Abstract
Resistance to single-agent chemotherapy and molecularly targeted drugs prevents sustained efficacy of treatments. To address this challenge, combination drug treatments have been used to improve outcomes for patients. Potential toxicity of combination treatments is a major concern, however, and has led to the failure of several clinical trials in different cancers. The use of cell-based models of normal tissues in preclinical studies enables testing and identifying toxic effects of drug combinations and facilitates an informed decision-making process for advancing the treatments to animal models and clinical trials. Recently, we established that combinations of molecular inhibitors of mitogen-activated protein kinase (MAPK) and phosphatidylinositol-3-kinase–protein kinase B (PI3K/Akt) pathways effectively and synergistically inhibit growth of BRAFmut and KRASmut colorectal tumor spheroids by blocking feedback signaling of downstream kinase pathways. These pathways are important for cell proliferation, however, and their simultaneous inhibition may cause toxicity to normal cells. We used a cellular spheroid model to study toxicities of drug combinations to human bone marrow and colon. Our results indicated that MAPK and PI3K/Akt inhibitors used simultaneously were only moderately toxic to bone marrow cells but significantly more toxic to colon cells. Our molecular analysis of proliferative cell activities and housekeeping proteins further corroborated these results. Overall, our approach to identify toxic effects of combinations of cancer drugs to normal cells in three-dimensional cultures will facilitate more informed treatment selections for subsequent animal studies.
Introduction
Despite advances in developing targeted cancer therapies, drug resistance remains a major challenge against sustained effects of treatments. Resistance to single-agent treatments with molecular inhibitors occurs through processes such as activation of a compensatory signaling pathway or overactivation of the target to enable cancer cells to survive and proliferate, leading to treatment failure. 1 To overcome limitations of single-agent therapies of cancer, combination treatments with two or more drugs are often used. This strategy is to block signaling of the initially active pathway and a second pathway that confers resistance to inhibition of the initially active signaling.2–4 Compared to monotherapies, drug combinations increase anti-tumor effects and prolong progression-free survival and overall survival. But unfortunately, and despite these benefits, combination therapies can cause significant and excessive toxicities. 5 Toxicity can occur in different forms and affect various organs. Two of the most common tissues that are prone to toxicity from chemotherapies are bone marrow and colon epithelium. This is because both tissues contain highly proliferative cells, and most cancer drugs either directly target highly proliferative cells through interfering with the cell cycle or indirectly inhibit signaling pathways that are critical to proliferation of normal cells.
Drug toxicity is evaluated in preclinical studies using cell-based and animal models. Historically, two-dimensional (2D) cell cultures have been used in the cancer drug discovery pipeline due to their ease of maintenance and drug treatments in cultureware that are compatible with automated liquid-handling and analysis platforms. It is widely recognized, however, that 2D cell cultures do not faithfully represent tumor cells or normal cells for studies of drug efficacy and toxicity, respectively. 6 Therefore, various three-dimensional (3D) cell culture technologies have been developed to reproduce the morphological and certain biologic characteristics of cells in their native environments. 7 Culturing cells as a compact 3D aggregate, known as a spheroid, is one of the most attractive techniques due to the ease of forming and maintaining spheroids with standard cell culture equipment. Here, we used spheroid cultures of normal bone marrow stromal and colon epithelial cells to investigate cytotoxicity of combination treatments with kinase inhibitors.
The mitogen-activated protein kinase (MAPK) pathway is an important signaling cascade with significant roles in cell proliferation, differentiation, stress responses, and apoptosis. 8 Similarly, the phosphatidylinositol-3-kinase–protein kinase B (PI3K/Akt) pathway is another central signaling cascade for cell survival and proliferation. 9 These two pathways are frequently dysregulated in various cancers and, thus, provide attractive targets for targeted therapies. 10 More than 50% of colorectal cancers are characterized by alterations and mutations in oncogenic pathways such as MAPK or PI3K/Akt. 11 Due to extensive crosstalk among these pathways in different cancers,12–14 their simultaneous inhibition generates significant anti-tumor effects.15–17 For example, our group recently demonstrated that combinations of inhibitors of MAPK and PI3K/Akt pathways synergistically inhibit growth and invasiveness of colorectal cancer cells and block their adaptive drug resistance.13,14 Building on these studies, we aimed to investigate potential toxicities of combined inhibition of these two oncogenic pathways to normal colon epithelial and bone marrow stromal cells in 3D spheroid cultures. Our results showed that this approach generates marginal toxic effects to bone marrow stromal cells but causes significant toxicity to colon epithelial cells, which we also confirmed through molecular analysis of cell proliferation and metabolism. Overall, we demonstrated the feasibility of conducting quantitative studies of normal cell cytotoxicity from cancer drug combinations using 3D cell cultures as a preclinical tool.
