage harvested from talar osteochondral lesions contains live chondrocytes and the appropriate extracellular matrix that could allow it to function as a single-stage augmentation for OLT repairs.
Methods
Institutional review board approval was obtained prior to initiation of this study. Thirty-one patients aged 18 to 70 years undergoing arthroscopic debridement and microfracture of OLTs at a single institution between 2022 and 2023 consented to being included in this study. Data collection and tissue sampling were performed prospectively but in a nonrandomized and noncontinuous manner. Inclusion criteria were patients with a preoperative MRI demonstrating an OLT who had failed conservative treatment. These patients were undergoing primary surgery for their OLT, which could be combined with other procedures as well. The most common additional procedures included 7 Brostrom procedures, 6 peroneal tendon debridements/repairs, 3 tibial cheilectomies, and 3 flatfoot reconstructions. Exclusion criteria were patients outside of the above stated age range, those with significant tibiotalar arthritis identified on weightbearing plain radiographs, and revision OLT procedures. Five fellowship-trained orthopaedic surgeons participated in the study. At the time of ankle arthroscopy, the OLT was identified and the damaged cartilage was freed from the underlying bone using curettes. The cartilage from the OLT was then collected either by using an arthroscopic shaver with a GraftNet autologous tissue collector (Arthrex; Naples, FL) attached in-line or by manual collection with an arthroscopic grasper. The choice of how the OLT was harvested was left to the discretion of the surgeon. No attempt was made to randomize GraftNet vs arthroscopic grasper or to try to end up with an equal number of patients in each group because comparing these 2 techniques was not the main aim of this study. The primary surgeon identified the OLT as either hyaline cartilage or fibrocartilage based on visual appearance and probing. The size of the OLT was then measured in 2 perpendicular planes using an arthroscopic probe ruler. The location of the lesion was recorded as well as the Outerbridge chondromalacia grade of the surrounding cartilage. Finally the surgeon characterized the bone underlying the OLT as hard, soft, or cystic during bone marrow stimulation using a microfracture awl.
The surgically removed cartilage samples were placed in saline or kept in the GraftNet device aseptically and processed immediately for chondrocyte isolation and histology. After being weighed, 7 of the samples that were collected by a grasper were minced into 1- to 2-mm3 pieces with an No. 11 blade scalpel. The remaining 24 specimens that were collected using an arthroscopic shaver and GraftNet were already in 1- to 2-mm3 fragments and did not require further mincing. All samples were digested with 1.2% collagenase (Thermo Fisher Scientific Inc, Waltham, MA) in Dulbecco’s modified Eagle medium (DMEM; Thermo Fisher Scientific) on a shaker, which was placed in an oven set to 37°C for 20-24 hours. The tissue lysate was filtered through a 70-µm cell-strainer, and centrifuged twice at 300g for 5 minutes. The resulting cell pellet was resuspended in phosphate-buffered saline (PBS). A sample of the cell solution (20 µL) was mixed with an equal amount of Trypan Blue, which stains the nuclei of dead cells blue. The stained samples were loaded onto chambered slides (20 µL/chamber). An automated cell counter (Quadcount, Accuris Instruments, Edison, NJ) was used to read the slides for total cell number and viability. The final cell number and viability of each cartilage sample was the average of duplicate measurements.
Two or 3 small cartilage fragments were taken from each cartilage sample and fixed with 4% paraformaldehyde. After being embedded within Optimal Cutting Temperature compound (OCT; Sakura Finetek USA, Inc, Torrance, CA), the cartilage fragments were cryosectioned at a thickness of 5 µm. The tissue sections were stained with Safranin O/Fast Green for histologic structure and aggrecan in the extracellular matrix. After staining with Weigert iron hematoxylin working solution (Sigma-Aldrich, St Louis, MO) for 10 minutes, the slides were subsequently stained in 0.05% Fast Green FCF solution (Sigma-Aldrich) and 0.1% Safranin O solution (Sigma-Aldrich) for 5 minutes, respectively.
