Abstract
Background:
For hallux rigidus, dorsal cheilectomy remains a treatment option even with advances in interposition techniques and devices. Cheilectomy aims to alleviate dorsal impingement and improve pain and function as well as range of motion. Cryopreserved umbilical cord allograft, with properties to mitigate inflammation and scar formation, has theoretical benefit for improving outcomes following cheilectomy. In this first prospective randomized and blinded cheilectomy study reported, we aimed to compare outcomes between cheilectomy alone and cheilectomy with umbilical cord allograft.
Methods:
Patients were randomized to cheilectomy alone (CA) or cheilectomy with cryopreserved umbilical cord (ie, amniotic membrane–umbilical cord [AM-UC]). Patients were evaluated with American Orthopaedic Foot & Ankle Society (AOFAS), Foot Function Index (FFI), and visual analog scale (VAS) pain outcomes collected preoperatively and at 6 months and 1 year postoperatively. In addition, radiographic range of motion data were collected using stress radiographs. Fifty-one patients (26 AM-UC, 25 CA) completed the study, with 5 bilateral surgeries in the AM-UC group and 2 in the CA group, totaling 31 and 27 feet, respectively.
Results:
The AM-UC group had statistically significant improved AOFAS and FFI scores at 1 year compared with the CA group, but there was no difference at 6 months. There was no significant difference between groups for VAS-pain scores at any time point, but overall VAS-pain improved in both groups from preoperative values. There was no significant difference in range of motion (total arc) between groups and changes in range of motion (total arc) in both groups from preoperative to 1 year postoperative were small.
Conclusion:
We present the results of the first randomized and blinded prospective study of cheilectomy surgery patients. When appropriately selected, cheilectomy remains a good option for patients with symptomatic hallux rigidus. Cryopreserved umbilical cord is a potential adjuvant to cheilectomy, with 1-year results showing improvements in functional outcome scores.
Level of Evidence:
Level II, prospective comparative study.
Introduction
Hallux rigidus is a progressive arthritic condition of the first metatarsophalangeal joint (MTPJ). Clinically, it is characterized by pain, decreased motion, and enlargement of the joint. Radiographically, there are the classic findings of osteoarthritis about the joint with joint space narrowing, subchondral cysts, osteophytes, and sclerosis. The hallux MTPJ is the most common joint affected by arthritic disease in the foot, next to knee and hip arthritis, and tends to affect younger patients. It affects 10% of adults and is present on radiographs in 20% to 48% of adults older than 40 years. 3
Hallux rigidus has a clinical and radiographic scoring system that is both descriptive and helpful in guiding treatment. It incorporates degree of radiographic change (joint space narrowing and osteophyte formation) and accounts for pain and range of motion. A key component of the clinical assessment is differentiating pain with dorsiflexion from pain at midrange motion, as that has implications for decision about treatment options. 10,17
Operative treatment options include various joint preserving and sacrificing options. Preserving motion and alleviating pain is the goal of treatment except for hallux metatarsophalangeal arthrodesis, which is the gold standard for end-stage disease. A mainstay of operative treatment for joint preservation is cheilectomy. Popularized by Mann et al, 20 the procedure involves joint debridement and osteophyte removal as well as removal of 20%-30% dorsal articular surface. Coughlin and Shurnas reported satisfactory results in >90% or patients with this treatment. 10 A meta-analysis, which found only Level III and V studies on cheilectomy (3 Level III, 15 Level IV, and 14 Level V) identified at least 80% good to excellent results in 11/15 Level IV studies. Although this demonstrates the abundance of literature, it is lacking high-level studies and as such, it only received grade C recommendation. 21
Arthritis of the hallux MTPJ is a progressive condition, and concerns exist about postoperative stiffness, recurrent pain, and need for revision surgery following cheilectomy. 3 There has been increased interest in adjuvants, modifications, and alternatives to cheilectomy to attempt to improve on results. Various soft tissue interpositions as well as a polyvinyl alcohol hydrogel implant have been described. 3,4,14,21 An alternative to mechanical adjuvants is biologic supplementation to attempt to mitigate postoperative inflammation and scar formation.
Cryopreserved amniotic membrane (AM) and umbilical cord (UC) allografts have gained interest as a tissue with biologic properties for potential orthopedic applications. For consistency, previous studies by our authors referred to the umbilical cord allograft (Clarix CORD 1 K; Amniox Medical Inc, Miami, FL) used in this study as amniotic membrane–umbilical cord (AM-UC), and that same abbreviation will be used in this article as it is the more well-known term. 6,14
Basic science research has identified AM-UC tissue as having the ability to provide scaffold for epithelial migration, downregulate inflammatory cascade, inhibit fibroblast differentiation, prevent infection, and remain immune-privileged. 15 There are also small series in foot and ankle literature demonstrating effectiveness to support total ankle arthroplasty incision healing as well as pain relief in Achilles tendinosis and plantar fasciitis. 6,8,16,27 In addition, safety was demonstrated across 124 various foot and ankle procedures. 11
Cryopreserved umbilical cord–amniotic membrane (UC-AM) allograft as a potential adjuvant to cheilectomy has been previously described. 14 Our hypothesis was that functional outcomes and range of motion would be improved when cheilectomy was supplemented with UC-AM. In addition, we hypothesized that supplementation with UC-AM would cause functional outcomes to improve quicker than cheilectomy alone secondary to the theoretical benefit of UC-AM tissue to decrease postoperative inflammation.
Methods
After obtaining institution review board (IRB) approval, eligible patients (Table 1) were recruited to the study and randomized to one of 2 treatment groups in a 1:1 ratio. Patients were indicated for surgery based on surgeon discretion, which across our practice meant dorsal impingement symptoms and lack of midrange pain, regardless of radiographic appearance. Patients were blinded throughout the follow-up period to treatment group, with treatment revealed on completion of the 12-month follow-up visit. Because of the nature of the operative intervention, the investigator, research staff, and surgeons were not blinded. Preoperative demographic data and patient-reported outcome measure scores were collected at enrollment. American Orthopaedic Foot & Ankle Society (AOFAS) total score was the primary outcome measure. Foot Function Index (FFI) and visual analog scale (VAS)–pain (walking, waking, and end of day) scores were also collected. Preoperative hallux MTPJ motion was also assessed using lateral radiographs in maximal forced dorsiflexion and plantarflexion, which was performed by the treating surgeon at enrollment (Figure 1).
