Abstract
Lisfranc injuries are a disruption of one or more of the tarsometatarsal joints and have an estimated incidence of 1/55 000 people. However, the total number of Lisfranc injuries could be underreported, because almost 20% of these injuries are initially missed. Because of the relative infrequency of these injuries, the current literature is inconsistent in regard to proper treatment. This article provides a review of Lisfranc complex injuries including relevant anatomy, diagnosis, treatment, classifications, operative approaches, and outcomes and complications. Based on existing evidence, it also proposes an algorithm the authors prefer for the evaluation and treatment of Lisfranc complex injuries.
Introduction
Lisfranc injuries encompass a wide spectrum of injuries from frank fracture dislocations to subtle ligamentous injuries only revealed with stress tests. These injuries can occur as a result of either high-energy trauma, such as motor vehicle accidents and falls from height, or low-energy trauma from sports activities. 68 The anatomy of the midfoot makes it difficult to diagnose subtle low-energy Lisfranc injuries on static plain radiographs. It has been reported that almost 20% of Lisfranc injuries are initially missed. 22,57 Missed or delayed diagnoses can be devastating; patients may develop progressive midfoot instability, arch collapse, forefoot abduction, or post-traumatic osteoarthritis (PTOA) that results in stiffness, chronic pain, and dysfunction of the foot and ankle complex. 47
Because Lisfranc injuries are relatively rare, only representing 0.2% of all fractures, with an estimated incidence of 1/55 000 people, there continues to be an ongoing debate on the ideal management of such injuries. 1,8,24 This review aims to present the current literature and use existing knowledge to develop updated diagnostic and treatment algorithms for Lisfranc injuries.
Anatomy
The Lisfranc joint complex is composed of metatarsals M1 to M5, cuneiforms C1 to C3, the cuboid, and interconnecting ligaments, capsules, and reinforcing tendons. The midfoot is inherently stable because of its osseous arrangement. The metatarsal bases form a Roman arch structure in the axial section, with M2 acting as a keystone, articulating proximally with C2, and wedged between C1 and C3 with on average 8 and 4 mm of contact, respectively (Figure 1). The importance of M2 to the Lisfranc joint complex is demonstrated by an increased rate of Lisfranc injuries with a shallower M2 mortise and with a smaller ratio of M2 length to foot length. 21,46 The midfoot is further stabilized by the keystone arrangement of the navicular in the medial column and the cuboid in the lateral column. 26
On top of the osseous stability, the midfoot has several key ligaments that add to its stability. At the tarsometatarsal joints, there are longitudinally oriented dorsal ligaments, transversely oriented interosseous ligaments, and obliquely oriented plantar ligaments. The intermetatarsal ligaments, which include dorsal, interosseous, and plantar components, transversely connect the M2 to M5 bases. There is no intermetatarsal ligament between M1 and M2. 26 The Lisfranc (C1M2) ligament is the strongest midfoot ligament and connects the medial and middle columns. 65 Biomechanical measurements have demonstrated that the dorsal Lisfranc ligaments provide relatively less stability. Thus, isolated injuries of dorsal Lisfranc ligaments can be managed nonoperatively. 60 Other contributors to midfoot stability include capsules; plantar muscles; tendons of the peroneus longus, tibialis anterior, and tibialis posterior; and the plantar fascia, which all maintain the arch of the foot.
There are 3 distinct tarsometatarsal synovial joint systems correlating to the 3 columns of the foot: C1-M1 (medial), C2-M2 and C3-M3 (middle), and Cu-M4-M5 (lateral) (Figure 1). 17 In vitro models have demonstrated that of the 3 columns, the lateral column has considerably more motion than the medial and middle columns. 42 As a result, the lateral column is generally not fused in the treatment of Lisfranc injuries and is allowed to remain mobile. Recently, Mason et al 36 described the lateral Lisfranc ligament, a transverse suspensory metatarsal ligament that connects the long plantar ligaments of both the transverse and longitudinal arches. In cadavers, they noted that this ligament allowed M2 through M5 to act as a single unit. They theorized that if the lateral Lisfranc ligament remains intact, fixation of the middle column is all that is necessary to overcome lateral instability.
