Abstract
This study was conducted to evaluate the predictive efficacy of tumor mutation burden (TMB) in patients with non–small-cell lung cancer (NSCLC) receiving PD-1 antibodies. Embase, PubMed, Ovid Medline, and the Cochrane Library were systematically searched until May 24, 2020. The keywords included “PD-1,” “TMB,” and “NSCLC.” Overall survival (OS) and progression-free survival (PFS) were summarized and combined using the hazard ratio (HR) and 95% confidence interval. Twenty-one studies with 9883 patients were included in the meta-analysis. The overall relapse rate ranged from 39.3% to 64.3% in the higher TMB group as compared with 0% to 40% in the lower TMB group. The median OS ranged from 2.9 to 23 mo in the higher TMB group as compared with 4.3 to 16.2 mo in the lower TMB group. Patients with a higher TMB had a better OS as compared with patients with a lower TMB (HR = 0.61, P < 0.001). Similarly, a higher TMB was also a good predictor of PFS in patients treated with PD1/PDL1 antibodies (HR = 0.55, P < 0.001). Our results suggest that among NSCLC patients receiving PD1/PDL1 antibodies, patients with higher TMB could have a better survival outcome.
Introduction
Lung cancer is a type of human malignant tumor disease with a current high incidence. Its morbidity and mortality rate ranks first among all types of malignant tumors and is increasing each year. 1 The most common type of lung cancer is non–small-cell lung cancer (NSCLC), which accounts for about 80% to 85% of cases. 2 In general, lung cancer has the characteristics of insignificant symptoms and delayed specific symptoms. Thus, there is a huge need to determine the best timing of patient treatment and improve their quality of life. Over the years, there have been a large number of clinical studies on the early diagnosis of NSCLC, which has resulted in remarkable results.3,4 Clinical treatment of NSCLC usually consists of surgery, and selective adjuvant chemotherapy is then performed based on the patient’s pathological type and clinical characteristics.2,4 However, according to relevant data, because of the inadequate understanding of lung cancer and insufficient attention paid to this disease, most patients are in the late stage when they are clinically diagnosed, losing the opportunity for surgery. Moreover, patients often suffer from severe side effects caused by repeated systemic intravenous chemotherapy, which in turn causes the patient’s compliance with treatment compliance to decline, results in patients feeling overcome with a loss of confidence in the disease, and seriously affects the survival rate of the disease and the patient’s quality of life after the disease. 5 It has been reported that the 5-y survival rate of NSCLC patients after diagnosis is only 15%. 1 In recent years, with the gradual deepening of the research on the molecular biology of NSCLC, lung cancer driver genes such as EGFR, TP53, ALK, and KARS have been discovered, and as a result, the incidence of lung cancer has been gradually revealed from the perspective of molecular biology. 6 The clinical application of molecular-targeted therapy for lung cancer at the end of the 20th century has achieved unprecedented benefits, thereby greatly improving the survival rate of advanced NSCLC and improving the quality of life of patients to a certain extent. However, in recent years, drug resistance after targeted therapy has become another major clinical problem, 7 resulting in some scholars believing that immunotherapy is expected to be another treatment approach for patients with NSCLC.
Programmed cell death 1 (PD1) is a protein expressed mainly after T cells are induced (mature T cells), and its ligands include PDL1 and PDL2, of which most tumor cells express PDL1. 8 When PDL1 is combined with PD1, it promotes the phosphorylation of tyrosine in the ITSM domain, which causes the dephosphorylation of downstream proteins PI3K and Syk, thereby inhibiting the activation of downstream ERK and AKT pathways and T-cell activation. The transcription and translation of the required cytokines and genes can negatively regulate the activity of T cells.9–11 The good and sustained efficacy of PD1/PDL1 antibody in a variety of tumors is one of the most important advances in the history of tumor therapy. At present, a variety of PD1 antibodies are approved by the Food and Drug Administration for various types of tumors, and NSCLC is one of them.12,13 Although PD1/PDL1 has achieved great success in cancer treatment, because the overall effectiveness of PD1/PDL1 antibody in most tumors is still low, expensive, and the treatment itself has toxic and side effects, accurately screening the beneficial population has become a hot issue in the field of immunotherapy research in recent years. 14 At present, the main curative effect markers include tumor mutation burden (TMB), PDL1 protein expression, interferon-γ–related gene markers, tumor-infiltrating lymphocytes, and copy number variation. 15 However, the use of TMB to predict the efficacy of PD1/PDL1 antibodies in NSCLC patients is still controversial. Thus, the aim of this systematic review and meta-analysis is to discuss the predictive effect of TMB in NSCLC patients treating with PD1/PDL1 antibodies.
Methods
This study was designed and reported in accordance with the Preferred Reporting Items for Systematic review and Meta-analysis (PRISMA) guidelines. 16
Search Strategy
Embase, PubMed, Ovid Medline, and the Cochrane Library were systematically searched to evaluate the predict efficacy of TMB in NSCLC patients treated with PD1/PDL1 antibodies. In addition, the related website and conference database, such as the World Conference on Lung Cancer, the European Society for Medical Oncology, and the American Society of Clinical Oncology were searched to screen for gray literature. Both keywords and medical subheadings (MeSH) terms were designed by an experienced librarian and used in the search strategy. The keywords included “tumor mutation burden,” “PD-1,” “nivolumab,” “atezolizumab,” “pembrolizumab,” “durvalumab,” “immune checkpoint blockade,” and “non-small cell lung cancer.” The ending date in the search strategy was May 24, 2020. All studies with titles and abstracts were imported into Endnote X6 (Thomson Reuters, Toronto, Canada) to determine duplication and for further literature screening.
