Abstract
Background:
Intraarticular corticosteroid injections (ICIs) are widely used to treat foot and ankle conditions. Although laboratory studies indicate certain corticosteroids and local anesthetics used in ICIs are associated with chondrotoxic effects, and selected agents such as ropivacaine and triamcinolone may have less of these features, clinical evidence is lacking. We aimed to identify the patterns of drug selection, perceptions of injectate chondrotoxicity, and rationale for medication choice among surgeons in the American Orthopaedic Foot & Ankle Society (AOFAS).
Methods:
An e-survey including demographics, practice patterns, and rationale was disseminated to 2011 AOFAS members. Frequencies and percentages were calculated for demographic data, anesthetic and steroid choice, rationale for injectate choice, and perception of chondrotoxicity. Bivariate analysis was used to identify practice patterns significantly associated with perceptions of injectate risk and rationale.
Results:
In total, 387 surveys were completed. Lidocaine and triamcinolone were the most common anesthetic and corticosteroid used (51.2% and 39.3%, respectively). Less than half of respondents felt corticosteroids or local anesthetics bear risk of chondrotoxicity. Respondents agreeing that corticosteroids are chondrotoxic were more likely to use triamcinolone (P = .037). Respondents agreeing local anesthetics risk chondrotoxicity were less likely to use lidocaine (P = .023). Respondents choosing a local anesthetic based on literature were more likely to use ropivacaine (P < .001).
Conclusion:
Corticosteroid and local anesthetic use in ICIs varied greatly. Rationale for ICI formulation was also variable, as the clinical implications are largely unknown. Those who recognized potential chondrotoxicity and who chose based on literature were more likely to choose ropivacaine and triamcinolone, as reflected in the basic science literature. Further clinical studies are needed to establish guidelines that shape foot and ankle ICI practices based on scientific evidence and reduce the variation identified by this study.
Level of Evidence:
Level IV, cross-sectional survey study.
Keywords
Introduction
Intraarticular corticosteroid injections (ICIs) are frequently used for the treatment of degenerative foot and ankle conditions. 12 ICIs are typically composed of a corticosteroid combined with a local anesthetic and are used for diagnostic and therapeutic purposes. 24 Despite a long history of widespread use among foot and ankle surgeons, there are currently no clinical practice guidelines for ICI use in the foot and ankle, and clinical evidence to inform the most safe and effective intraarticular injection practices is lacking.2,11,18,30,43
However, several in vivo and in vitro studies investigating the most commonly injected medications have found that certain corticosteroids and local anesthetics are associated with pronounced chondrotoxic effects.5,6,8,13-15,22,25,37,38,40,41 These studies found the extent of chondrotoxicity to be time- and dose-dependent, and several studies found deleterious effects were exaggerated when local anesthetics and corticosteroids were combined.5,15,37,38,40 Despite the substantial body of laboratory research with concerning clinical implications, very few clinical studies have attempted to validate these findings, and the limited evidence is incongruent.29,30,35,43 As such, whether the chondrotoxic effects identified by laboratory studies translate to human in vivo joint conditions remains unclear.
This study aimed to describe the current ICI medication selection of orthopaedic surgeons within the American Orthopaedic Foot & Ankle Society. We sought to identify the types and volumes of corticosteroids and local anesthetics most commonly used by the membership, perceptions of injectate chondrotoxicity, and rationale for medication selection. Based on our experience, we hypothesized that medication selection patterns would be variable and largely based on personal experience. These data will be helpful for understanding current drug selection choices and designing future prospective clinical trials to determine best practices for foot and ankle intraarticular injections.
Methods
Institutional review board approval was obtained to begin this study. On receiving approval of the AOFAS research committee, an anonymous REDCap survey was disseminated to 2011 AOFAS active surgeon members via email. 19 Both US and international members were included. After the initial invitation was emailed to the membership, 3 follow-up reminders were sent at 1-week intervals.