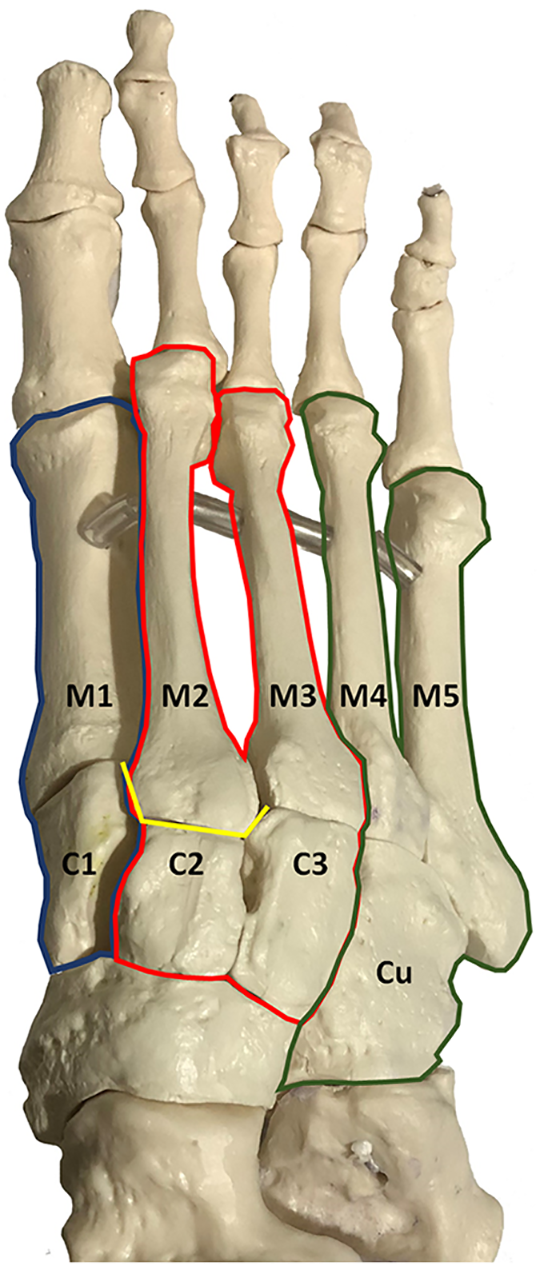
Model of the foot demonstrating the keystone configuration of M2 between C1 and C3 in yellow. The 3 columns of the foot are highlighted, with the medial in blue, the middle in red, and the lateral in green.
Diagnosis
Generally, Lisfranc complex injuries from high-energy trauma have obvious clinical and imaging evidence that make it unlikely to miss, the exception being polytrauma patients with distracting injuries. However, diagnosis of low-energy Lisfranc injuries can be difficult without appropriate history, physical examination, and imaging.
History
Lisfranc injuries occur via direct midfoot crush injuries or indirect injury due to forefoot twisting and axial loading of the plantarflexed foot, with indirect injury the more common mechanism of injury. 40 Lisfranc injuries occur 2 to 4 times more often in males than in females. In contact sports such as football, the twisting injury can occur without contact. 37 Patients generally present with swelling and pain that is localized to the TMT joint region.
For patients with delayed presentation from initial injury with a mechanism consistent with Lisfranc injury, it is important to maintain a high index of suspicion. Swelling may not be evident, but additional history that may provide important clues include pain and limitations of athletic activity as well as reduced walking tolerance. 7
Physical Examination
On physical examination, swelling and/or point tenderness will be evident around the TMT joint region. Special attention should be paid to identifying any plantar ecchymosis, which is strongly suggestive of Lisfranc injury. 53 Examination should also include provocative maneuvers to evaluate for instability—squeezing of the midfoot, pronation, supination, abduction, adduction, single limb weightbearing, and passive motion in sagittal and coronal planes of all 3 columns of the midfoot. 39,56 In the clinic setting, provocative maneuvers can be performed under fluoroscopy, as discussed in the following section, to help guide diagnosis and treatment.
Imaging
All patients with suspected Lisfranc injuries will need standard, 3-view plain films of the foot—an anteroposterior of both feet on one cassette, 30-degree internal oblique, and lateral views. All films should be weightbearing and ideally bilateral for comparison, if tolerated by the patient. With nonweightbearing films, subtle Lisfranc injuries are missed at a reported rate of 20% to 50%. 22,41
The following signs of instability should be evaluated on the weightbearing plain films (Figure 2): The medial border of M2 is not aligned with the medial border of C2 on the anteroposterior radiograph >2 mm gapping difference is noted between the base of M1 and M2 or between C1 and the base of M2 on the anteroposterior radiograph
18
The medial and lateral borders of M1 are not aligned with the medial and lateral borders of C1 on the oblique radiograph
62
The lateral border of M3 is not aligned with the lateral border of C3 on the oblique radiograph The medial border of M4 is not aligned with the medial border of Cu on the oblique radiograph Step-off of the dorsal cortex from M1 to C1 on the lateral radiograph. The plantar border of C1 dips below the plantar border of M5 on the lateral radiograph
18
“Fleck sign”—any avulsion fragment from the lateral edge of C1 or the medial edge of the base of M2.