Immunohistochemistry for collagen types I and II was performed on separate tissue sections. After heat-induced antigen retrieval in Tris-EDTA buffer (10 mM Tris base, 1 mM ethylenediaminetetraacetic acid [EDTA], 0.05% Tween 20, pH 9.0), the randomly selected tissue sections were blocked with 3% hydrogen peroxide and normal goat serum (1:50 dilution) sequentially. The primary antibody of collagen type I (rabbit anti-human; Abcam, Cambridge, MA) was applied onto tissue sections at 1:200 dilution and incubated in a moisture chamber at 4°C overnight. After washing with Tris-buffered saline, the slides were treated with a secondary biotinylated goat anti-rabbit antibody (1:200 dilution) at room temperature for 1 hour, followed with ABC reagents (VECTASTAIN Elite ABC system, Vector Laboratories, Burlingame, CA). Peroxidase substrate 3,3-diaminobenzidine (DAB) was used for chromogenic detection of collagen type I. Cell nuclei were counterstained with hematoxylin. The primary antibody of collagen type II (mouse anti-human 2B1.5, Thermo Fisher Scientific, 1: 200 dilution) was used after the slides were treated with 2% bovine testicular hyaluronidase (Sigma-Aldrich) and normal goat serum (1:50). A secondary antibody (goat anti-mouse, 1:200 dilution) conjugated with Alexa Fluor Plus (Thermo Fisher Scientific) was used for detecting collagen type II. The slides were mounted with VECTASHIELD (Vector Laboratories) with 4′,6-diamidino-2-phenylindole (DAPI) for nuclear staining. For antibody specificity and negative control, the primary antibody was replaced with either IgG of the antibody-host animals or PBS during the staining procedure.
Images of Safranin O/Fast Green staining and immunohistochemistry of collagen types I and II were taken under a light or fluorescent microscope, respectively. The staining intensity of types I and II collagen, and aggrecan was assigned a grade 0 for no staining, grade 2 for strong staining, and grade 1 for intensity between no staining and strong staining. A grade for staining area was also assigned for types I and II collagen, and aggrecan (0: <5% of the section; 1: <50% of the section; and 2: >50% of the section). OLT specimens with only type II collagen staining and no type I collagen were classified as hyaline cartilage. Specimens with both type I and type II collagen present were considered fibrocartilage. OLTs with only type I collagen staining and no type II collagen were categorized as fibrous tissue.
A PhD statistician assisted with data analysis. Descriptive statistics for characteristics of patients and intraoperative measurements were presented as means and SDs, and frequencies for categorical variables. To assess the association between these factors and each outcome, a nonparametric Kruskal-Wallis test was used for categorical variables and Pearson correlation was used for continuous variables. A variable with P value ≤.05 indicated a significant association with outcomes. All analysis was performed using SAS, version 9.4 (SAS Institute Inc, Cary, NC, USA).
Results
There was an average of 41.9 × 104 cells present in the cartilage harvested from each OLT (range, 0.4 × 104 to 189.0 × 104 cells). The mean weight of the OLT samples was 0.134 g (range, 0.016-0.578 g). Cell density was calculated by dividing the number of cells by the weight of the specimen. The average cell density was 3.41 × 106 cells per gram of tissue. The average size of the OLTs was 74 mm2 (range, 6-440 mm2).
On average, 71.2% (range, 30.3%-93.7%) of the cells in each OLT sample were still viable after being mechanically minced into 1- to 2-mm3 fragments. Of the 31 specimens, 24 were harvested using the arthroscopic shaver with GraftNet and 7 were removed with a grasper and minced using a scalpel. Because of the small number of specimens that were minced by scalpel, no significant differences were able to be found between the 2 groups (Table 1).
Comparison of Cell Viability Between Cartilage Samples Collected With GraftNet vs Samples Removed With Grasper and Minced With Scalpel.

Nine of the OLTs were located on the lateral dome of the talus, 19 were located medially and three were central lesions. Lateral OLTs had significantly higher cell density than medial lesions (5.8 × 106 ± 2.9 × 106 cells/g vs 2.5 × 106 ± 2.6 × 106 cells/g, P = 0.02) (Table 2).