Selection Criteria
A study was included if it satisfied the following criteria: (1) the study included NSCLC patients treating with PD1/PDL1 antibodies and (2) the study mentioned the effect of TMB in predicting the survival outcome or treatment response. The survival outcome was defined as the overall survival (OS) rate and progression-free rate (PFS), and the treatment response was described as objective response rate (ORR).
The exclusion criteria were (1) the mention of other malignancies treated with PD1/PDL1 antibodies; (2) inclusion of NSCLC patients who were not treated with PD1/PDL1 antibodies; (3) survival outcome or response was not reported or could not be extracted; (4) the study type was review, comments, or case report; and (5) publication in a language other than English.
Literature Screening, Data Extraction, and Quality Assessment
Two investigators (Y.Z. and M.Y.) independently screened the titles and abstracts based on the inclusion and exclusion criteria. If the titles and abstracts could not be determined, the full text was further assessed. The third investigator (Y.Y.) was included in the discussion for any disagreement due to the titles and abstracts.
The data were extracted by two researchers independently and were imported into a standard Excel form. The information was as follows: study characteristics (author, year published, recruitment period, country, institution, study design, study source, etc.), the included patient data (median age, gender, smoker history, histology of lung cancer, tumor stage, etc.), TMB cutoff, PD1/PDL1 antibodies, and outcome (OS, PFS, and ORR). The HR and 95% CIs associated with OS and PFS were extracted from either univariate or multivariate Cox regression analyses.
Two investigators independently assess the quality of the included articles. For case-control and cohort studies, the Newcastle-Ottawa Quality Assessment Scale (NOS) was used, in which a score of 6 to 9 indicated high quality and a score of 0 to 5 indicated low quality. 17
Statistical Analysis
All statistical analyses were performed by Stata 15.0 software (Stata Corporation, College Station, TX). Survival analysis was combined with HR with 95% CIs. If the HR was not described explicitly in the univariate or multivariate analysis, we calculated the time-to-event data through the survival curve based on Tierney’s method. 18 The likelihood χ2 test and I2 statistics were used for detecting heterogeneity across studies (I2 ≥ 50% indicating the presence of heterogeneity). When the heterogeneity existed among studies, the random effect model was conducted to calculate the pooled HRs; otherwise, the fixed effect model was used. A P value less than 0.05 was regarded as significant.
Results
Literature Selection
We identified a total of 4463 studies based on the search strategy. Twelve studies were found from the conference database and Google Scholar. After deleting the duplicated studies, 3984 studies were screened by abstracts and titles. After excluding the unrelated articles, 21 studies were finally included in our systematic review and meta-analysis.12,13,19–37 Figure 1 shows the flowchart.
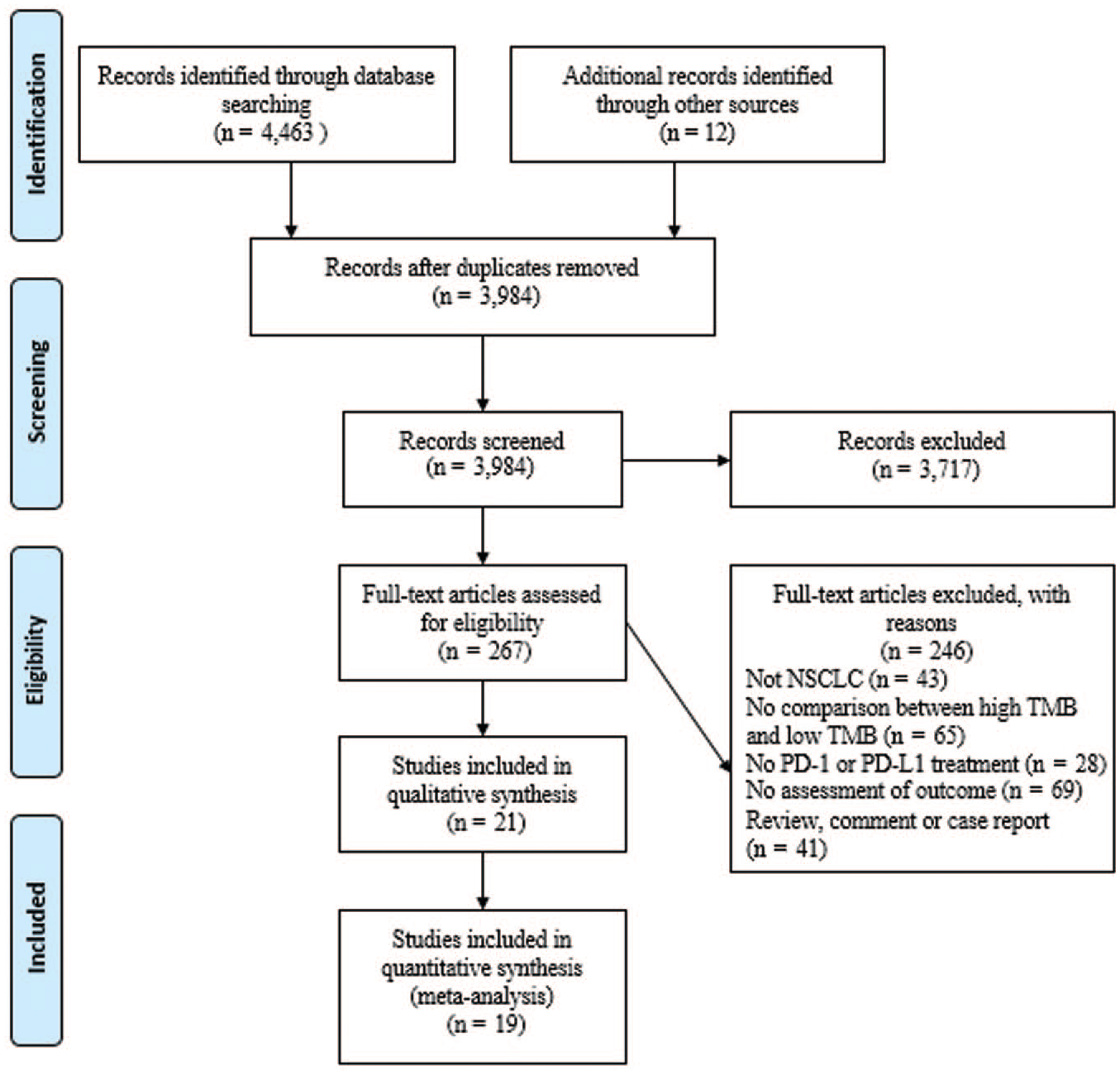
Flowchart of literature screening.