The survey queried surgeons about their current practice of and rationale for choosing a corticosteroid and local anesthetic for intraarticular injections, the typical dose/volume they inject into the first metatarsophalangeal (MTP) and ankle joint, as well as their perceptions of risk regarding chondrotoxicity of corticosteroids and local anesthetics. Participants were additionally presented with a list of possible complications resulting from intraarticular injections and asked to select any they had observed during their career. 1 Demographics such as practice setting, foot and ankle fellowship training, and number of years in practice after training were collected. The full survey can be viewed in Appendix.
Statistical Analysis
Only fully completed surveys were included in analysis. Frequencies and percentages were calculated for demographic data, anesthetic and steroid choice, volume of local anesthetic injected, primary rationale for injectate choice, perception of chondrotoxicity, and observed complications as a result of intraarticular foot or ankle corticosteroid injections.
Univariate analysis by chi-square test or Fisher exact test was used to identify practice patterns associated with perceptions of injectate risk and rationale. Agreement scale responses regarding perception of injectate chondrotoxicity were collapsed to produce three categories for analysis: “agree,” “neither agree nor disagree,” and “disagree.” Anesthetic volume was categorized as follows: injection of <1 mL, 1 mL, or >1 mL for the first MTP and <2 mL, 2 mL, or >2 mL for the ankle joint. Dependent variables included injectate choice and local anesthetic volume injected. Independent variables included injectate rationale and perception of local anesthetic/steroid chondrotoxicity. P values less than .05 were deemed a priori as statistically significant. Statistical analysis was performed using IBM SPSS Statistics, version 27.
Results
Of 2011 actively practicing AOFAS members, 387 (19.2%) completed the survey. Most respondents reported working in private practice (56.6%) and had 11 or more years of experience (63.3%). Nearly all respondents completed a foot and ankle fellowship (95.1%) (Table 1).
Description of Survey Respondents (N = 387).

Abbreviation: F&A, foot and ankle.
Local Anesthetic Use
Perceived chondrotoxicity of anesthetics
Perceptions of local anesthetic chondrotoxicity varied. Less than half of respondents (42.1%) agreed or strongly agreed that local anesthetics bear risk of chondrotoxicity, 39.3% reported neither agreeing nor disagreeing, and 18.4% disagreed or strongly disagreed local anesthetics may be chondrotoxic (Figure 1).

Respondent perceptions of risk of chondrotoxicity associated with local anesthetics and corticosteroids.
Local anesthetic choice
Lidocaine was the most commonly used local anesthetic, with 43.7% of respondents typically using lidocaine 1% (Table 2). Bupivacaine was used by 27.7% of respondents, and ropivacaine was used by 4.9% of respondents. All respondents who reported injecting a combination of local anesthetics (14%) reported injecting lidocaine.
Local Anesthetic Use and Significant Associations With Rationale and Perception of Risk.

Abbreviation: LA, local anesthetic.
Volume of anesthetic injected
Sixteen (4.1%) respondents did not inject the first MTP joint with local anesthetic, and only 1 respondent did not inject the ankle joint with local anesthetic. Respondents most commonly reported injecting 1 mL of anesthetic agent into the first MTP joint (43.2%), with 33.1% injecting less than 1 mL and 19.4% injecting more than 1 mL (Figure 2A). Responses regarding volume of local anesthetic injected into the ankle joint also varied; 30.5% of respondents injected less than 2 mL, 26.6% injected 2 mL, and 42.6% reported injecting more than 2 mL (Figure 2B).

(A) Volume of local anesthetic injected into the first MTP joint. (B) Volume of local anesthetic injected into the ankle joint.
Rationale for anesthetic choice
Respondents reported basing their local anesthetic choice primarily on clinical experience (28.7%), what they learned in training (28.2%), and the anesthetic action (21.4%). Only 7.2% based their choice on literature they reviewed, and 3.6% reported no specific basis for their choice (Figure 3).