Materials and Methods
Cell Culture
The CCD 841 CoN cell line (ATCC CRL-1790) was purchased from ATCC (Manassas, VA), and the HS5 cell line was generously provided by Dr. Gary D. Luker (University of Michigan, Ann Arbor, MI). Eagle’s minimal essential medium (EMEM; Sigma-Aldrich, St. Louis, MO) and Dulbecco’s modified Eagle medium (DMEM; Sigma-Aldrich) were supplemented with 10% fetal bovine serum (FBS; Sigma-Aldrich) and 1% streptomycin/penicillin/amphotericin B (ThermoFisher, Waltham, MA) and used to culture CCD 841 CoN and HS-5 cells, respectively. Cells were kept in a humidified incubator at 37 °C supplemented with 5% CO2 and subcultured when they were confluent. CCD 841 CoN cells were subcultured at a ratio of 1:2–1:4, and HS-5 cells were subcultured at a ratio of 1:8–1:12. To dissociate the cells from culture flasks, a 0.25% trypsin solution (ThermoFisher) was used. Trypsin was neutralized by adding a respective complete growth medium. The resulting cell suspension was centrifuged down at 1000 rpm for 5 min at 4 °C, and the supernatant was removed. The cells were suspended in 1 ml of medium for counting.
Spheroid Culture
To form spheroids, our previously established aqueous two-phase system (ATPS) technique was used. Briefly, bio-ultra 35 kDa polyethylene glycol (PEG; Sigma-Aldrich) and 500 kDa dextran (DEX; Pharmacosmos, Holbaek, Denmark) were dissolved in a complete growth medium to obtain solutions with concentrations of 5.0% (w/v) PEG and 12.8% (w/v) DEX. Next, 30 µL of the PEG solution was dispensed in each well of a round-bottom ultralow-attachment 384-well plate (Corning, Corning, NY). A suspension of 6.6×107 cells/ml was prepared and thoroughly mixed with the 12.8% (w/v) DEX solution at a ratio of 1:1 (v/v). The resulting solution had a DEX concentration of 6.4% (w/v) and a cell density of 3.3×107 cells/ml. A Bravo SRT robotic liquid handler (Agilent, Santa Clara, CA) was used to dispense 0.3 µL of the solution in each well, resulting in a cell density of 103 cells/well. This process was done one column at a time to reduce the required number of cells. The DEX–cell solution was robotically mixed after printing eight columns to ensure uniformity of the cell suspension. The cultures were maintained for 48 h to allow the cells to form spheroids.
Metabolic Activity of Spheroids
A Prestoblue reagent (ThermoFisher) at 10% of total well volume was used to measure the metabolic activity of cellular spheroids throughout a 10 d period after formation of spheroids. After 3 h, the fluorescence intensity was measured using a Synergy H1M plate reader (BioTek Instruments, Winooski, VT) at 560 nm excitation and 590 nm emission wavelengths. The measured fluorescence signals were normalized to the signal from the first time point. To complement these measurements, phase images of the spheroids were captured using an inverted microscope (Axio Observer, Zeiss, Oberkochen, Germany) equipped with a high-resolution camera (AxioCam MRM, Zeiss). An average diameter of spheroids was determined by averaging maximum and minimum conjugate diameters using Fiji (https://fiji.sc/) distribution of the ImageJ software [National Institutes of Health (NIH), https://imagej.nih.gov/ij/]. 18
Drug Preparation and Combination Treatments
Trametinib, SCH772984, and dactolisib were purchased from Selleckchem (Houston, TX). Stock solutions were prepared by dissolving the compounds in DMSO at concentrations of 10 mM, 3.4 mM, and 1.5 mM, respectively. To ensure that dactolisib was completely dissolved in DMSO, the mixture was heated at 60 °C for 30 min, as recommended by the manufacturer. The drug solutions were stored at −80 °C. Drug concentrations in combination treatments were kept the same as in our previous study to directly compare the responses of normal and cancer cells. The concentrations were selected based on a 50% lethal dose of each drug against HCT116 colorectal cancer cells (HCT-LD50) and arranged in a 7×7 combination matrix. The matrix was assembled by combining multiples of this HCT-LD50 concentration (4, 2, 1, 0.5, 0.25, 0.125, and 0) for each compound. Drugs were added 48 h after printing of cells (day 2) to allow sufficient time for spheroid formation. Drug solutions with 4× concentrations of the combinations (i.e., 16, 8, 4, 2, 1, 0.5, and 0) were first prepared. From these solutions, 15 µL of solution of a MAPK pathway inhibitor and 15 µL of dactolisib solution were added to each well containing 30 µL of culture medium, effectively diluting each drug by fourfold. This arrangement enabled simultaneous screening of both combination and single-agent treatments in a 384-well plate. To avoid potential effects of evaporation of medium, spheroids in the outermost wells were not used. Metabolic activity of cells was measured 4 d later (day 6) using a Prestoblue assay. To obtain cell viability of spheroids, the fluorescence signal from each treated sample was divided by the vehicle control average at the same timepoint. Fraction of cells affected (Fa) was calculated by subtracting the normalized metabolic activity from unity for each treatment. Each pair of concentrations had six replicates.
