Abstract
Background:
Although nicotine and tobacco use are well established to have adverse health effects, the effects on tendons are less well understood. The purpose of this study was to investigate the effect of chronic nicotine exposure on Achilles tendon (AT) and supraspinatus tendon (SS) physiologic and mechanical properties in a rodent model.
Methods:
Sprague-Dawley rats (n = 20) were exposed to either 0.9% saline or 36 mg/mL nicotine through an implantable osmotic pump for 12 weeks. At 12 weeks, the AT was imaged in vivo with contrast-enhanced ultrasound (CE-US) to assess vascularity. Bilateral AT and SS were then harvested for ex vivo biomechanical analysis. Statistical analysis was performed using Student t test and Mann Whitney U test.
Results:
AT CE-US demonstrated a significantly increased wash-in rate in the nicotine group compared to the saline group, indicating an increase in tissue perfusion rate in the nicotine group. AT percent stress relaxation, a measure of tendon viscoelasticity, was significantly increased in the nicotine group compared to the saline group. Similarly, there was a trend toward increased SS percent stress relaxation in the nicotine group compared to the saline group. No differences in other mechanical properties were observed.
Conclusion:
AT perfusion rate increased and both AT and SS viscoelasticity were altered with chronic nicotine exposure at a clinically relevant dose that models the average smoker in the United States (14 cigarettes per day). Further studies are necessary to link these properties with tendon degeneration and injury.
Clinical Relevance:
Chronic nicotine exposure affects tendon health. Patients should be counseled as such.
Introduction
According to the Centers for Disease Control and Prevention, up to 20% of the population in the United States are tobacco users. 7 Nicotine, a potent stimulant, is one of the main active ingredients in tobacco products. Previous research has used nicotine exposure as a surrogate for smoking. It has been shown to affect the musculoskeletal system in intervertebral disk degeneration, osteoporosis, arthritis, fracture healing, and delayed ligament healing. 10 Although smoking and nicotine have been described to increase the incidence of tendon injury in the clinical literature, there is a paucity of literature describing their effects on tendon physiologic and mechanical properties. 1,5,9
It is critical for orthopedic surgeons to understand the effects of chronic nicotine exposure on tendon health in order to improve patient outcomes. In a survey of patients undergoing orthopedic surgery, 64% of patients quit smoking and 16% reduced the rate of their smoking preoperatively after being informed of the risks. 20 Furthermore, smoking has been previously found to be predictive of poor outcomes following tendon repair. 15 Thus, knowledge of the effects of smoking on tendon health may be effectively used to modify patient behavior preoperatively and may also alter protocols postoperatively.
There are many anatomic variations in tendon tissue that affect their properties. These variations may affect treatment and rehabilitation protocols. Different tendon types have distinct anatomic environments (eg, intrasynovial vs extrasynovial, upper vs lower extremity) that have been previously shown to affect the mechanical load the tendon experiences, and mechanism in which the tendon is injured, and healing response. 2,4,8,14,18 For example, the Achilles tendon (AT) is an extrasynovial tendon of the lower extremity that experiences larger magnitudes of load compared to the supraspinatus tendon (SS), which is an intrasynovial tendon of the upper extremity. It is more common for AT injuries to occur within the midsubstance of the tendon whereas SS injuries more typically occur at the tendon’s insertion. AT typically undergoes healing from tendon to tendon while SS undergoes healing from tendon to bone. Basic science literature describes how early active range of motion is beneficial for tendon to tendon healing but detrimental for tendon to bone healing in a rat model. 4,6 Given these differences, it is imperative that orthopedic surgeons understand the effects of nicotine on various tendon types in order to customize treatment protocols for various scenarios encountered in clinical practice.
The purpose of this study was to establish an animal model to assess the effect of chronic nicotine exposure on AT and SS physiologic and mechanical properties. We hypothesized that chronic nicotine exposure is associated with degenerative changes to the tendon’s histology and detrimental to the tendon’s mechanical properties. Our secondary hypothesis was that AT and SS would be differentially affected by the chronic nicotine exposure because of their anatomic differences: AT is an extrasynovial tendon of the lower extremity, and SS is an intrasynovial tendon of the upper extremity. Ultimately, this model may provide insight into the mechanism predisposing tendon injury in tobacco users. This model may be used in future experiments to study interventions that alter nicotine’s effect on both uninjured and injured tendons.