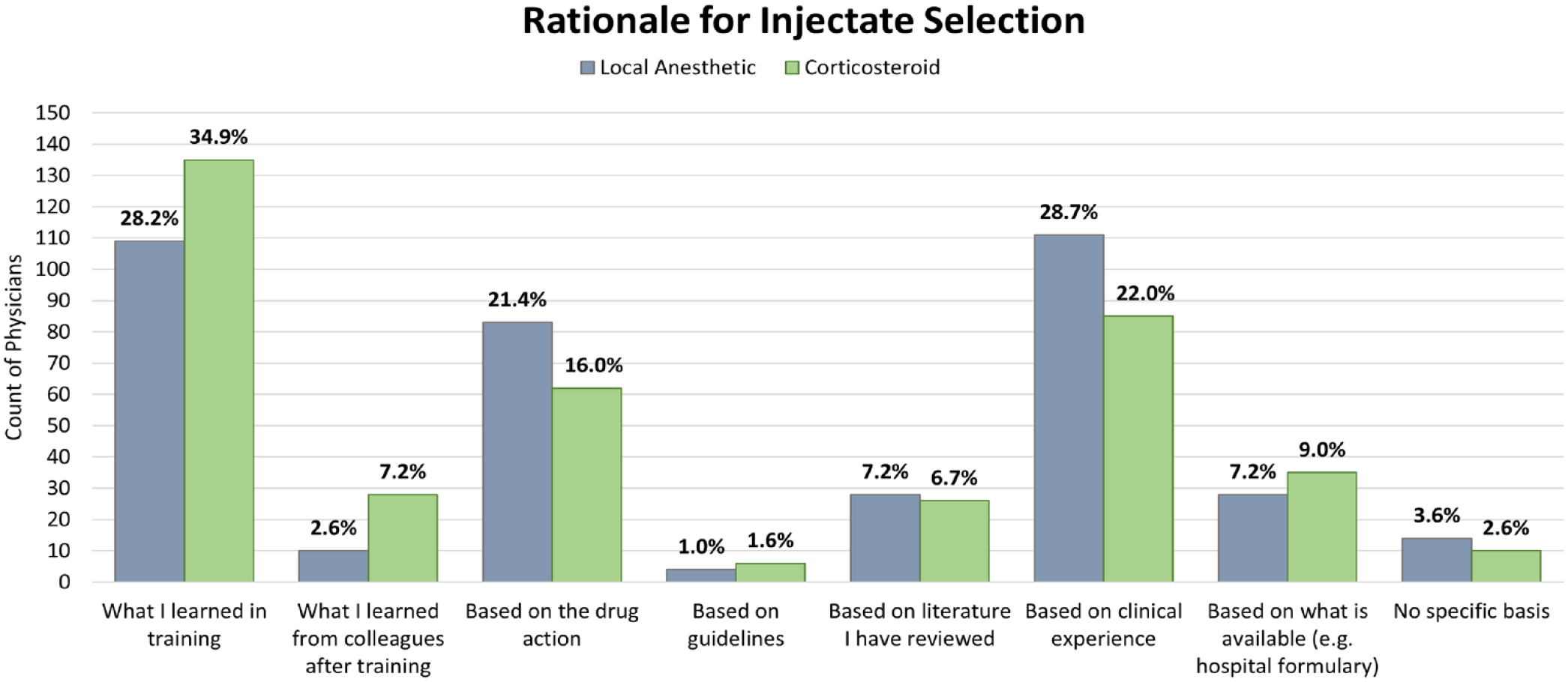
Rationale for injectate selection.
Associations between anesthetic use and perceived injectate risk and rationale
Respondents who disagreed that local anesthetics bear risk of chondrotoxicity were less likely to report using bupivacaine (P = .021), and those who agreed that local anesthetics risk chondrotoxic effects were less likely to use lidocaine (P = .019) (Table 2). Those who reported basing their choice primarily on what they learned in training were more likely to use lidocaine (P = .037), whereas those who based their decision on literature they reviewed were more likely to use ropivacaine (P <.001). Respondents who agreed local anesthetics risk chondrotoxicity were more likely to inject less than 1 mL into the first MTP (P = .005) and less likely to inject 1 mL into the first MTP (P <.001), whereas those who were ambivalent about the risk of local anesthetic chondrotoxicity were more likely to inject 1mL (P = .005).