Western Blotting
To obtain sufficient protein mass, spheroids from half of a 384-well plate were used for each condition. Spheroids were harvested and transferred into 15 mL conical tubes. Following centrifugation and removal of the supernatant, spheroids were washed with cold phosphate-buffered saline (PBS) and lysed in 300 µL of a radioimmunoprecipitation assay (RIPA) buffer solution [50 mM Tris–HCl, 150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate, and 0.1% sodium dodecyl sulfate (SDS), pH 7.4±0.2] with protease inhibitor (complete mini, Roche Diagnostics, Indianapolis, IN) and phosphatase inhibitor (ThermoFisher). To ensure proper lysing of cells, the solutions were sonicated twice, each time for 5 s at an amplitude of 20–30%. The sonication was repeated if large vesicles were observed under a microscope. Then, the lysate was centrifuged at 14,000 RCF (relative centrifugal force) for 5 min at 4 °C, and the supernatant was used as the protein solution. Protein concentration in each sample was quantified using a bicinchoninic acid (BCA) protein assay (ThermoFisher). The solutions were then diluted to the concentration of the least concentrated sample, ensuring equal loading conditions. From the normalized solutions, a constant mass of protein (8–20 µg) was loaded into each cell of a 4–15% polyacrylamide gel (Bio-Rad, Hercules, CA) for electrophoresis. The proteins were transferred onto a nitrocellulose membrane using electroblotting. Next, the membranes were blocked for unspecific bonding using 5% nonfat dry milk for 1 h. Solutions of rabbit monoclonal antibodies for glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and β-actin (Cell Signaling Technology, Beverly, MA) were prepared in the blocking buffer at concentrations suggested by the manufacturer. The membranes were incubated with the primary antibodies overnight at 4 °C; washed three times, each time for 15 min; and incubated with a horseradish peroxidase (HRP)-conjugated anti-rabbit immunoglobulin G (IgG; Cell Signaling Technology) for 1 h. After a washing step, membranes were developed using an ECL (enhanced chemiluminescence) detection kit (GE Healthcare, Chicago, IL) and imaged using the FluorChem E imaging system (ProteinSimple, San Jose, CA). Each Western blot experiment was repeated at least twice.
Histological Examination of Spheroids
Following our established technique, 19 spheroids were harvested 4 days after treatments (6 days after printing), placed in Petri dishes, and fixed with 4% solution of formaldehyde for 15 min at room temperature. Spheroids were fixed and washed with phosphate-buffered saline (PBS) three times, each time for 5 min. To prevent crystal formation during freezing, samples were incubated with 100 µL of a 30% (w/v) sucrose solution overnight at 4 °C. Next, cryomolds were half-filled with a tissue freezing medium (General Data, Cincinnati, OH) and flash-frozen on dry ice. Spheroids were gently transferred to the middle of the cryomolds and sandwiched by adding more tissue freezing medium on top. The samples were immediately flash-frozen and kept at −80 °C for storage. To section the spheroids, a cryostat (Leica CM1860, Leica, Wetzlar, Germany) was used to obtain slices ~15 µm thick. The cryosections were placed on Superfrost Plus microscopic slides (Fisher Scientific, Pittsburgh, PA) and stored at −20 °C. To stain the samples, they were first brought back to the room temperature, washed with PBS for 2 min, and blocked with 5% (v/v) donkey serum in a blocking buffer (PBS with 0.3% Triton X-100). Subsequently, they were stained with a primary Ki-67 immunohistochemical (IHC)-specific rabbit monoclonal antibody (Cell Signaling Technology) at concentrations recommended by the manufacturer in an antibody dilution buffer [PBS with 0.1% bovine serum albumin (BSA) and 0.3% Triton X-100]. Samples were washed three times with PBS, each time for 5 min, and incubated in rhodamine-conjugated anti-rabbit IgG (Jackson ImmunoResearch, West Grove, PA) as the secondary antibody for 1 h at room temperature. To stain the DNA, samples were also incubated in a 2 µg/ml Hoechst (Life Technologies, Carlsbad, CA) solution for 15 min, and then rinsed three times with PBS. To prevent loss of fluorescence, an antifade reagent (ThermoFisher) was added in the last step, and samples were sealed using coverslips and transparent nail polish. Fluorescent images were captured using a confocal microscope (Fluoview FV1000, Olympus, Tokyo, Japan) and processed using Fiji.
