Abstract
Background:
A novel biointegrative implant was developed for proximal interphalangeal joint (PIPJ) arthrodesis to treat hammertoe deformity. Composed of continuous reinforcing mineral fibers bound by bioabsorbable polymer matrix, the implant demonstrated quiescent, gradual degradation with complete elimination at 104 weeks in animal models. This prospective trial assessed the implant’s safety, clinical performance, and fusion rate of PIPJ arthrodesis for hammertoe correction.
Methods:
Twenty-five patients (mean age 63.9±7.5 years) who required PIPJ arthrodesis were enrolled at 3 centers. Outcomes included radiographic joint fusion, adverse events, pain visual analog scale (VAS) score, Foot and Ankle Ability Measure (FAAM) Activities of Daily Living (ADL) score, and patient satisfaction. Patients were evaluated 2, 4, 6, 12, and 26 weeks postoperatively.
Results:
Twenty-two patients (88%) achieved radiographic fusion at 26 weeks. All joints (100%) were considered clinically stable, with no complications or serious adverse events. Pain VAS improved from 5.3±2.5 preoperatively to 0.5±1.4 at 26 weeks postoperatively. FAAM-ADL total scores and level of functioning improved by mean 19.5±19.0 points and 24.4±15.7 percentage points, respectively, from preoperation to 26 weeks postoperation. Improvements in pain VAS and FAAM scores surpassed established minimal clinically important differences. All patients were very satisfied (84%) or satisfied (16%) with the surgery. Patient-reported postoperative results greatly exceeded (72%), exceeded (20%), or matched (8%) expectations.
Conclusion:
This prospective, multicenter, first-in-human clinical trial of a novel biointegrative fiber-reinforced implant in PIPJ arthrodesis of hammertoe deformity demonstrated a favorable rate of radiographic fusion at 12 and 26 weeks, with no complications and good patient-reported clinical outcomes.
Level of Evidence:
Level IV, prospective case series.
Introduction
Hammertoe deformity results from biomechanical dysfunction, 4,15 leading to flexion deformity of the proximal interphalangeal (PIP) joint and hyperextension of the metatarsophalangeal (MTP) joint. When nonoperative treatment fails, surgery is considered to relieve pain, realign the toe, and correct deformity, primarily by PIP joint arthrodesis. 19,43 However, consensus on optimal fixation is lacking. 14
Kirschner wire (K-wire) fixation is considered the standard treatment for correction of hammertoe deformity. 21,28,44 Although inexpensive and easy to insert, percutaneous K-wires protrude from the toe during the initial 6-week recovery period and require removal. 24 Complications include wire site infection, wire migration, wire breakage, and nonunion or malunion. 24,28,44,46 Additionally, K-wire fixation with only 1 wire does not provide rotational stability, which can lead to malalignment, relapse of deformity, and a need for revision surgery. 21,24 To address the limitations of K-wire fixation, a variety of intramedullary internal fixation devices that do not require removal have been developed to improve the standard of care. These PIP joint fixation devices include permanent implants made from a variety of metals (eg, steel, titanium) 10,12,16,17,22 or polyetheretherketone (PEEK). 3 Allograft implants have also been developed. 25,31,34 Most of these implants have not demonstrated improved complication and revision rates when compared to K-wire fixation. 14,19,21,33,34,44,45
A novel class of biointegrative fixation implants has been developed to provide strong, secure fixation while gradually integrating into native tissue. These implants are engineered from continuous reinforcing mineral fibers bound together by a bioabsorbable polymer [poly(
A biointegrative fiber-reinforced implant has been designed to operatively treat hammertoe deformity through PIP joint arthrodesis. The implant bridges the proximal phalanx to the middle phalanx to support fusion of the resected joint. An animal model of physiological load-bearing osteotomy fixation demonstrated bone fixation and fusion with the fiber-reinforced implant were equivalent to those of standard stainless-steel implants. 6 Animal studies also demonstrated gradual, progressive degradation of the fiber-reinforced implant with complete elimination by 104 weeks. 6
The purpose of this prospective clinical study was to assess the safety, fusion rate, and clinical performance of this novel, biointegrative, fiber-reinforced fixation implant in PIP joint arthrodesis of hammertoe deformity. The primary aim of this clinical investigation was to determine the radiographic fusion rate and serious adverse events. In addition, patient-reported clinical outcomes and satisfaction were assessed.