Corticosteroid Use
Perceived chondrotoxicity of corticosteroids
There was no consensus on potential chondrotoxicity of corticosteroids, with 46% agreeing or strongly agreeing that corticosteroids bear risk of chondrotoxicity, 34.1% neither agreeing nor disagreeing, and 19.9% disagreeing or strongly disagreeing (Figure 1).
Corticosteroid choice
The most used corticosteroid was triamcinolone (39.3%), followed by methylprednisolone (31.3%) and betamethasone (20.7%). Only 7.8% of respondents reported injecting dexamethasone (Table 3).
Corticosteroid Use and Significant Associations with Rationale and Perception of Risk.

Dose of corticosteroid injected
Examination of reported steroid dosages revealed data that could not be reliably correlated with steroid choice. For example, several respondents who reported most commonly using betamethasone or dexamethasone also reported typically injecting 20- or 40-mg doses of steroid into the first MTP (dosages equivalent to >100 mg of methylprednisolone or triamcinolone). Because of this limitation, we were unable to transform data using dose and potency equivalents or examine relationships between dose injected and perception of chondrotoxicity and injectate rationale.
Rationale for corticosteroid choice
Rationale for corticosteroid choice was heterogenous, with the greatest proportion of respondents basing their selection on what they learned in training (34.9%). Other frequently reported rationales were “based on clinical experience” (22.0%) and “based on the drug action” (16.0%). Only 6.7% of respondents based their corticosteroid selection on literature they had reviewed (Figure 3).
Associations between corticosteroid use and perceived injectate risk and rationale
Respondents basing corticosteroid selection on what they learned in training were less likely to use betamethasone (P = .048), and those who based their decision on what they learned from colleagues after training or the drug action were more likely to use betamethasone (P = .026 and P = .040, respectively). Those who agreed corticosteroids bear risk of chondrotoxicity were more likely to use triamcinolone (P = .037), and those who disagreed corticosteroids risk chondrotoxicity were less likely to use triamcinolone (P = .009) (Table 2).
Observed complications from intraarticular corticosteroid injections
The most frequently observed complication resulting from a foot or ankle ICI was a postinjection flare (70.3%). The majority of respondents had also observed a temporary increase in blood sugar (59.4%) and skin bleaching (59.2%) during their career. Twenty-three of respondents (5.9%) had never observed a complication because of an ICI in their practice (Table 4).
Complications Observed Resulting From Foot or Ankle Intraarticular Injection.

n represents the number of respondents who reported observing the listed complication during their career.
Discussion
This study identified the current ICI drug selection patterns of active surgeon members of the AOFAS. We found that foot and ankle surgeons use a variety of different medications and base their injectate choice primarily on what they learned in training or clinical experience. Those who agreed local anesthetics or corticosteroids risk chondrotoxicity and those who chose local anesthetics based on literature were more likely to choose medications previously reported to have less chondrotoxic potential.
The present study calls attention to the need for wider education on the available evidence and additional clinical studies exploring the safety and efficacy of the most commonly used medications for ICIs in the foot and ankle. These studies will be necessary for refining ICI drug selection practices, as the variability in selection and rationale identified herein demonstrates a need for more robust clinical evidence to support surgeons in practicing evidence-based medicine.
A substantial body of literature describes the chondrotoxic effects of local anesthetics,2,6,7,8,13,18,21-23,25,28,32,33 particularly when combined with corticosteroids.5,15,21,27,29,30,37,38,40,41 Several of these studies concluded that ropivacaine is less chondrotoxic than bupivacaine or lidocaine, indicating that ropivacaine may have the greatest margin of safety for clinical use.2,21-23,27,29,33,40 We found that 42.1% of surgeons acknowledged the risk of chondrotoxicity with local anesthetics; however, only 4.9% typically used ropivacaine for ICIs. Most respondents (78.9%) injected exclusively lidocaine or bupivacaine, with an additional 14% using lidocaine in combination with another local anesthetic. We hypothesize that although many AOFAS members may be aware of the relative chondrotoxicities of local anesthetics, perhaps few use ropivacaine because of its higher cost as compared to the other anesthetic agents, its longer onset time, lack of robust clinical data, or an inability to choose what anesthetic agent is available. Nevertheless, these results suggest many AOFAS members’ anesthetic choice is not swayed by the available literature. Indeed, we found that only 7.2% of respondents chose based on literature they reviewed, whereas more than half of respondents reported basing their anesthetic choice on what they learned in training or their clinical experience. While we await more robust clinical evidence on the optimal ICI medications, surgeons who are able to choose may wish to use the lowest-risk option based on what is known from the basic science literature.