Materials and Methods
Study Design
Prior to the commencement of the study, Institutional Animal Care and Use Committee approval was obtained. Twenty adult male Sprague-Dawley rats (Charles River Laboratories) with a mean 12 weeks of age (range 10-13) and mean weight 398 g (range 367-425) were used in this study. After arrival to the facility, the rats were allowed a 1-week acclimation period. Rats were housed in a conventional facility with 2 animals per cage, 12-hour light/dark cycles, were fed standard chow, and were provided water ad libitum. Rats were randomized to be exposed to either 0.9% saline (n = 10) or 36 mg/mL of nicotine ((-)-Nicotine, N3876
Contrast-Enhanced Ultrasound Acquisition
To assess the effect of nicotine exposure on tendon vascularity, unilateral right AT of 10 Sprague Dawley rats (n = 5 saline exposed, n = 5 nicotine exposed) were evaluated with contrast-enhanced ultrasound analysis (CE-US) in vivo, before sacrificing the animals, after 12 weeks of exposure. SS was not able to be assessed using CE-US because of the overlying acromion, which obscured visualization of the tendon. All data collection and analysis was performed by a single, blinded, operator (C.N.) to control for user variability. Animals were anesthetized using isoflurane, hair was removed from the right hind limb for ultrasonographic visualization, and a tail vein catheter was inserted and secured. The animal was placed on a heated imaging table with the ankle secured at 90° of plantarflexion. AT vascularity was assessed with adaptations to previously described methods. 3,11 Briefly, the signal of a 21-MHz ultrasound transducer (MS250, VisualSonics, Toronto, ON), with an axial resolution of 75 µM, was attached to a mount to image AT from the myotendinous junction to calcaneus. Video was recorded at 1 frame per second and contrast gain of 35 dB along the sagittal plane of the entire AT for 10 minutes following a bolus injection of 100 µL of microbubble contrast agent through the tail vein (Definity; Lantheus Medical Imaging, Billerica, MA) to capture the full wash-in and wash-out of the contrast in the tissue. The video clip, with a resolution of 2232 x 840 pixels, was loaded into a contrast analysis program (VevoCQ, VisualSonics, Toronto, ON) and a region of interest was traced around the entire tendon area. Perfusion parameters were then derived from this model, including peak enhancement, rise time, time to peak, wash-in rate, wash-in area under the curve, and wash-in perfusion index as previously described. 3,11
Tendon Histology
The animals were sacrificed after 12 weeks of exposure, at which time the right AT and SS complexes of the rats used in the contrast-enhanced ultrasound assay (n = 5 saline exposed, n = 5 nicotine-exposed animals) were harvested en bloc and fixed for histologic assays in 4% paraformaldehyde. The AT and SS from each animal were then processed using standard paraffin techniques and sectioned sagittally at 7 µm. Sections were stained with hematoxylin-eosin (H&E) to evaluate cellularity and cell shape. An adjacent separate set of sections were stained with Safranin-O and Fast Green (SAF-O) to evaluate glycosaminoglycan (GAG) content. 4 Stained sections were imaged at the midsubstance of each tendon using a microscope (Nikon Eclipse 90i, Melville, NY) at ×200 magnification. Cell density (cells/mm2) and cell shape (aspect ratio 0-1, with 0 being spindle shaped and 1 being a circle) were quantified using a bioquantification software system (BioquantOsteo II, Nashville, TN) as previously described. 4 SAF-O staining was grossly assessed and compared between groups by a single blinded observer.