Methods
Study Design
This multicenter, prospective, single-arm, open-label study was conducted at a hospital in Slovenia and 2 centers in Spain between December 2018 and October 2019. Patients aged 18-75 years who required PIP joint arthrodesis for correction of hammertoe deformity of the second, third, or fourth toe and were able to provide written, informed consent were eligible for inclusion in the study. Further inclusion and exclusion criteria are shown in Table 1. Recruitment lasted until 25 patients were treated with the fiber-reinforced hammertoe fixation implant (Figure 1). This study adhered to the Declaration of Helsinki and was approved by the local ethics committee at each institution.
Study Inclusion and Exclusion Criteria.


Patient disposition chart.
During the screening visit, the patient demographic data, medical history, medication information, and preoperative laboratory data were recorded. A foot and toe assessment and a pain assessment were performed (ie, “baseline”). Radiographs were taken and evaluated, and computed tomography (CT) scans were ordered at the surgeon’s discretion.
Patient Demographics
Twenty-nine patients were screened for this study (Figure 1). Three patients were excluded because of vitamin D deficiency. One patient was excluded as unable to comply with the follow-up schedule. Thus, 25 patients were enrolled, including 24 women (96%). The mean age of the cohort was 63.9 ± 7.5 (range: 48.6-74.2) years (Table 2). The hammertoe deformity affected the second toe in 24 patients (96%) and the third toe in 1 patient (4%). All patients (100%) were available for follow-up at 12 weeks and 26 weeks.
Patient Demographics Preoperatively.

a Shown as count and percentage.
Study Device
The biointegrative hammertoe fixation implant (OSSIOfiber ® Hammertoe Fixation Implant, OSSIO Ltd, Caesarea, Israel) is composed of continuous, reinforcing, natural mineral fibers (SiO2, Na2O, CaO, MgO, B2O3, and P2O5; approximately 50%), bound with PLDLA polymer matrix (approximately 50%). The implant used in this study has a hexagonal cross section with a nominal dimension of 2.9 mm (Figure 2A). The ribbed design permits implant fixation in the phalangeal canal of the toe. The internal structure of the implant consists of layers of oriented continuous fibers (Figure 2B) that provide mechanical strength through the bone healing process. Gradual integration of the implant begins as early as 4 weeks and is completed by 24 months.

(A) The biointegrative, fiber-reinforced hammertoe fixation implant used in this study has a hexagonal cross-section of 2.9 mm. (B) Scanning electron microscope cross section of implant demonstrates continuous mineral fibers surrounded by polymeric material.
Operative Procedure and Postoperative Care
Two foot and ankle surgeons performed all surgeries. On the day of the surgery, patient eligibility was confirmed, medication information was recorded, and a foot and toe assessment was performed. For the surgery, local and/or regional anesthesia was administered. Following the site standard of care, some patients received prophylactic antibiotic.
During the surgery, fluoroscopy imaging was taken as required, based on the surgeon’s clinical judgment. The surgeon prepared the PIP joint for arthrodesis in the usual manner, with resection of both sides of the joint surface of the middle and proximal phalanges. The proximal phalanx diaphyseal canal and the middle phalanx diaphyseal canal were drilled to the appropriate depth. Using the implant holder, the proximal end of the fiber-reinforced hammertoe fixation implant was introduced into the proximal phalanx, parallel to the long axis of the drill hole to prevent bending (Figure 3). The middle phalanx was manually reduced over the distal end of the implant while applying slow, steady pressure until bone-to-bone contact was reached (Figure 3). After confirming that the implant was properly fitted and fixated to the bone, the surgeon completed the procedure using routine soft tissue closure. Any adverse events during the procedures were recorded. Dorsoplantar, lateral, and lateral oblique radiographs were taken immediately following the procedure. All patients were fitted with a protective shoe for 6 weeks.
