Abstract
Background:
The purpose of this systematic review was to evaluate outcomes following intra-articular injection of hyaluronic acid (HA) for the treatment of hallux rigidus.
Methods:
During April 2024, a systematic review of the MEDLINE, EMBASE, and Cochrane Library databases was performed based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Data collected and analyzed were number of patients, patient age, follow-up, subjective clinical outcomes, complications, and failures.
Results:
Five studies were included. In total, 218 patients (218 feet) underwent intra-articular injection of HA at a weighted mean follow-up time of 4.4 ± 1.4 months (range, 3-6). There was an improvement in postinjection visual analog scale (VAS) pain at rest scores, VAS pain during activity scores, American Orthopaedic Foot & Ankle Society (AOFAS) scores, and Foot Health Status Questionnaire (FHSQ) scores. In total, 21 complications (10.0%) were observed, the most common of which was transient postinjection pain in 20 patients (9.6%). There were 7 patients (3.2%) who underwent secondary procedures (3.2%). One randomized control trial (RCT) demonstrated no difference in outcomes between an intra-articular injection of HA compared to an intra-articular injection of saline. One RCT demonstrated superior FHSQ scores following between an intra-articular injection of HA compared to an intra-articular injection of triamcinolone acetonide.
Conclusion:
This systematic review suggests that intra-articular injection of HA for the treatment of hallux rigidus may lead to improved clinical outcomes with a low complication rate at short-term follow-up. However, the low level and quality of evidence underscores the need for further high-quality studies to be conducted to identify the precise role of HA in the treatment of hallux rigidus.
Introduction
Hallux rigidus is a common degenerative disorder of the first metatarsophalangeal joint (MTPJ), affecting approximately 25% of patients aged >50 years. 3 This progressive arthritic disease results in first MTPJ pain and gradual loss of first MTPJ range of motion (ROM). 4 Common radiographic findings include joint space narrowing, osteophyte formation, sclerosis, and subchondral cyst formation. Treatment is predicated on the severity of the disease as determined by the Coughlin and Shurna classification system. 6 Conservative management is indicated in early-stage disease, which entails oral analgesics such as nonsteroidal anti-inflammatory drugs, intra-articular injections, physical therapy, orthotics, and activity modification. For patients who fail conservative management and/or present with more severe disease, surgical intervention is warranted. 5 Cheilectomy is the gold standard treatment for grade 2-3 hallux rigidus, with first MTPJ arthrodesis being the gold standard treatment for grade 4 hallux rigidus. 19 Other surgical strategies for grade 2-4 hallux rigidus include interpositional arthroplasty, metatarsal osteotomies, and first MTPJ arthroplasty; however, their results are less predictable in comparison to cheilectomy and arthrodesis.4,5,19
Intra-articular injections that can be used for the treatment of mild- to moderate-grade hallux rigidus include corticosteroids, platelet-rich plasma, and hyaluronic acid (HA).2,8,18 HA is a high-molecular-weight polysaccharide biopolymer that is native to the synovial fluid. 13 In recent years, there has been a growing interest in the use of HA in orthopaedic surgery, especially as a nonoperative treatment modality for hip and knee osteoarthritis.1,7,22,23 Preclinical studies have demonstrated that HA is effective in restoration of joint viscoelasticity, reducing pain and providing an anti-inflammatory effect to the joint. 23 With regard to the available clinical evidence, HA has demonstrated to be an effective nonsurgical strategy in the setting of knee osteoarthritis, providing adequate pain relief, improved ROM, and delaying a total knee arthroplasty (TKA).1,7
Despite early promising studies, there is no consensus regarding the efficacy and precise role of HA in the management of hallux rigidus. Thus, the purpose of this systematic review was to evaluate the clinical outcomes following intra-articular injections of HA for the treatment of hallux rigidus. In addition, we sought to evaluate the quality and level of evidence (LOE) of the included studies.