Tendon Mechanics
AT and SS tendon-bone complexes (n = 10 saline exposed, n = 10 nicotine exposed for each tendon type) underwent mechanical testing as used extensively in our laboratory. 2,4 Briefly, all tendons were removed en bloc and fine dissected under a stereomicroscope (Leica Microsystems, Inc, Buffalo Grove, IL) to remove any remaining muscle and non-tendinous connective tissue. All tendons were marked with Verhoeff’s stain for optical strain tracking. The cross-sectional area (CSA) of the tendons was measured with a custom-made laser-based device and specimens were then prepared for mechanical testing. AT and SS bone-tendon units were gripped using custom fixtures in a physiologic orientation to load along the longitudinal axis of the tendon. Specimens were attached to a testing frame and submerged in phosphate-buffered saline bath maintained at 37º celsius. AT specimens were then subjected to a protocol that consisted of 10 cycles of preconditioning, stress relaxation for 10 minutes at 5% strain, and ramp to failure at 0.1% strain/second. SS specimens were subjected to a protocol that consisted of 10 cycles of preconditioning, stress relaxation for 10 minutes at 5% strain, and ramp to failure at 0.3% strain/second. Images for optical strain measures were captured at 1 frame per second using a camera (102F; Basler, Exton, PA) and zoom lens (Micro-NIKKOR AF 200 mm lens; Nikon, Melville, NY). Custom-code (MATLAB, MathWorks, Natick, MA) was used to track stained regions on the tendon throughout the ramp to failure. From these tests, mechanical properties including stiffness, percent relaxation, and failure load were computed. Material properties including modulus and maximum stress were also computed. Samples that failed at the grip during mechanical testing were excluded from maximum load and stress calculations.
Statistics
All data are presented as mean ± standard deviation. All data sets were assessed for normality using the Shapiro-Wilk test. The data were examined for outliers. Outliers in the non-normally distributed data were defined as being at least 3 times the interquartile range or having corroborating experimental evidence of some experimental difficulty (eg, an observation during experimentation that there may have been a dissection/specimen preparation error). Outliers in the normally distributed data were defined as being at least 2 standard deviations from the mean (sample sizes excluding outliers are denoted in the results section for each assay). The Mann-Whitney U test was used to compare non-normally distributed data. Student t test was used to compare groups for all normally distributed data. The level of significance was set at P < .05.
Results
Serum Cotinine
Both groups initially showed no detectable serum cotinine prior to osmotic pump implantation (n = 10 in saline group, n = 10 in nicotine group). Serum levels of cotinine demonstrated a controlled systemic release of nicotine after osmotic pump implantation (Table 1).
Serum Cotinine Levels Over Time.a

an = 10/group. Data are mean ± standard deviation.
Contrast-Enhanced Ultrasonography
CE-US showed an increase in tissue perfusion rate with nicotine exposure of AT (n = 4 in each saline and nicotine groups) as evidenced by a significant increase in wash-in-rate (P = .049) and trend toward a decrease in rise time (P = .054) and time to peak (P = .059). There was no difference detected in peak enhancement, wash-in area under the curve, and wash-in perfusion index in these samples (Table 2).
Achilles Tendon Contrast-Enhanced Ultrasonograph.a
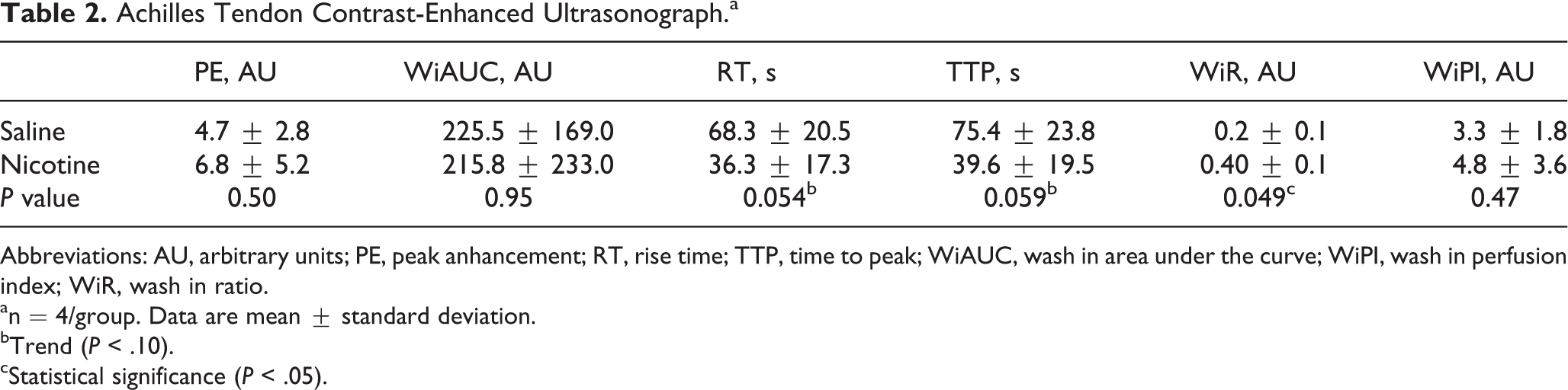
Abbreviations: AU, arbitrary units; PE, peak anhancement; RT, rise time; TTP, time to peak; WiAUC, wash in area under the curve; WiPI, wash in perfusion index; WiR, wash in ratio.