Association Between OLT Location and Cell Number, Cell Density, and Cell Viability.

Abbreviation: OLT, osteochondral lesions of the talus.
Eighteen OLT samples (58%) were hyaline cartilage, 11 were fibrocartilage (35%), and 2 were fibrous tissue (6%) based on type I/type II collagen immunohistochemical staining. Aggrecan staining showed 12 samples with no loss of aggrecan, 15 samples with partial loss, and 4 with complete loss of aggrecan within the extracellular matrix relative to normal cartilage standards. Normal cartilage was available from our research laboratory’s library of cartilage fragments collected from other patients with acute intra-articular fractures.
Further analysis showed that OLT samples that were observed to have surrounding cartilage with a worse Outerbridge chondromalacia grade had a significantly higher number of total cells as well as higher cell density (P = .008 and .009, respectively) (Figure 1 and Table 3). This was likely due to either early arthritis where cartilage injury can cause an increase in chondrocyte proliferation as the body tries to repair the damage, or due to the presence of fibrocartilage where there are an increased number of fibroblasts. Finally, younger patients had a significantly higher number of cells harvested than older patients (P = .04). No other differences were found in cell number or cell density with respect to sex, dimension of the lesion, whether the tissue was hyaline or fibrocartilage, or bone quality under the OLT. Cell viability did not appear to be statistically associated with any of these variables (Table 4).
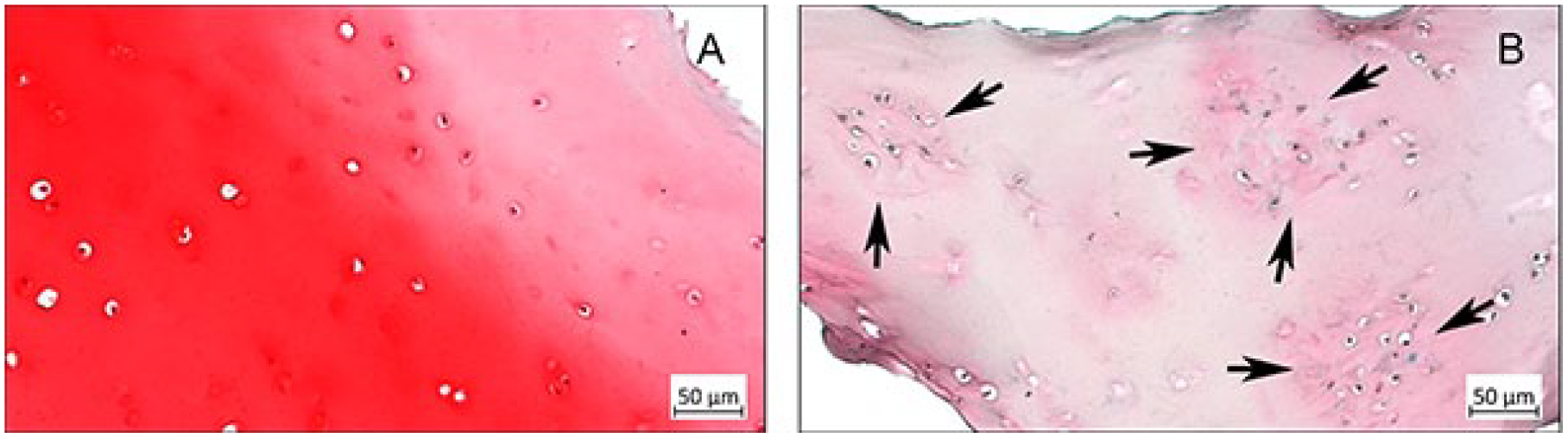
Histology of tissue samples with low and high grades of chondromalacia. (A) Hyaline cartilage with sparsely distributed chondrocytes in a case of Outerbridge grade 0. (B) Histologic osteoarthritic signs (chondrocyte clusters, indicated with arrows, and severe deletion of aggrecan) in a case of Outerbridge grade 3. Safranin O/Fast Green stain.
