Abstract

Keywords
Introduction
Scedosporium boydii (formerly known as Pseudallescheria boydii) is an opportunistic fungal pathogen that has been isolated from soil near polluted and clean parts of streams and ponds. 3 Prior to 2010, it was thought to be the anamorph (asexual) form of the fungi Scedosporium apiospermum until declared 2 separate species. 7 Three types of infections are recorded to be caused by S boydii: (1) localized disease after trauma, (2) asymptomatic and symptomatic colonization of cavities (ear) and lungs of patients with predisposing pulmonary disorders, and (3) systemic invasive disease in immunocompromised patients. 8 Fungal osteomyelitis is notoriously difficult to diagnose and treat because of its insidious clinical presentation, the absence of characteristic laboratory markers, and failure to suspect fungi as the causative infectious agent. Multimodal treatment strategies call for surgical debridement, drainage of joints or abscesses, removal of infected prosthetic joints, and systemic antifungals. 6
We are presenting a rare case of fungal osteomyelitis with S boydii following an open fracture that was successfully treated with antifungal chemotherapy and multiple surgical debridements in lieu of the original prognosis, amputation. We believe this to be the first reported case of an environmentally acquired infection involving S boydii following an open fracture. Our review of literature published prior to February 2022 in PubMed, MEDLINE, and EBSCO databases using search terms “Scedosporium apiospermum” or “Pseudallescheria boydii” and “osteomyelitis” yielded a 1997 case of distal tibial osteomyelitis following osteophyte and hardware removal after ankle fracture. 10 Also of note is a case of posttraumatic Pseudallescheria/Scedosporium apiosperma osteomyelitis of the tibia; however, this case was published after the 2010 species delineation. 4
Case Report
A 67-year-old woman sustained a left ankle grade 3 open trimalleolar fracture in the woods near a pond. Unable to walk, she dragged herself through the soil to return to her car. An open reduction with internal fixation (ORIF) was performed and complicated by a bacterial ankle joint infection. The infection was treated with multiple surgical debridements as well as outpatient intravenous antibiotics for 6 weeks via peripherally inserted central catheter.
Six months after the ORIF, she noticed sudden swelling and pain in the ankle. Two months later, a bone biopsy was conducted with debridement and hardware removal, which showed nonviable bone with mild chronic inflammation and cultures positive for Scedosporium boydii. She was treated with intravenous micafungin for approximately 3 weeks and then transitioned to oral voriconazole for the next year. Thirteen months after the debridement and biopsy, she was started on isavuconazole because of the persistence of pain and swelling in the joint. Isavuconazole was continued for 10 months before she was referred to our office for a second opinion from the orthopaedic surgeon. Prior to referral, the patient had been counseled that medical therapy could not cure her infection, and she had begun attending amputation support groups.
On presentation, the patient was afebrile with a C-reactive protein less than 0.5 mg/dL, an erythrocyte sedimentation rate of 11 mm/h, and a white blood cell count of 6.4 103/μL. Radiographs demonstrated erosive processes on both sides of the joint (Figure 1). Debridement was repeated along with placement of a gentamicin-impregnated methyl methacrylate spacer because of the patient’s history of bacterial infection; cultures from surgery remained positive for S boydii. The minimum inhibitory concentration (MIC) suggested that voriconazole or posaconazole would be superior to isavuconazole. The patient transitioned back to voriconazole a month later, which she took until her symptoms improved 7 months later. Side effects reported from the voriconazole were blurred vision and seeing colored lights, which ophthalmology determined was not ocular toxicity.

Anteroposterior, lateral, and oblique radiographs of the left ankle with diffuse lytic destruction of the distal tibia, distal fibula, and talus taken on January 9, 2017 (33 months after open fracture). Periosteal reaction of medial aspect of distal tibia (white arrows: lytic areas; dashed arrow: periosteal reaction). Radiographic remnants of 4 to 5 screws from previous internal fixation visible throughout distal fibula.
Three months after the cessation of fungal chemotherapy, distal tibia and talus bone biopsy cultures revealed no growth. The methyl methacrylate spacer was removed and an ankle fusion and femoral head allograft was performed 6 months later once surgery cultures were negative (Figure 2). The patient was seen at 2-week, 5-week, 3-month, and 6-month postoperative follow-up with no complications. The patient was interviewed 42 months after the spacer removal and ankle fusion and reported no recurrence of or new symptoms.
