Abstract
The clinical team attending to a patient upon a diagnosis is faced with two main questions: what treatment, and at what dose? Clinical trials’ results provide the basis for guidance and support for official protocols that clinicians use to base their decisions upon. However, individuals rarely demonstrate the reported response from relevant clinical trials, often the average from a group representing a population or subpopulation. The decision complexity increases with combination treatments where drugs administered together can interact with each other, which is often the case. Additionally, the individual’s response to the treatment varies over time with the changes in his or her condition, whether via the indication or physiology. In practice, the drug and the dose selection depend greatly on the medical protocol of the healthcare provider and the medical team’s experience. As such, the results are inherently varied and often suboptimal. Big data approaches have emerged as an excellent decision-making support tool, but their application is limited by multiple challenges, the main one being the availability of sufficiently big datasets with good quality, representative information. An alternative approach—phenotypic personalized medicine (PPM)—finds an appropriate drug combination (quadratic phenotypic optimization platform [QPOP]) and an appropriate dosing strategy over time (CURATE.AI) based on small data collected exclusively from the treated individual. PPM-based approaches have demonstrated superior results over the current standard of care. The side effects are limited while the desired output is maximized, which directly translates into improving the length and quality of individuals’ lives.
Introduction
In the past 50 years, clinical practice has been going through a transition fueled by technological developments. Biomedical engineering,1–9 material science,10–18 and artificial intelligence (AI)19–24 have been transforming the entire medical landscape: diagnostics,25–33 drug development,24,34–36 drug delivery,37–46 and data analytics.47–49 The medical team has access to ever-growing information about their patients prior to and at the time of treatment. Like never before, doctors can practice evidence-based medicine and informed decision making.
AI has emerged as a prominent tool for health data analytics.20,48,50–52 Among others, it has been used in diagnostics,47,53–55 prognosis,56–58 and identifying personalized care strategies.59–61 Classical AI approaches consist of training an algorithm on population data to identify statistical patterns that can be then used to diagnose and treat an individual based on his or her specific demographics and a specific disease history. 62 Among other examples, those approaches have demonstrated success in classifying electrocardiography signals toward the detection of arrythmia 63 and radiology images toward correct diagnosis. 64 Population-based AI approaches can further reveal the most impactful features affecting selected clinical outcomes based on the identified statistical patterns. For example, support vector regression and multiple linear regression have been used to identify the features that are the most prominent predictors of tacrolimus blood concentration.65,66 The versatility of AI approaches is one of the main factors behind the onset of its widespread application in healthcare. However, as of now, good quality training datasets—representative, diverse, and of sufficient size—are not always available for particular indications or patient scenarios. 52 Unrepresentative training datasets introduce bias to AI-derived knowledge through overrepresentation of a particular symptom or a demographic group. 50 For example, if the training dataset contains only patients with well-defined symptoms and strong evidence, then the AI’s performance might be suboptimal when encountering more complex disease manifestations, common in real-world scenarios. There is also a growing understanding of cultural, ethnic, and gender biases in training datasets that limit the applicability of AI-derived knowledge to selected subpopulations. 67 The collection, annotation, and integration of medical data needed for practicing personalized medicine via population-based AI approaches is resource intensive, and currently there is no sufficient infrastructure to treat individuals based on AI-derived knowledge in a sustainable and egalitarian fashion. 50
A phenotypic personalized medicine (PPM) platform offers an alternative to personalized medicine driven by big data, systems biology, and omics. This approach aims to select the best combination therapy—both the drug(s) and the dose(s)—by identifying a relationship between the input stimuli and a phenotypic response based solely on the treated individual’s data: medical log and readouts.
PPM has evolved from the feedback system control (FSC) platform.68–79 Neural networks were used to correlate multisignal combinations and their effects through a learning process, and led to an observation that the relationship between dose and treatment efficacy is represented by a smooth quadratic surface. 72 In its current form, PPM consists of two complementary approaches: (1) maximize the desired outcome via selecting drugs for combination therapy and their initial doses with the quadratic phenotypic optimization platform (QPOP), and (2) dynamically recommend the most effective dosing approach with CURATE.AI. In contrast to AI-driven and traditional techniques used for drug combination and dose selection, like statistical metamodeling 80 and pharmacokinetic/pharmacodynamic-driven precision dosing,81,82 PPM does not require a large amount of information from the population and from the individual to identify drug interactions and guide optimal combinatorial therapy. In this review, we focus on CURATE.AI and its current applications and future directions.
