Abstract
We present a low-cost clinically viable ventilator design, AmbuBox, using a controllable pneumatic enclosure and standard manual resuscitators that are readily available (AmbuBag), which can be rapidly deployed during pandemic and mass-casualty events with a minimal set of components to manufacture and assemble. The AmbuBox is designed to address the existing challenges presented in the existing low-cost ventilator designs by offering an easy-to-install and simple-to-operate apparatus while maintaining a long lifespan with high-precision flow control. As an outcome, a mass-producible prototype of the AmbuBox has been devised, characterized, and validated in a bench test setup using a lung simulator. This prototype will be further investigated through clinical testing. Given the potentially urgent need for inexpensive and rapidly deployable ventilators globally, the overall design, operational principle, and device characterization of the AmbuBox system have been described in detail with open access online. Moreover, the fabrication and assembly methods have been incorporated to enable short-term producibility by a generic local manufacturing facility. In addition, a full list of all components used in the AmbuBox has been included to reflect its low-cost nature.
Introduction
As of June 15, 2020, the total number of cases of coronavirus disease 2019 (COVID-19) surpassed 7,900,000 with more than 430,000 fatalities worldwide. 1 Acute respiratory distress syndrome (ARDS) has so far been the most common complication in COVID-19 patients requiring intensive care unit (ICU) admission.2–6 While mild cases of ARDS may respond to non-invasive ventilation (NIV) such as high-flow nasal cannula, moderate to severe cases of ARDS often require intubation and ventilator support. 7 Current projections estimate that up to 1 million ventilators are needed in the United States to manage the COVID-19 pandemic, not including the anticipated surges expected in the coming months as quarantining restrictions are lifted. This is much higher than the number of ventilators estimated to be available in the United States, ranging from 60,000 to 160,000 units. 8 Furthermore, many US hospitals are estimated to be functioning currently at or near their ventilator capacity prior to this pandemic.9,10
Mechanical ventilation, originally developed in the early twentieth century within the context of the polio pandemic, has been revolutionary and evolutionary with respect to delivering optimized respiratory care for critically ill patients. 11 While mechanical ventilators have been significantly improved, they have also become more complicated. This is a natural consequence of “feature creep,” the addition of features into a device to improve the experience of users, such as healthcare providers and patients. Although this is considered a process that improves the overall function of ventilators, feature creep in fact increases the complexity of the machines in a nonlinear way. 12 For every added feature, numerous additional algorithms and software adjustments need to be made to accommodate this feature, making regulatory hurdles more challenging, and contributing to the overall increasing cost and building complexity of the machines. As a result, a modern ventilator typically used in an ICU can cost between $25,000 and $50,000. 12 When considering new medical equipment, however, evidence-based, patient-centered value is of utmost importance, 13 and not all added premium features on modern ventilators have data-driven evidence to show added clinical benefits. Such features may be instead economically driven, especially when considering that in the United States, there seems to be a predilection in obtaining the latest medical technology just because it is available. 14
In contrast, the need and use of ventilators in developing countries and rural settings is also a challenging issue that has and will continue to exist. Shah et al. studied trauma and critical care–related issues in Nepal, where they noted that electrical shortage was a major infrastructural deficiency that existed in all parts of the country. 15 With respect to equipment, lack of pulse oximetry and ventilators was a problem in most hospitals, both large and small.
To address these issues, three categories of low-cost ventilators are currently under active development: sleep apnea machines converted to ventilators, conventional ventilator designs using components from nonclinical supply chains, and bag valve mask (BVM)-based (AmbuBag) ventilators. 16
In the first category, commercial sleep apnea machines are converted into ventilators by adding an oxygen concentrator and virus filter. Both CPAP (continuous positive airway pressure) and BiPAP (bilevel positive airway pressure) machines have been studied. Researchers and clinicians from University of California, Berkeley and University of California, San Francisco have launched a project (Ventilator SOS) to promote the donation of sleep apnea machines to convert into ventilators for the COVID-19 pandemic. 17 ResMed and GE Healthcare have received US Food and Drug Administration (FDA) Emergency Use Authorization (EUA) to use their sleep apnea machines for ventilator development in the COVID-19 pandemic, among a list of others. 18 Sleep apnea machines are expensive, however, and can be difficult to access in resource-deprived settings, and this limits the ability of a prototype of this nature to be rapidly deployed.
In the second category, researchers are designing conventional ventilators with off-the-shelf components from nonclinical supply chains. A number of academic groups and nonmedical companies have led the efforts in this direction, including VITAL (Ventilator Intervention Technology Accessible Locally) from the Jet Propulsion Laboratory of NASA, 19 Vent4US from Stanford University, 20 Mechanical Ventilator Milano (MVM) from Elemaster S.p.A. Tecnologie Elettroniche, 21 RespiraWorks from UC Berkeley, 22 the European Organization for Nuclear Research (CERN), 23 and so on. Among them, VITAL and MVM devices have recently received FDA EUA. 18 This design strategy simplifies existing ventilator designs and circumvents the currently strained ventilator component supply chain. As an example, researchers have substituted more premium components such as proportional valves and flow sensors with inexpensive pinch valves20,22 and differential pressure sensors, respectively. 24 These prototypes still require numerous components, however, which may not be suitable for mass production and rapid deployment in a short period of time.