an = 4/group. Data are mean ± standard deviation.
bTrend (P < .10).
cStatistical significance (P < .05).
Histology
No histologic differences were detected on quantitative analysis H&E staining of cell density or cell shape between the saline and nicotine groups for both AT and SS. There were no differences in GAG staining detected by SAF-O between saline and nicotine groups for both AT and SS groups (n = 5 in each saline and nicotine group, Figure 1).
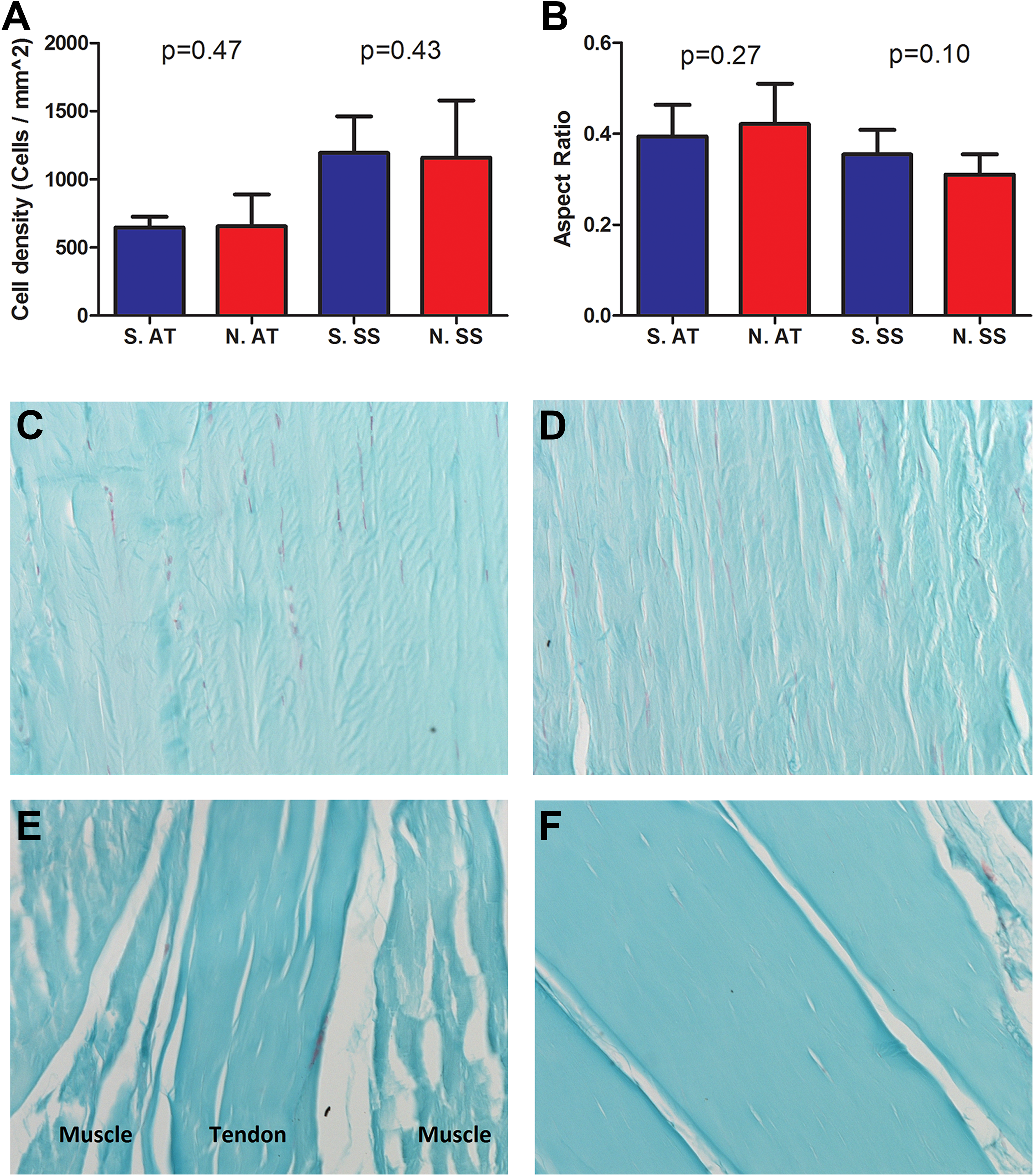
Histology: (A) Cell density (cells/mm2) and (B) cell shape aspect ratio, with a value of 1 representing round cells and a value of 0 representing a spindle-shaped cell. Groups include Saline AT (S. AT), Nicotine AT (N. AT), Saline SS (S. SS), and Nicotine SS (N. SS). Graphs denote mean with error bars denoting standard deviation. (C, D) Representative SAF-O images of AT midsubstance exposed to either saline and nicotine, respectively, at ×200 magnification. (E, F) Representative SAF-O images of SS midsubstance exposed to saline and nicotine, respectively, at ×200 magnification.
Biomechanics
CSA analysis of AT and SS detected no difference between saline and nicotine groups (n = 10 saline group and n = 9 nicotine group; Table 3). Biomechanical results showed changes in tendon viscoelasticity with nicotine exposure as evidenced by a significant increase in stress relaxation in nicotine-exposed AT (n = 10 saline group and n = 9 nicotine group, P = .04) and trend toward an increase in stress relaxation in nicotine-exposed SS (n = 9 saline group and n = 10 nicotine group, P = .06). All SS tendons failed at the grip and thus were excluded from analysis in maximum load and maximum stress. No differences in maximum load or maximum stress were detected between the saline and nicotine groups in the AT (n = 6 saline group and n = 7 nicotine group). No differences were detected in stiffness or modulus between saline and nicotine groups in both AT (n = 9 in both saline and nicotine groups) and SS (n = 9 in both saline and nicotine groups) (Table 3).
Biomechanics: CSA results of AT (n = 9 or 10/group) and SS (n = 10/group).a

Abbreviations: AT, Achilles tendon; CSA, cross-sectional area; SS, supraspinatus tendon.
a% Relaxation of AT (n = 9 or 10/group) and SS (n = 8 or 9/group). Maximum load of AT (n = 6-7/group) with samples excluded that failed at the grip. Maximum stress of AT (n = 6/group) with samples excluded that failed at the grip or were outliers from CSA analysis. All SS samples failed at the grip and thus were excluded from maximum load and maximum stress analysis. Stiffness of AT (n = 10/group) and SS (n = 8 or 9/group). Modulus of AT (n = 9/group) and SS (n = 8 or 9/group). Stiffness and modulus outlier samples with evidence of experimental difficulty were excluded from analysis. Data are mean ± standard deviation.
bStatistical significance (P < .05).
cTrend (P < .10).