40
Be aware of potential mimics, such as a free-standing type os intermetatarseum, which is well corticated and smooth in shape
10
Medial column tangential line—a line tangential to the medial aspect of the navicular and medial cuneiform that does not intersect the base of M1 on the anteroposterior abduction stress view
15
Associated signs suggesting Lisfranc injury—for example, compression fracture of cuboid, metatarsal base or neck fractures, and metatarsophalangeal joint dislocation/subluxation
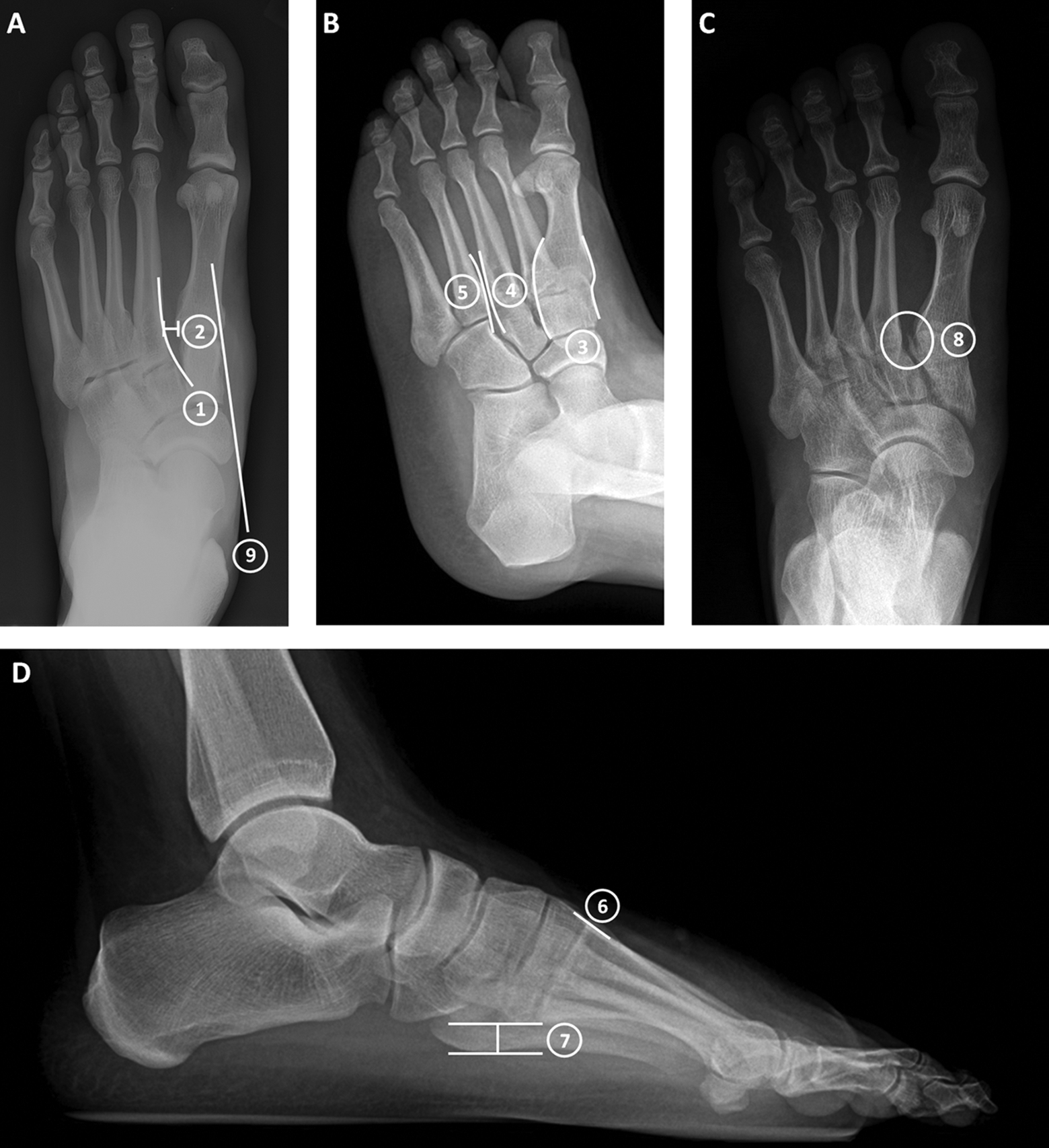
Radiographic signs of instability: (A) Left foot weightbearing anteroposterior radiograph demonstrating (1) continuity of medial borders of M2 and C2, (2) gapping between M1 and M2 bases, and (9) medial column tangential line. (B) Left foot weightbearing oblique radiograph demonstrating (3) continuity of medial and lateral borders of M1 and C1, (4) continuity of lateral borders of M3 and C3, and (5) continuity of medial borders of M4 and Cu. (C) Left foot weightbearing anteroposterior radiograph demonstrating (8) Fleck sign. (D) Left foot weightbearing lateral radiograph demonstrating (6) continuity of dorsal cortices of M1 and C1 and (7) normal relationship of M5 and C1 plantar cortices.
Unfortunately, the diagnostic accuracy of radiographs, even those that are obtained weightbearing, remains low. Studies comparing radiographic diagnoses to CT scans or intraoperative findings have noted only 61% to 68.9% agreement. 51,58 As a result, patients with negative radiographic evaluation for Lisfranc injury, but with continued clinical concern, should obtain further imaging by dynamic stress radiograph, computed tomography (CT), or magnetic resonance imaging (MRI).
CT is ideal for detecting subtle fracture and persistent displacement not evident on plain radiographs (Figure 3). 32 When compared to plain radiographs, where the complex anatomy of the midfoot leads to overlapping projections, CT identifies an additional 51% of tarsal fractures and 38% of metatarsal fractures. 49 CT allows for better visualization of fracture patterns and comminution of joint surfaces. It can also be used to evaluate for pathologic widening of C1 and M2 by measuring the narrowest transverse gap between the articular surfaces. 58 However, it should be noted that conventionally, CT is nonweightbearing and nondynamic. Thus, it is less useful in identifying pure ligamentous injuries. Weightbearing CT is increasing in popularity, but to date, no studies have evaluated its utility in Lisfranc injuries.

(A) Axial CT slice of the right foot demonstrating Fleck sign. (B) Axial CT slice of the right foot demonstrating comminution of metatarsal and cuboid fractures.
For Lisfranc injuries, MRI is useful for evaluation of ligament integrity, occult fractures, or edema. 9,58 Because of a limited number of cases, no MRI-based grading scheme has yet been described. However, MRI has demonstrated good ability to determine stability, correctly classifying 90% of Lisfranc joint complex injuries when compared to intraoperative findings. 50 The one known MRI finding that strongly predicts instability, with a sensitivity of 94% and a specificity of 75%, is a disruption or grade 2 injury of the plantar ligament between C1 and the bases of M2 and M3. In these cases, surgery is recommended and thus obviates the need for manual stress radiographs.