Study Inclusion and Exclusion Criteria (Determined by Treating Surgeon and Reviewed by Study Personnel).

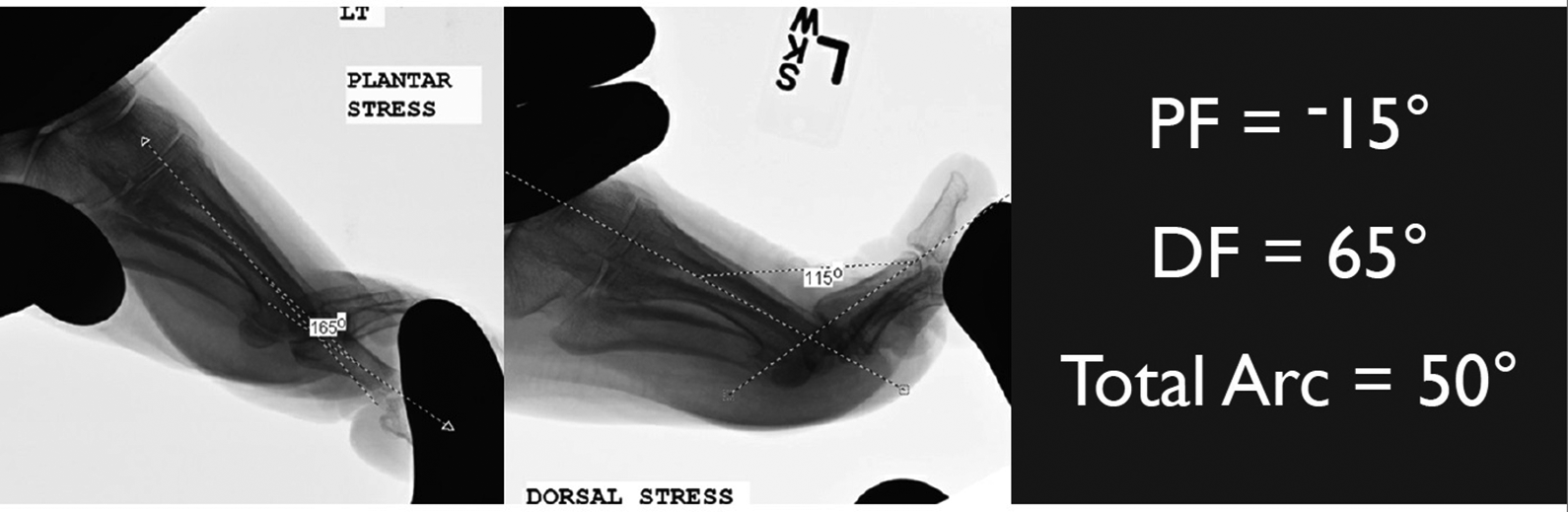
Imaging software (iSite PACS, Philips; Amsterdam, Netherlands) was used to measure maximal metatarsophalangeal joint dorsiflexion and plantarflexion angles determined under stress examination by treating surgeon wearing leaded gloves. DF, dorsiflexion; PF, plantarflexion.
Seventy-six patients (83 feet) were randomized following eligibility review, consent and enrollment. Fifty-one patients (7 bilateral) completed the study with 31 feet treated with cryopreserved umbilical cord (UC-AM) and 27 feet treated with cheilectomy alone (CA). PI or patient withdrawal was due to inadvertent unblinding, missed follow-up appointments, insurance/facility approval, or patient cancelation of surgery (Figure 2).
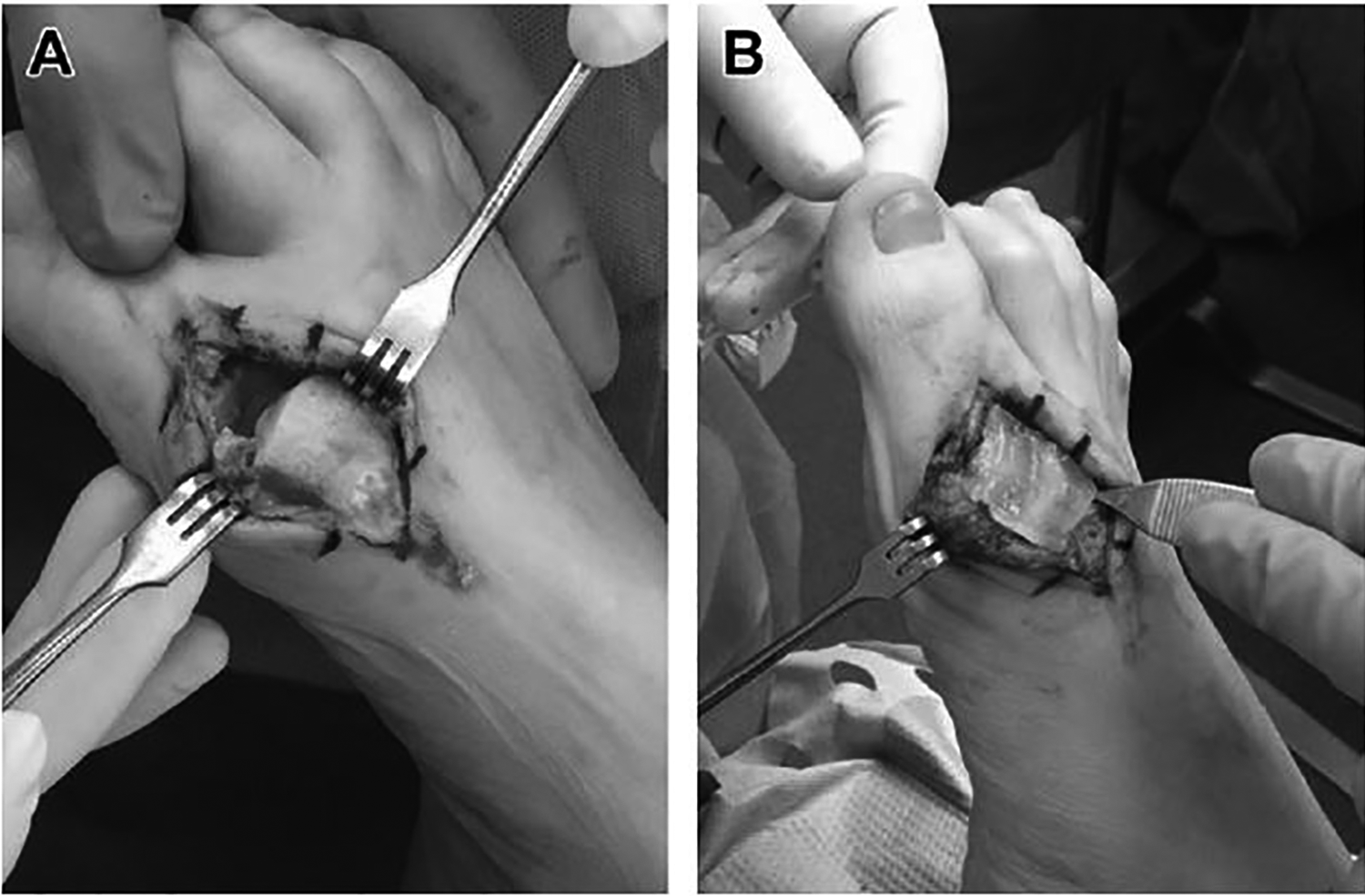
Intraoperative photographs of cheilectomy site and UC-AM implantation. (A) A standard cheilectomy site, and (B) the UC-AM graft in place before soft tissue closure. Reprinted with permission from Ferguson & Ellington. 14
One group (CA) underwent dorsal cheilectomy alone according to treating surgeons’ preferred method. 10 The hallux MTPJ was approached via standard midline dorsal approach. A capsulotomy was performed and extensor hallucis longus was mobilized and protected laterally. Osteophytes were debrided from dorsal metatarsal head and proximal phalanx. The medial and lateral gutters were debrided of loose bodies and hypertrophic synovium. A microsagittal saw was used to complete dorsal cheilectomy to remove approximately 25% of the metatarsal head (Figure 3A). Fluoroscopy was used to confirm adequate resection.