Nineteen of 25 patients (76%) underwent concomitant procedures for various first ray deformities, including but not limited to scarf, Akin, or Chevron osteotomies 20 and first MTP joint fusion. 38

Hammertoe correction procedure. (A) The implant is inserted into the proximal phalanx. (B) Implantation into the proximal phalanx is completed. (C) The middle phalanx is mounted onto the distal end of the implant.
Outcomes Measures
The primary performance endpoint was radiographic PIP joint fusion at 12 and 26 weeks, rated as either fusion (≥50% osseous bridging) or no fusion (<50% osseous bridging) as assessed on radiographs and CT scans by the 2 surgeons who performed the surgeries and 2 other foot and ankle surgeons who had no direct contact with the patients. Radiographs were taken preoperatively, immediately postoperatively, and at 2, 6, 12, and 26 weeks follow-up. CT scans were taken at the 12- and 26-week follow-ups.
The primary safety endpoint was the rate and nature of serious adverse events throughout the study period. Any adverse events were recorded intraoperatively, immediately postoperatively, and at all follow-up visits.
Secondary endpoints included several patient-reported outcome measures: a visual analog scale (VAS) for pain with 0 representing no pain and 10 representing worst pain; the Foot and Ankle Ability Measure and Activities of Daily Living subscale (FAAM-ADL) questionnaire (total score, and percent level of functioning) 30 ; and a satisfaction questionnaire. When responding to the outcome questionnaires, patients were reminded to report solely based on the afflicted hammertoe, to minimize any bias due to concomitant procedures.
Patients returned for follow-up visits at 2, 4, 6, 12, and 26 weeks postoperatively. Medication information, any adverse events, and pain VAS scores were recorded at each visit. A foot and toe assessment was also performed and healing progress assessed. The FAAM-ADL questionnaire was completed preoperatively (ie, baseline) and at 12 and 26 weeks postoperatively. The satisfaction questionnaire was completed at 26 weeks’ follow-up.
Data Collection and Statistical Analysis
A cohort of 25 patients with 26-week follow-up data was determined sufficient to evaluate the safety and performance of the fiber-reinforced hammertoe fixation implant. The information provided by this cohort provides adequate data to compare with published literature for this indication. As such, this study was not intended to statistically test hypotheses and was not powered for statistical significance.
Data were collected using an electronic data capture system (DataFax DF/Net Research, Inc, Seattle, WA) and analyzed using a Statistical Analysis Software, version 9.4 (SAS Institute Inc, Cary, NC). Continuous variables are recorded as mean, standard deviation (SD), median, and range. Categorical variables are recorded as count and percentage.
Safety analysis was performed on all patients for whom the study procedure was initiated. The performance analysis was performed on all patients who completed at least 1 postprocedure visit and excluded patients with a major entry violation that likely affected outcome, as determined by the Medical Monitoring Board.
Results
Fusion Rate and Safety Parameters
At 12 and 26 weeks, 22 patients achieved radiographic fusion of the PIP joint as defined on radiographs (Figure 4) and CT scans (Figure 5), for an overall fusion rate of 88% at final follow-up. All 25 treated PIP joints were evaluated as clinically stable by the end of the clinical investigation, and none required further clinical intervention.