Because the primary design challenge still remains, several universities have been pursuing further simplified solutions by automating a standard manual BVM (e.g., AmbuBag). A list of the representative BVM-based ventilator projects with relevant specifications and features is summarized in Table 1 , including MIT’s E-Vent project, which provides open access to the detailed design files. BVM-based ventilators have the distinct advantages of availability, minimal number of components, simple mechanism, low cost, and the capability for rapid deployment. The majority of currently available BVM-based designs, however, all rely on the machining or 3D printing and assembly of a series of mechanical transmission components. Machined parts can be challenging to mass produce during lockdown periods, while 3D-printed parts may experience limited lifespan and eventually catastrophic failure. In addition, these mechanically driven systems require manual loading and alignment of the AmbuBag within the machine arms to achieve optimal compression of the bag to achieve adequate oxygenation and ventilation. Alternatively, OxVent, a joint research effort from University of Oxford and King’s College London, takes a different route, using pneumatic control and compressed air to compress the AmbuBag and deliver a breath. The OxVent design seals the AmbuBag inside of the chamber permanently, which is problematic in clinical situations in which manual bagging is required, because there is no way to switch the automated bagging back to a manual mode if a control failure occurs. One potential advantage of permanently sealing the chamber, however, is that it could offer higher reliability (e.g., avoid leaking) for the pressurized compartment. 25
A List of Representative Projects on BVM-Based Ventilators for COVID-19.
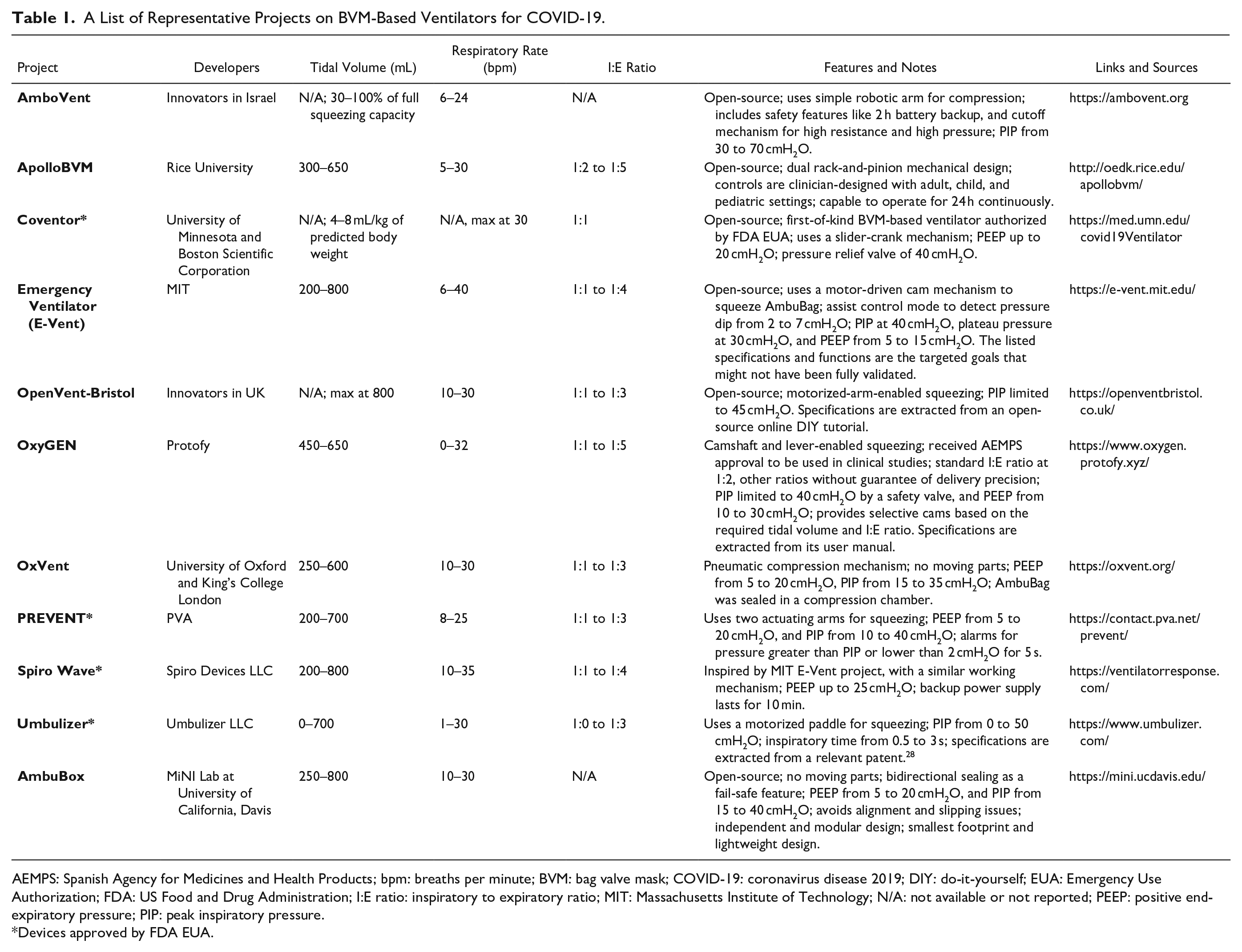
AEMPS: Spanish Agency for Medicines and Health Products; bpm: breaths per minute; BVM: bag valve mask; COVID-19: coronavirus disease 2019; DIY: do-it-yourself; EUA: Emergency Use Authorization; FDA: US Food and Drug Administration; I:E ratio: inspiratory to expiratory ratio; MIT: Massachusetts Institute of Technology; N/A: not available or not reported; PEEP: positive end-expiratory pressure; PIP: peak inspiratory pressure.
