Abstract
Free tissue transfer for dural coverage can be challenging for various reasons. In the case of malignancy, patients often have received significant doses of radiation to the head and neck leading to fibrosis and osteoradionecrosis. Not only will free tissue transfer need to accommodate an often large defect but will need to protect intracranial contents. Recipient vessel quality and patency is often affected by comorbidities such as diabetes mellitus and coronary artery disease and can be compounded by radiation. Due to these factors, more proximal vessels in the head and neck are often pursued but due to insufficient length, often require vein grafts or arteriovenous loops to reach the donor vessels for anastomosis. This requires larger incisions and harvesting of lengthy veins. In this study, we discuss a technique of harvesting a small dorsal hand vein, that is hidden well in a hand crease, to create an arteriovenous loop between the superficial temporal vessels. Benefits include exploration of the recipient superficial temporal vessels prior to craniectomy without creating additional incisions, readily accessible recipient vessels to reach donor vessels without harvesting lengthy vein grafts, and allowing for arterialization of the superficial temporal vein leading to decreased venous congestion and thrombosis. While this can be done in 1 stage, we perform this in 2 stages to avoid an increased number of anastomoses and increased risk of flap failure. We present the case of an elderly male with multiple comorbidities and scalp osteoradionecrosis secondary to malignancy who this technique was successfully performed on.
Introduction
Arteriovenous loops in microsurgery have long been described in the literature, especially in relation to extremity reconstruction. Success in reconstructive microsurgery is predicated on several factors such as overall health of the patient as well as quality of donor and recipient vessels. Extensive zones of traumatic injury, history of radiation, and atherosclerosis of vessels from diabetes mellitus and coronary artery disease can compromise the success of free tissue transfer. 1 In head and neck reconstruction, specifically of the neurocranium, free tissue transfer is often necessary to recreate a soft tissue defect and protect the intracranial contents. Flap selection is often predicated on location and size of the defect, vascular pedicle length, as well as size, location, and quality of the recipient vessels in the head and neck. 2,3 While there are several options for recipient vessels in the head and neck, selecting vessels in close proximity to the defect can be challenging for the reasons stated above. In attempts to overcome this, several techniques have been developed such as the use of vein grafts and arteriovenous loops to often larger, more proximal vessels in the head and neck region. 4,5,6,7 Below, we discuss the creation of a superficial temporal arteriovenous loop for staged free tissue transfer for dural coverage and its benefits.
Patient History
A 75 year old Caucasian male with diabetes mellitus, hypertension, and hyperlipidemia presented for evaluation of a non-healing 2.5 × 2 × 0.1 cm right scalp wound 2 years after excision of squamous cell carcinoma with adjuvant radiation to the area. CT and MRI imaging were performed preoperatively and the patient was found to have osteoradionecrosis of the area with possible underlying tumor involvement of the dura.
First Stage—Creation of Arteriovenous Loop
Through a preauricular facelift incision, the superficial temporal artery and vein were identified within dense radiation fibrosis and isolated. A 4 cm dorsal hand vein was harvested and using microscope magnification, 2 hand-sewn anastomoses in an end-to-end fashion were performed with interrupted 9-0 nylon suture to create an arteriovenous loop from the superficial temporal artery to the superficial temporal vein. A small piece of silicone sheeting was placed under the arteriovenous loop prior to closure to facilitate easier dissection at the second stage. The patient was admitted to a non-ICU bed and had doppler monitoring hourly postoperatively and was discharged home on postoperative day 1 with a palpable thrill in the arteriovenous loop (Figure 1).