Characteristics of the Patients in the Included Studies
The characteristics of the NSCLC patients are summarized in Table 1 . A total of 9883 patients were treated with PD1/PDL1 antibodies due to NSCLC. Most of the studies were from the United States, and the year of publication ranged from 2015 to 2020, with patient recruitment occurring in 2014 to 2018. Among the included patients, 42.7% were female and 71.4% of patients had a history of smoking. Most patients were diagnosed as advanced stage, and 71.8% of patients had nonsquamous NSCLC.
Characteristics of Included Studies.
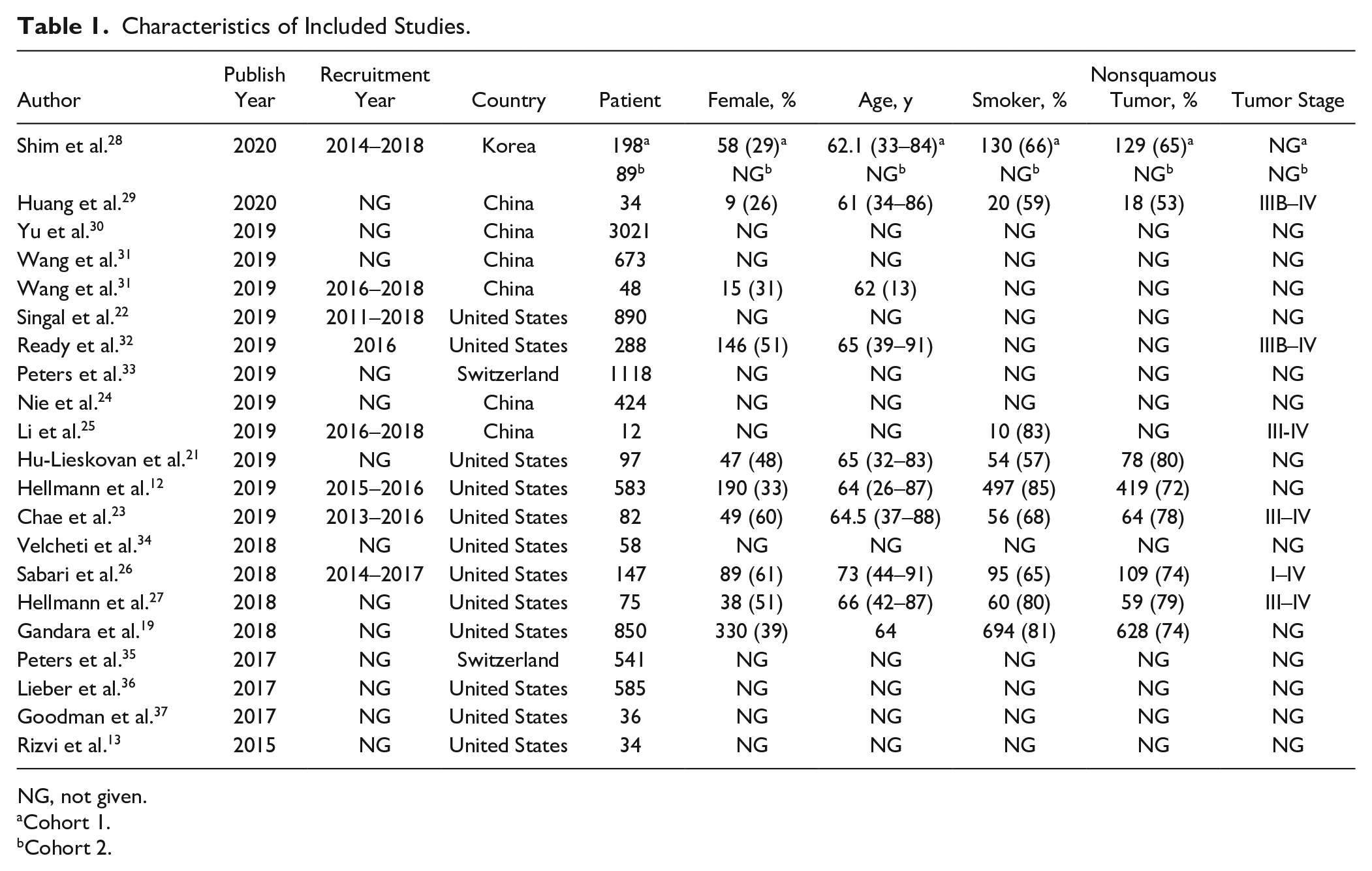
NG, not given.
Cohort 1.
Cohort 2.