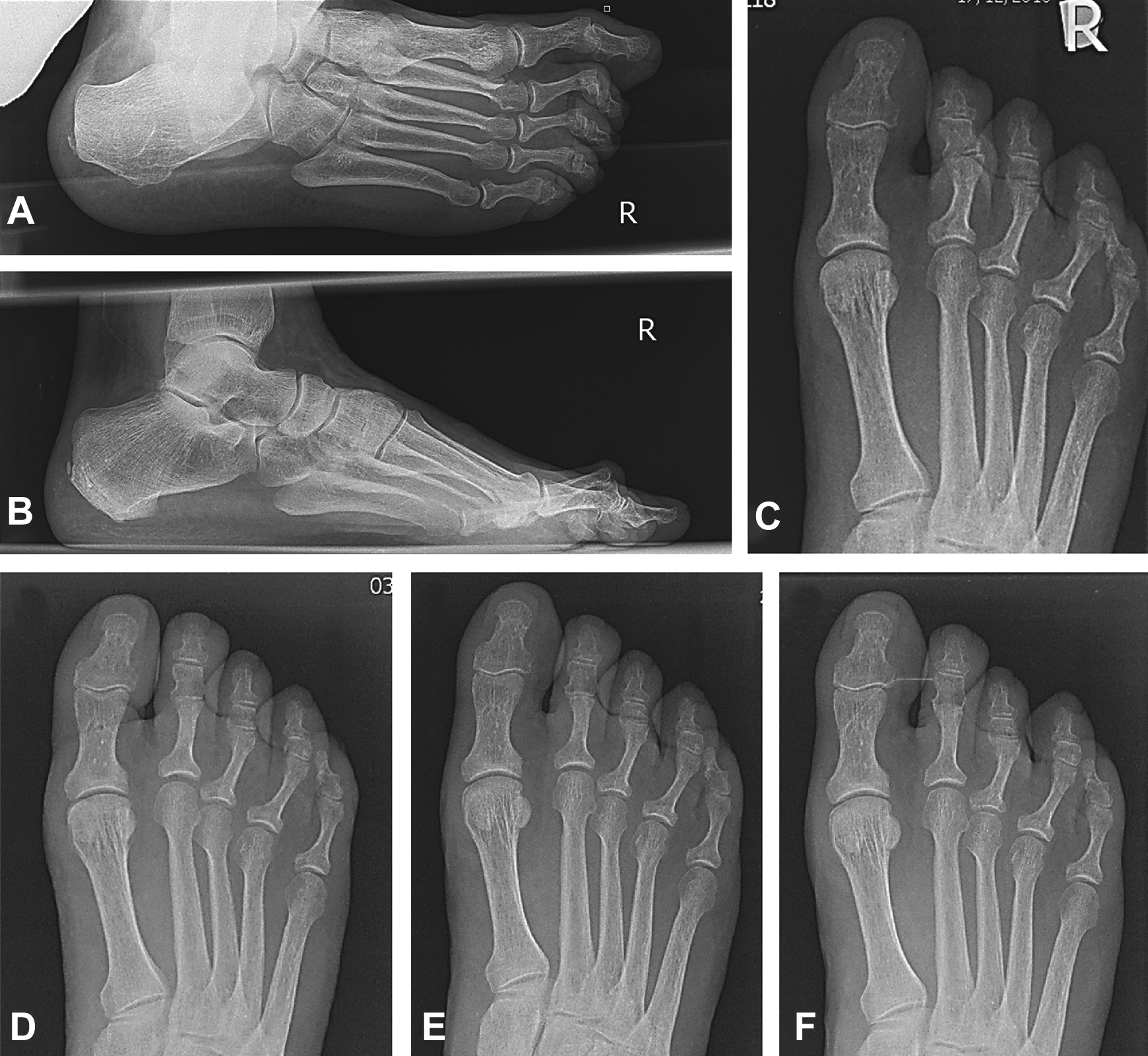
Weightbearing radiographs of a 68-year-old woman presenting with a hammertoe deformity of the second toe in the right foot. Preoperative radiographs: (A) lateral oblique, (B) lateral, and (C) dorsoplantar views. Postoperative radiographs at (D) 2 weeks, (E) 12 weeks, and (F) 26 weeks, demonstrating union.