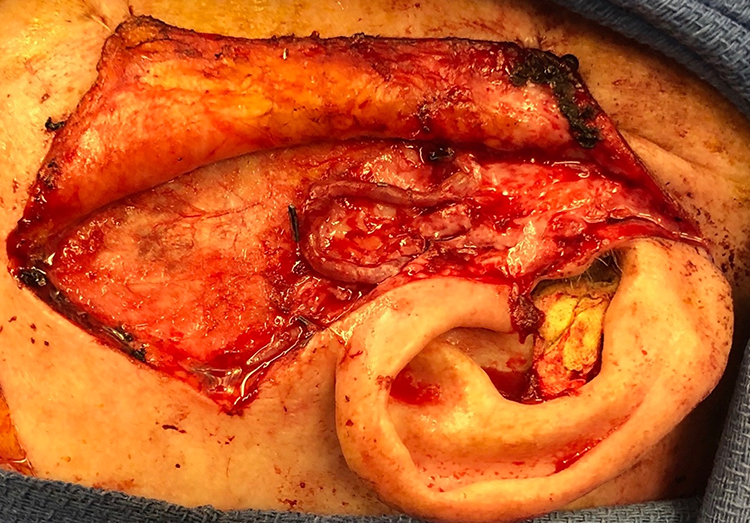
Arteriovenous loop between superficial temporal artery and vein.
Second Stage—Free Tissue Transfer
A 2-team approach was taken in conjunction with neurosurgery 1 week later. Neurosurgery proceeded with craniectomy and resection of tumor in the epidural space. A free latissimus muscle was harvested for adequate soft tissue coverage of the resulting defect as well as for proper pedicle length (Figure 2). Under microscope magnification, the silicone sheet was removed. The loop was divided and a 3 mm coupler was used for the end-to-end venous anastomosis. The recipient artery did have a slight size mismatch (3 mm) compared to the donor artery (2 mm). The donor artery was spatulated and a hand sewn end-to-end anastomosis was performed with interrupted 9-0 nylon suture. The muscle was inset and a split-thickness skin graft placed over this (Figure 3). The patient was monitored in the neurosurgical ICU with hourly doppler monitoring and was discharged from the hospital on postoperative day 9 with a well perfused flap and no complications. At his 2-month follow-up visit, the flap was well healed with complete skin graft incorporation (Figure 4). The patient was offered a cranioplasty but declined this as he was satisfied with his result.
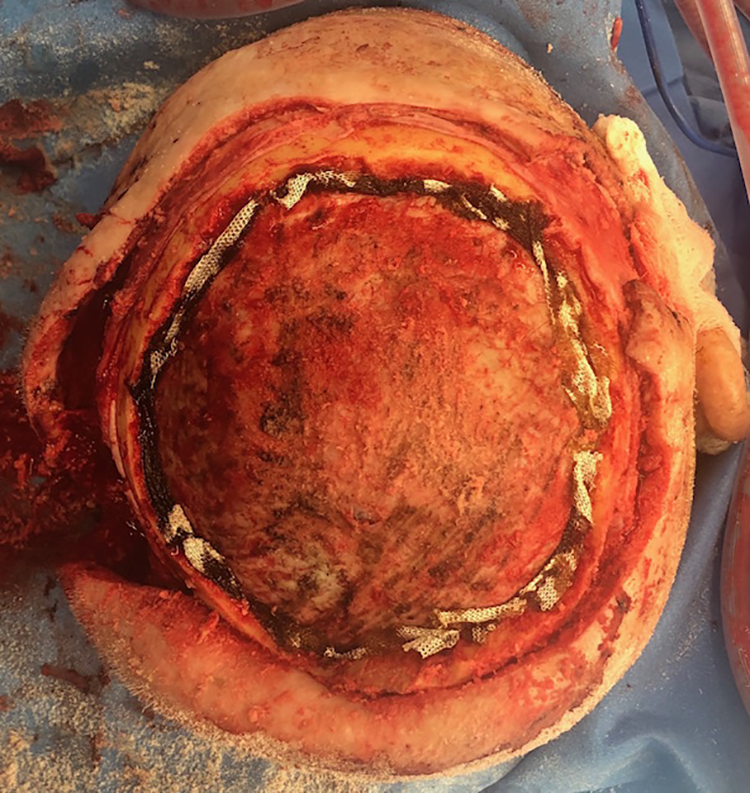
Large soft tissue and calvarial defect to be reconstructed with free tissue transfer.