Materials and Methods
Search Strategy
During April 2024, a systematic review of the MEDLINE, EMBASE, and Cochrane Library databases was performed based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 11 The following search terms were used: (hyaluronic or hyaluronate or hyaluronan or hyaluronic acid) and (hallux or hallux rigidus or metatarsophalangeal joint). The inclusion criteria consisted of studies evaluating clinical outcomes following the use of intra-articular HA injection for the treatment of hallux rigidus, with a minimum of 5 patients per cohort, a minimum of 3 months’ follow-up, and studies written in English. Cadaveric studies, in vivo studies, systematic reviews, and case reports were excluded. After the search was completed, the titles, abstracts, and article full texts were screened by 2 independent reviewers (2 orthopaedic researchers) applying the previously mentioned criteria. A senior author resolved any disagreements that arose.
Assessment of Level of Evidence and Methodologic Quality
The LOE was assessed based on the guidelines published by the Journal of Bone & Joint Surgery. 25 Assessment of the quality of clinical evidence and risk of bias for nonrandomized studies was completed using the Risk Of Bias In Non-randomized Studies-of Interventions (ROBINS-I) tool. 20 This evaluates 7 domains through which bias could potentially be introduced. These domains include bias in study participant selection, potential confounders, intervention classification, missing data, result reporting, deviation from described interventions, and outcome measurements. A final domain gives an overall assessment of the bias, of which the options for a domain-level risk-of-bias judgment are “low,” “moderate,” “serious,” or “critical” risk of bias, with an additional option of “no information.”