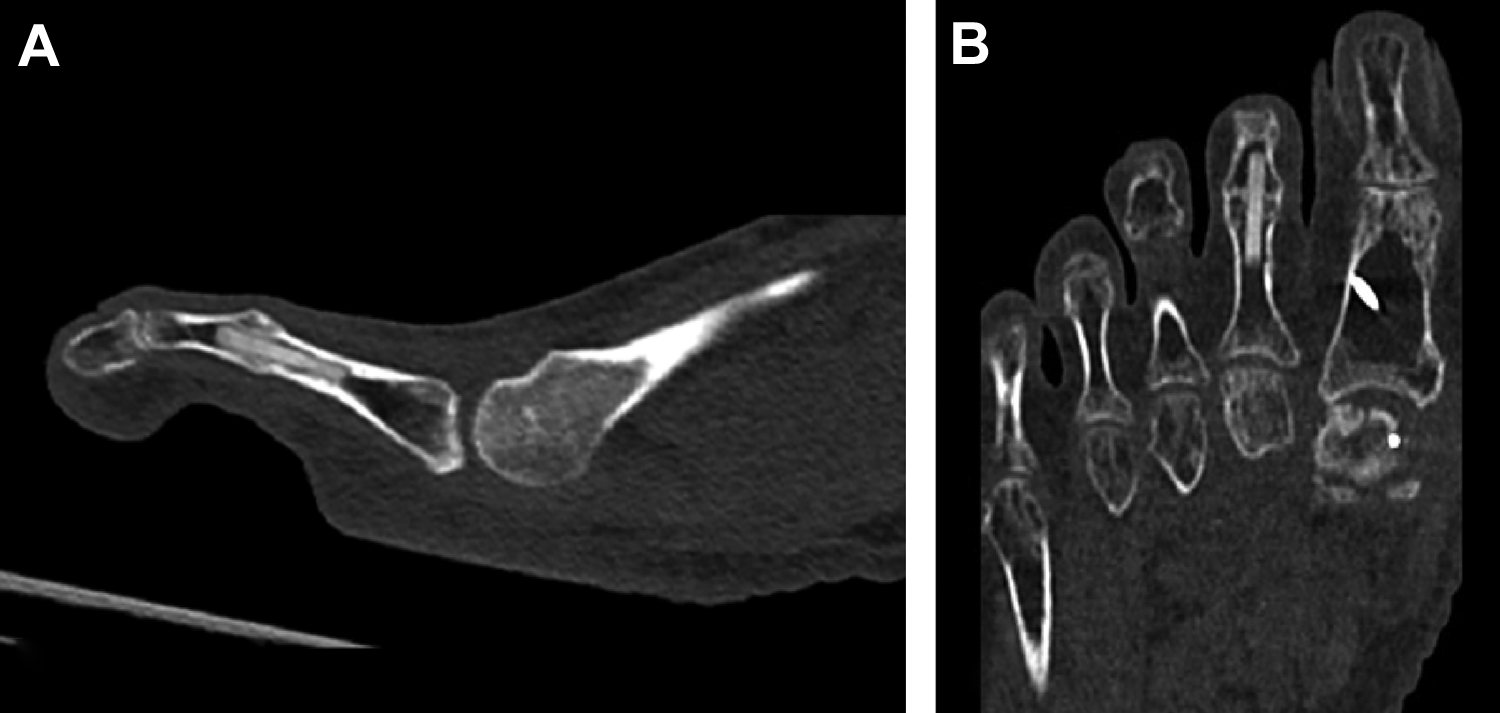
Postoperative computed tomographic scans of the second toe of a 59-year-old woman, at 26 weeks following proximal interphalangeal (PIP) joint arthrodesis, demonstrating union: (A) sagittal and (B) coronal axes.
All 25 patients underwent the hammertoe correction procedure using the fiber-reinforced hammertoe fixation implant, with no observed serious adverse events related to the device or the procedure.
One patient reported swelling at the PIP fusion site 43 days post-procedure, which was assessed clinically as being of mild severity. The period of protective shoe wear was extended to 3 months, and the swelling resolved by the end of the clinical investigation. The patient had not achieved bony fusion at 26 weeks.