This inconsistency in decision-making rationale surrounding ICI practices has been previously observed. A study examining local anesthetic and corticosteroid use trends for large joint and bursa injections among sports medicine physicians found less than half chose a corticosteroid based on literature review. 9 In addition, a survey of the American Society for Surgery of the Hand and the American Shoulder and Elbow Surgeons found only 9% of respondents selected injectates based on scientific reference. 26 Like the present study, both authors cited training as a main rationale.
Given the variation in rationale, it is unsurprising that there is no consensus in injection practices among AOFAS members or within and across other orthopaedic subspecialties.3,13,26,39 Kegel et al 26 found a 365-fold difference in steroid dose used in trigger finger injections alone, and Blankstein et al 3 found widespread variation in ICI practices in the setting of knee osteoarthritis nonoperative management. Skedros et al 39 compared ICI use for shoulder pain across surgical and nonsurgical specialties, whereas the study found no significant difference between dose equivalents of corticosteroids used across specialties (orthopaedic surgeons, primary care sports medicine physicians, physical medicine and rehabilitation physicians), there was significant variation in the volumes of local anesthetic used.
We found that AOFAS members with concern regarding the potentially chondrotoxic effects of local anesthetics tended to introduce a lesser amount of anesthetic agent into the joint. Although responses varied widely, we found surgeons most commonly injected 1 and 2 mL of local anesthetic into the first MTP joint and ankle joint, respectively. Volume injected into the first MTP ranged from 0.25 up to 5 mL (1 respondent), and volume injected into the ankle joint ranged from 0.3 to 10 mL. No studies to date have explored the effect of local anesthetic volume on clinical outcomes, and as such, it remains unclear whether the time- and dose-dependent chondrotoxic effects demonstrated in the laboratory have implications for optimizing injectate volume. Of note, a series of in vivo and ex vivo experiments using human cartilage suggest local anesthetic volume may have little to no clinical impact; synovial local anesthetic concentrations 10-15 minutes after lidocaine injection into the knee were more than 40-fold lower than the injected concentration, indicating mechanisms that reduce intraarticular local anesthetic concentrations act so quickly that concerns regarding prolonged exposure with larger volumes may be unfounded. 35
In vitro and animal in vivo studies have found corticosteroids can similarly damage chondrocytes.14,38,40,41,44 However, basic science studies have also shown that triamcinolone, and in some cases methylprednisolone, may provide protection for chondrocytes when administered in low doses.16,44 Clinical studies comparing the most commonly used corticosteroids are limited and have been performed almost exclusively in the knee, hip, and shoulder joints.10,11,20 According to some reports, triamcinolone may be the safest and most efficacious2,20; however, other studies have been unable to identify differences in the radiographically measured joint space or patient-reported pain levels when comparing triamcinolone to saline 36 or methylprednisolone. 29 One study comparing triamcinolone and betamethasone for knee osteoarthritis found that triamcinolone was more effective at reducing pain and local tenderness than betamethasone, although unvalidated outcomes were used and assessed only 1 week after injection. 42 The efficacies of triamcinolone, methylprednisolone, betamethasone, and dexamethasone have yet to be compared in the treatment of foot and ankle pathologies. Our study found that triamcinolone was the most frequently used corticosteroid (39.3%) among the AOFAS followed by methylprednisolone (31.3%) and betamethasone (20.7%). More than one-third chose a corticosteroid based on what they learned in training, and those who agreed corticosteroids are chondrotoxic were more likely to use triamcinolone than any other corticosteroid.