Bone scintigraphy is not used routinely. In one study of 15 athletes, bone scintigraphy demonstrated 100% sensitivity in detecting low-grade Lisfranc injuries. 41 However, bone scintigraphy is nonspecific and may demonstrate increased uptake in cases of chronic instability or osteoarthritis.
When imaging is equivocal, it may be necessary to consider a stress examination under anesthesia. Stress maneuvers performed under fluoroscopy allow dynamic evaluation of the foot, which is advantageous over previously discussed static imaging modalities in identifying instability. 39 Stress maneuvers are similar to those used in the physical examination—squeezing of the midfoot, pronation, supination, abduction, adduction, and shucking of M1 in dorsal and plantar directions (Figure 4). 15,16 In 2 different cadaver studies, abduction stress maneuver was superior to weight-bearing radiographs in diagnosis of Lisfranc instability with diagnostic accuracy reported to be 75-100%. 15,25

Right foot intraoperative fluoroscopy demonstrating (A) no stress maneuver, (B) abduction stress maneuver, and (C) adduction stress maneuver.
Classifications
Numerous classification systems have been created to describe Lisfranc injuries. For high-energy Lisfranc fracture-dislocations, the modified Hardcastle classification as described by Myerson is the most commonly used to characterize injury morphology of total incongruity, partial incongruity, and divergent types. 24,40 Myerson also noted the existence of the proximal variant, where injury is not isolated to the tarsometatarsal joint, but extends proximally to affect the intercuneiform and naviculocuneiform joints. 40 Regarding low-energy Lisfranc injuries, Nunley and Vertullo proposed a classification to grade severity by combining clinical findings, weightbearing radiographs, and bone scintigraphy. 41
Unfortunately, the existing classification systems offer little utility in clinical practice. They demonstrate only a moderate degree of interrater reliability and do not add prognostic value. 63 Even Nunley and Vertullo concede that excellent outcomes are related to anatomic reduction of the Lisfranc joint. 41
Treatment
The end goal following foot and ankle injury is to achieve a stable and painless plantigrade foot. Specifically, to restore stability following Lisfranc injuries, anatomic reduction of the Lisfranc complex is essential. Because Lisfranc injuries generally involve a component of instability that can lead to deformity, the role of nonoperative management in Lisfranc injuries is limited. In the literature, nonoperative management has been reserved for Lisfranc injuries without evidence of instability, such as those with <2 mm diastasis or extra-articular fractures with stability confirmed by weightbearing and stress views. 12,41
Operative management is generally indicated for all other Lisfranc injuries. However, optimal operative treatment remains ambiguous. Because there is a wide spectrum of severity, instability patterns, and concomitant joint conditions, there has been no concrete evidence supporting any particular treatment modality. Ongoing debates exist in regard to fusion vs fixation, the role of suture buttons, transarticular screws vs extra-articular plates, operative timing, and need for removal of hardware.
Historically, open reduction and internal fixation was the accepted standard treatment for acute Lisfranc injuries. Fusion was primarily used as a salvage procedure in situations where patients were initially treated with internal fixation and subsequently developed PTOA. 38,54 However, more recently, primary fusion has been proposed as definitive management for those patients who have extensive articular cartilage damage that makes PTOA inevitable. 6 Primary fusion would avert a secondary procedure, additional rehabilitation, and increased financial burden. In multiple systematic reviews and meta-analyses comparing fixation vs fusion, it has been demonstrated that patients treated with internal fixation had a significantly higher rate of additional surgery, most commonly for hardware removal or secondary fusion, compared with those treated with primary fusion. However, no significant differences were identified for patient outcome scores. 3,34,59
The major criticism of the studies included in these systematic reviews and meta-analyses are that all manner of Lisfranc injuries are included to bolster the study power. However, the increased heterogeneity does confound the data. Primary fusion demonstrates superior outcomes compared to fixation when evaluating pure ligamentous or high-energy Lisfranc injuries with severe joint surface damage only. 28,33 Other situations where primary fusion has been recommended include patients with delayed presentations and for obese or elderly patients in whom fixation has a higher rate of failure.
Currently, fixation remains the standard of care in the young athletic population. Because the athlete has a higher demand, maintenance of motion in the medial column is thought to be necessary for restoration of full function. 39 Besides limitations to motion, the higher-demand athlete with arthrodesis may place an excessive burden on surrounding structures, which increases the risk for pseudoarthrosis, stress fracture, or development of transfer metatarsalgia. 41 In a survey of AOFAS surgeons, less than 70% of surgeons recommend return to play for running, football, soccer, and basketball after Lisfranc fusion. 66 Cochran et al presented evidence that challenges this dogma. 13 They compared active duty military personnel with low-energy Lisfranc injuries, where 14 were treated with fusion and 18 were treated with internal fixation. Primary fusion patients had an earlier return to full military activity and better fitness test scores after 1 year. However, the major limitation of the study was that it was not randomized. Acute cases were generally treated with internal fixation, whereas those with delayed presentation were generally treated with fusion.