Statistical Analysis
One-way analysis of variance (ANOVA) was used to assess significance in GraphPad prism software (GraphPad, San Diego, CA). p-values of less than 0.05 were used to define statistical significance.
Results
Microprinting of Spheroids and Their Metabolic Activity Measurements
Our robotic ATPS enables precision microprinting of tumor spheroids without exerting mechanical, chemical, or thermal stresses on cells.
20
Facilitated by the immiscibility of the two aqueous phases, cells in the DEX phase nanodrop spontaneously aggregate and form a single spheroid in each well of 384-well plates (
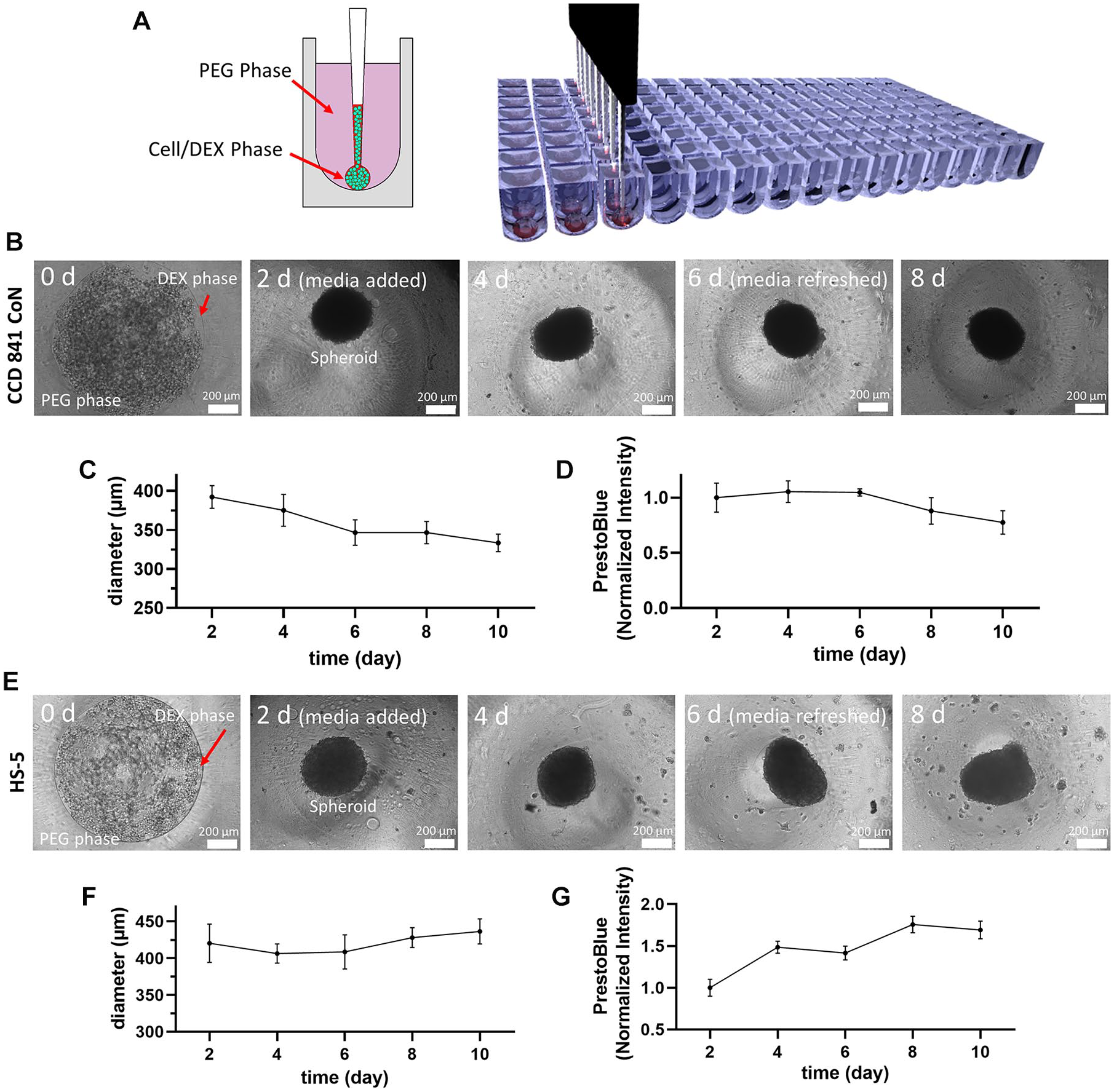
(
Combination Drug Treatments of Spheroids of Cancer and Normal Cells
In previous studies, we used KRASmut and BRAFmut colorectal tumor spheroids and demonstrated that single-agent treatments with inhibitors of the MAPK pathway activate compensatory signaling through the PI3K/Akt pathway, rendering cells resistant to the treatment. Then, we established that combining inhibitors of these pathways (trametinib or SCH772984 as MEK inhibitors and dactolisib as a PI3K inhibitor) synergistically blocks cross-talk between the pathways and inhibits proliferation of HCT116 and HT29 spheroids.13,14 Due to the potential increase in the toxicity of drug combinations compared to when a drug is used alone, we aimed to determine toxicities of combinations of MAPK and PI3K/Akt inhibitors to normal colon and bone marrow stromal cells.