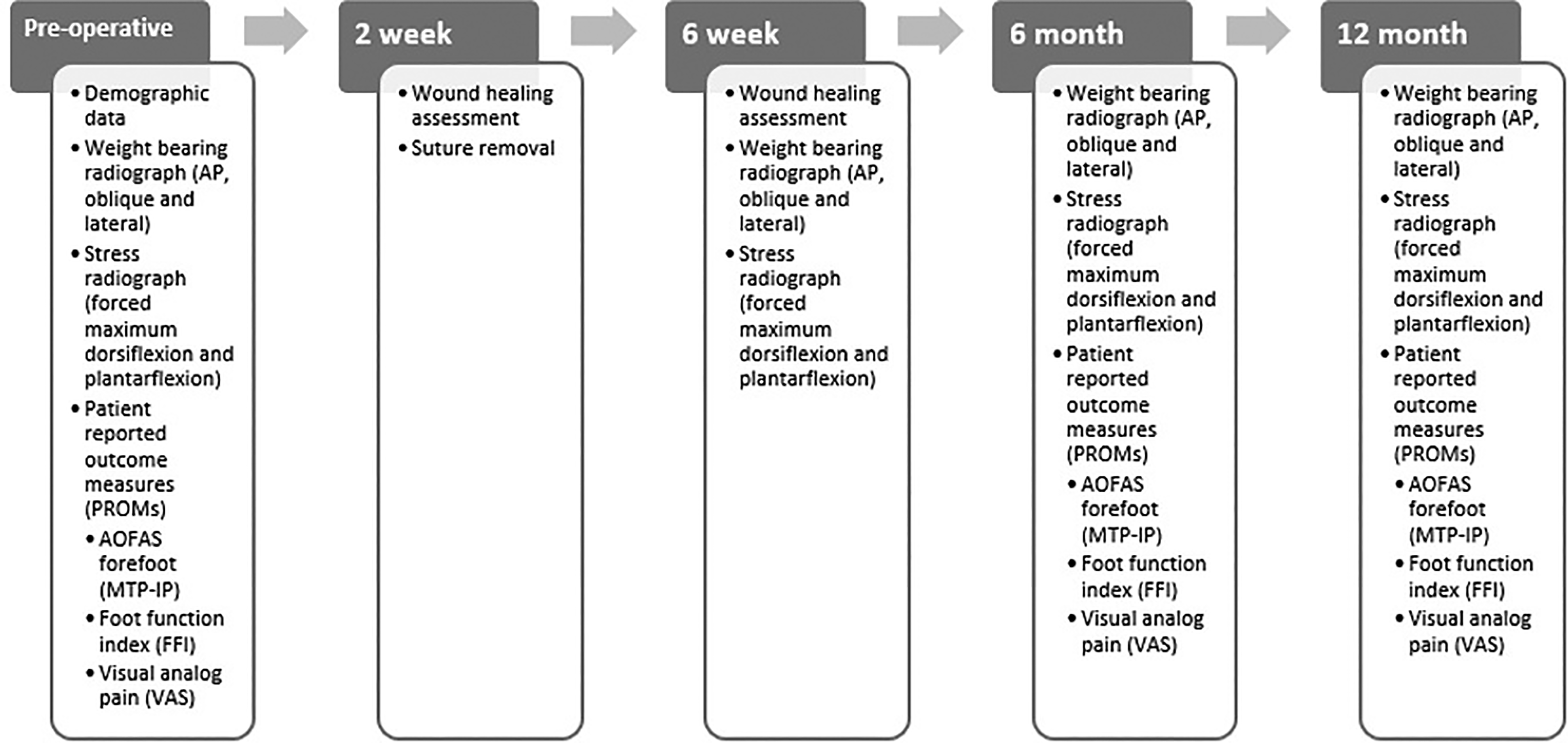
Flow chart showing clinical and patient reported outcome measure assessments and timepoints.
The other group (UC-AM) underwent dorsal cheilectomy in a similar fashion as CA with implantation of cryopreserved umbilical cord (Clarix CORD 2.5 × 2.5 cm 1 K; Amniox Medical Inc, Miami, FL) as described by the principal investigator previously. 14 The cryopreserved AM-UC was thawed per protocol and placed in an on-lay fashion over the exposed cancellous bony surface of the cheilectomy site (Figure 3B). The capsule was repaired with 2-0 Vicryl with a “stay stitch” passing through the cryopreserved umbilical cord.