Patient-Reported Outcome Measures
The mean pain VAS score improved from 5.3 ± 2.5 preoperatively to 0.4 ± 0.6 at 6 weeks and 0.5 ± 1.4 at 26 weeks (Figure 6). The protective shoe was discontinued at 6 weeks. A transient slight elevation in pain score was observed in 7 patients at the following visit and was not unexpected as patients returned to full normal activity.

Pain visual analog scale (VAS) scores preoperatively and at weeks 2, 4, 6, 12, and 26 postoperative (n = 25).
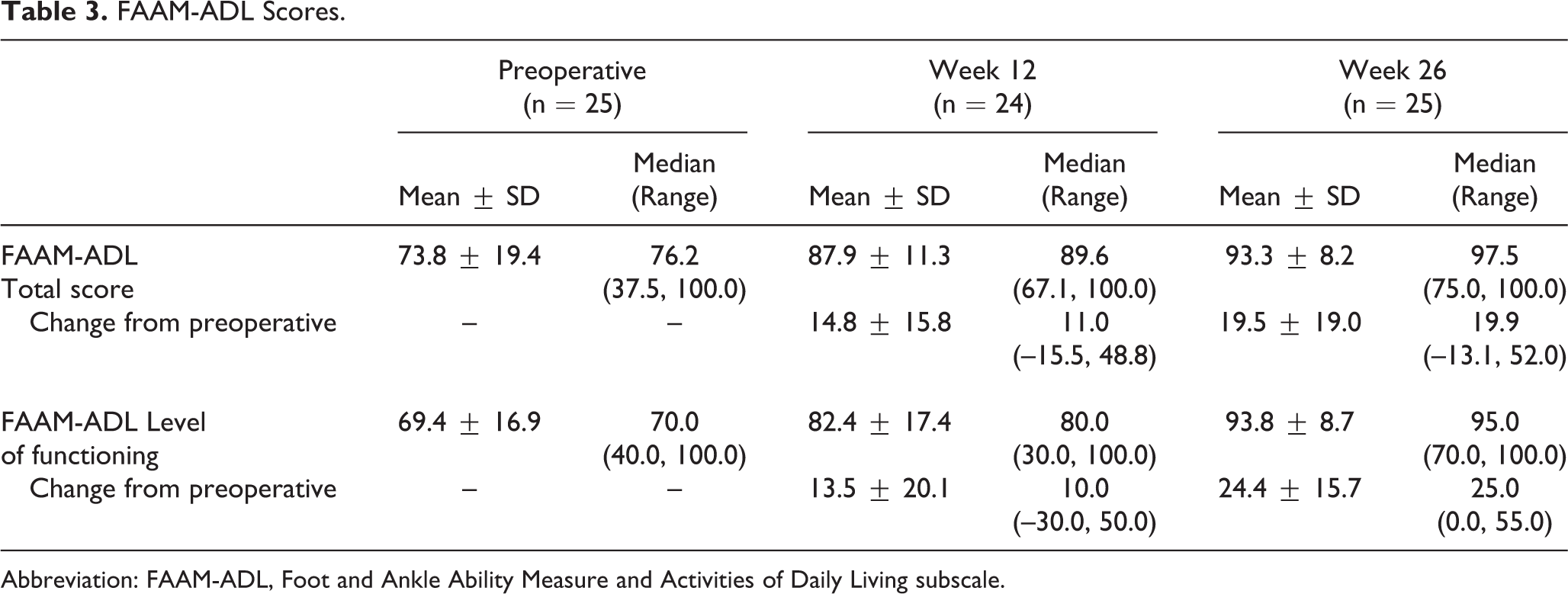
The mean FAAM-ADL total score was 73.8 ± 19.4 preoperatively and improved to 93.3 ± 8.2 at 26 weeks postoperation (Table 3), with a mean change of 19.5 ± 19.0 points. One patient was instructed to keep wearing the postoperative shoe past the 12-week visit, and the FAAM-ADL was therefore not completed by this patient at this visit. The FAAM-ADL percentage level of functioning also improved, from 69.4 ± 16.9 preoperatively to 93.8 ± 8.7 at 26 weeks, for a mean improvement of 24.4 ± 15.7 percentage points (Table 3).