Discussion
There are more than 50 million tobacco users in the United States. Multiple studies have shown that nicotine and smoking impairs the musculoskeletal system’s ability to heal. 10 Prior studies have shown that smoking predisposes the rotator cuff to injury and negatively impacts the tendon-to-bone interface’s ability to recover after injury. 5,9 However, there is a paucity of literature describing the effects of nicotine on tendon midsubstance and whether the various anatomic environments of tendons (such as intrasynovial vs extrasynovial tendons) alters the effect of nicotine exposure on tendon properties. It is important for orthopedic surgeons to understand the variety of effects in order to better counsel patients how to prevent injury. Thus, the purpose of this study was to establish an animal model to assess the effect of chronic nicotine exposure on AT and SS physiologic and mechanical properties. We hypothesized that chronic nicotine exposure is associated with changes in vascularity and detrimental to the tendon’s mechanical properties. Our secondary hypothesis was that AT and SS would be differentially affected by chronic nicotine exposure because of their anatomic differences (ie, extrasynovial vs intrasynovial tendon, lower vs upper extremity).
This study has several limitations. We recognize the limitations of an animal model and translatability to humans. Nicotine exposure and serum cotinine levels are highly variable in the clinical setting. They are affected by variations in smoking habits, time of day, activity, sex, and ethnicity. However, the animal model used is also this study’s strength. We used a model that could be rigorously controlled for consistent nicotine exposure, activity level, sex, and genetic background. The dose and duration of nicotine exposure used were comparable with previous rodent studies. 5 The serum cotinine levels in the rats exposed to nicotine in this study were comparable to those found in the average smoker in the United States (14 cigarettes per day) but below that of a heavy smoker (>20 cigarettes a day). 13 Thus, our model does not show the effect of nicotine exposure at high doses. Furthermore, we recognize nicotine is one component of many compounds found in cigarettes. Tar and carbon monoxide, among other components of cigarettes, may also affect tendon properties. However, nicotine is the active ingredient of cigarettes that is commonly delivered in other forms such as electronic cigarettes, patches, or gum for recreational and therapeutic purposes. Although these other delivery forms of nicotine are thought to be less detrimental, our study shows that the effect of nicotine in isolation does indeed alter tendon properties. Thus, clinicians may inform patients that nicotine in itself may alter tendons and that smoking-cessation treatment such as nicotine patches or gum may alter tendon properties.
We assessed the histologic effects of chronic nicotine exposure on uninjured tendon tissue, and future experiments may assess the effects of nicotine on the relationship between tendon structural and mechanical properties. Another limitation was that we had small differences in strain rates during mechanical testing between the AT and SS tendons, which should be considered when comparing these 2 tendon types to each other. The different strain rates were used to remain consistent with our laboratory’s prior protocols. Regardless and importantly, previous literature shows that small changes in strain rate (such as those in the current study) do not affect the shape of the stress-strain curve and failure properties. 12 Thus, the effects of the different strain rates used in mechanical testing of AT and SS was likely minimal, and comparisons between the 2 tendons may be made. However, this study provides baseline characteristics of uninjured tendon chronically exposed to nicotine that are necessary for comparisons in future experiments assessing treatment strategies and injury response.
In this study, chronic nicotine exposure increased the AT perfusion rate, whereas SS was unable to be assessed with CE-US in this model because of the overlying acromion obscuring the tendon. The increase in nicotine-exposed AT perfusion rate is evidenced by a trending decrease in rise time and time to peak and a significant increase in the wash-in rate. The histology assays were limited in that they only assessed for degenerative changes in the tenocytes (cellularity and cell shape) but did not assess changes in collagen content, collagen orientation, or perform immunohistochemistry assays to detect vascularity that may have contributed to the more directly observed changes in vascular and viscoelastic properties of the nicotine exposed tendons. There is a paucity of research describing the effects of nicotine on tendon vascularity. In one study, CE-US detected a significant increase in vascularity in tendinopathic AT when compared to healthy patients. 17 Thus, we hypothesize the changes in vascularity found in our study’s AT may suggest angiogenesis or possibly early tendinopathic changes to the tendon’s tenocytes following chronic nicotine exposure. Future studies are needed to further elucidate the effects of nicotine exposure on tendon vascularity and possible tendinopathic changes.
Contrary to our hypothesis, no differences in AT and SS tenocyte histology were found between saline- and nicotine-exposed groups with H&E and SAF-O staining in this study. We did not detect differences in histologic tendinopathic changes when assessing for cellularity, cell shape, and GAG content. Future studies are necessary to assess histologic changes in vascularity and structural properties such as collagen content and orientation.