Both groups were treated with the same postoperative protocol. Patients were weightbearing as tolerated in a postoperative shoe following surgery. Range of motion was initiated/encouraged beginning 5 days postoperatively. Patients were followed for 12 months postoperatively with clinical assessments and data collection obtained at 2 weeks, 6 weeks, 6 months, and 12 months (Figure 4). Wound healing was assessed at 2 and 6 weeks, and clinical notes were reviewed for details. Radiographs (weightbearing and stress range of motion) were obtained at 6 weeks, 6 months, and 1 year and clinical outcomes (AOFAS, FFI, VAS-pain) were assessed at 6 months and 1 year.
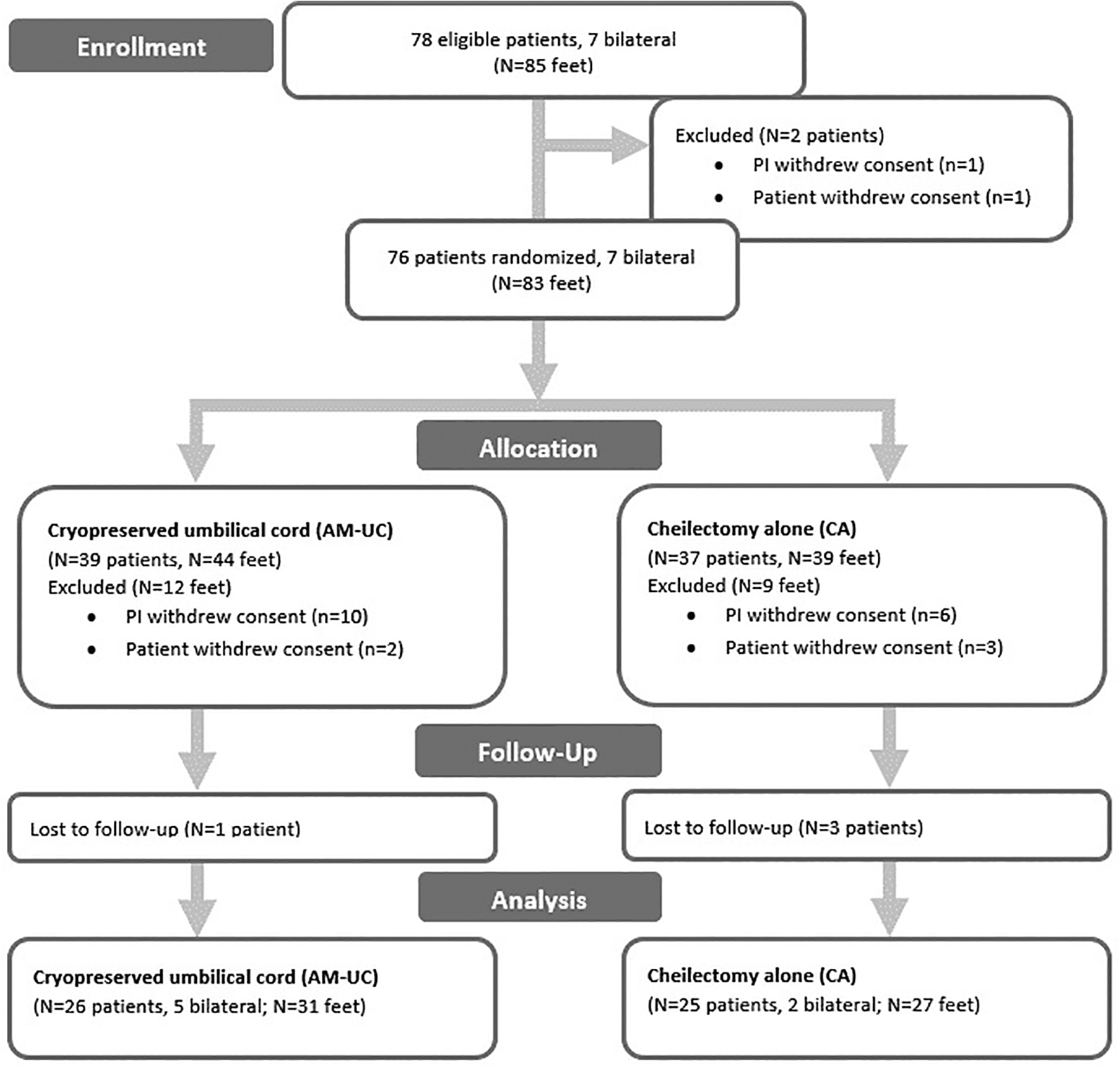
Flow chart showing details of enrollment, study group allocation, patients lost/withdrawn, and included in analysis.
Power analysis with G*Power software (Universität Düsseldorf, Germany) determined 27 feet per group were needed to detect a significant difference between AOFAS scores of 95 (AM-UC) and 85 (CA). Data were analyzed using statistical analysis software (SAS version 9.4, Cary, NC). AOFAS, FFI, and VAS scores were analyzed using Wilcoxon signed-rank test. Range of motion data were analyzed using 2-way analysis of variance with Tukey adjusted least squares means test. A P value of less than .05 was considered statistically significant.
Results
There were no reoperations in either group. There were 5 superficial wound issues within the first 6 weeks that were managed with close observation (1 AM-UC, 2 CA) or local wound care and oral antibiotics (2 AM-UC) at the treating surgeon’s discretion and resolved without further sequela.
AOFAS, FFI, and VAS-pain scores were not normally distributed; thus, median values are reported. Range of motion data were normally distributed and mean values are reported. Demographic data (Table 2) and preoperative VAS-pain scores were similar between groups (Table 2). AOFAS total score was slightly higher in the CA group than in the UC-AM group (69 vs 65, P = .05). FFI total score was lower in the CA group than in the UC-AM group (23.6 vs 28.7, P = .02).
AOFAS and FFI total scores improved in both groups from preoperative to 1 year (Table 3). AOFAS total scores improved from 65 to 87 at 6 months and 90 at 1 year in the UC-AM group and from 69 to 85 at 6 months and 81.5 at 1 year in the CA group. No significant difference was seen in 6-month results (P = .53), but 1-year results showed greater improvement in AOFAS scores in the UC-AM group (25 points) compared with the CA group (12.5 points, P = .003). There was also improvement between groups seen from 6 months to 1 year, with the UC-AM group gaining 3 points and the CA group losing 3.5 points (P = .005). FFI total scores improved from 28.7 to 5.9 at 1 year (22.8 points) in the UC-AM group and 23.6 to 4.9 (18.7 points) at 1 year in the CA group (P = .05). No significant difference was seen in the preoperative to 6-month results (P = .11) or in the 6-month to 1-year results (P = .47).