FAAM-ADL Scores.
Abbreviation: FAAM-ADL, Foot and Ankle Ability Measure and Activities of Daily Living subscale.
All patients were either very satisfied (21/25; 84%) or satisfied (4/25; 16%) with the results of the procedure. Eighteen patients (72%) reported that their postoperative results greatly exceeded their expectations, 5 (20%) reported the results exceeded their expectation, and 2 (8%) reported the results matched their expectations. When asked about their ability to be more mobile, the surgery greatly exceeded, exceeded, or matched expectations for 10 (40%), 7 (28%), and 4 (16%) patients, respectively; 4 patients did not identify this as an expectation. Most patients were very likely (23/25; 92%) or likely (1/25; 4%) to choose this implant surgery again; 1 patient was somewhat likely to choose the surgery again.
Discussion
Use of a novel, biointegrative, fiber-reinforced implant for PIP joint correction arthrodesis in the treatment of hammertoe deformity was safe, with no serious adverse outcomes and an 88% fusion rate at 26 weeks postoperation. Patients reported substantial clinical improvements in pain and function, high satisfaction with the study device and procedure, and fulfilment of their expectations.
The radiographic fusion rate of 88% at 26 weeks’ follow-up with the fiber-reinforced implant is consistent with the higher end of published fusion rates for other intramedullary PIP joint implants. 2,9,10,12,16 -18,22,23,26,27,34,35,39 -42,45 Three studies of absorbable intramedullary implants in PIP joint arthrodesis reported fusion rates of 79%, 26 83%, 27 and 85%. 45 In 8 studies of intramedullary nitinol implants for PIP joint arthrodesis to correct hammertoe deformity, fusion rates ranged from 44% 34 to 100%, 2 and averaged around 79%. 2,9,23,34,35,40 -42 Reported fusion rates for 1-piece and 2-piece metal implants ranged from 22% 39 to 94% 10 and averaged 74% in 647 toes across 7 studies. 10,12,16 -18,22,39 In studies evaluating the use of allograft bone matrix devices for PIP joint arthrodesis, reported radiographic fusion rates were 37%, 34 96%, 31 and 97%. 25
Reported fusion rates for K-wire fixation are somewhat inconsistent, ranging from 7% 39 to 93% 11 with an average of 61% in a total of 604 toes across 8 studies of correction of hammertoe deformity. 2,11,13,22,29,34,39,42 It is important to note that complications of K-wire fixation include wire site infection, wire migration, and wire breakage, 24,28,44,46 which are not a concern with other implants. Furthermore, the lack of rotational stability with K-wire fixation with only 1 wire can lead to malalignment, relapse of deformity, and a need for revision surgery. 21,24
The pain VAS mean score of 0.4 at the 26-week follow-up also compares well with studies of other implants. 2,39 In a retrospective comparative study of PIP joint fusion for correction of hammertoe deformity, Richman et al 39 reported mean pain VAS scores of 1.7 and 1.8 at the 12-month follow-up for 1-piece implant (n=54) and K-wire (n=95) fixation, respectively. Angirasa et al 2 reported mean pain VAS scores of 0.33 following nitinol implant fixation and 1.6 following K-wire fixation for PIP joint arthrodesis to correct hammertoe deformity.
Patients in this study demonstrated good functional and clinical outcomes, with substantial improvements from preoperative evaluation to final follow-up. As the first study to obtain initial safety and performance data for this device, this study was not powered to determine statistical significance. Nevertheless, the observed changes from preoperative evaluation to 26 weeks surpassed established minimal clinically important differences (MCIDs) for both pain VAS and FAAM-ADL scores. Study patients achieved an approximately 90% reduction in pain VAS scores, where a reduction of ≥30% has been considered clinically successful. 5 The FAAM-ADL total score, which has an MCID of 8 points, 5 improved a mean of 19.5 ± 19.0 points in this study.