The current internal fixation technique with the “home run” screw and intercuneiform screws, was popularized by Lewis & Anderson. 31 But the first mention of the “home run” screw was in 1990 by Sangeorzan et al. They described a 3.5- or 4.0-mm cortical lag screw that followed the course of the Lisfranc ligament from C1 to the base of M2. 54 The combination of the “home run” screw and intercuneiform screws reduces the medial to the middle column. An alternative method of inserting the “home run” screw was proposed by Panchbhavi. 43,44 Instead of the screw entering from a medial to lateral orientation, a cannulated partially threaded 4.0- to 5.0-mm screw can be inserted from the base of M2 into C1. The advantage of this technique is easier targeting, better purchase, and easier removal if broken.
Hansen proposed that the medial and middle column contain nonessential joints and may be fused by placement of permanent implants (Figure 5). The lateral column contains essential joints, and so it is necessary to reconstruct and preserve the joint. 23 Kirschner wires are commonly used for provisional fixation and maintenance of reduction in the lateral column. Often, Kirschner wires are buried subcutaneously to decrease infection risk, but their superficial location allows them to easily be removed at about 6 weeks’ time. 12

(A) Right foot anteroposterior radiograph demonstrating dorsal bridge plating and Lisfranc screw fixation. (B) Right foot intraoperative fluoroscopy demonstrating primary fusion of the medial and middle columns and temporary fixation of the lateral column.
More recently, the suture button has been considered as an alternative to the traditional “home run” screw of the Lisfranc ligament. The purported advantage of the suture button is maintenance of midfoot flexibility, while still providing a similar level of stability. In one study, 7 dancers and high-level athletes who presented with Lisfranc injury late after failing conservative management were treated with suture button fixation. All returned to sport in 6 months and at minimum 15 months of follow-up had improved AOFAS scores from 65 to 97. 11 A cadaver study demonstrated similar levels of stability when comparing screw and suture button fixation for an isolated transection of the Lisfranc ligament. 45
Extra-articular bridge plating has been proposed as an alternative to transarticular screws. Previous cadaveric studies have demonstrated 2% to 6% disruption of the articular surface with transarticular screws and purported that extra-articular bridge plating provides stiffer fixation and less displacement when compared to transarticular screws. 2,20,35 However, these factors have not manifested in reality. No differences in functional outcomes have been linked to type of implant. In fact, most studies conclude that anatomic reduction is more crucial to predicting functional outcomes. 30 There exists a single retrospective review, specifically of Lisfranc fracture dislocations, where AOFAS scores were significantly better in those treated with bridge plating compared to those treated with transarticular screws or a combination technique. 27 However, in that study, the plate fixation group was noted to be trending toward better anatomic reduction. In terms of technique, Ardoin and Anderson recommended that locking screws be used in the distal and proximal holes and nonlocking screws be used at the holes close to the joint so that the screws can be directed away from the joint as needed. 4
Optimal timing of surgery is still not clearly defined. The general rule has been the sooner the better, so long as swelling is manageable. In this manner, the patient can begin the long rehabilitation process. Myerson and Cerrato recommend operating within 6 weeks. From their experience, satisfactory reduction of displaced fractures and dislocations are more difficult to obtain the longer one waits from the initial injury. At 2 months’ time, they state that reduction is not possible without resection of the scar that has formed in the first web space. 39 However, no study has been able to definitively prove that outcomes are worse after 6 weeks. Hardcastle et al treated 27 cases operatively, with the majority of those treated prior to 6 weeks having good results, whereas 2 patients treated after 6 weeks had only fair results. 24 In contrast, Kuo et al 29 were unable to determine any difference in outcome scores between those with an acute vs. delayed diagnosis. Furthermore, Feng et al performed staged management of missed Lisfranc injuries at a mean duration of 4.8 months after initial injury. Of their 15 patients, the 13 in which they were able to maintain reduction had comparable AOFAS scores to those treated acutely in Kuo’s cohort. 19
Operative Approaches
The most commonly adopted approach reported is the dorsal double incision (Figure 6). The 2 incisions are separated by a skin bridge of at least 4 to 5 cm. 5 This approach does have concern with skin necrosis. Furthermore, the large skin bridge required can make it difficult to obtain adequate exposure. Although a transverse incision would avoid this issue, the incision would cross over the neurovascular structures. It would also be difficult to accurately locate the level of interest.