Preoperative Demographics, Outcome Scores, and Range of Motion Compared Between Study Groups.
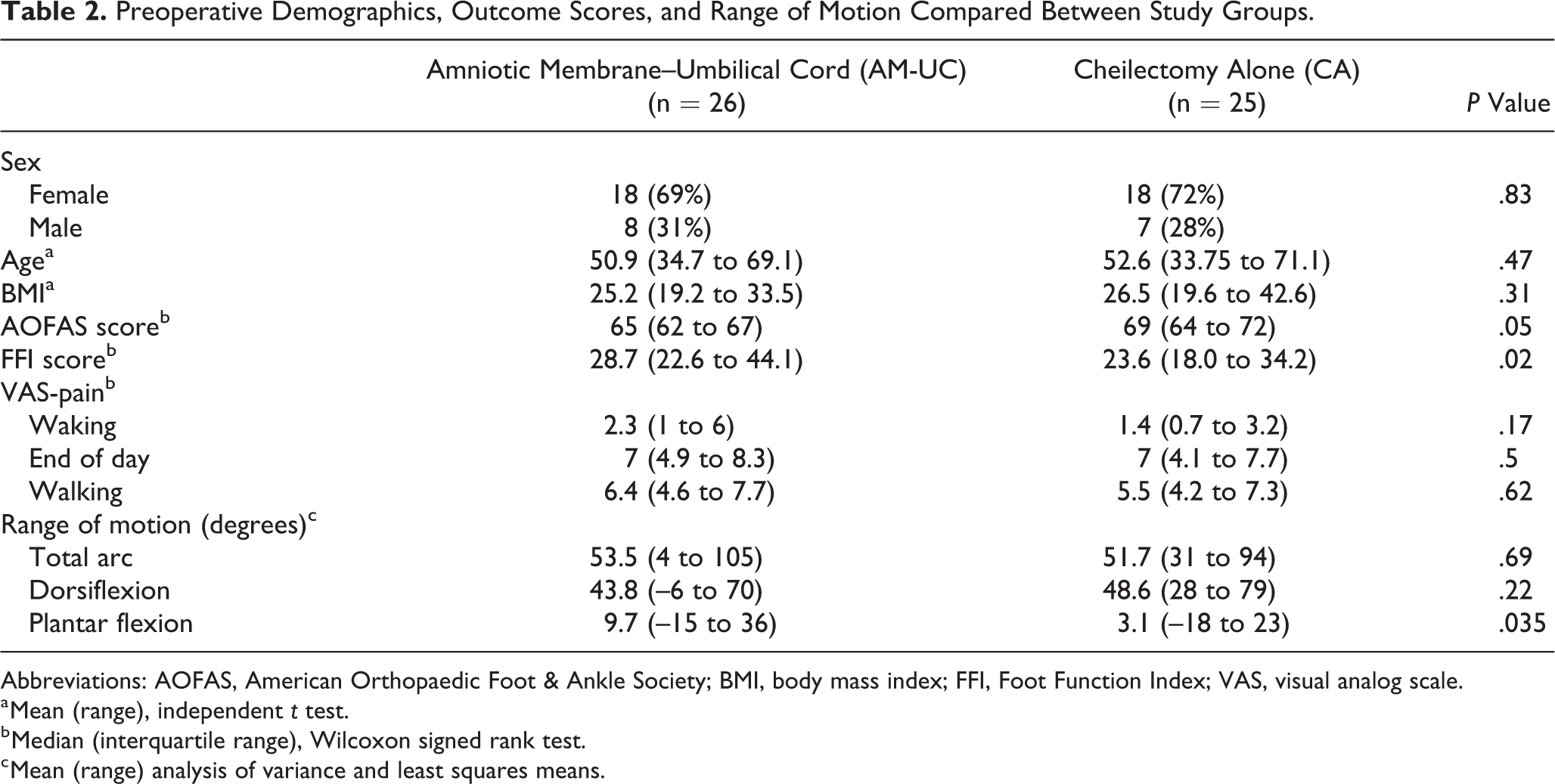
Abbreviations: AOFAS, American Orthopaedic Foot & Ankle Society; BMI, body mass index; FFI, Foot Function Index; VAS, visual analog scale.
a Mean (range), independent t test.
b Median (interquartile range), Wilcoxon signed rank test.
c Mean (range) analysis of variance and least squares means.
AOFAS and FFI Scores Compared Between Study Groups.a

Abbreviations: AOFAS, American Orthopaedic Foot & Ankle Society; FFI, Foot Function Index.
a Values are median with interquartile range Q1, Q3; Wilcoxon signed-rank test.
VAS-pain scores (waking, walking, and end of day) improved from preoperative to 1 year in both groups, but there was no difference between groups (Table 4). There was also minimal change in scores between 6 months and 1 year.
VAS-Pain Scores (on Waking, End of Day, and With Walking) Compared Between Study Groups.a

Abbreviations: Pre, preoperative; VAS, visual analog scale.
a Values are median with interquartile range Q1, Q3; Wilcoxon signed-rank test.
Total arc range of motion did not change between preoperation and 1 year postoperation (Table 5). At 6 months, the UC-AM group initially gained more dorsiflexion (at the expense of plantarflexion) compared to the CA group (dorsiflexion 10.7 vs 5.7 degrees, P = .008). Overall dorsiflexion gained was not different at 1 year (4.6 vs 5.8 degrees, P = .13) except that UC-AM group did demonstrate persistent loss of plantarflexion compared with the CA group (8.7 vs 3.3 degrees lost, P = .04).