For randomized control trials (RCTs), the methodologic quality of clinical evidence and risk of bias was evaluated using the Risk of Bias 2 (RoB 2) tool. 21 The RoB 2 tool is used to determine the risk of bias in randomized trials in 5 different bias domains and 1 domain for an overall assessment of bias. These domains include bias from the randomization process, bias due to deviations from intended interventions, bias due to missing outcome data, bias in measurement outcomes, and bias in selecting reported results. The options for a domain-level risk-of-bias judgement are “low,” “some concerns,” and “high” risk of bias.
Data Extraction and Evaluation
Two independent reviewers independently extracted and assessed the data from each individual study. Data on the injection procedure were collected, which included volume of HA injected, concentration of HA injected, proprietary HA brand, and number of HA injections performed. Subjective outcomes, satisfaction, complications, failures, and reoperations were additionally evaluated.
Results
The search generated 36 studies. Of these, 5 met the inclusion and exclusion criteria (Figure 1).
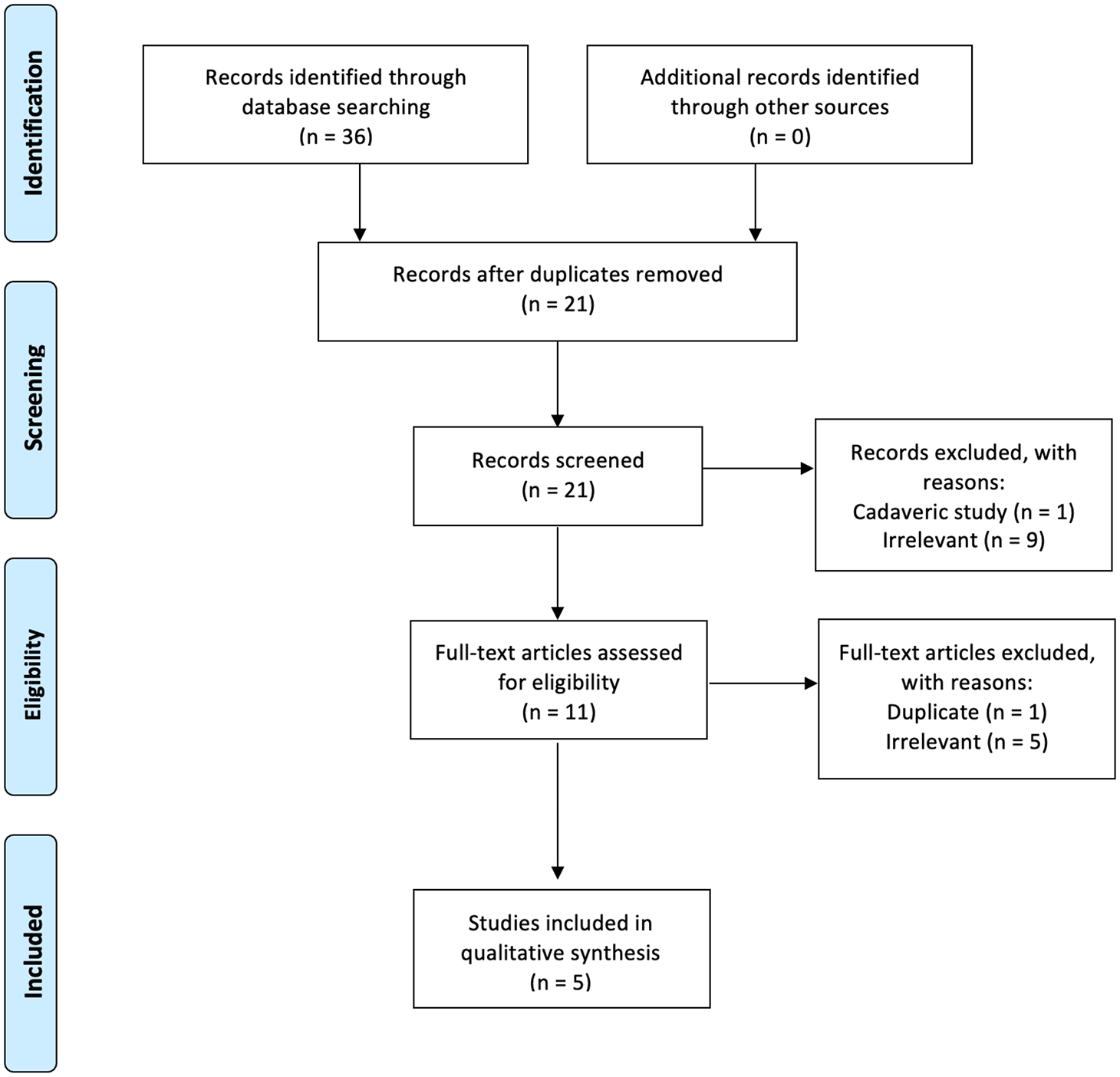
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram.
Study Characteristics and Patients Demographics
Study characteristics and patient demographic data are listed in Table 1. Two studies were LOE I,15,17 and 3 studies were LOE III.8,12,16 According to the ROBINS-I tool, there were 3 studies with moderate risk of bias (Figure 2).8,12,16 Using the RoB 2 tool, there was 1 study of low risk of bias 15 and 1 study with some concerns of bias (Figure 3). 17
Study Characteristics and Patient Demographics.

Abbreviations: BMI, body mass index; LOE, level of evidence; M/F, male/female; n/r, not recorded; R/L, right/left.