Association Between Outerbridge Chondromalacia Grade of the Cartilage Surrounding the OLT and Cell Number, Cell Density, and Cell Viability.

Abbreviation: OLT, osteochondral lesions of the talus.
Associations Between Patient Factors and Cell Number, Cell Density, and Cell Viability (P Values).

Abbreviation: OLT, osteochondral lesions of the talus.
Values in bold indicate significance.
The operating surgeon was able to correctly identify whether the OLT tissue was hyaline cartilage or fibrocartilage in only 58% of cases based on visual inspection and probing arthroscopically. In the remaining 42% of cases, the surgeon incorrectly identified fibrocartilage as hyaline cartilage in 10 patients and vice versa in 3 patients.
Discussion
This study shows that an average of 419 000 cells can be obtained from the cartilage removed during arthroscopic surgery for OLTs with a viability of 71.2% after mincing it into small 1- to 2-mm3 pieces. A significantly higher total number of cells was harvested from samples taken from younger patients and from lesions with more severe chondromalacia surrounding the OLT. Lateral OLTs and lesions with a worse surrounding chondromalacia grade had significantly higher cell density.
Previous studies have examined the possibility of using detached cartilage harvested from OLTs as a source of chondrocytes for 2-stage ACI or MACI procedures. Giannini et al determined that the cells harvested from their OLT samples had a viability of 99.9%. Cell number was not reported and the authors did not describe immediately mincing cartilage specimens prior to analysis in their methods. Histology showed high concentrations of proteoglycan, and immunohistochemistry was positive for type II collagen and negative for type I collagen. 12 Kreulen et al found an average of 3.13 × 105 cells in the OLT specimens with a weight of 0.187 g for a cell density of 1.67 × 106 cells/g. Viability after in vitro cell culture for 4 weeks was 92.3%. However, these cartilage samples were not subjected to mechanical mincing, and it is unclear how long after harvesting the assessment of the chondrocytes occurred at the Genzyme laboratory. 15 Candrian et al harvested damaged ankle cartilage fragments from 6 patients with OLTs. The samples were minced and chondrocytes were isolated at a cell density of 1.7 × 106 cells/g. Histology and immunohistochemistry of the damaged cartilage fragment showed diffuse staining for type I collagen and only faint, scattered staining for type II collagen and glycosaminoglycan. 2 All of these studies were primarily focused on the suitability of the explanted chondrocytes for in vitro tissue culture expansion and later reimplantation during a second-stage surgery.
The ability of chondrocytes to migrate out of their lacunae to repair a chondral defect is thought to be limited because of the rigidity of the extracellular matrix that they are embedded within. 20 Lu et al, however, demonstrated that increasing the surface area of the cartilage fragments by mincing them into 1-mm3 pieces permitted chondrocytes to migrate out of the matrix and proliferate in order to repair chondral defects in an animal model. 20 Several other in vivo animal studies that implanted minced autologous cartilage in a single-stage procedure to repair osteochondral defects have shown higher-quality cartilage repair tissue compared to microfracture alone and equivalent results to 2-stage autologous chondrocyte implantation.3,11,18,27
Delman et al first described particulated autograft cartilage implantation (PACI) as a single-stage repair technique for osteochondral lesions in the ankle. Cartilage that was harvested from the patient’s OLT was minced on the back-table with an No. 11-blade scalpel into 1- to 2-mm3 fragments. These autologous fragments were reimplanted during the same procedure into the chondral defect without microfracture and sealed with fibrin glue in 8 patients with promising results. 7 Roth et al 23 published a technique tip using a similar procedure that avoided using the degenerative cartilage and only harvested from the edge of the OLT. Shim et al retrospectively reviewed 32 patients who underwent PACI with mean clinical follow-up of 17 months. Their surgical technique differed from the 2 previous articles because the authors microfractured the subchondral bone prior to implantation of the minced cartilage. Foot Function Index (FFI) and Foot and Ankle Outcome Scale (FAOS) scores both improved significantly. Follow-up MRI at 1 year demonstrated significant improvement in MOCART scores and second-look arthroscopy at 11 months showed the repair cartilage to be ICRS grade I to II (normal to nearly normal). 26
It is currently unknown whether it is more important to deliver a maximal number of viable cells to the repair site or to provide more hyaline-like tissue with mature differentiated chondrocytes and type II collagen during a single-stage procedure. If this was determined through future studies, it may be possible for surgeons to only select optimal cartilage fragments for immediate reimplantation. However, this study shows that fellowship-trained orthopaedic foot and ankle surgeons are only slightly better than 50% successful at distinguishing hyaline cartilage from fibrocartilage arthroscopically. There are currently no other viable methods beyond visual inspection to distinguish hyaline cartilage vs fibrocartilage at the time of surgery.