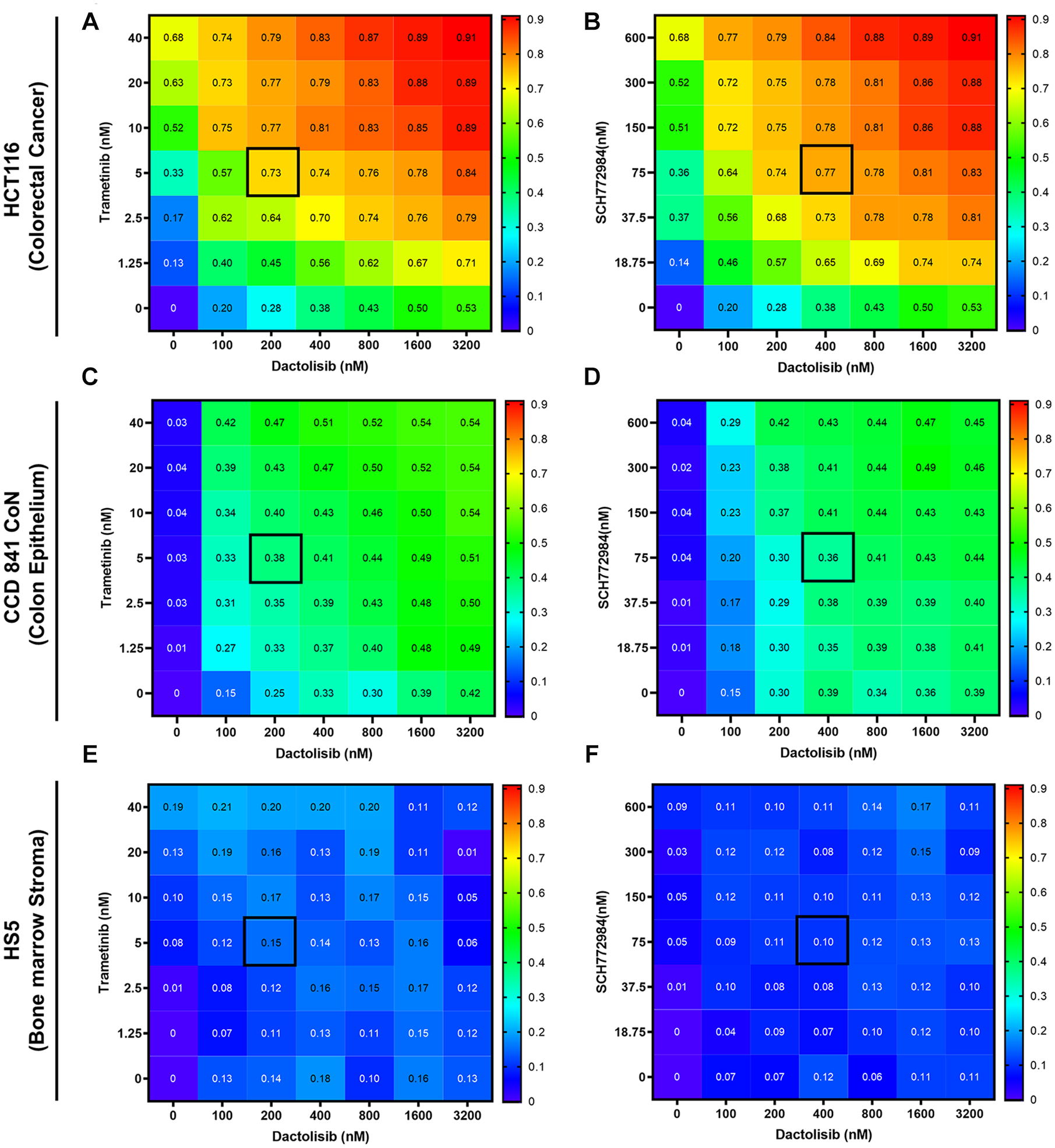
Combination treatments with phosphatidylinositol-3-kinase (PI3K) and mitogen-activated protein kinase (MAPK) pathway inhibitors. Heatmap plots show effects of combination treatments with trametinib–dactolisib and SCH772984–dactolisib on (
Molecular Analysis of Toxicity of Combination Treatments to Normal Cells
To validate our phenotypic results of toxicity of combinations of MAPK and PI3K/Akt inhibitors to normal colon epithelial and bone marrow stromal cells, we performed a Western blot analysis of two major housekeeping proteins, GAPDH and β-actin, in drug-treated spheroids. GAPDH is an important enzyme for cellular energy metabolism, whereas β-actin regulates cell growth and motility.23,24 We used 5 nM trametinib–200 nM dactolisib and 75 nM SCH772984–400 nM dactolisib combinations that were highly effective against HCT116 cells and generated low-to-moderate toxicity to the normal cells. Consistent with the metabolic activity results, trametinib–dactolisib and SCH772984–dactolisib treatments of CCD 841 CoN cells significantly reduced expression levels of GAPDH by 70% and 29% and β-actin by 52% and 36% ( Fig. 3a–3b ), respectively. In contrast, the treatments showed only moderate effects on the bone marrow stromal cells. That is, trametinib–dactolisib and SCH772984–dactolisib reduced GAPDH levels in HS5 cells by 27% and 7% and β-actin levels by 28% and 20% ( Fig. 3c–3d ), respectively.