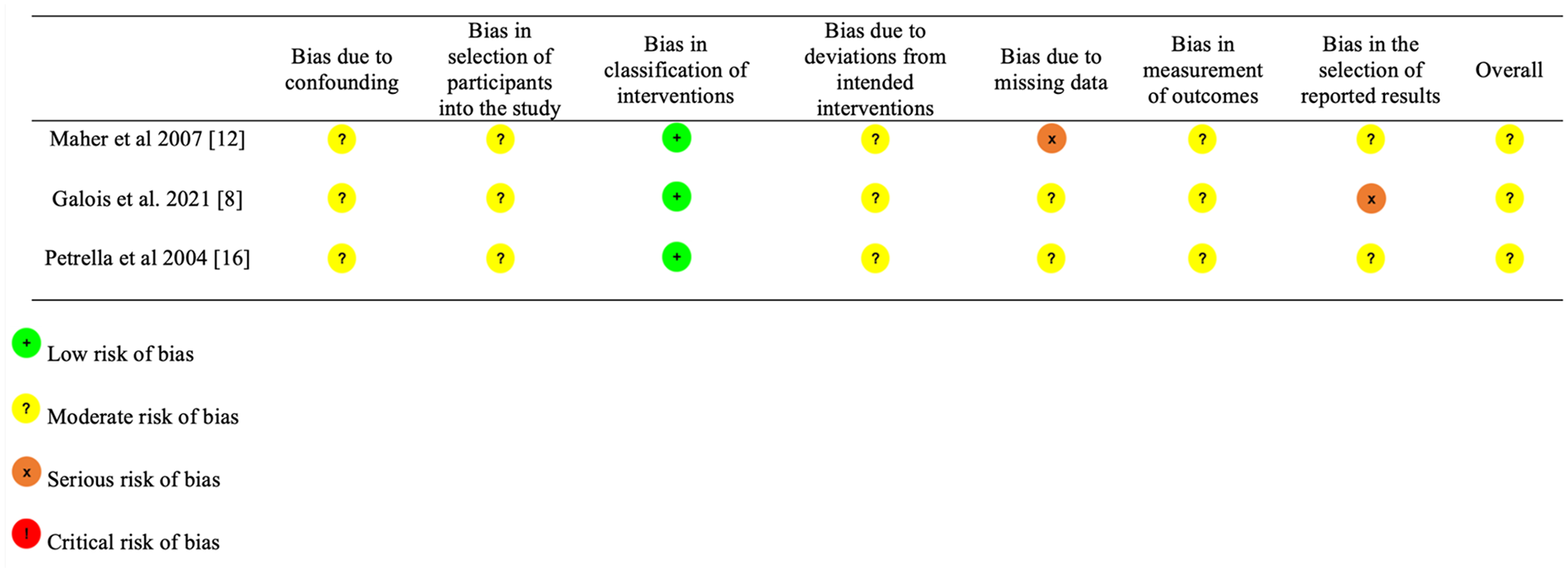
Risk of Bias in Non-randomized Studies–of Interventions (ROBINS-I).

Risk of Bias 2 (RoB 2).
In total, 218 patients (218 feet) underwent intra-articular HA injection for the treatment of hallux rigidus. The weighted mean postprocedure follow-up time was 4.4 ± 1.4 months (range, 3-6 months). The weighted mean age was 60.3 ± 7.2 years (range, 53.7-71). In total, 115 patients (56%) were male, and there were 134 right feet (71.7%). The weighted mean BMI was 26.0 ± 1.5 (range 24.9-27.0).
There were 2 RCTs that met the inclusion criteria.15,17 In total, there were 95 patients (95 feet) that underwent either an intra-articular injection of saline 15 or triamcinolone acetonide (TA) 17 for the treatment of hallux rigidus. The weighted mean postprocedure follow-up time was 5.4 ± 2.9 months (range, 3-6 months). The weighted mean age was 56.8 ± 5.2 years (range, 53.3-62.7). In total, 50 patients (53%) were male and there were 35 right feet (46%).
Procedure Characteristics
There was marked heterogeneity in the procedure techniques and injection protocols used across the included studies. The volume of HA injected was 1.0 mL in 4 studies8,12,16,17 and 0.8 mL in 1 study. 15 The concentration of HA was 10 mg/mL in 3 studies,12,16,17 8 mg/mL in 1 study, 15 and 16 mg/mL in 1 study. 8 A single intra-articular injection of HA was performed in 2 studies,8,17 whereas 2 studies performed serial injections at 1 month and/or 3 months following the initial injection of HA if the patient was still symptomatic.12,15 One study performed weekly intra-articular HA injections for a total of 8 consecutive weeks. 16 A variety of proprietary HA brands were used, including Ostenil Mini, Synvisc, and Happymini.