CURATE.AI: Dose Recommendation
CURATE.AI is an AI-derived platform that maps the relationship between an intervention intensity (input) and a phenotypic result (output) for an individual, based exclusively on that individual’s data (
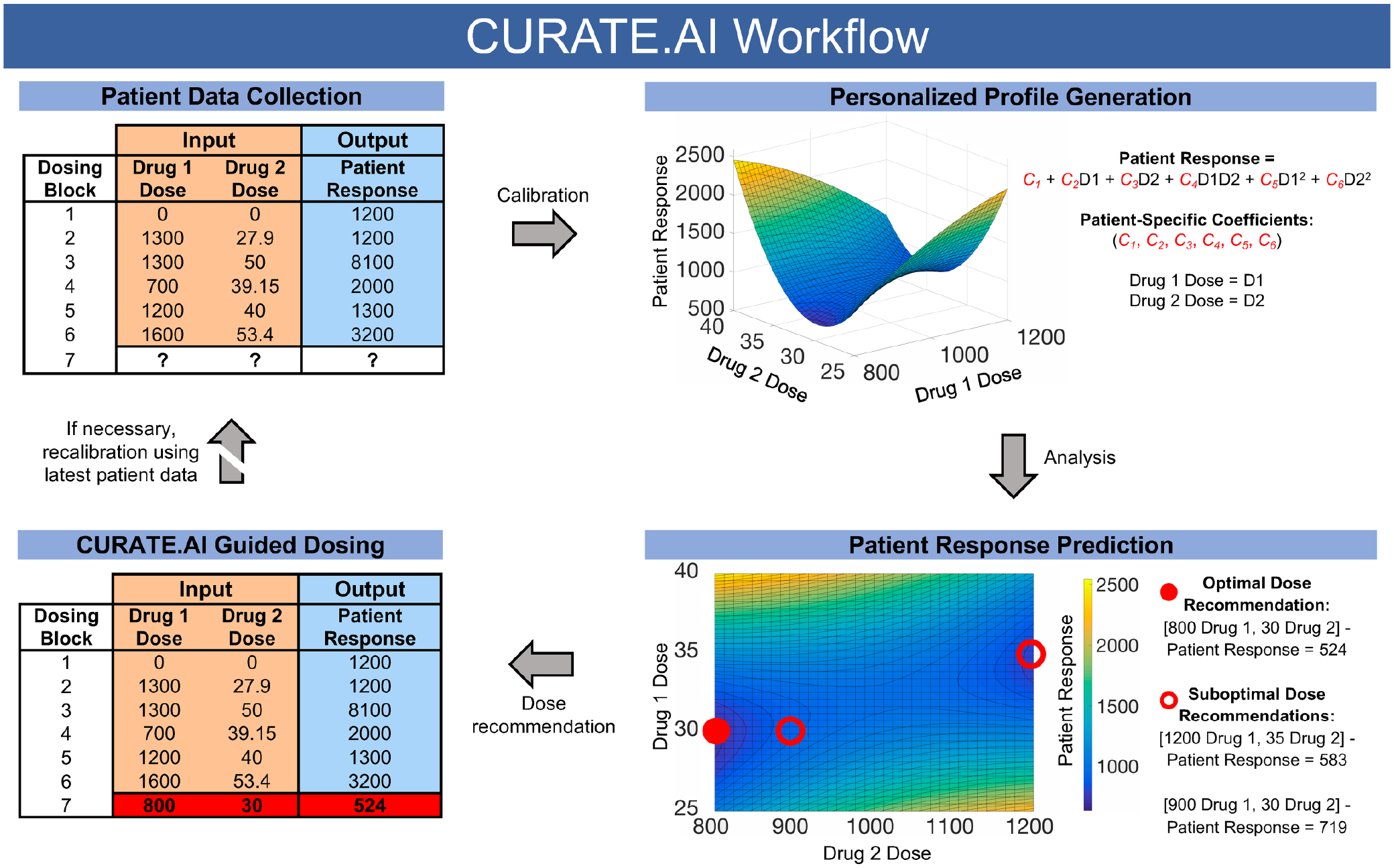
CURATE.AI-guided dosing workflow for a two-drug optimization. Through calibration of patient data from an initial physician-guided six-dosing block period, CURATE.AI generates a personalized profile using a second-order quadratic correlation between drug doses (input 1, input 2), and a phenotypic patient response (output). The generated second-order equation contains patient-specific coefficients (C1, C2, C3, C4, C5, C6), which CURATE.AI then analyzes to predict dose combinations that guide patient responses toward a preferable outcome. In this particular case, CURATE.AI aims to minimize the phenotypic output. CURATE.AI recommends several effective dose combination options for physician review (open circles) while highlighting an optimal dose (closed circle). In the case of external interventions such as regimen changes, CURATE.AI recalibrates the generated personalized profile using the latest available patient data to accommodate changes in patient state. Though drug doses serve as inputs in this case, inputs may be other interventions with varying intensities.
How Does It Work?