The device demonstrated a good safety profile, with no complications, no serious adverse events, and no safety-related withdrawals from the study. Only 1 patient experienced delayed swelling around the device site, which resolved with an extended period of protective shoe wear.
Quiescent bio-integration is a major challenge for a non-permanent orthopedic fixation implant. 6,36 The implant must be mechanically strong and robust enough to provide stable fixation, and yet be able to gradually integrate into the surrounding bone. 36 The biointegrative implant evaluated in this study is uniquely engineered of continuous reinforcing mineral fibers bound together by a bioabsorbable polymer PLDLA matrix to achieve high mechanical strength and gradual, progressive integration with the surrounding anatomy. 6 Its mineral content is substantially higher than that of other bioabsorbable implants and is composed entirely of minerals found in native bone. 7 Animal studies demonstrated the fixation performance of fiber-reinforced implants and fusion results to be comparable to standard stainless steel implants in a physiological load-bearing osteotomy fixation model. 6 Furthermore, the implant demonstrated a gradual and progressive degradation profile through complete elimination at 104 weeks, with the absence of local or systemic adverse tissue response. 6
The fiber-reinforced implant also provides safe and artifact-free imaging on radiographs, computed tomography, and magnetic resonance imaging. This newly introduced technology facilitates the use of a well-established operative technique while demonstrating high radiographic fusion rates, with no permanent implant material left behind. Thus, hardware removal procedures, which are quite common with some operative procedures, as well as pin site infection, pin migration, and pin breakage complications of K-wire fixation are avoided.
Limitations of this study include the small sample size and lack of a control group. Concomitant procedures at the great toe may have contributed slightly to the large improvements observed here, although patients were asked to complete the questionnaires based only on the operated hammertoe. Nevertheless, the prospective design of this trial is a strength, and the substantial improvements observed in patient-reported clinical outcome measures far surpassed the MCIDs of these measures.
Conclusion
This prospective, multicenter, first-in-human clinical trial of a novel, fiber-reinforced implant introduces a biointegrative alternative for PIP joint correction arthrodesis for treatment of hammertoe deformity. The implant demonstrated a favorable rate of fusion at 26 weeks, with no complications and substantial patient-reported improvements in pain and function.
Supplemental Material
Supplemental Material, FAO966311-ICMJE - Prospective, Multicenter, Clinical and Radiographic Evaluation of a Biointegrative, Fiber-Reinforced Implant for Proximal Interphalangeal Joint Arthrodesis
Supplemental Material, FAO966311-ICMJE for Prospective, Multicenter, Clinical and Radiographic Evaluation of a Biointegrative, Fiber-Reinforced Implant for Proximal Interphalangeal Joint Arthrodesis by Luke D. Cicchinelli, Jurij Štalc, Martinus Richter and Stuart Miller in Foot & Ankle Orthopaedics
Footnotes
Acknowledgments
The authors thank Dagmar Gross for assistance with preparation of this manuscript.
Ethics Approval
Ethical approval for this study was obtained from The National Medical Ethics Committee (NMEC) (Approval number: 0120-607/2017/8) in Slovenia; and Comité Autonómico de ética de la Investigación de Galicia (CAEI de Galicia) (Registration code: 2018/331) in Spain.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Luke D. Cicchinelli, DPM, reports personal fees from Ossio Ltd. (Israel), grants from Ossio Ltd. (Israel), during the conduct of the study. Jurij Štalc, MD, reports other from OSSIO Ltd. (Israel), during the conduct of the study. Martinus Richter, MD, PhD, and Stuart Miller, MD, report personal fees from OSSIO Ltd. (Israel), during the conduct of the study. ICMJE forms for all authors are available online.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by OSSIO Ltd (Israel).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