It is also unclear whether surgeons should perform microfracture of the subchondral bone during single-stage particulated autologous cartilage transfer as described by Shim et al, 26 or if microfracture should be avoided altogether as reported by Delman et al. 7 Lu et al 20 postulated that by avoiding the marrow-derived mesenchymal stem cells released by microfracture that may have osteogenic potential, the repair tissue can be driven more by chondrocytes. Finally, no studies have determined whether the cartilage fragments should be implanted into the lesion and fixed with fibrin glue alone as outlined by the above studies, or if readily available allograft, xenograft, or synthetic scaffold materials should be used in conjunction with the autologous cartilage.
Our study group is limited by a small sample size of only 31 specimens. The patients were also not stratified for other comorbidities such as diabetes, smoking history, inflammatory arthritis, or BMI. It is likely that surgical harvesting of the OLT cartilage did not represent 100% capture, and some tissue may have been lost with the use of the grasper or harvester. This in turn would affect cell number and weight measurements. However, our cartilage handling for cell counting, viability measurements, and immunohistochemistry appear consistent with other historical references in the literature.
Conclusions
This study shows that a large number of viable cells can be harvested from osteochondral lesions of the talus. This suggests that minced cartilage from the lesion itself may be able to function in a single-stage repair augmentation role. Certain patient characteristics including age, location of the lesion, and the quality of the neighboring cartilage can affect both cell number and density. Future prospective level I studies with longer follow-up comparing this technique to microfracture alone, allograft scaffolds, particulated juvenile allograft cartilage, and MACI are necessary to help clinicians select the optimal treatment for a given individual’s OLT in the future.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114241278967 – Supplemental material for Suitability of Minced Cartilage From Osteochondral Lesions of the Talus for Immediate Autograft Reimplantation
Supplemental material, sj-pdf-1-fao-10.1177_24730114241278967 for Suitability of Minced Cartilage From Osteochondral Lesions of the Talus for Immediate Autograft Reimplantation by Emilie R. C. Williamson, Zijun Zhang, Morgan Motsay, Maggie Manchester, John T. Campbell, Rebecca A. Cerrato, Patrick J. Maloney, Lew C. Schon and Clifford L. Jeng in Foot & Ankle Orthopaedics
Footnotes
Ethical Approval
Ethical approval for this study was obtained from the Mercy Hospital Institutional Review Board (approval number 1750582).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: John T. Campbell, MD, reports research grant from Paragon28; OMEGA educational grant; AOFAS fellowship grant; editorial board, Foot & Ankle International; and chair, AOFAS Fellowship Match & Accreditation Committee. Rebecca A. Cerrato, MD, reports consultancy fees from Stryker and Acumed. Patrick J. Maloney, MD, reports consultancy fees from Arthrex and Paragon28; institutional research support from Arthrex, Paragon28, and Stryker. Clifford L. Jeng, MD, reports consultancy fees from Smith & Nephew. Disclosure forms for all authors are available online.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Arthrex (Naples, FL), who provided the GraftNet autologous tissue collectors, as well as financial support used for processing the cartilage samples (IIRR-01218).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