Mechanical testing showed that AT and SS viscoelasticity were altered, though not other properties measured, following chronic nicotine exposure. For comparison, Ichinose and colleagues reported an increase in elastic modulus in the rat rotator cuff after chronic nicotine exposure to both low and high doses of nicotine with serum cotinine measurements consistent with a heavy smoker (>20 cigarettes per day). This may be because they found a decrease in CSA following nicotine exposure compared to saline exposure. Given elastic modulus is calculated from stiffness divided by CSA, the effect in CSA altered their elastic modulus results. Moreover, Ichinose and colleagues did not find differences in maximum load or stress. 9 Furthermore, in a clinical study that analyzed the Achilles tendon with ultrasonography, it was found that smoking was associated with a thinner tendon and increased strain ratio. 1 In our study, we did not detect a difference in AT or SS CSA, which may explain why we did not observe changes in modulus that was observed in these previous studies. Furthermore, our rats received a dose of nicotine equivalent to the average American smoker (14 cigarettes per day) where the previous studies had nicotine doses for heavy smoker (>20 cigarettes per day). Thus, the changes in CSA and modulus of the tendon may be nicotine dose dependent, requiring high doses of nicotine to observe these effects. However, in our study we did detect a significant difference in percent relaxation, a viscoelastic tendon property, between saline and nicotine groups in AT (P < .05) and a trend in SS (P < .10). Previous studies have described using measurements of tendon viscoelasticity as a tool to assess for tendinopathy when values deviate from a normal healthy tendon. 19 Thus, changes in the viscoelastic properties of tendons may be a marker for other changes in tendon properties.
Contrary to our secondary hypothesis that chronic nicotine exposure would affect AT and SS differently, we found similar trends between both tendons. Chronic nicotine exposure altered the viscoelastic properties of both tendons but did not change other biomechanical properties measured. Furthermore, chronic nicotine exposure increased the tissue perfusion rate of AT, which may be surrogate for tendinopathic changes. In conclusion, chronic nicotine exposure alters the viscoelastic and tissue perfusion properties of uninjured tendons. Further studies are necessary to elucidate whether these changes predispose tendons to degeneration and injury.
Supplemental Material
Supplemental Material, FAO842529-ICMJE - Chronic Nicotine Exposure Alters Uninjured Tendon Vascularity and Viscoelasticity
Supplemental Material, FAO842529-ICMJE for Chronic Nicotine Exposure Alters Uninjured Tendon Vascularity and Viscoelasticity by Daniel Gittings, Corinne Riggin, James Boorman-Padgett, Stephanie Weiss, George Fryhofer, Daniel Farber, David Steinberg and Louis J. Soslowsky in Foot & Ankle Orthopaedics
Footnotes
Acknowledgments
This study was funded by the AFSH Resident / Fellow Fast Track Grant (Award 1271), NIH/NIAMS supported Penn Center for Musculoskeletal Disorders (P30 AR069619), NSF Graduate Research Fellowship, and Rheumatology training grant (4T32AR007442-29). The authors thank Zakary Beach, Courtney Nuss, Harina Raj, and Snehal Shetye for their contributions.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Daniel Gittings, MD, reports grants from AFSH Resident/Fellow Fast Track Grant, during the conduct of the study. Corinne Riggin, BS, reports grants from NSF Graduate Research Fellowship, and Rheumatology training grant, during the conduct of the study. Louis Soslowsky, PhD, reports grants from NIH/NIAMS supported Penn Center for Musculoskeletal Disorders (P30 AR069619), during the conduct of the study; grants from OrthoFix, grants from Bonti, and grants from Marine Polymer, outside the submitted work. ICMJE forms for all authors are available online.
Funding
The author(s) declared receipt of the following financial support for the research, authorship, and/or publication of this article: AFSH Resident / Fellow Fast Track Grant 1271, NIH / NIAMS P30AR069619, NSF Graduate Research Fellowship, and Rheumatology training grant 4T32AR007442-29.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