Devices approved by FDA EUA.
In this study, we propose and demonstrate a low-cost clinically viable ventilator design, AmbuBox, using a controllable pneumatic enclosure and standard manual resuscitators that are readily available, which can be rapidly deployed during mass-casualty and pandemic situations with a minimal set of components to manufacture and assemble. The current AmbuBox ventilator is arguably the simplest and smallest ventilator, with a modular and independent design and bidirectional sealing for the AmbuBag; it is assembled with laser-cut and off-the-shelf parts, while maintaining high accuracy and long-lasting stability. We have demonstrated the AmbuBox for adjustable control of tidal volume (TV) from 250 to 800 mL within 10%, respiratory rate (RR) from 10 to 30 bpm (breaths per minute) within 2 bpm, and pressure monitoring within 2 cmH2O. The positive end-expiratory pressure (PEEP) can be adjusted from 5 to 20 cmH2O, and the peak inspiratory pressure (PIP) limit can be set from 15 to 40 cmH2O in addition to the relevant alarms for high and low pressures. The AmbuBox ventilator is a viable ventilator design to fill the gap between the conventional ventilators and AmbuBags, by retaining the critical functions of a ventilator for emergent use during the current COVID-19 pandemic and in mass-casualty situations, as well as emergent clinical use in low-resource settings.
Materials and Methods
Figure 1a shows the testing system of AmbuBox with key components labeled. The AmbuBag (1, AirFlow manual resuscitator adult size, SunMed Medical, Marlton, NJ) was connected with an extension tubing (2) and a HEPA filter (3, ISO-Gard HEPA Light, Teleflex, Morrisville, NC) into a test lung (4, QuickLung Breather, IngMar Medical, Pittsburgh, PA). The exhausted air from the test lung was drained from a manually adjustable PEEP valve (5, VP700, SunMed Medical) connected with the AmbuBag. A pressure sensor (6, Go Direct Gas Pressure Sensor, Vernier, Beaverton, OR) and a spirometer (7, Go Direct Spirometer, Vernier) were connected inline between the extension tubing and the filter with an adapter that was originally used for oxygen enrichment in CPAP machines. The AmbuBag was bidirectionally sealed in a laser-cut acrylic AmbuBox chamber (8). An additional Vernier pressure sensor (9) was connected to monitor the chamber pressure. Compressed air from the wall was connected to a pressure regulator (10, SAW2000M-N02BG, PneumaticPlus, Torrance, CA) and then attached to a three-way solenoid valve (11, Masterflex 3-way solenoid pinch valve, Cole-Parmer, Vernon Hills, IL). One end of the solenoid valve was connected with atmosphere, while the other end was connected to the chamber with an adapter and a one-way valve (12), which was detached from the AmbuBag. A microcontroller unit (MCU) with adjustment knobs and an accessory LCD display were used to control the solenoid valve and monitor the conditions in the simulated patient airway and the AmbuBox chamber.

Overview of the testing system and assembly of AmbuBox. (
Figure 1b–1d shows the process of the bidirectional sealing of AmbuBag into the acrylic chamber. As shown in Figure 1b , the AmbuBox chamber was made of two components: an upper cap and a lower cap. Each of the two caps was prepared by laser ablation (Epilog Mini 18/24 laser system, Epilog Laser, Golden, CO) of five pieces of 0.5-inch-thick acrylic sheets, with the interlock structures designed for assisted and simplified assembly. As an alternative to using a standard laser cutter to make these components, in resource-deprived situations one could alternatively use other, easier-to-access tools such as a power saw or hand saw. The caps were assembled and bonded using acrylic cement (cat. no. 10315, IPS Corporation, Compton, CA). Each of the caps was designed with curved edges at the ends to best fit the contour of the AmbuBag. Four clasp clamps were installed to hold and seal the two caps tightly with rubber bands attached to both of the two heads of the AmbuBag to assist sealing, while reserving the function for convenient separation of the caps to quickly access the AmbuBag in situations of emergency or system failure.