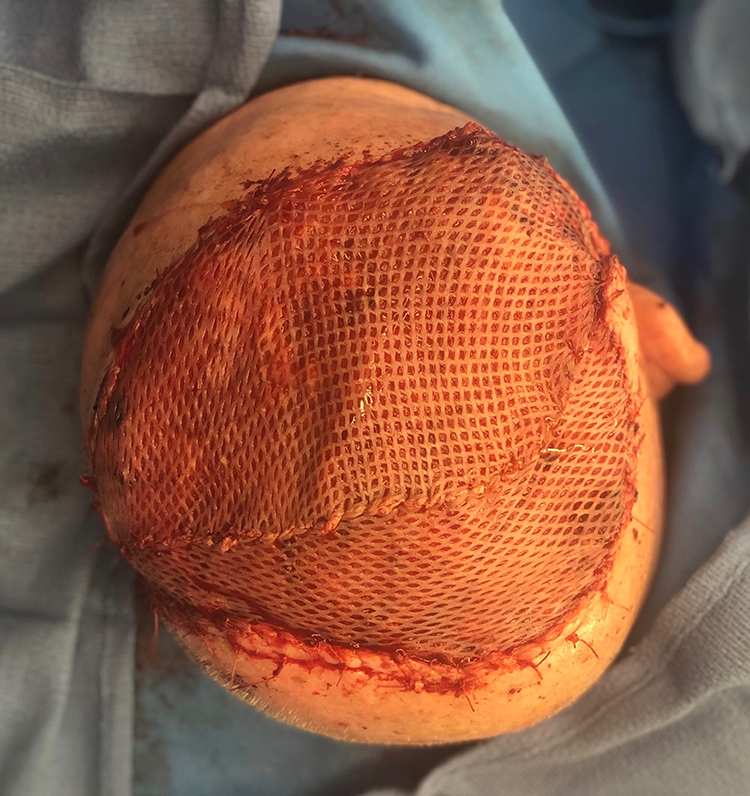
Free latissimus muscle with split-thickness skin graft with recipient vessels generated from superficial temporal AV loop.
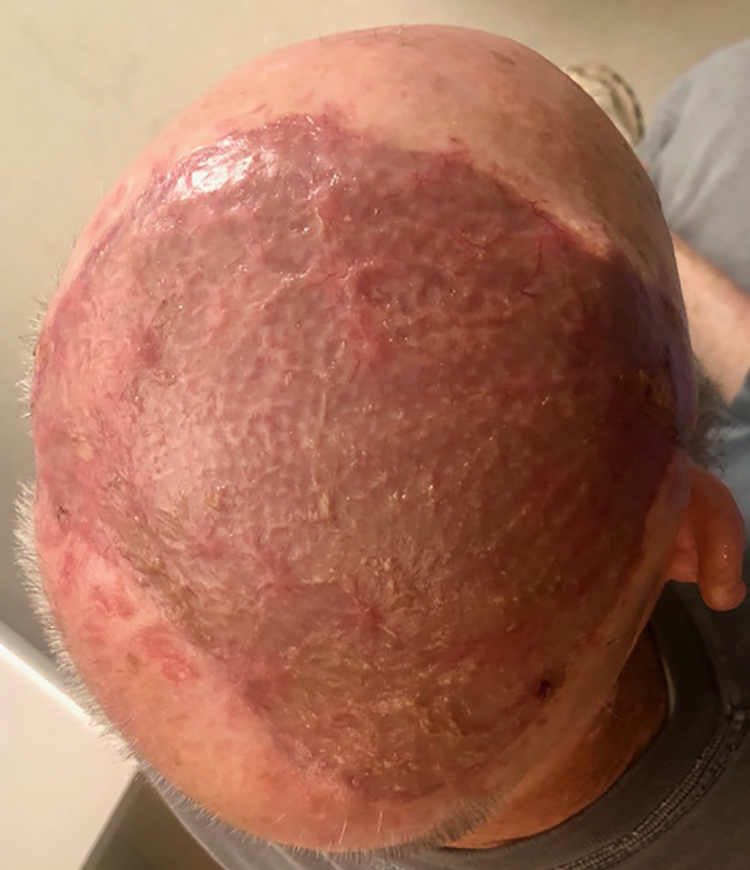
Two-month postoperative photo depicting a well healed free latissimus muscle flap with split-thickness skin grafting.