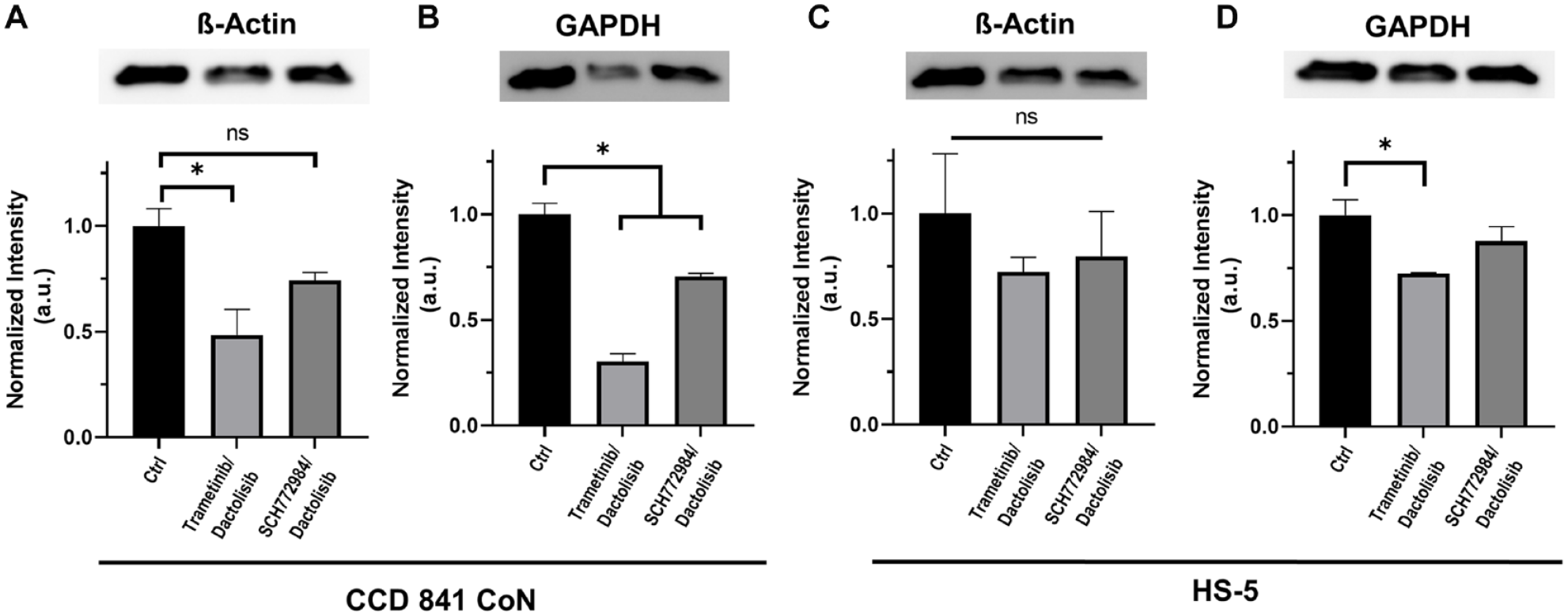
Expression of housekeeping proteins [glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and β-actin] in normal cell spheroids under treatment with 5 nM trametinib–200 nM dactolisib or 75 nM SCH0772984–400 nM dactolisib, compared to the respective vehicle controls. *p < 0.05. ns, Not significant.