In detail, the electronic control unit consists of an MCU module (Arduino Mega R3, Arduino, www.arduino.cc), a 1602 LCD display, two adjustment knobs, and accessories such as solid-state relay (KF0602D, Kyotto, Kytech Electronics, Shenzhen, China) and a power supply adapter (Chanzon, Shenzhen, China). In the first stage, the pressure sensors and spirometer from Vernier were used for the characterization of the AmbuBox ventilator, due to their high precision and convenient graphical user interface (GUI) for data display, in addition to their capability for Bluetooth-enabled wireless data transmission. One snapshot of the collected data in real time is shown in
Results and Discussion
In this part, we first described the operating principles of AmbuBox. Next, the minimal functions of a clinically viable ventilator were discussed, and we identified that tidal volume and respiratory rate are the two most critical parameters clinicians emphasize. Based on the operating principles of the AmbuBox, we then investigated the inflation/deflation and inspiration/expiration dynamics of the AmbuBox, by real-time monitoring the pressure and flow inside both the AmbuBox chamber and the simulated-patient breath circuit, to correlate the dynamics of AmbuBox with the two critical parameters aforementioned. Third, we conducted a thorough parametric study of the dependence of tidal volumes and respiratory rates on the various control parameters and patient conditions, such as the inflation duration, and the compliance and resistance of the patient lung, as tabularized in
Operating Principles
The AmbuBox ventilator functions through an automated pneumatically driven squeezing and releasing of an AmbuBag. Specifically, the AmbuBox consists of three components (
Fig. 2a
): (1) a standard AmbuBag-type manual resuscitator with its accessories (yellow-colored), (2) a laser-cut chamber (blue-colored) with airtight and bidirectional sealing to the AmbuBag, and (3) a pneumatic control unit with solenoid valves and pressure- and flow-sensing feedback (green-colored). The AmbuBag can be placed bidirectionally inside the AmbuBox, where it is squeezed by pressurizing the AmbuBox via a positive-pressure air source (e.g., compressed gas line, gas cylinder, or air pump). The solenoid valve allows for precise control of the duration of inflation and deflation of the AmbuBox. As a result, the AmbuBox pushes oxygenated air from the AmbuBag through