Range of Motion (Degrees) on Stress Radiographs Compared Between Study Groups.a
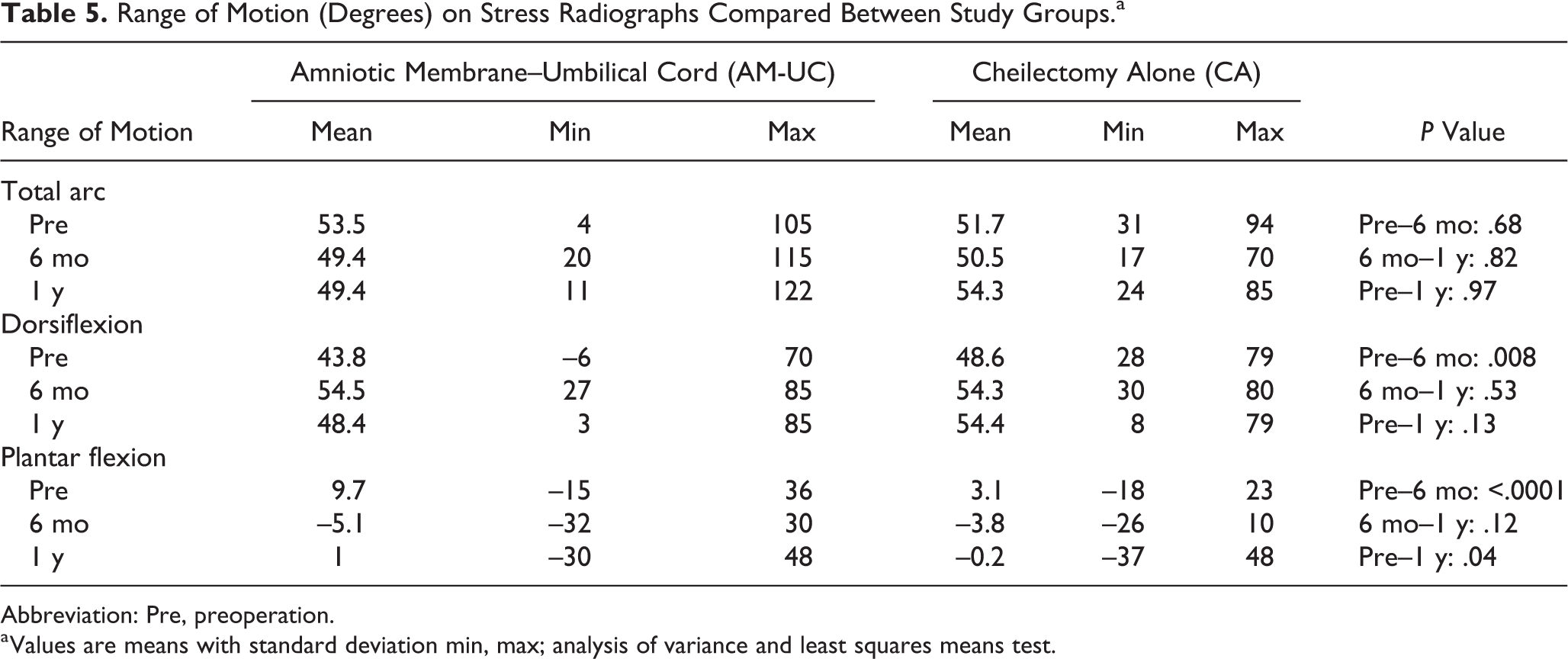
Abbreviation: Pre, preoperation.
a Values are means with standard deviation min, max; analysis of variance and least squares means test.
Following completion of the study, all charts were retrospectively reviewed for joint injections and/or additional procedures to the involved hallux (Table 6). Average follow-up from date of surgery to chart review was 45 months and was not different between groups (P = .51). One patient in the bilateral UC-AM group had follow-up in-office particulate UC-AM injection on one side and ultimately underwent bilateral polyvinyl alcohol implant placement. One patient in the CA group had 2 follow-up in-office corticosteroid injections and was scheduled for hallux MP joint fusion. Another 2 patients (3 feet) in the UC-AM group had follow-up in-office corticosteroid injections, with one performed during the study period at 6-month follow-up. Overall, 5 feet (3 patients) in the UC-AM group vs 2 feet (2 patients) in the CA group had an injection following surgery. Two feet (1 patient) in the UC-AM group had a second hallux surgery vs 1 in the cheilectomy alone group, though another was scheduled for hallux metatarsophalangeal fusion.
Retrospective Chart Review of Study Participants After Completion of Trial.
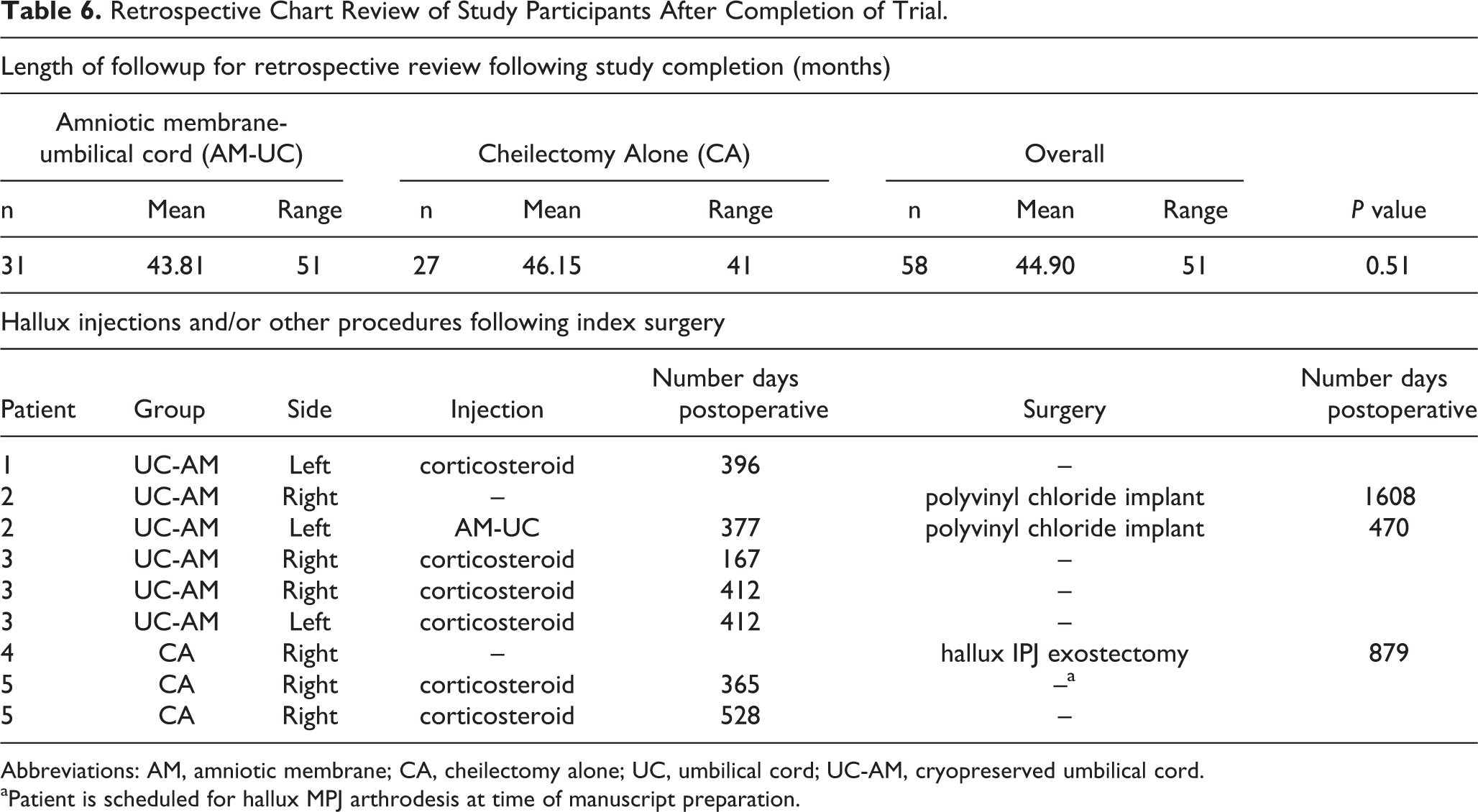
Abbreviations: AM, amniotic membrane; CA, cheilectomy alone; UC, umbilical cord; UC-AM, cryopreserved umbilical cord.