With regard to the control cohorts, 1 study performed an intra-articular injection of 0.8 mL of 0.9% sterile saline, with the option of repeating the injection at 1 month or 3 months following the index injection if the pain continued. 15 One study performed an intra-articular injection of 1.0 mL of TA. 17
Subjective Clinical Outcomes
Clinical and functional outcomes are listed in Table 2. Three different subjective clinical scoring tools were recorded across the 5 studies in the HA cohorts, including the visual analog scale (VAS), the American Orthopaedic Foot & Ankle Society (AOFAS) score, and the Foot Health Status Questionnaire (FHSQ). The VAS score was the most commonly used scoring tool across 3 studies.8,16,17 The weighted mean preinjection VAS-pain during activity score was 6.5 ± 0.4 (range, 6.1-6.9) and the postinjection VAS-pain during activity score was 2.8 ± 0.5 (range. 2.4-3.3). The VAS-pain at rest score was reported in 2 studies.16,17 The weighted mean preinjection VAS-pain at rest score was 5.2 ± 1.5 (range, 4.1-6.2), and the postprocedure score was 2.8 ± 0.3 (range, 2.6-3.0). The AOFAS score was reported in 1 study. 15 The mean preinjection AOFAS score was 51 ± 18.8 and the mean postinjection score was 77.6 ± 15.8.
Subjective Clinical Outcomes.

Abbreviations: AOFAS, American Orthopaedic Foot & Ankle Society; FHSQ, Foot Health Status Questionnaire; n/r, not recorded; VAS, visual analog scale.
One RCT compared mean postinjection VAS-pain during activity and VAS-pain at rest scores between the HA cohort and the TA cohort. 17 There was a statistically significant difference in VAS-pain during activity scores between the HA cohort (2.4 ± 2.4) compared with the TA cohort (3.7 ± 2.0) in favor of the HA cohort at final follow-up. There was no statistically significant difference in VAS-pain at rest scores between the HA cohort (2.6 ± 2.4) compared to the TA cohort (3.4 ± 1.7) at final follow-up. There was a statistically significant difference in AOFAS scores between the HA cohort (77.6 ± 15.8) compared to the TA cohort (64.3 ± 16.9) in favor of the HA cohort at final follow-up.
One RCT compared mean postinjection FHSQ scores between the HA cohort and the saline cohort. 15 There was no statistically significant difference in FHSQ scores between the HA cohort (68.0 ± 21.4) compared with the saline cohort (71.4 ± 18.7) at final follow-up.
Range of Motion
First MTPJ ROM was recorded in 2 studies.15,16 One study reported that the mean preinjection first MTPJ total ROM was 6.3 ± 4.1 degrees, which improved to 28.7 ± 7.5 degrees postinjection. 16 One study reported that the mean preinjection first MTPJ dorsiflexion was 59.3 degrees, which decreased to 58.9 degrees postinjection. 15
Complications, Secondary Surgical Procedures, and Failures
Data concerning complications, secondary procedures, and failures are listed in Table 3. Complications were reported in all 5 studies. In total, 21 complications (10.0%) were observed in the patient cohort. Complications included transient postinjection pain in 20 patients (9.6%) and infection in 1 patient (0.5%). In the control cohort, 1 patient (1.1%) experienced transient postinjection pain. In the HA cohort, 7 patients (3.2%) underwent secondary surgical procedures. In the control cohort, 9 patients (9.5%) underwent secondary procedures. There were 39 failures (18.7%) recorded in the HA group and 9 failures (9.5%) in the control group.
Complications, Failures, and Secondary Surgical Procedures.