Effects of Combination Treatments on Proliferation of Normal Cells
We performed immunostaining of cryosectioned spheroids for Ki-67 protein, which is used as a marker of proliferative cells. Nontreated spheroids of CCD 841 CoN and HS5 cells showed a uniform distribution of Ki-67+ cells throughout the sections (
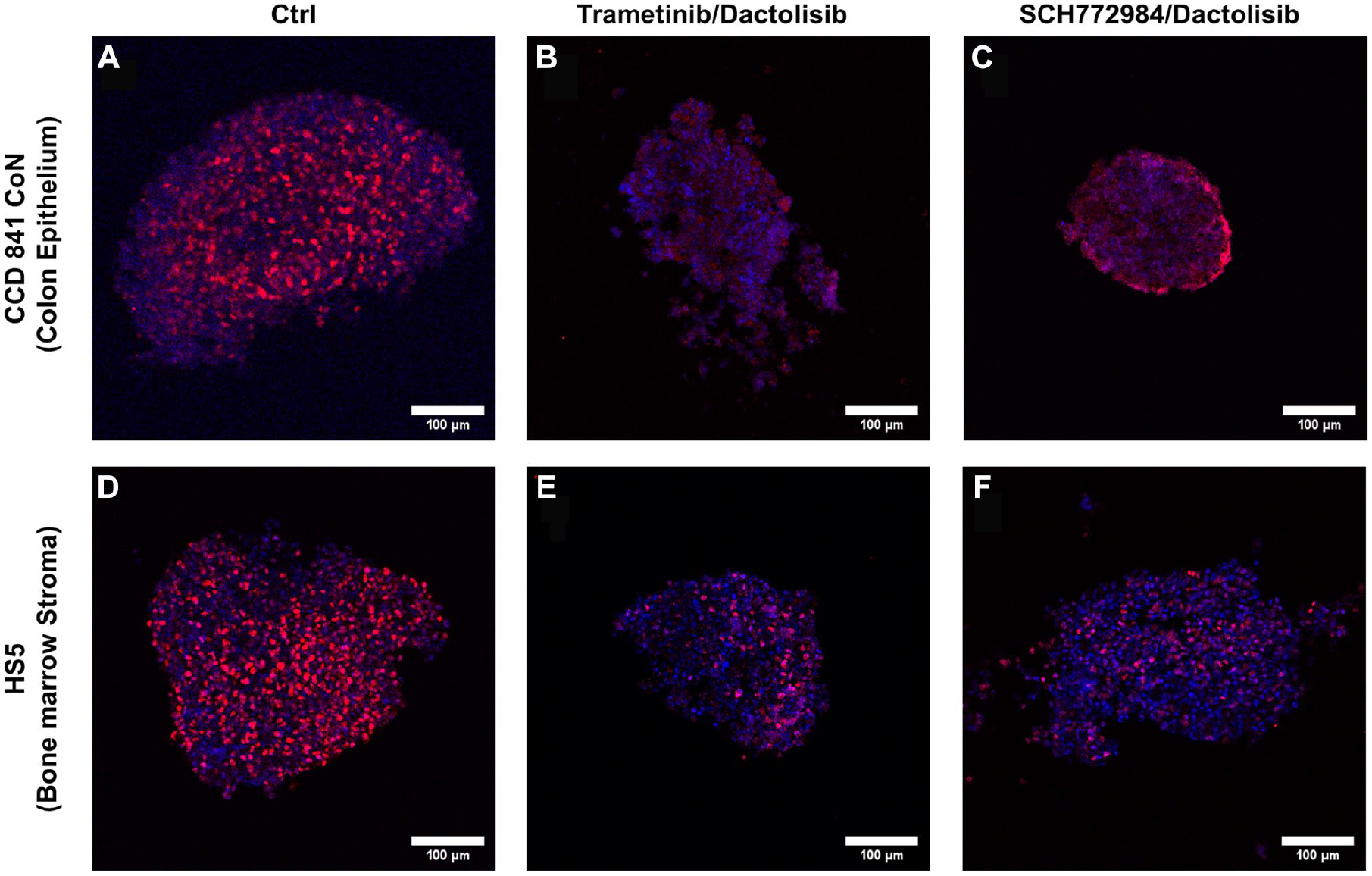
Immunostained cryosections of normal cell spheroids showing the distribution of (
Discussion
Currently, only about 10% of drug candidates reach clinical trials, 25 and more than half of those entering phase III trials ultimately fail. 26 The lack of efficacy and the excessive toxicity are the major causes of failure of drug candidates in clinical trials. Although drug-induced toxicity is monitored in all phases of clinical trials, failure of ~20% of compounds in phase III due to toxicity highlights a need to more reliably study toxicity in preclinical stages of drug discovery. 27 Historically, mouse models have been the gold standard to assess drug toxicity. Monitoring the animal weight in the course of treatments and immunohistochemical analysis of various tissues after treatments are terminated are routinely used. Besides the high costs and ethical issues associated with them, 28 animal models do not reliably predict drug toxicity in humans. 29 This recognition has led to efforts to develop humanized cell-based models to study the efficacy and potential toxicity of candidate drugs. Three-dimensional cultures of cell lines or primary cells as spheroids and organoids, and in microfluidic devices, are among the most widely used models.30–34
A major obstacle in effective treatment of cancer patients is resistance to conventional chemotherapy and molecularly targeted therapies. Although most drugs initially generate anti-tumor effects, resistance makes treatments less effective or ineffective altogether. While heterogeneity of cells within a tumor is a major cause of drug resistance and tumor relapse, cancer cells can also develop adaptive responses to the treatment such as activation of compensatory survival pathways and overactivation of the target. 13 To overcome resistance to single-agent chemotherapy, combination therapies that involve simultaneous use of two drugs have been pursued.4,14 This approach has proved effective in blocking drug resistance and improving outcomes. 35 Drug combinations, however, often cause greater off-target toxicities than single-agent treatments. 4 Several clinical trials were discontinued due to excessive toxicities to patients. For example, PI3K/Akt/mTOR and RAS/RAF/MEK/ERK pathways are two of the most commonly dysregulated signaling cascades in human cancers.36,37 Due to extensive cross-talk between these pathways, targeting them simultaneously has been an attractive strategy. 38 While early-stage clinical trials for these combinations showed promising anti-tumor effects,39,40 significant toxicity led to frequent dose interruptions and reductions, or stopping of the trials.41–44 This highlights a need for modeling drug-induced toxicities in preclinical studies. Organs-on-a-chip devices that create several interconnected compartments representing different organs are very promising tools for these investigations, 45 albeit their complexity and low throughput limit their broad use in preclinical studies.