the extension tubing into an inspired breath for the patient. The expired air from the patient is exhausted from a standard adjustable PEEP valve, while passing through a HEPA filter to capture aerosols from the patient, protecting health care workers. A gauge pressure transducer and a differential pressure-based flowrate-detecting orifice are connected along the breathing circuit of the patient to monitor any aberrancies in the airway pressure and flowrate. Similarly, a gauge pressure transducer and a flow sensor are installed between the pneumatic controller and the AmbuBox chamber to monitor the pressure and flowrate during the inflation and deflation of the AmbuBox chamber. A standard MCU with a simple user interface allows adjustment of respiratory rate
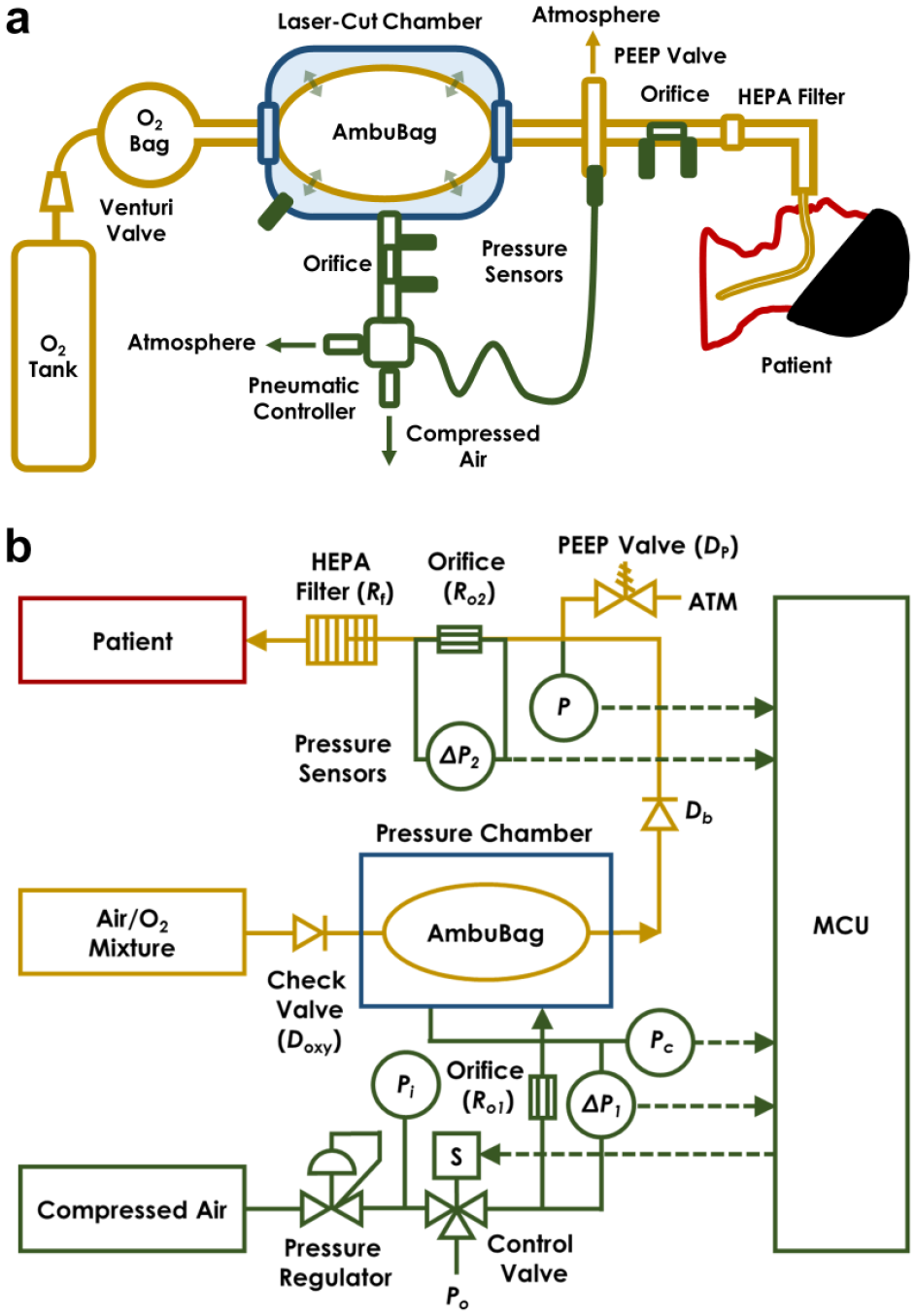
Concept of AmbuBox ventilator. (
To model the functionality of the proposed AmbuBox ventilator, a pneumatic-electronic analogical circuit has been developed, as illustrated in
Figure 3
. Briefly, the operation of the AmbuBox ventilator can be separated into three processes, namely the inflation of the AmbuBox chamber (inspiration of the patient), the expiration of the patient, and the deflation of the chamber. It is worth mentioning that all three processes contribute to the setting of respiratory rate and tidal volume, two of the most critical parameters for a ventilator design. In the initial process, as shown in
Figure 3a
, high-pressure
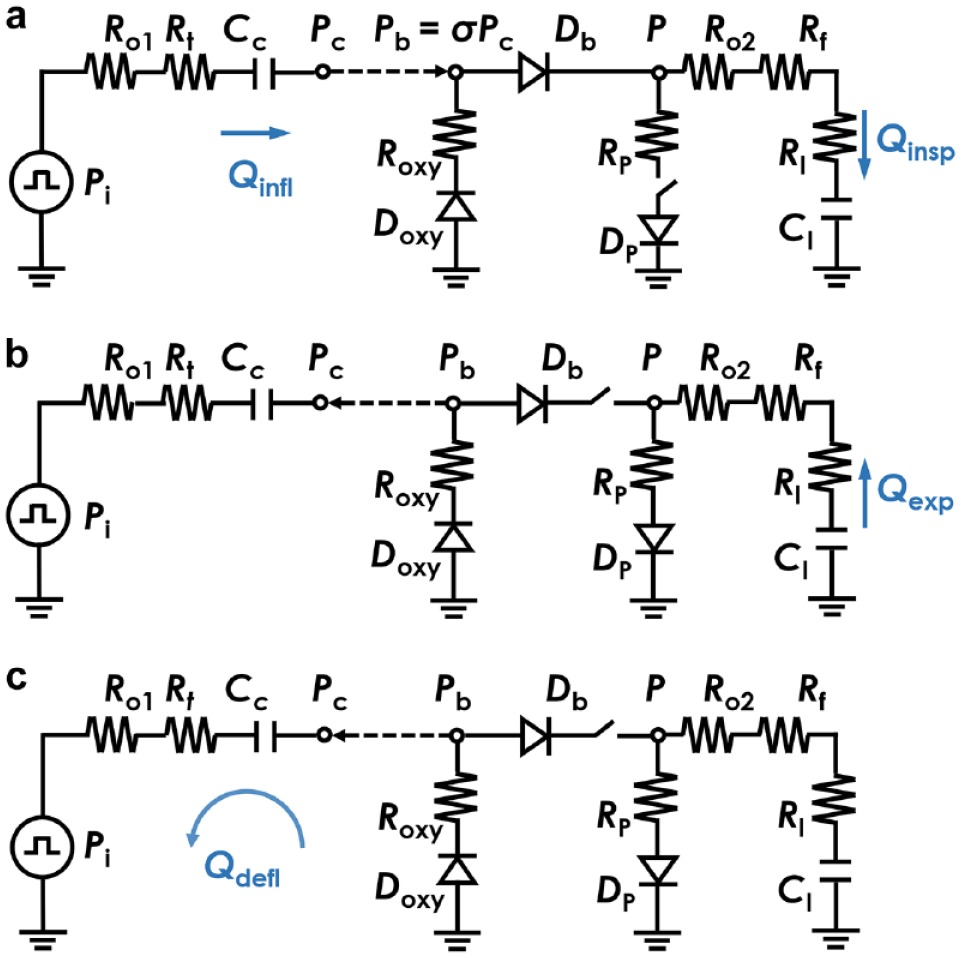
Pneumatic circuit model of three processes in AmbuBox. (
Minimal Functions of a Clinically Viable Ventilator
By referring to the emergency use ventilator (EUV) guideline 26 and literature, in addition to the clinical demand at University of California Davis Medical Center (UCDMC), the following requirements of the AmbuBox ventilator have been identified to make a minimal viable product (MVP) for emergent clinical settings. The minimal requirements include (1) two controls: respiratory rate from 10 to 30 bpm within 2 bpm, and tidal volume from 250 to 800 mL within 10%; (2) adjust and monitor two pressures within 2 cmH2O (PEEP pressure from 5 to 20 cmH2O, and PIP pressure from 15 to 40 cmH2O); and (3) alarms for pressures that are too high or too low (with safety presets). It is worth noting that the proposed ventilator is aimed to be used for the resuscitation of sedated patients. In such scenarios, only periodic ventilation is required.