The PD1/PDL1 antibodies treatment approach and the common cutoff of the TMB are listed in Table 2 . Six studies did not specify the PD1/PDL1 antibodies in detail. Six studies used nivolumab, 5 studies used pembrolizumab, seven studies used atezolizumab, and two studies used durvalumab. The TMBs were either from the tumor tissue or blood sample. Seven studies only used tumor tissue, four studies used blood sample, two studies adapted either tumor tissue or blood sample, and four studies adapted both tumor tissue and blood sample as a matched pair. The common cutoff of TMB was 6, 7, 10, 16, and 20 mutations/Mb. A higher TMB was usually related to a better ORR and a longer OS and PFS as compared with a lower TMB in NSCLC patients treated with PD1/PDL1 antibodies. The ORR ranged from 39.3% to 64.3% in the higher TMB group as compared with 0% to 40% in the lower TMB group. The median OS ranged from 2.9 to 23 mo in the higher TMB group as compared with 4.3 to 16.2 mo in the lower TMB group. Similarly, the median PFS was 7.1 to 17.1 mo in the higher TMB group as compared with 2.6 to 5.8 mo in the lower TMB group.
Prognostic Predictors of Tumor Mutation Burden.
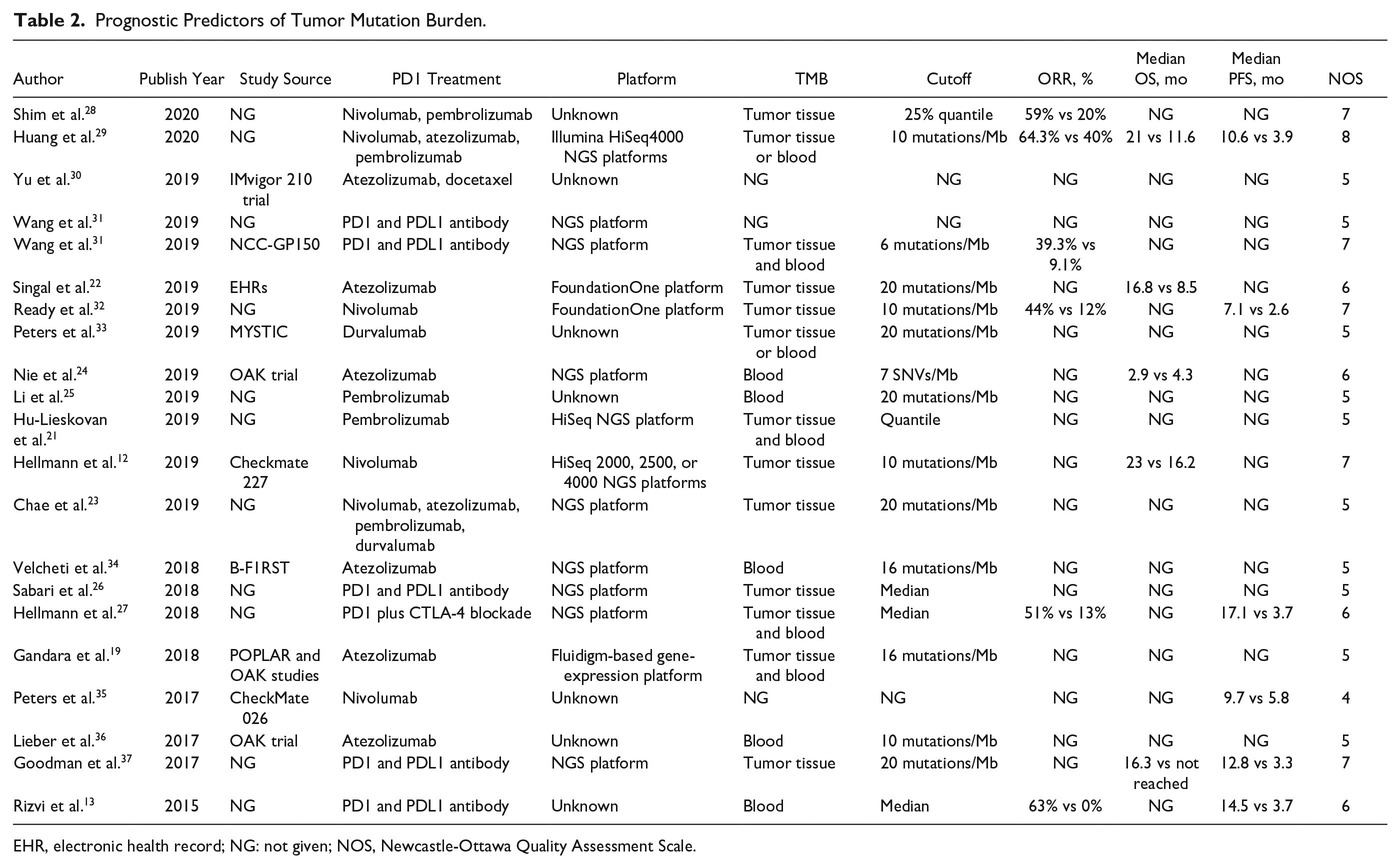
EHR, electronic health record; NG: not given; NOS, Newcastle-Ottawa Quality Assessment Scale.
Quality Assessment of Included Studies
The assessment of quality between studies was based on the NOS, which is shown in Table 2 . Fourteen studies were regarded as median quality with a score of 5 to 6, and 6 studies were regarded as high quality with a score greater than 7.