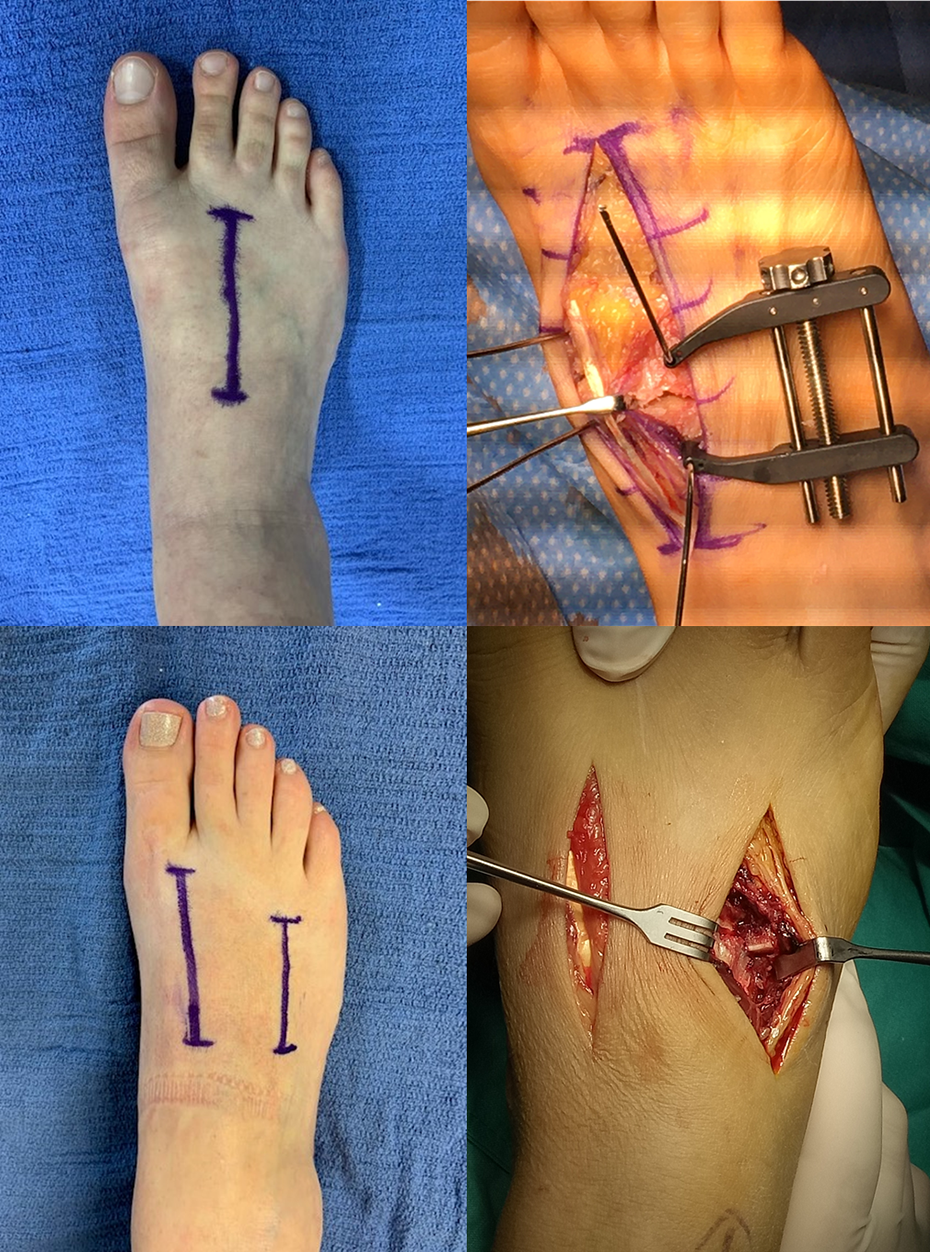
Dorsal single incision (top) vs dorsal dual incision (bottom).
Philpott et al presented a modified single dorsal approach using tissue windows to access all 3 columns. 48 For their cohort of 150 patients, they reported 14% wound-related complications. At first glance, the rate reported appears to be higher than that seen in the literature. However, the patient cohort had 16% open injuries and 75% high-energy mechanisms, both of which are associated with a higher complication rate. Furthermore, the study definition of wound-related complications is broad and included an aggregate of all complications previously reported in the literature. When compared individually, all complications fell within previously reported ranges—superficial infection 5%, delayed healing 3%, dehiscence 3%, complex regional pain syndrome 1%, and impaired sensation 1%. They concluded that a single dorsal incision offers a viable alternative to the double dorsal incision with comparable wound complication rates and superior exposure.
Outcomes and Complications
The literature demonstrates that the quality of anatomic reduction is most closely correlated with patient outcomes. Richter et al evaluated anatomic reduction by assessing restoration of length to the medial and lateral columns as well as the shape of the longitudinal arch. 52 They demonstrated that acceptable anatomic reductions had higher functional scores when evaluated by the AOFAS-ET, AOFAS-Midfoot, Hannover Scoring System, and Hannover Questionnaire.
Acceptable anatomic reduction has been demonstrated with both closed and open reduction. In a systematic review of 106 patients, Stavrakakis et al demonstrated good functional outcomes for patients treated with closed reduction and percutaneous fixation. 61 The main benefit of percutaneous fixation like other minimally invasive operative techniques is a reduction in soft tissue damage and minimization in morbidity from traditional open surgeries. 67 However, when compared to closed reduction, open reduction better achieves anatomic restoration. Direct visualization likely helps not only with anatomic reduction, but also in identification of the instability pattern for more appropriate treatment. In one study of 28 patients, 33% of patients undergoing closed reduction had acceptable reduction, whereas 86% of patients undergoing open reduction had acceptable reduction. 55 Additionally, the type of hardware used for fixation influences maintenance of reduction. Loss of reduction was seen in 37.5% of patients treated with Kirschner-wire fixation, whereas no loss of reduction was seen in those treated with rigid fixation with plates and screws.