Dynamics of AmbuBox
To further clarify the correlation between the pneumatic controls of the three processes and the resultant respiratory rate and tidal volume, the typical transient pressure, the flowrate, and the cumulative volume along the breath circuit of the simulated patient and the AmbuBox chamber are plotted in
Figure 4
. The testing conditions and preset parameters are as follows: Compliance C is 20 mL/cmH2O, resistance R is 20 cmH2O/(L/s), PEEP pressure is 5 cmH2O, input compressed air pressure
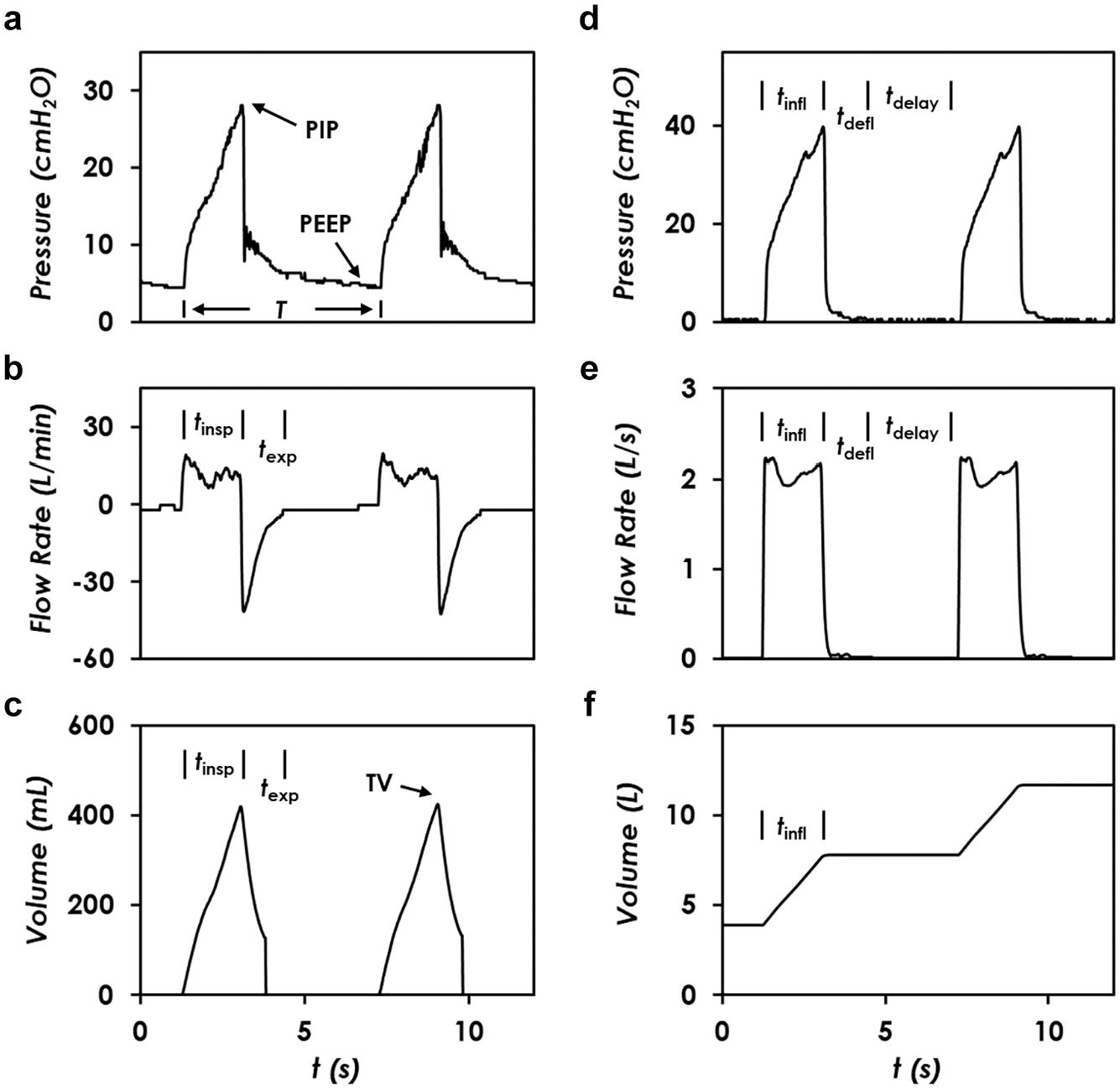
Real-time monitoring of the pressure and flow conditions in patient airway and AmbuBox chamber. Readings from the patient’s breath circuit, including (
The detailed correlation of respiratory rate and tidal volume with the specifications of the AmbuBox are discussed below. As shown in
Figure 4a
, the respiratory rate (RR; in units of bpm) correlates with the breath period
Parametric Studies of Tidal Volume and Respiratory Rate
First, we investigated how tidal volume varied at different respiratory rates. As shown in
Figure 5a
, under the testing conditions of C = 20 mL/cmH2O, R = 20 cmH2O/(L/s), PEEP = 5 cmH2O,
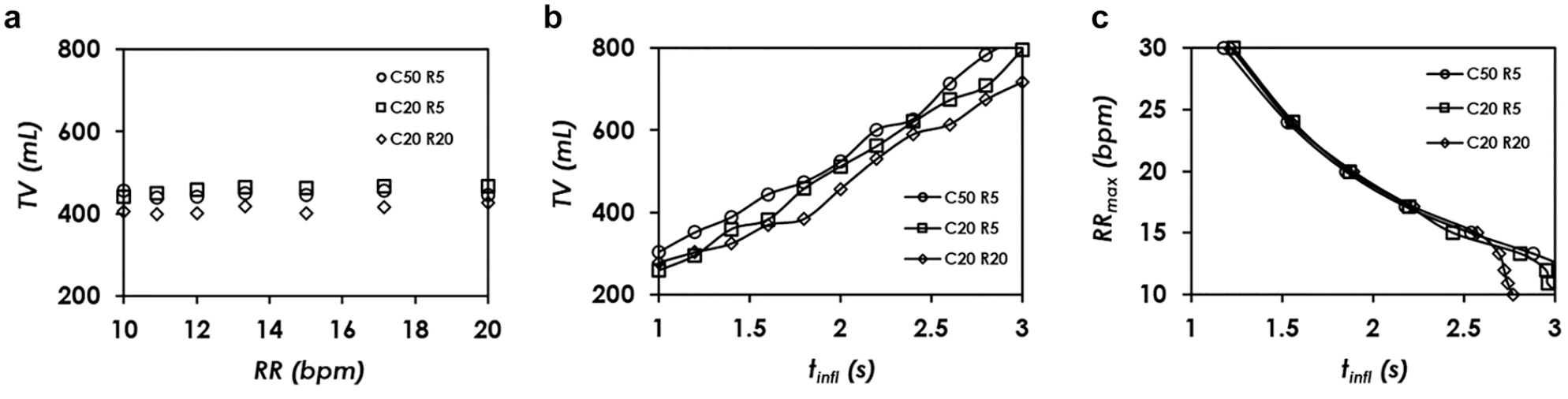
Parametric studies of the AmbuBox ventilator. (
Demonstration
Following the thorough characterization of AmbuBox, we demonstrated the real-time adjustment of tidal volume, respiratory rate, and PEEP pressure using an AmbuBox, together with its long-term stability, as shown in
Figure 6
. First, the tidal volume can be conveniently adjusted by increasing the inflation duration. As shown in
Figure 6a–6c
, we increased the inflation duration from 1 s to 2 s, and the corresponding tidal volume was raised up from 270 mL to 520 mL, under the testing conditions of C = 20 mL/cmH2O, R = 20 cmH2O/(L/s), PEEP = 5 cmH2O,
Lastly, we tested the long-term stability and consistency of the AmbuBox ventilator, as shown in
Figure 6j–6l
, under the testing conditions of C = 20 mL/cmH2O, R = 20 cmH2O/(L/s), PEEP = 5 cmH2O,