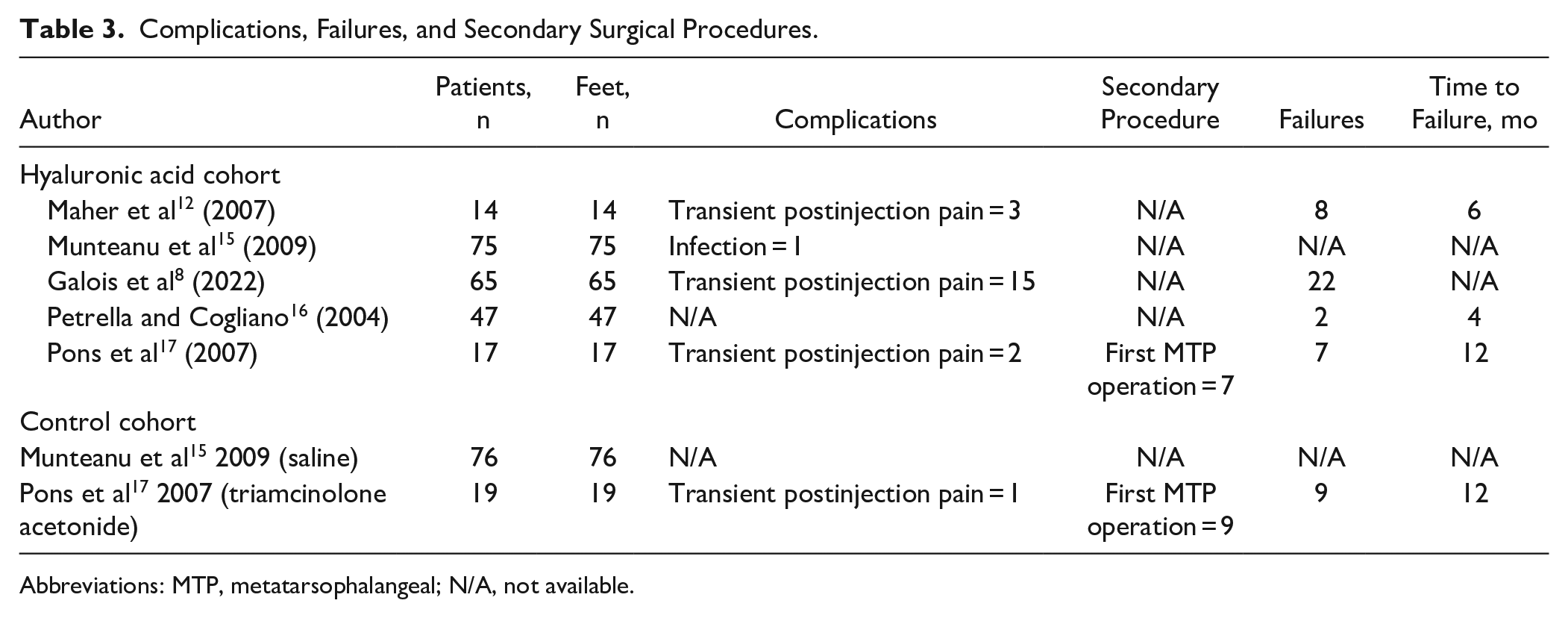
Abbreviations: MTP, metatarsophalangeal; N/A, not available.
Discussion
The most important finding of this systematic review was that intra-articular injection of HA for the treatment of hallux rigidus leads to improved clinical outcomes at short-term follow-up. There was a low complication rate, reinforcing that intra-articular injection of HA is a safe treatment strategy with low associated morbidity. However, the symptomatic relief following intra-articular HA injections is likely to deteriorate in the long term. In addition, there was limited high-quality evidence to determine superiority of HA over placebo. The low number of studies together with the marked heterogeneity between the included studies underscores the need for further, high-quality, comparative studies to be conducted to elucidate the optimal role of intra-articular HA injection for the treatment of hallux rigidus.