Discussion
Dural coverage in patients with a prior history of radiation and osteoradionecrosis often necessitates free tissue transfer for adequate soft tissue reconstruction and protection of intracranial contents. Recipient vessel selection in these patients is crucial for microsurgical success but can be complicated by radiation as well as extensive comorbidities, such as diabetes mellitus, in these often elderly patients. Often, the superficial temporal vessels are in close proximity to the defect needing reconstruction but are not readily usable to due to the quality of the vessels. This has led to the use of vein grafts with an increased risk of thrombosis, twisting, or kinking with subsequent flap loss. 4 Veins are often harvested from large incisions in the lower and upper extremities. Arteriovenous loops in head and neck reconstruction have had better outcomes. However, these require more proximal incisions to larger proximal vessels. 5 –7
This is the first description of the use of an arteriovenous loop of the superficial temporal vessels. Using this technique allows for exploration of the superficial temporal vessels prior to craniectomy without creating any additional incisions on the head and neck. The donor site for the vein graft is often well tolerated and can be hidden in a hand crease. This donor site is significantly less morbid than the donor site for saphenous vein. Clegg et al describe using a similar technique as ours but with use of the saphenous vein for the arteriovenous loop to the facial vessels. Their patient suffered from COPD, hyperlipidemia, tobacco use, hypertension, and diabetes mellitus and while their 2-stage utilization of an arteriovenous loop was similarly successful to ours, their donor site was more extensive than ours in a patient with significant comorbidities. 8 Similarly Tan et al describe the use of saphenous vein AV loops in a single stage with free latissimus dorsi flaps. They cite that one downside of the use of the saphenous vein is that it prevents its use in the future in the event that a coronary bypass surgery is needed. In addition, due to the large caliber, they also site size discrepancy in performing microanastomoses. 9
While we performed this technique in 2 stages, it is feasible to consider using this technique in a 1-stage approach in the proper patient. Performing this technique in 2 stages allows for only 2 anastomoses being created at a time and avoids possible flap loss if performed as a single stage procedure with 4 anastomoses in patients with extensive comorbidities that may affect vessel patency. 10 While this does require 2 general anesthetics, the first operation is substantially shorter and can theoretically be done on an outpatient basis. In addition, it also allows the second stage to be faster and technically easier as the vein has been given time to arterialize via the loop and will be larger allowing for an easier anastomosis with decreased venous congestion and thrombosis. 11 And while the vein caliber will be appropriately sufficient with this technique, it will theoretically be smaller than the saphenous vein and avoid size mismatch that is often seen with saphenous vein grafts. We also find that with a successful arteriovenous loop this decreases vasospasm that can occur with the temporal artery. This technique also promotes efficiency as at the second stage, the vessels are already readily accessible. This was also emphasized by Steiner et al who utilized AV loops in patients without sufficient recipient vessels. Similar to our case series, they performed reconstruction with a 2-stage approach to decrease flap loss secondary to AV loop complications such as hemorrhage, inadequate blood flow, and thrombosis. 12
Conclusions
This technique is currently being explored in other patients at our institution to avoid more extensive dissections in the head and neck region and limit extent of vein harvesting. Several advantages exist with this technique including decreasing donor site morbidity, avoiding vessel size mismatch while creating readily accessible vessels of appropriate caliber, decreasing complications associated with vein grafts, and decreasing vasospasm commonly seen with the temporal artery. This newly described technique presents a useful approach to complex dural coverage in 2 stages while potentially decreasing free tissue transfer loss due to radiation and patient comorbidities that may affect microsurgical vessel patency and quality.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