Our approach to use cellular spheroids and study cancer drug toxicity to normal cells allows screening of large arrays of drugs and their combinations and quantitative assessment of drug responses of cells. Our results showed that simultaneous inhibition of MAPK and PI3K/Akt pathways generates minimal cytotoxicity to bone marrow stromal cells across a wide range of concentrations. The combination treatments, however, led to significant toxicity to the colon epithelial cells. The MAPK inhibitors had a very marginal effect on both normal cells. This is consistent with the tolerability of US Food and Drug Administration (FDA)-approved MAPK inhibitors such as trametinib for certain cancers.46,47 The toxicity of the combination treatments to the epithelial cells was mainly caused by the PI3K inhibitor, dactolisib, and in a dose-dependent manner. This agrees with a report of gastrointestinal toxicities associated with dactolisib in treatments of advanced solid tumors.48,49 Furthermore, toxicity to the normal colon epithelial cells increased at the same dactolisib concentration and by increasing the MEK inhibitor dose, potentially due to an additive effect. Because single-agent treatments using trametinib and SCH0772984 affected the viability of normal cells very marginally, calculating combination indices using Chou’s method was not feasible without significant extrapolation to determine synergy between the drug pairs. Although cytotoxicity to the colon epithelial cells was relatively and significantly less than that to HCT116 colorectal cancer cells, it diminished proliferative Ki-67+ cells and downregulated housekeeping proteins such as GAPDH and β-actin. In addition, our system reflects only the acute effects of combination therapies on normal cells. Although longer term cultures can potentially be used to assess drug toxicity during cyclic treatment regimens, the decline in the viability of normal colon epithelial cells after 6 days of culture limits the use of this cell line for long-term studies. Other approaches such as organoid culture of normal cells or organotypic models provide alternative preclinical models for long-term toxicity tests.
Despite using a simple 3D model of normal cells, our study demonstrated the feasibility of high-throughput screening of drug combinations to evaluate potential toxicities to normal cells and the differences in their sensitivities to specific targeted therapies. Our results were consistent with those from clinical trials of simultaneous inhibition of MAPK and PI3K/Akt pathways in patients with solid tumors. Nevertheless, care must be taken when drawing correlations between results from preclinical assays and clinical trials due to complex processes such as drug pharmacokinetics and pharmacodynamics (PK/PD) and regenerative capabilities of different tissues in patients that cannot be accurately modeled in vitro. This and similar studies will be very useful in informing the effectiveness of different cancer drugs and their combinations against tumor cells and differences in vulnerabilities of different tissues to such treatments to narrow down treatment selections to those most effective against cancer cells and with reduced toxicity to normal tissue. Furthermore, future improvements of this model to develop 3D models of primary cells derived from patients or induced pluripotent stem cells (iPSCs) can also serve preclinical studies of off-target toxicities of cancer drugs to identify combination treatments with minimal toxic effects to normal cells and tissues.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants CA216413 and CA225549 from the National Institutes of Health and 1801591 from National Science Foundation.