Hyaluronic acid is an unsulfated, unbranched negatively charged glycosaminoglycan that is naturally produced in human synovial tissue. 9 HA can exist as a low-molecular-weight compound or a high-molecular-weight compound (105-107 Da), each with contrasting biological and structural functions. 17 The high-molecular-weight form of hyaluronic acid (HMWHA) serves a structural function by binding water at a rate of 10 to 10 000 times its own molecular weight. 13 In a fully hydrated state, HMWHA exhibits osmotic activity, occupying space as both a shock absorber and lubricant. 13 From a biological perspective, HMWHA chains demonstrate chondroprotective effects within the joint because of its antiangiogenic, anti-inflammatory, and immunosuppressive capabilities. 13 As a result of the tribologic, rheologic, and chondroprotective properties of HA, there has been a growing interest in the use of intra-articular HA injections for the management of osteoarthritis.
This current systematic review found improvements in subjective clinical outcome scores following intra-articular injections of HA in patients with hallux rigidus, regardless of the patient reported outcome measurement used. Statistically significant improvements in postoperative visual analog scale (VAS) scores, American Orthopaedic Foot & Ankle Society (AOFAS) scores and FHSQ scores were recorded at final follow-up. However, the only validated scoring tool for hallux rigidus is the Foot and Ankle Outcome Score, 10 which was not used in any of the included studies. Additionally, the Foot and Ankle Ability Measure scoring tool, a high-quality scoring tool frequently used to assess subjective outcomes following foot and ankle procedures, was also not used in any of the included studies. 14 Thus, the reported improvements in subjective clinical outcomes following intra-articular HA must be interpreted with caution, especially in light of the high ceiling effect associated with the AOFAS scoring tool.
There was discordance in the literature regarding the effect of intra-articular HA on restoration of first MTPJ ROM. Munteanu et al 15 found no statistically significant improvement in first MTPJ ROM at final follow-up; however, the authors did not perform first MTPJ traction and/or manipulation under anesthesia following the intra-articular HA injection. Conversely, Petrella et al 16 reported a statistically significant improvement in first MTPJ total ROM from 6.3 ± 4.1 degrees preinjection to 32.8 ± 3.1 degrees postinjection at 16-week follow-up following intra-articular HA injection under semitraction. However, at final follow-up, the first MTPJ ROM slightly deteriorated to 28.7 ± 7.5 degrees. This suggests that in order to improve first MTPJ ROM following intra-articular HA, manipulation of the first MTPJ must be performed postinjection. However, patients must be informed that hallux rigidus is a relentless, progressive disorder; thus, the initial improvement in first MTPJ ROM is likely to slowly deteriorate a number of months postinjection.
Overall, the complication rate following intra-articular HA injection was 10.0%. The most common complication reported was transient first MTPJ pain, which typically resolved within 3-7 days. This indicates that the tenderness experienced at the first MTPJ was due to disturbance of the local soft tissue structures from the high volume of fluid that penetrated the joint capsule during the injection, rather than due to potentially pro-inflammatory, toxic components within HA. Additionally, the infection rate was low, with only 1 infection (0.5%) observed in this cohort, further reinforcing the safety and low morbidity associated with this injection.
The longevity of pain relief following intra-articular HA injection for hallux rigidus was short-lived. Overall, there were 39 failures (18.7%) which occurred at a weighted mean time of 7.3 ± 4.2 months following the index injection. In addition, only 1 study reported outcomes at 1 year follow-up, which found that 7 patients in their cohort (46.6%) required operative intervention due to symptomatic recurrence. 17 These data conflict with the use of HA for knee osteoarthritis. Altman et al 1 reported that the median time to TKA was 114 days for patients who did not receive intra-articular HA compared with 484 days for patients who received intra-articular HA. Furthermore, Dasa et al 7 found that the time to TKA following 1 course of intra-articular HA was 375.6 days. In addition, Waddell et al 24 demonstrated that the median time to TKA following intra-articular injection of HA was 638 days.