Despite anatomic reduction with rigid fixation, there is no guarantee of a painless fully functional outcome. In a study of 11 patients following operative treatment of unilateral closed Lisfranc injuries, 10 patients had anatomic reduction. Yet, 8 of 11 still had evidence of PTOA and 9 of 11 had decreased relative range of motion. 64 Similar results were reported in a retrospective study of 92 adults after Lisfranc injury, with about 25% of the patients developing PTOA. 29 The amount of PTOA is directly proportional to the area of damage on the articular surface at the time of initial injury. 40,54 Besides joint destruction, persistent instability also plays a role in development of PTOA and poor long-term outcomes. Kuo et al demonstrated that a subgroup of patients with pure ligamentous Lisfranc injury had poorer outcomes regardless of anatomic reduction and screw fixation. 29 Both of these factors help to explain why patients with a late presentation do poorly. Not only do these patients have a reduced chance of achieving an anatomic reduction but they may already have developed aberrant soft tissue healing that affects overall ligamentous stability.
Algorithm Preferred by Authors for Evaluation and Treatment of Lisfranc Injuries
It is generally accepted that if the patient can tolerate weightbearing, has no detectable radiographic signs of instability on weightbearing radiographs, and has no arch collapse, then the patient can be managed nonoperatively (Figure 7). 14,31,39 The patient should be placed in a short leg cast or immobilization boot for 1-2 weeks of nonweightbearing. Progressive protected weightbearing is permitted if repeat weightbearing films are stable 2-3 weeks after initial acute injury and patient has reduced pain and swelling in the foot. At 6 weeks, the boot can be discontinued. Firm orthotic arch support should be prescribed at that time. Exercise may start at 3 months if there is no pain on stress. Return to sports may start at 6-8 months.
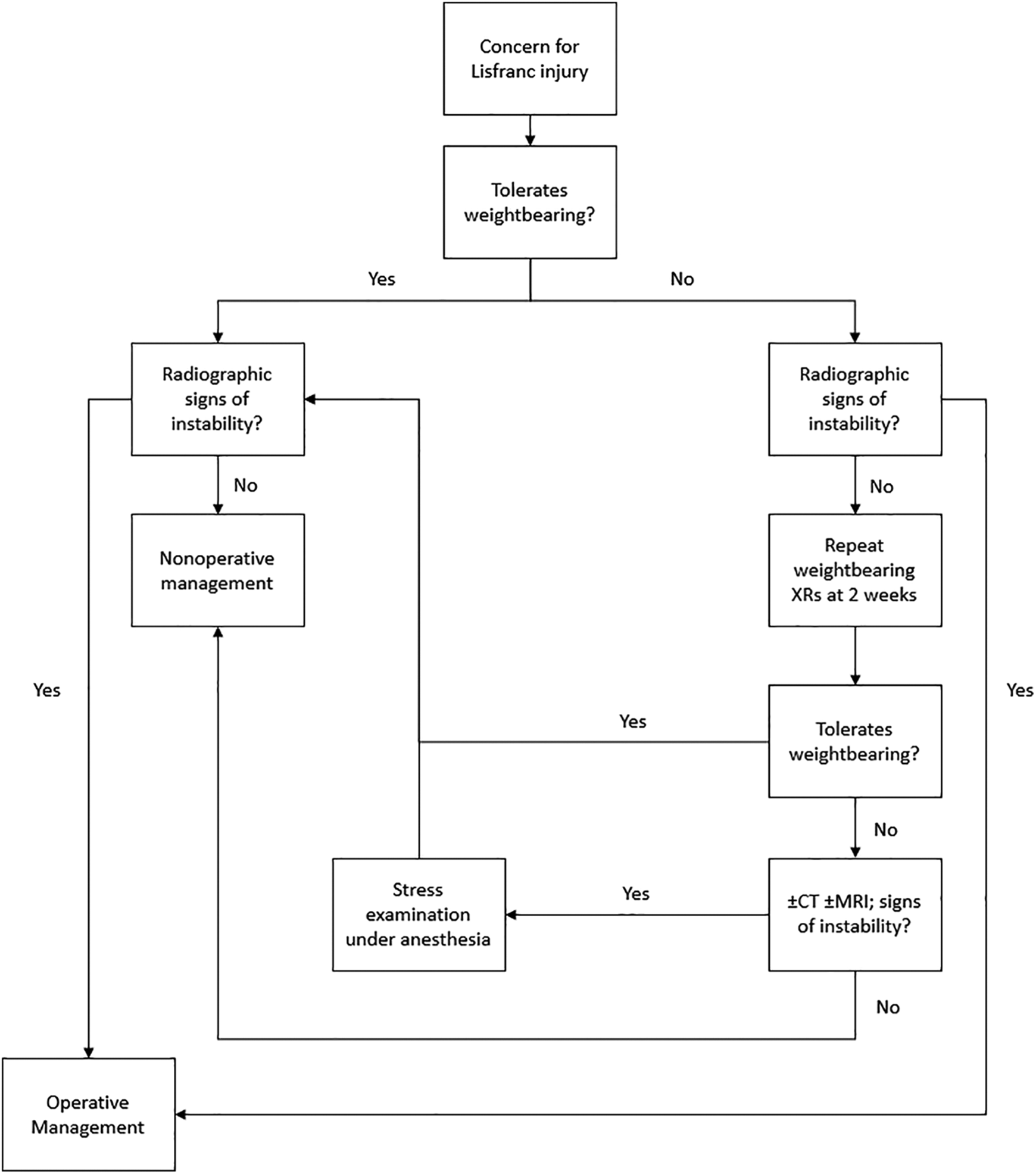
Authors’ preferred algorithm for triaging patients with Lisfranc complex injuries to nonoperative or operative treatment.