Predictive Effect of TMB in NSCLC Patients Treated with PD1/PDL1 Antibodies
There were a total of 8 studies with 10 cohorts reporting the relationship between TMB and OS. The patients with higher TMB had a better OS as compared with patients with a lower TMB (HR = 0.62, 95% CI = 0.55–0.69, I2 = 0%, fixed-effect model, P < 0.001; Figure 2 ). Similarly, 13 studies reported the association between TMB and PFS. A higher TMB was also a good predictor of PFS for patients treated with PD1/PDL1 antibodies (HR = 0.55, 95% CI = 0.44–0.68, I2 = 64.9%, random-effect model, P < 0.001; Figure 3 ).
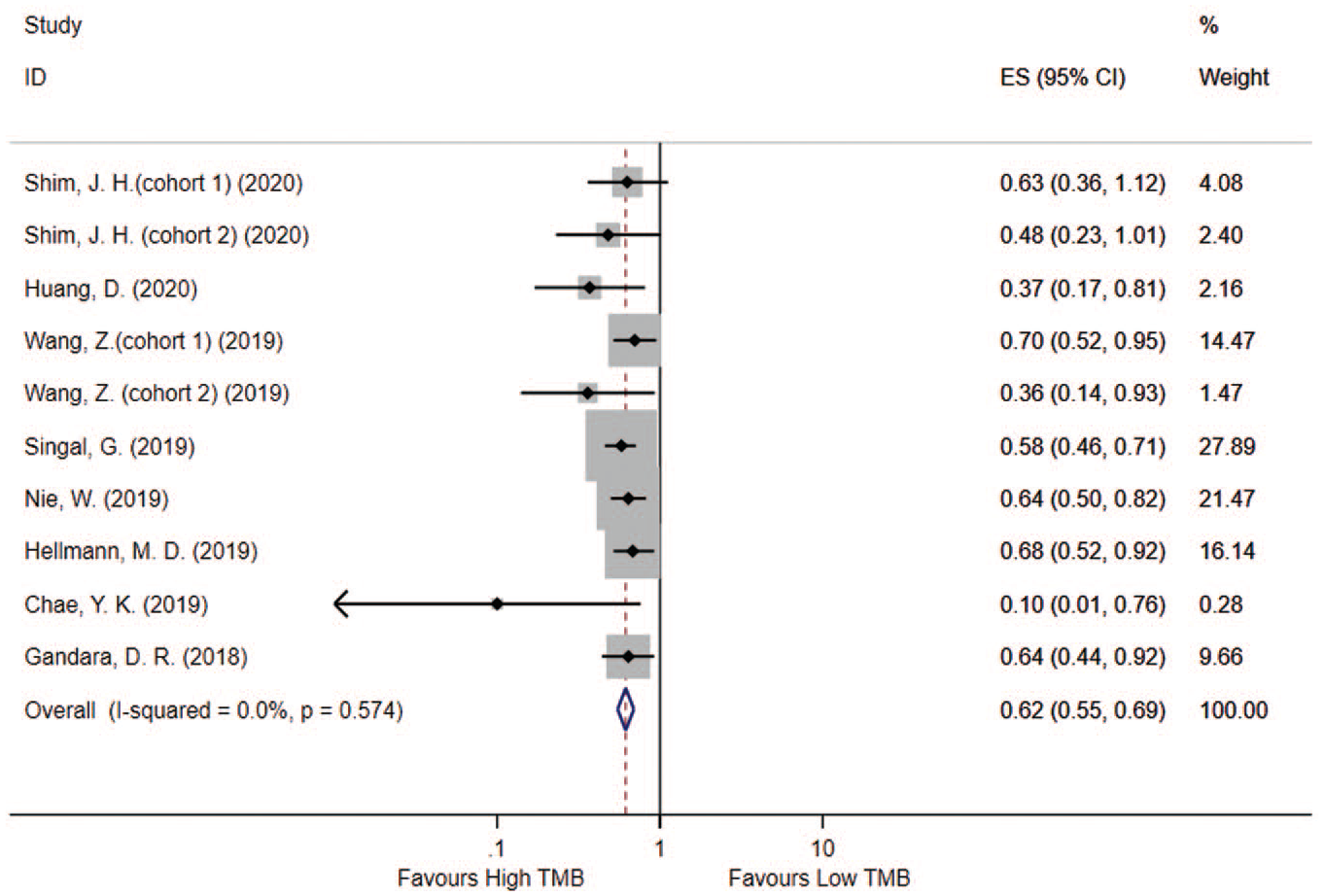
Predictive effect of tumor mutation burden (TMB) in evaluating the overall survival rate for non–small-cell lung cancer patients receiving PD1/PDL1 antibodies.