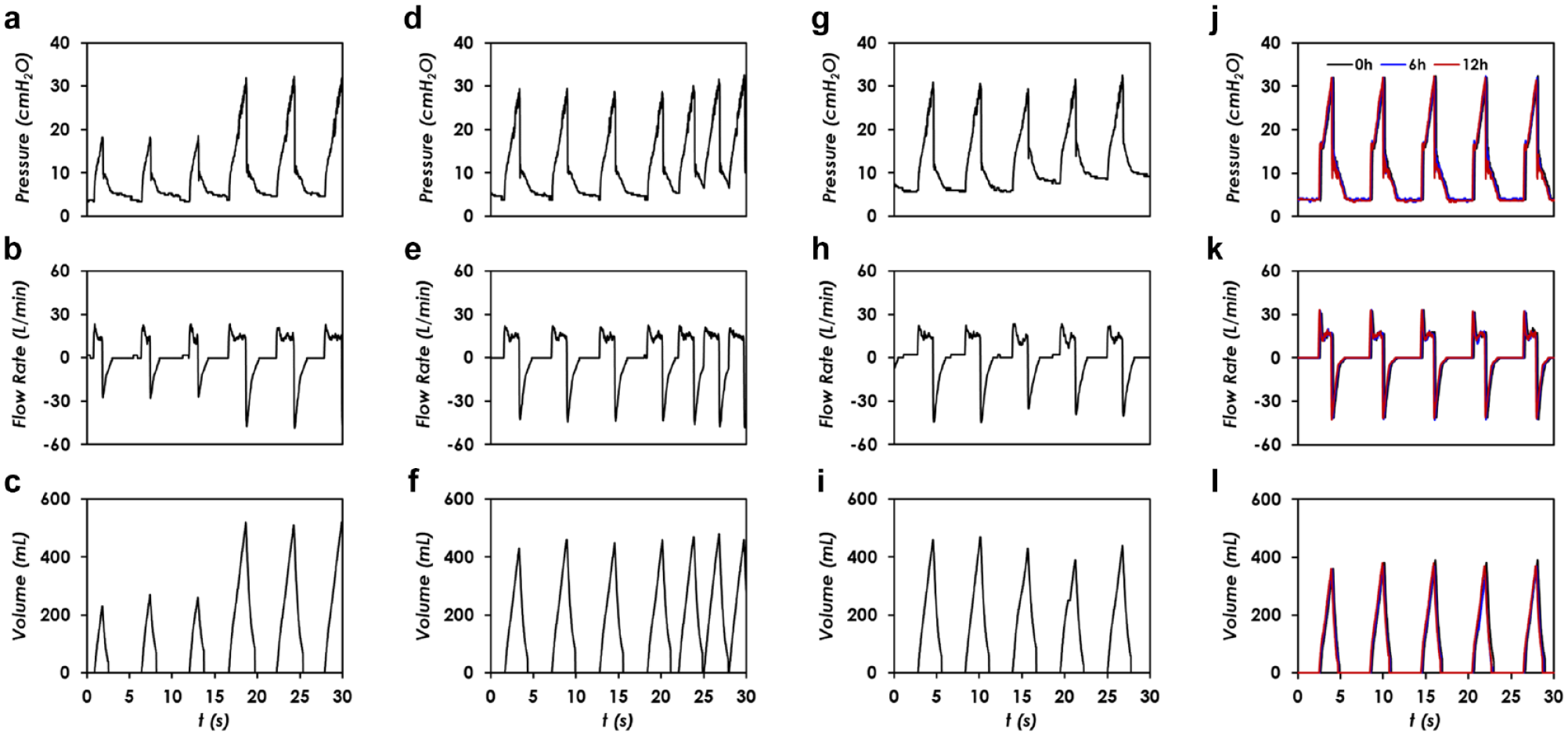
Demonstration of AmbuBox ventilator. (
In this article, we have presented a clinically viable and rapidly deployable low-cost ventilator, AmbuBox, for emergent care during the COVID-19 pandemic. The AmbuBox uses pneumatic control of compressed air to automate the bagging of a manual resuscitator, or BVM (AmbuBag). A pneumatic-electronic analogy model has been proposed to elucidate the dynamics of three processes during ventilation: the inflation of the AmbuBox chamber (inspiration of the simulated patient), the expiration of the simulated patient, and the deflation of the AmbuBox chamber. The AmbuBox device offers several distinct advantages over existing counterparts: (1) the simplest ventilator design with high control accuracy, with adjustable tidal volumes from 250 to 800 mL with an accuracy of 10%, respiratory rates from 10 to 30 bpm within adjustment of 2 bpm, adjustable PEEP pressure from 5 to 20 cmH2O, and the capability to monitor airway pressure with an accuracy of 2 cmH2O, in addition to alarms for pressures higher or lower than safety presets; (2) easy to use with a failure protection mechanism, enabled by the capability of bidirectional sealing; (3) long-term stability and reliability, owing to the unique pneumatically driven working principle that eliminates any moving parts; (4) a user-friendly design that excludes any issues observed in mechanical designs, such as alignment and slipping; (5) an independent and modular design with two parts (mechanical and electronic modules) working separately and enabling transitions from manual compression of the AmbuBag to an automated clinical grade ventilator; (6) the smallest footprint and most lightweight design, with all the parts capable of being packed into a standard toolbox kit; and (7) low cost and prompt manufacturability with standard laser cuts and off-the-shelf components for convenient assembly. Benefiting from its simple yet functional design, the AmbuBox uses a controllable pneumatic enclosure and a standard manual resuscitator and can be rapidly deployed with a minimal set of components to manufacture and assemble, enabling it to be a promising candidate for emergent use during the COVID-19 pandemic and mass-casualty events, as well as regular clinical use in low-resource settings.