aPatient is scheduled for hallux MPJ arthrodesis at time of manuscript preparation.
Discussion
Cheilectomy remains a treatment option in the operative algorithm for symptomatic hallux rigidus. Multiple studies across the last 2-3 decades have demonstrated improved outcomes from this procedure. Our experience anecdotally aligned with these findings. However, research and our experience also indicate that some patients do not have the desired result, most commonly because of persistent pain and stiffness as well as complaints related to postoperative swelling. In the first prospective randomized and blinded cheilectomy trial reported, we aimed to compare outcomes of patients undergoing dorsal cheilectomy alone and dorsal cheilectomy supplemented with cryopreserved UC-AM allograft. The UC-AM group still received standard cheilectomy, which was supplemented with UC-AM. This is not an interposition, but rather an adjuvant that was hypothesized to improve functional outcomes by decreasing postoperative inflammation and scarring.
Our results show that functional outcomes (AOFAS and FFI) were improved more in the UC-AM group compared to cheilectomy alone. However, this difference was not significant until 1 year. This disproved our hypothesis that supplementation with UC-AM would improve results quicker. To our knowledge, no other study has looked at time to maximal recovery, and our data would suggest that it takes up to a year following cheilectomy with or without amniotic tissue supplementation. It is unclear why the cheilectomy alone group’s AOFAS score declined slightly from 6 months to 1 year. Sidon et al, 25 when looking at outcomes of 169 feet following cheilectomy, found that in 75% of cases that experienced recurrence (21/28 feet), it occurred prior to 2 years.
Our functional outcome measures are consistent with those reported in literature following cheilectomy. Feltham et al 13 reported an average AOFAS score of 80 for 67 patients who underwent cheilectomy, with higher scores seen in patients 60 years and older. Coughlin and Shurnas 10 reported improvements in the average AOFAS score from 45 to 90 for 80 patients who underwent cheilectomy. In their study, preoperative AOFAS scores were determined retrospectively from chart review, whereas in our study it was obtained at the time of preoperative visit, which could account for the differences in our cohort. Regardless, their patient cohort’s postoperative AOFAS scores are similar to our cohort.
Similarly, Easley et al 12 reported that the average AOFAS score improved from 45 to 85 for 66 patients who underwent cheilectomy. Lau et al 19 reported an average postoperative AOFAS score of 78 and an FFI score of 21 when they compared 24 cheilectomies with 11 interpositional arthroplasties. The AOFAS score is similar to our cohort, but postoperative FFI was better (lower value is better) in our cohort.
Chan et al 9 found that when evaluating patients undergoing hallux valgus surgery, the minimal clinically significant difference was 8.4 for AOFAS scores when differentiating good to excellent outcomes from fair to poor outcomes. The AOFAS score improvements in our study were 25 for amniotic tissue and 12.5 for cheilectomy alone, which would suggest that the difference in our study was not just statistically significant but also clinically significant.
VAS-pain was improved in both groups, and there was no difference between groups, and the improvement was seen by 6 months and did not change significantly afterward. Similarly, total arc of motion was not different between groups and not different from preoperation to postoperation. It does appear that there was a shift of the arc of motion, where dorsiflexion was gained at the expense of plantarflexion, which was interestingly worse in the UC-AM group. This could have been related to the fact that they also started with worse plantarflexion prior to surgery. This would argue that improved outcome measures following cheilectomy are about more than pain and range of motion as these were not different between groups despite the differences in AOFAS and FFI scores.
Nonetheless, the lack of a large improvement in range of motion is different from that seen in literature previously and disproved our hypothesis that range of motion would improve with AM-UC supplementation. When looking at dorsiflexion alone, 4.6 and 5.8 degrees of dorsiflexion were gained in AM-UC and cheilectomy groups, respectively, in our study. Alternatively, Canseco et al 7 reported improved range of motion in addition to improved gait mechanics with 20 cheilectomy patients who underwent pre- and postoperative gait analysis. In an anatomic study, Heller and Brage showed dorsiflexion improved from 68 to 78 degrees following cheilectomy. 18 Nawoczenski et al showed that dorsiflexion during gait improved approximately 10 degrees in their gait analysis study of 15 cheilectomy patients, with additionally noted improvements in plantar pressure loading distribution. 22 Easley at al 12 reported clinically assessed dorsiflexion improved 20 degrees in 52 cheilectomy patients. Coughlin and Shurnas similarly reported goniometer-assessed dorsiflexion improvement of 24.5 degrees. 10 This is an important consideration when counseling patients preoperatively as it has been our practice to advise patients that range of motion does not reliably improve following cheilectomy.
Minor wound problems were encountered in both groups, which improved with local wound care. This is different from that seen elsewhere in the literature, where amniotic tissue has been shown to decrease wound complications, notably in total ankle arthroplasty. 6 Similarly, the use of amniotic tissue for wound healing applications has been well documented in several Level I studies in the wound healing literature. 24,28 This potentially could relate to the early initiation of range of motion at 5 days postoperatively, which can irritate an operative incision.