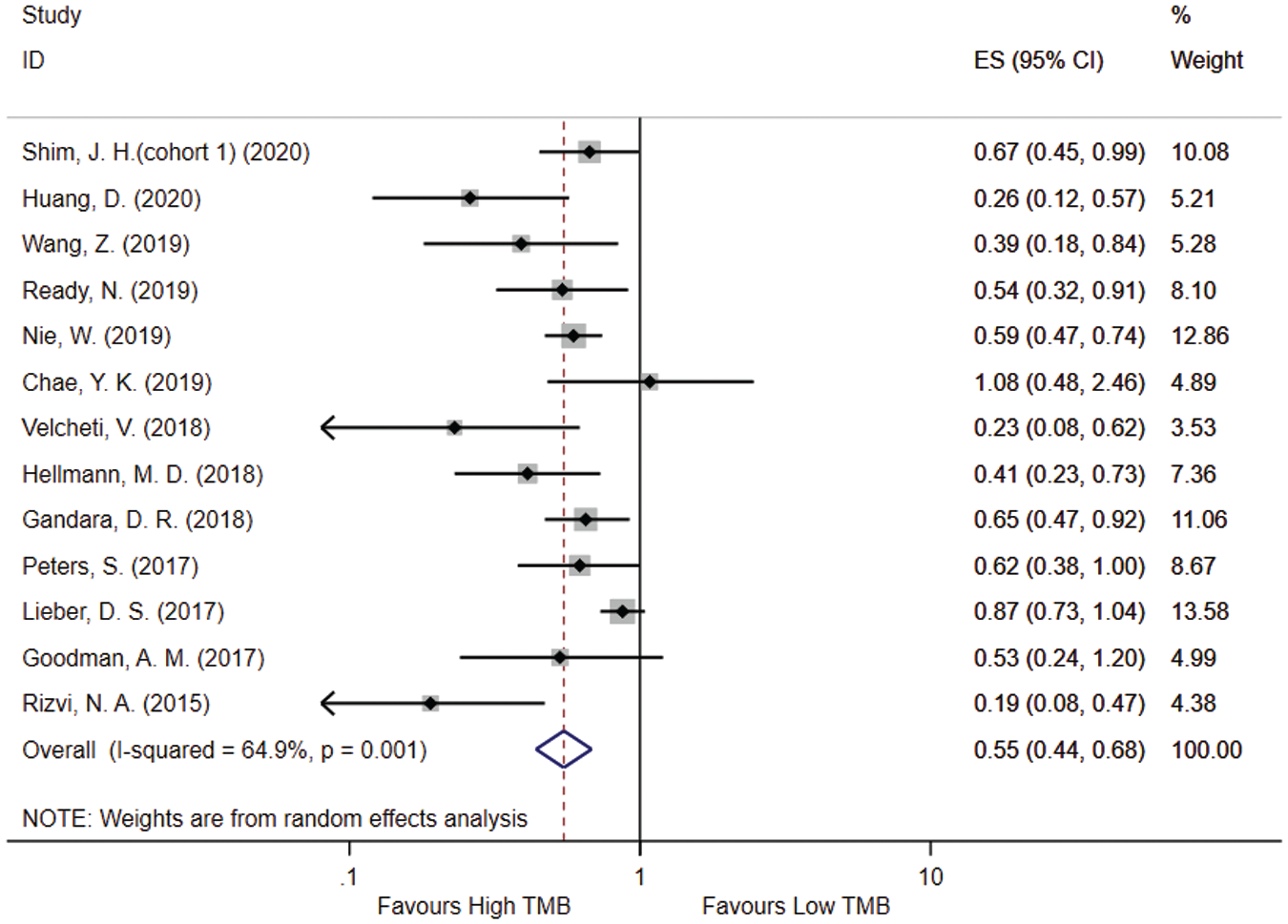
Predictive effect of tumor mutation burden (TMB) in evaluating the progression-free survival rate for non–small-cell lung cancer patients receiving PD1/PDL1 antibodies.
Subgroup Analysis of the Predictive Effect of TMB in NSCLC Patients Treated with PD1/PDL1 Antibodies
Table 3 shows the subgroup analysis. We further analyzed the relationship between TMB and OS and PFS in terms of different region, platform, detection methods, combination or single treatment, and mutation cutoff. In both the eastern and western countries, a higher TMB was related to a better efficacy of PD1/PDL1 treatment (all P < 0.001). For patients from eastern countries, the HR of PFS tended to be lower as compared with western countries (0.57-fold vs 0.71-fold) but similarly in the HR of OS.
Impact of Different Cutoffs of TMB in Relationship to the Outcome.
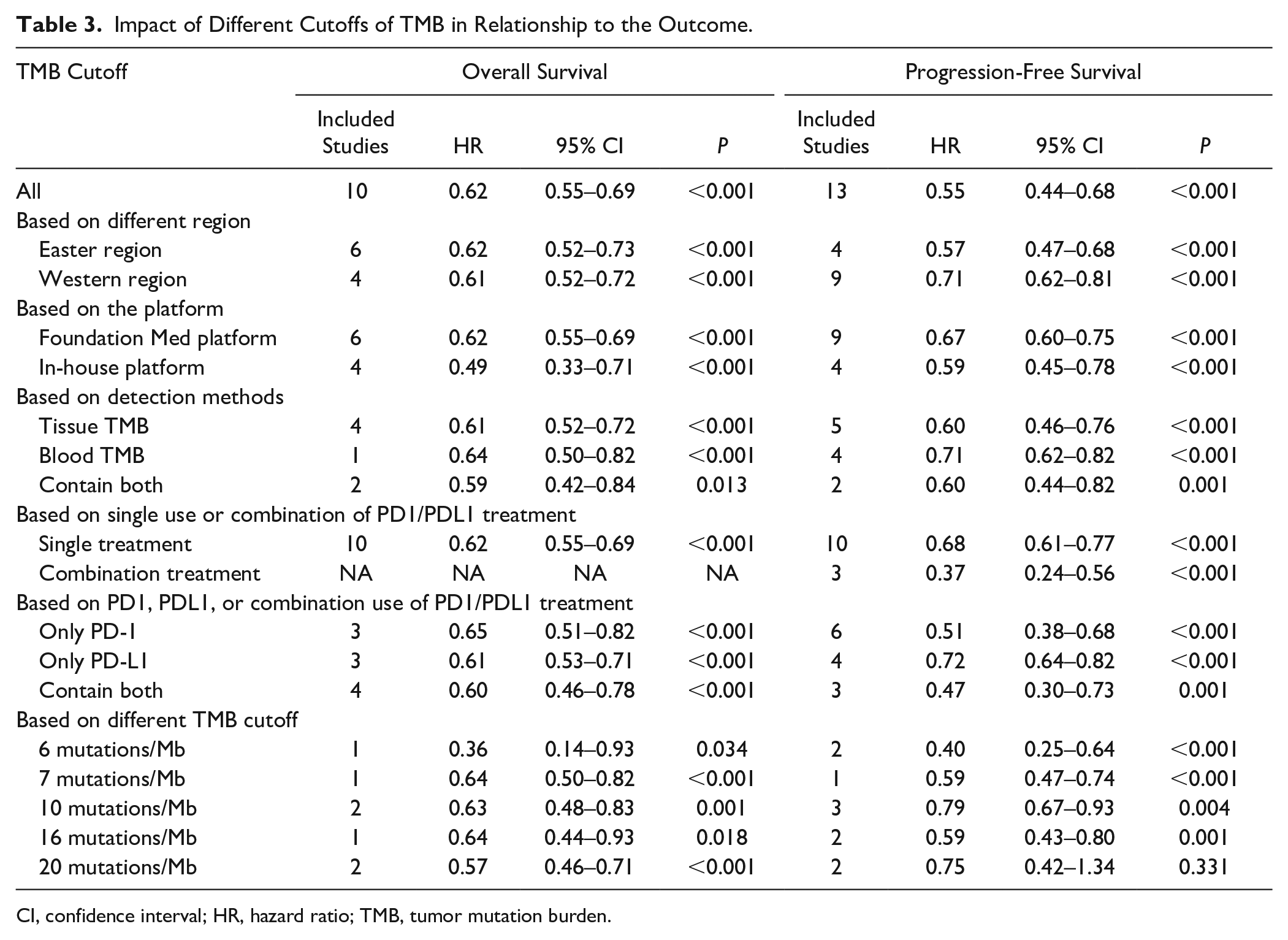
CI, confidence interval; HR, hazard ratio; TMB, tumor mutation burden.
In terms of the platform used for detecting TMB, studies using the foundation Med platform seemed to have a similar predicting efficacy of TMB as compared with those studies using an in-house platform for both OS and PFS, with an HR ranging from 0.49 to 0.67, and all P < 0.001.
Different studies used different sources of tissue to detect TMB in tumors. There was almost a similar predictive effect between tumor tissue and blood sample for the detection of TMB (HR = 0.61, 95% CI = 0.52–0.72, P < 0.001 in tumor tissue; HR = 0.64, 95% CI = 0.50–0.82, P < 0.001 in blood sample). In addition, in those studies that used both tumor tissue and blood sample, the predictive effect was similar studies using only one method (HR = 0.59, 95% CI = 0.42–0.84, P = 0.013).