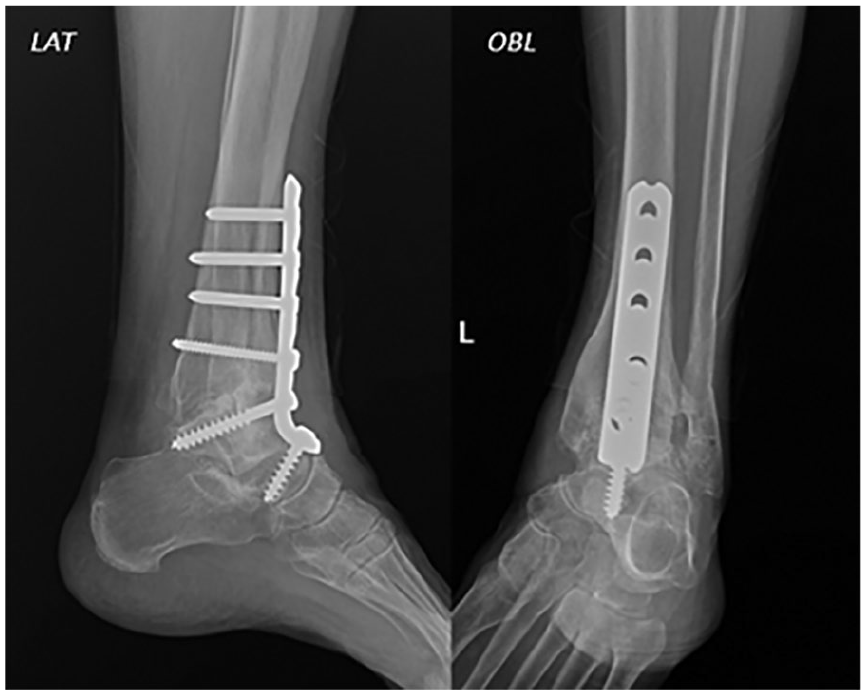
Lateral and oblique radiographs of the left ankle with internal fixation ankle fusion device after resolution of infection taken on February 15, 2019 (58 months after open fracture).
Discussion
Management and treatment of fungal osteomyelitis is difficult because of the rare nature of the disease and its delayed presentation. Little to no evidence-based treatments for fungal osteomyelitis exist, but the Infectious Disease Society of America have guidelines for common fungal pathogens causing vertebral osteomyelitis and consist of debridement, irrigation, and antifungal therapy. Scedosporium boydii has demonstrated its ability to form biofilms, resulting in an increased resistance to azole antifungal drugs. 9
Diagnosing fungal osteomyelitis requires a high index of suspicion because routine fungal testing is not standard of care. Systemic inflammatory markers normally used to monitor osteomyelitis or joint infections are typically minimally elevated or normal in cases of fungal infections. Detection and diagnosis of fungal osteomyelitis can be done by amplifying the DNA through polymerase chain reaction (PCR), specialized stains (eg, Calcofluor white, potassium iodide), and fungal cultures. 3 Panfungal PCR assays have recently proved useful in the early accurate diagnosis of invasive fungal disease. These tests are becoming commercialized and approved by the US Food and Drug Administration for specific common fungal species and offer simultaneous detection of common azole resistance mutations. 5 Another emerging method of invasive fungal disease diagnostics is the matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS), which is now widely used in clinical microbiology laboratories for rapid and accurate diagnosis of different species of bacteria and fungi. However, MALDI-TOF MS has only reported high identification rates when using in-house databases, which are expensive and time-consuming to create. 1 MALDI-TOF MS also requires isolation of the specimen and culture growth to acquire sufficient data for identification. Rapid and accurate diagnosis can help facilitate early intervention. For our patient, the diagnosis of S boydii osteomyelitis was made a year after the fracture.
The antifungal chemotherapy regimen is complex as no consensus exists on what level of MIC clinically predicts successful therapy. 2 In our case, the success of infection resolution can likely be attributed to the repeated debridements because the patient remained culture positive after nearly 2 years of antifungal therapy. Amputation was offered because of the poor prognosis associated with resistant fungal osteomyelitis. However, the patient sought a second opinion, and therapy continued; after 3 years, cultures were negative.
Summary/Conclusion
When considering all discussed factors, S boydii osteomyelitis can be exceptionally difficult for the orthopaedic surgeon and, because of its rare nature, low on the differential diagnosis list. Recent decades have shown an emergence of nonfumigatus Aspergillus infections and infections by rare molds with resistance to azoles, so this clinical scenario may become more common. 2 Our patient was informed that her case was hopeless, with amputation being the only option; however, today she walks pain free. We are hopeful that more diagnostic techniques—other than standard cultures—will soon be readily available in clinical laboratories for invasive fungal disease.
Footnotes
Ethics Approval
Ethical approval was not sought for the present study because written consent was obtained from the patient for this case study to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. ICMJE forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