Although this systematic review demonstrated improvement in subjective clinical outcomes following intra-articular HA injection, 4 of the 5 included studies (80.0%) did not include an adequate control cohort. Only 1 RCT by Munteanu et al 15 assessed clinical outcomes following intra-articular HA injections compared with an adequate placebo control cohort (saline). At the 3-month follow-up, the authors found no statistically significant differences in FHSQ pain scores between the HA cohort (68.0 ± 21.4) compared with the saline cohort (71.4 ± 18.7) at final follow-up. Additionally, there was no difference in the failure rates, complication rates, or secondary surgical procedure rates between the 2 cohorts. However, the mean follow-up period for this study was 3 months; thus, it is uncertain if the intra-articular HA injection led to sustained pain relief and functional improvement in this cohort compared with the placebo group. Thus, further RCTs with longer-term follow-up are warranted.
One RCT included in this review evaluated outcomes following intra-articular HA injection compared with intra-articular TA injection for the treatment of hallux rigidus. 17 TA is a potent glucocorticoid that modulates synovial inflammation, and has been used for decades in the setting of knee osteoarthritis. 1 There were comparable outcomes between the HA cohort and the TA cohort for the first 2 weeks postinjection. However, by 4 weeks postinjection, there were superior VAS-pain scores during activity and better AOFAS scores in the HA cohort compared with the TA cohort, which was sustained until the 3-month follow-up. The short-lived benefits of TA can be attributed to its short half-life of 24-36 hours in addition to its rapid drug efflux from the joint into the systemic circulation. A major limitation of intra-articular injection of TA is the risk of chondrotoxicity associated with repeat injections, which directly contrasts with the chondroprotective effects of HA. Although the results of this study favor intra-articular HA injection over intra-articular TA injection for providing symptomatic relief in patients with hallux rigidus, the small patient cohort and lack of adequate control group warrants further research to be conducted.
This study must be interpreted in light of its limitations and biases. First, the number of studies included was low with small patient cohorts. Additionally, outcomes following intra-articular HA injection were assessed at short-term follow-up; thus, the midterm and long-term outcomes are yet to be determined. Furthermore, only 1 study in this review included an adequate placebo control group. There was significant heterogeneity between the studies with regard to number of injections performing, performing manipulation of the first MTPJ following the injection, and the patient reported outcome measurements used, thus limiting accurate comparisons between the included studies. Finally, the inclusion criteria were limited to papers published exclusively in English that were available in the MEDLINE, Embase, and Cochrane library databases.
This systematic review demonstrated that intra-articular injection of HA for the treatment of hallux rigidus may lead to improved clinical outcomes with a low complication rate at short-term follow-up. However, there is limited evidence supporting the use of intra-articular injections of HA in comparison to placebo. Future high-quality, comparative studies are warranted to identify the precise role of intra-articular HA injection for the treatment of hallux rigidus.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114241265109 – Supplemental material for Limited Evidence to Support the Use of Intra-Articular Injection of Hyaluronic Acid for the Management of Hallux Rigidus: A Systematic Review
Supplemental material, sj-pdf-1-fao-10.1177_24730114241265109 for Limited Evidence to Support the Use of Intra-Articular Injection of Hyaluronic Acid for the Management of Hallux Rigidus: A Systematic Review by James J. Butler, Hayden Hartman, Amanda Mener, Nathaniel P. Mercer, Grace W. Randall, Stephen Petropoulos, Andrew J. Rosenbaum and John G. Kennedy in Foot & Ankle Orthopaedics
Footnotes
Ethical Approval
Ethical approval was not sought for the present study because this is a systematic review where we did not obtain access to any patient records.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: John G. Kennedy, MD, MCh, MMSc, FFSEM, FRCS(Orth), is a consultant for In2Bones and Arthrex and receives financial support from the Ohnell Family Foundation, Tatiana Rybak, and Mr Winston Fisher. Disclosure forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