Research Data
4_Movie_S1_of_AmbuBox for AmbuBox: A Fast-Deployable Low-Cost Ventilator for COVID-19 Emergent Care
4_Movie_S1_of_AmbuBox for AmbuBox: A Fast-Deployable Low-Cost Ventilator for COVID-19 Emergent Care by Zecong Fang, Andrew I. Li, Hongcheng Wang, Ruoyu Zhang, Xiyan Mai and Tingrui Pan in SLAS Technology
Supplemental Material
Supplemental_Material_for_AmbuBox_by_Fang,_et_al – Supplemental material for AmbuBox: A Fast-Deployable Low-Cost Ventilator for COVID-19 Emergent Care
Supplemental material, Supplemental_Material_for_AmbuBox_by_Fang,_et_al for AmbuBox: A Fast-Deployable Low-Cost Ventilator for COVID-19 Emergent Care by Zecong Fang, Andrew I. Li, Hongcheng Wang, Ruoyu Zhang, Xiyan Mai and Tingrui Pan in SLAS Technology
Footnotes
Acknowledgements
The authors would like to acknowledge the timely prototyping services supported by University of California, Davis Engineering Student Design Center (ESDC) and Drs. Clifford and Gavin Pereira. The authors would like to acknowledge Brian J. Smith and Krystal Craddock from UCDMC Department of Respiratory Care for providing valuable advice on buildup and measurement of the breath circuit, and for acquiring QuickLung for testing. The authors would like to acknowledge Bill Cranston from Tri-Anim Health Services for providing sample manual resuscitators and Sam Ling from PreciGenome for providing solenoid pinch valves for testing.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by 2020 Seed Fund Award 2020-0000000096 from CITRIS and the Banatao Institute at the University of California. ZF would like to acknowledge the fellowship support by Superfund Research Program at UC Davis from the National Institutes of Health (2P42ES004699).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