No unplanned return trips to the operating room occurred during the study period, and no patients were converted to arthrodesis, though one patient was scheduled for arthrodesis at the time of manuscript preparation. Evaluating revision rates is limited because of the short follow-up of this study. Previous reported rates of conversion to arthrodesis ranged from 2/20 (10%), 4/67 (6%), to 6/169 (4%). 13,22,25 In a meta-analysis, Roukis reported 62/706 (8.8%) being converted to arthrodesis. 23
The strength of this study is the prospective randomized design. We used established outcome measures to evaluate differences, and patients were blinded until completion of the trial at 1 year.
There are several limitations in our study. Surgeon blinding is challenging in any operative study. Outcome tools were obtained by a staff member from the research office, but range of motion stress radiographs were obtained by the treating surgeon. Radiographic measurements were all made by someone not involved in the patients’ care however—either a foot and ankle fellow or senior resident who ultimately completed a foot and ankle fellowship. Certainly, there was likely variability in how each treating surgeon obtained the stress radiographs as it was obtained by manual manipulation. This measured arc of motion also does not necessarily represent functional motion during gait. This was demonstrated by Vulcano et al, 26 who showed that clinical range of motion was on average 13 degrees less than the measured radiographic dorsiflexion. In addition, they assessed radiographic motion using standing, maximal joint dorsiflexion. One reason range of motion was not improved in the AM-UC group potentially relates to the fact that the natural history of osteoarthritis is unchanged. AM-UC to our knowledge has beneficial clinical applications for healing, but there is no study that has shown it affects articular cartilage.
In addition, as a study that investigated a degenerative arthritic condition, our follow-up is short, with the study concluding after 1 year. Sidon et al 25 recently evaluated outcomes following cheilectomy, and in their survival analysis, they found that 16% of patients had recurrence of pain and when present, it occurred within 2 years in 75% of cases. To attempt to address this, all charts were retrospectively reviewed at the time this article was prepared. Data from this secondary review should be interpreted with caution, because follow-up after the study period was not routinely performed and represents significant selection bias.
The treatment groups were also not comparable preoperatively. The amniotic membrane group had worse AOFAS and FFI scores potentially creating a bias that would allow for more improvement in their postoperative scores. This is a limitation of the randomization process that could not be controlled for once the study was completed and demographic data were analyzed. However, we feel the data are still meaningful because the change from preoperative to postoperative was analyzed rather than the raw score. This difference in preoperative scores relates to heterogeneity of the enrolled patients.
It can also be argued that AOFAS and FFI are not ideal outcome measures for higher-functioning patients with forefoot complaints. Agel et al 2 and Baumhauer et al 5 described a ceiling effect when these are used in patients that function above the threshold of basic activities of daily living. In addition, the AOFAS position statement on outcomes measures favors alternative tools for the evaluation of patients. 1 However, these guidelines were not available when patients were enrolled in our study, and we believe there is still validity as we obtained prospectively collected pre- and postoperative scores allowing comparison.
Lastly, patients were not scored according to either of the reported classification schemes, which limits the ability to potentially stratify results based on severity, which has been previously reported in other studies to affect outcomes. Although patients were not formally scored at enrollment, we reserved cheilectomy for Coughlin and Shurnas grade III disease or less and surgeons only enrolled patients that met indications for cheilectomy. Sidon et al supported this treatment algorithm in their long-term follow-up study as did Coughlin and Shurnas for grade III disease with >50% preserved articular cartilage. 10,25 Nonetheless, even with including grade III patients, without stratifying them, this could have impacted our results as grade III is known to have worse outcomes following cheilectomy. 12
Cost analysis was not performed as part of this study because the cost of AM-UC allograft was covered by the study. However, the authors acknowledge that cost is an important consideration when considering adjuvants to procedures. Further research would be needed on this cohort to see if it delays arthrodesis or if there are a subset of patients that benefit more from AM-UC than others to help guide its use in the future.
Conclusions
We present the results of the first randomized and blinded prospective study of cheilectomy surgery patients. There was improvement in pain, AOFAS, and FFI scores in all patients with statistically significant improvement at 1 year in AOFAS and FFI scores in the AM-UC group compared with the CA group. This was despite minimal change in range of motion in both groups. When appropriately selected, cheilectomy remains a good option for patients with symptomatic hallux rigidus. Cryopreserved AM-UC tissue is a potential adjuvant to cheilectomy to modulate inflammation and scarring, with 1-year results showing improvements in functional outcome scores.
Supplemental Material
Supplemental Material, sj-pdf-1-fao-10.1177_2473011420967999 - Cheilectomy With or Without Cryopreserved Amniotic Membrane–Umbilical Cord Allograft for Hallux Rigidus
Supplemental Material, sj-pdf-1-fao-10.1177_2473011420967999 for Cheilectomy With or Without Cryopreserved Amniotic Membrane–Umbilical Cord Allograft for Hallux Rigidus by Sara Heintzman Galli, Chad M. Ferguson, W. Hodges Davis, Robert Anderson, Bruce E. Cohen, Carroll P. Jones, Susan Odum and J. Kent Ellington in Foot & Ankle Orthopaedics
Footnotes
Ethics Statement
Ethical approval for this study was obtained from institutional review board at the Orthocarolina Research Institute (OCRI) (#Pro00020300).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W. Hodges Davis, MD, reports grants from Tissue Tech, during the conduct of the study; other from Amniox; and Amniox: Paid consultant; Research support. Robert Anderson, MD, reports grants from Tissue Tech, during the conduct of the study; other from Amniox; and Amniox: Paid consultant. Bruce E. Cohen, MD, reports grants from Tissue Tech, during the conduct of the study; other from Amniox, other from Tissue Tech; and Amniox: Paid consultant. Tissue Tech: Stock or stock Options. Carroll P. Jones, MD, reports grants from Tissue Tech, during the conduct of the study; Amniox, outside the submitted work; Amniox: Paid consultant. J. Kent Ellington, MD, reports other from Amniox, during the conduct of the study. Susan Odum, PhD, reports grants from Tissue Tech, during the conduct of the study. ICMJE forms for all authors are available online.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research support toward this study was received from TissueTech (Miami, FL).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