If the patient is unable to tolerate weightbearing, but there are no detectable radiographic signs of instability on nonweightbearing radiographs, then repeat weightbearing radiographs should be attempted at the 2-week follow-up. If the patient is still unable to tolerate weightbearing or presents with persistent swelling, pain, and plantar ecchymosis despite being able to tolerate weightbearing, then the patient should undergo a stress examination under anesthesia. The patient should be consented for possible operative reduction and fixation vs primary arthrodesis pending findings. The stress maneuver under anesthesia involves rotational stress by pronation/supination of the forefoot, sagittal stress by dorsiflexion/plantarflexion of the 3 columns of the foot, and coronal stress by abduction/adduction of the forefoot and midfoot and squeezing of the forefoot. Each of these maneuvers is assessed for the above radiographic signs of instability. Any findings of instability will be subjected to operative management.
If the patient is unable to tolerate weightbearing and there are detectable radiographic signs of instability, then the patient is managed operatively. A stress examination under anesthesia should still be performed to determine the instability pattern and determine appropriate operative care. A CT scan should be obtained preoperatively for any patient with a high-energy mechanism and radiographs that demonstrate a complex midfoot fracture or dislocation with comminution.
MRI will be obtained for those patients with an equivocal clinical examination prior to stress examination under anesthesia or those not willing to undergo stress examination under anesthesia. If only the dorsal ligaments of the Lisfranc joint are involved, we will manage the patient nonoperatively. 50 This is also like conventional CT, which is a static examination and will need dynamic stress examination under anesthesia to confirm the instability pattern and operative management.
In the operating room, stress examination is routinely performed to confirm all patterns of instability. If anatomic closed reduction can be achieved, fractures can be fixed with percutaneous screws (Figure 8). However, generally open reduction and internal fixation with either plate or screws is required. If joint surfaces are preserved and there are metatarsal base fractures, then dorsal joint sparing plates are used. Otherwise, if joint surfaces are preserved without metatarsal base fractures, then screws can be used. If joint structures are destroyed, partial fusion of the nonessential joints of the medial and middle column and percutaneous fixation with Kirschner wires at the fourth and fifth tarsometatarsal joints and subsequently removed at the 6-week follow-up. We use 3.5-mm cortical screws in noncompression mode for transarticular fixation and Lisfranc extra-articular screw fixation. We prefer to place the Lisfranc screw retrograde from the base of the lateral second metatarsal to the medial cuneiform. 43 Length of the intercuneiform screw is determined by the instability involvement. We will use medial column plating if there is extended instability medially to the naviculocuneiform joint.

Authors’ preferred algorithm for determining type of hardware to use in operative Lisfranc complex injuries.
For high-energy Lisfranc injuries with obvious fracture dislocation and complete ligamentous disruption, primary arthrodesis of the medial and middle columns is preferred unless there is a large intra-articular fragment with an intact Lisfranc ligament attached to the fracture piece. If patients choose to preserve the joint with fixation over fusion, a more conservative postoperative protocol should be adopted with nonweightbearing for 12 weeks. Fusion should be avoided in pediatric patients with open physes.
For young athletes with low-energy Lisfranc injuries, internal fixation is preferred over primary arthrodesis unless there is obvious destruction of the joint surfaces or late presentation of greater than 6 weeks. Flexible fixation with a suture button should be considered in the isolated Lisfranc ligament injury for those athletes requiring high performance and flexibility of the foot.
Postoperatively, the patient is placed in a Bulky Jones splint and made nonweightbearing for 2 weeks. Sutures are removed at 2 weeks. The patient is placed in a short leg cast until 6 weeks with a cast change typically performed at 4 weeks. At 6 weeks, the patient is transitioned to a boot and progressive weightbearing is initiated. For elite athletes, the patient is placed in a boot at 2 weeks and rehabilitation is initiated. 4 Custom-molded shoe orthosis will be required until 6 months.
For patients treated with internal fixation, hardware removal should occur between 4 and 6 months. We recommend only removing the transarticular screws of the tarsometatarsal joint. The Lisfranc screw and intercuneiform screws generally remain to prevent later diastasis.
Conclusion
Lisfranc complex injuries are rare, but serious, injuries with long-term consequences. Missed or delayed diagnoses have increased rates of morbidity. A high index of suspicion should always be maintained for patients with a mechanism consistent with a Lisfranc injury presenting with pain and swelling of the midfoot. Subtle Lisfranc complex injuries can be difficult to diagnose. It is important to obtain appropriate imaging and utilize the numerous stress maneuvers described to effectively evaluate for injury. We present a treatment algorithm based on the current body of evidence. Treatment is focused on anatomic reduction and rigid fixation, which have been correlated with improved patient outcomes. Much of the existing literature is limited by its weak evidence level. Further investigations are needed to improve the treatment of Lisfranc injuries with the goal of achieving improved long-term outcomes.
Supplemental Material
Supplemental Material, sj-pdf-1-fao-10.1177_2473011420982275 - Updates on Lisfranc Complex Injuries
Supplemental Material, sj-pdf-1-fao-10.1177_2473011420982275 for Updates on Lisfranc Complex Injuries by Alan Yan, Stephen R. Chen, Xin Ma, Zhongmin Shi and MaCalus Hogan in Foot & Ankle Orthopaedics
Footnotes
Ethics Approval
Ethical approval was not sought for the present study as no patients or cadaveric specimen were involved in this review article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. ICMJE forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