In terms of combination and single treatment, all studies evaluating OS used only the PD1/PDL1 treatment. However, for evaluating PFS, three studies combined the PD1 treatment with CTLA4 antibodies. Interestingly, TMB was more efficient for patients receiving combination immune checkpoint treatments (HR = 0.37, 95% CI = 0.24–0.56, P < 0.001). We also separately compared those studies that included only patients treated with PD1, PDL1, or a combination of both antibodies. The patients treated with PD1 antibodies seemed to have a lower HR of PFS as compared with patients treated with PDL1 antibodies (0.51 vs 0.72).
The use of different cutoffs defining high TMB and low TMB was one heterogeneity for the predictive effect of long-term outcome. In our meta-analysis, there was no trend showing that a higher cutoff had a better predictive effect. Most studies included used 10 mutations/Mb as the cutoff for high TMB and low TMB (HR = 0.63 for OS and HR = 0.79 for PFS, all P < 0.01). There was no significant difference in TMB classified as 20 mutations/Mb because of the small number of included studies, but all results showed that a higher TMB had a better outcome for both OS and PFS in NSCLC patients treated with PD1/PDL1 antibodies.
Discussion
In this systematic review and meta-analysis, we demonstrated that NSCLC patients with a higher TMB have a better survival outcome when treated with PD1/PDL1 antibodies. To the best of our knowledge, this is the first meta-analysis using a forest plot to summarize the relationship between TMB and survival outcome. However, further studies are needed to evaluate the best cutoff of TMB for clinical use.