Regarding corticosteroid dosing for ICIs, one recent review concluded a 20-mg dose of triamcinolone appears to provide noninferior relief to a 40-mg dose in small and intermediate-sized joints. 11 We found that 38.2% of those who reported injecting the ankle joint with corticosteroid reported injecting 40 mg or more of triamcinolone or methylprednisolone, indicating a substantial proportion of respondents may be injecting a higher dose of corticosteroid than may be necessary to achieve the desired clinical result. However, it is important to note that the evidence indicating a 20-mg dose may be as effective as a 40-mg dose for small or intermediate joints is limited, including just 2 randomized controlled trials that were performed in the distal radioulnar joint and the radiocarpal joint.4,31 Further research is necessary to identify the ideal corticosteroid dose to maximize clinical benefit while minimizing risks.
Postinjection flares have also been reported to be common by other authors.1,21 Importantly, these are not to be confused with infection. Although the etiology for such flares is not yet fully understood, proposed mechanisms include a rapid intracellular ingestion of the microcrystalline corticosteroid ester or a reaction to the corticosteroid crystals themselves. 21 Accordingly, our group frequently recommends 1-2 days of oral NSAIDs following the injection. Prior studies have not identified any associations between corticosteroid type and rate of postinjection flare.17,34
This study has limitations including selection and nonresponse bias, inherent to electronic questionnaires and all studies with incomplete response rates. We acknowledge that surgeons choosing to respond to a survey regarding injections may have strong views. Although we had a low response rate (19.2%), this is comparable to previous membership survey studies.3,9,24,26 Second, only AOFAS surgeon members were queried, which may not be generalizable to all providers who perform foot and ankle ICIs in clinical practice. Third, data regarding injection frequency or patient selection was not obtained. However, prior studies have explored injection frequency,3,24 and in our experience, surgeons largely do not adapt their drug choice based on patient demographics or disease severity. Lastly, steroid dosage data were not consistent enough for rigorous analysis, which prevented us from exploring potential relationships between dose injected and perception of chondrotoxicity and injectate rationale. Despite these limitations, we feel the results of our survey provide a useful description of the current ICI practice patterns of actively practicing AOFAS members and can be helpful for designing prospective clinical trials to determine best practices for foot and ankle intraarticular injections.
Conclusion
In summary, our survey identified the current practice patterns for ICIs within the AOFAS. Corticosteroid and local anesthetic use for ICIs varied greatly among the membership. Respondents who recognized potential chondrotoxicity and chose based on literature were more likely to choose ropivacaine and triamcinolone, reflecting the available basic science literature. This study adds to a small body of work highlighting a common theme: rationale for ICI formulation is variable as the clinical implications are largely unknown. Although the chondrotoxicity and long-term safety of ICIs remains controversial, choosing medication based on the available evidence may help to reduce potential risks associated with this common treatment modality. This study further underscores the need for additional studies to determine clinical implications and to shape ICI practices based on scientific evidence.
Supplemental Material
sj-pdf-1-fao-10.1177_24730114231216990 – Supplemental material for Intraarticular Injections in the Foot and Ankle: Medication Selection Patterns and Perceived Risk Of Chondrotoxicity
Supplemental material, sj-pdf-1-fao-10.1177_24730114231216990 for Intraarticular Injections in the Foot and Ankle: Medication Selection Patterns and Perceived Risk Of Chondrotoxicity by Emily B. Parker, Kalei A. Hering, Christopher P. Chiodo, Jeremy T. Smith, Eric M. Bluman and Elizabeth A. Martin in Foot & Ankle Orthopaedics
Footnotes
Appendix
Acknowledgements
The authors would like to thank the American Orthopaedic Foot & Ankle Society for allowing the dissemination of this survey to its members. The authors would also like to thank the AOFAS members who took the time to participate in this study.
Ethical Approval
Ethical approval for this study was obtained from the Mass General Brigham Institutional Review Board (IRB protocol number: 2021P002829).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. ICMJE forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