Among all new treatment strategies, immunotherapy is one of the most promising. A number of clinical studies have proven that the use of drugs targeting immune checkpoint receptors, especially PD1/PDL1 antibodies, is associated with a significantly better survival benefit of patients with advanced NSCLC as compared with chemotherapy.38–40 T-cell failure limits optimal tumor control after chronic tumor antigen exposure. This process is due to the upregulation of immune checkpoint inhibitory receptors after T-cell activation. PD1 is an inhibitory cell surface receptor expressed by T cells, B cells, natural killer cells, and monocytes. 41 The effector function of T cells expressing PD1 in the tumor microenvironment can be downregulated after being activated by PDL1 or PDL2 expressed by tumor cells. PDL1 is widely expressed in hematopoietic cells, nonhematopoietic cells, and tumor cells, whereas PDL2 is mainly expressed in dendritic cells and macrophages. 41 The therapeutic blockade of the PD1 immune checkpoint axis includes monoclonal antibodies (MABS) against PD1 and PDL1.42,43 The use of MABS to block PD1 or PDL1 can prevent the downregulation of T-cell effector functions, thereby allowing T cells to mediate the death of tumor cells. One of the important features of tumors is escaping immune surveillance, and tumor cells always use the PD1–PDL1/2 pathway to escape immune cell attacks. 44 Therefore, the development of screening methods to determine the best suitable patients for PD1/PDL1 antibodies has become important in patients treated with immunotherapy. 44
TMB is the total number of mutations in the coding region of the somatic cell. In 2015, Rizvi et al. 13 was the first to report that immunotherapy patients with elevated TMB can achieve long-term remission (>18 mo). This effect, which is clinically close to “functional cure,” is extremely important for the OS of patients. 13 The mechanism of the association between TMB and the benefit of immunotherapy is not fully understood. A main hypothesis is that the mechanism is linked to the formation of neoantigens, that is, tumor-specific nonself peptides caused by somatic nonsynonymous mutations. 37 Increased TMB increases tumor antigenicity, and tumor antigenicity is the prerequisite for the effectiveness of PD1/PDL1 antibodies. TMB has been proven to be a good predictor of efficacy in multiple immunotherapy clinical trials in recent years.12,20,22,25 Chae et al. 23 conducted a study and demonstrated that the higher TMB was correlated with smoking history and the number of DNA repair mutations, which may be associated with the prolonged OS for patients treated with PD1/PDL1 antibodies. In our study, we demonstrated that patients with higher TMB may have a significant benefit in long-term survival outcome in terms of both OS and PFS, which was the same finding reported in most previous studies. Some studies have also discussed the relationship between PDL1 expression and TMB in patients being treated with PD1/PDL1 antibodies. The clinical trial CheckMate-026 found that in NSCLC patients receiving immunotherapy, the ORR and PFS of the high TMB group were significantly better than those of the low TMB group. Those patients with highly expressed PDL1 (>50%) and with high TMB had the best benefit, whereas patients with highly expressed PDL1 (>50%) but with a low TMB did not have a significant benefit. 45 Another study also demonstrated that even if the PDL1 result is negative, a high TMB was still a predictor for NSCLC patients receiving immunotherapy. 26 Nevertheless, further studies are needed to elucidate the potential relationship between PDL1 expression and TMB in tumor patients.
The source of TMB detection is an important question for NSCLC patients because most patients are first diagnosed in the advanced stage and because of the lack of tumor tissue. Therefore, several studies discussed the effect of circulating/blood TMB as a noninvasive approach to guide immunotherapy in clinical practice.19–21 In their study, Gandara et al. 19 suggested that the tumor tissue TMB was correlated with blood TMB, and there was only a small fraction of observed variance between blood TMB and tumor tissue TMB. 19 Wang et al. 20 also demonstrated that a blood TMB of 6 or higher had an optional diagnostic effect as compared with tumor tissue TMB, with a sensitivity of 0.88 and specificity of 0.71. In our systematic review and meta-analysis, we demonstrated that both tumor tissue TMB and blood TMB had the same effect for the prediction of the long-term outcome of NSCLC patients treated with PD1/PDL1 antibodies.
The best cutoff for defining high TMB and low TMB remains an important question that deserves discussion. Because TMB is a continuous value, the cutoff value needs to be determined to judge the level of TMB for clinical decision making, but there is no unified conclusion thus far. There are different tumor types, detection methods, and platforms, and most studies use the upper third, upper quarter, or median of their own detection data as the cutoff value.19,21,24,27 The common cutoff was listed in our study, which ranged from 6 to 20 mutation/Mb. Gandara et al. 19 compared different cutoff values to assess the efficacy in predicting long-term outcome. They used every 2 mutations/Mb as a cutoff, starting at 4 mutations/Mb, and found that the cutoffs were suitable for prediction if the TMB was less than 20 mutations/Mb. In addition, they determined that a blood TMB of ≥16 mutations/Mb seemed to derive the most clinical benefit from atezolizumab. In our study, we also demonstrated that the TMB had a predictive effect if the TMB was <20 mutations/Mb. Nevertheless, future studies still need to discuss the TMB cutoffs, as such a discussion could improve the benefit of TMB for clinical use.
However, there are still some other questions regarding the clinical use of TMB for NSCLC patients treated with PD1/PDL1 antibodies. The first question is the cost-effective problem. At present, TMB is based on second-generation sequencing. The detection methods include full exon sequencing or target gene capture sequencing based on a gene combination to predict TMB. The cost of clinical testing is about 10,000 RMB in China. Such expensive testing costs greatly limit the promotion and popularization of TMB clinical applications. Second, it takes several weeks from tissue detection to final report, which gives the patients a long time to wait; therefore, the PD1/PDL1 antibodies could not be used during this period. In addition, insufficient tumor tissue or a negative result in the blood sample leads to the inability to analyze the occurrence of TMB. Thus, one mission for future research is the development of a set of economical, fast, and accurate TMB prediction methods that are not inferior to second-generation sequencing.
There were some limitations in our study. First, this was not a meta-analysis based on individual patient data, which might lead to heterogeneity among the different studies. Second, different studies used different cutoff values to define high TMB and low TMB. Although we used subgroup analysis to assess the effect of different cutoffs, the number of included studies was still small. Third, we included some conference studies, and full-text could not be provided. Thus, we could not extract some important or negative data from the analysis, which could possibly have induced selection bias.
Conclusion
We demonstrated that among NSCLC patients receiving PD1/PDL1 antibodies, those with a higher TMB could have a better survival outcome and treatment response. Both tumor tissue TMB and blood TMB had a satisfactory predictive effect for evaluating the long-term outcome. The best cutoffs for TMB should be defined as less than 20 mutations/Mb.
Footnotes
Authors Contributions
Yuhui Zheng, Meihong Yao, Yinghong Yang, designed the meta-analysis; Yuhui Zheng and Meihong Yao, performed literature screening; Yuhui Zheng and Meihong Yao, conducted the quality assessment; Yinghong Yang, performed the statistical analysis; Yuhui Zheng, Meihong Yao, and Yinghong Yang, wrote and revised the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