CURATE.AI is based on an observation that the relationship between a varying input and a measurable output in a complex system is described by a quadratic polynomial equation, and that the response space within the clinical range has a smooth surface. 72 Three data pairs (input : output) are needed to calibrate the CURATE.AI profile for one drug; 6 data pair triples (input 1, input 2 : output) are needed to calibrate the CURATE.AI profile for two drugs; 10 data pair quadruples (input 1, input 2, input 3 : output) are needed to calibrate the CURATE.AI profile for three drugs; and so on. The change in the system’s state that affects drug interactions at any stage between the uptake and the phenotypic output is the basis for recalibration: a profile shift within the response space or the creation of a new profile.
What Is It Used For?
The validity of the CURATE.AI approach has been successfully demonstrated for single-drug optimization;
83
combination treatment,84,85 oncology,84,85 and immunosuppression;
83
and both retrospective analysis83,84 and prospective dosing83,85 (
CURATE.AI-Based Studies for Personalized Treatment.
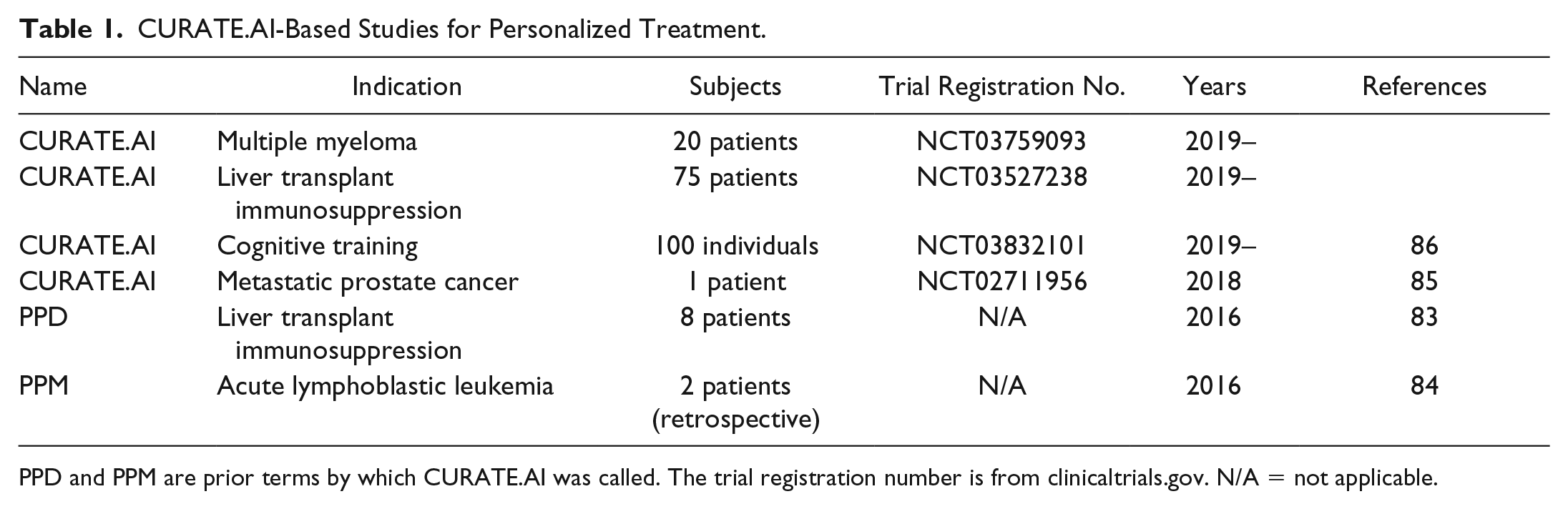
PPD and PPM are prior terms by which CURATE.AI was called. The trial registration number is from clinicaltrials.gov. N/A = not applicable.
CURATE.AI application for a prospective, single-drug optimization was demonstrated by Zarrinpar et al. to manage immunosuppression after liver transplant. 83 In a series of four N-of-1 clinical trials personalized PPD (parabolic personalized dosing; currently termed CURATE.AI) profiles were used to recommend the tacrolimus dose (input) to reach and sustain a desired tacrolimus blood level (output), which is recognized as a clinical predictor of successful immunosuppression.87,88 The PPD profiles were generated for each patient based on at least three data pairs (input : output), exclusively from that patient’s medical log. The desired tacrolimus trough level was specified by a clinician for each patient individually and was dynamically changing over the course of the treatment. Additionally, as typical for immunosuppression in real-world scenarios, the patients underwent changes to their treatment regimens with inclusion or exclusion of other drugs, and medical interventions like dialysis. This added temporal complexity of the system was addressed by CURATE.AI in two ways: (1) generation of the profile for each individual exclusively from that individual’s data, and (2) dynamic recalibration of the profile upon identifying change in the system’s state. The results of the study showed that CURATE.AI succeeded in guiding patients’ management toward significantly less variability in tacrolimus trough levels and enhanced adherence to the desired range as compared with the physician-guided dosing.
CURATE.AI application for a prospective, combination drug optimization was demonstrated by Pantuck et al. in oncology. 85 In the N-of-1 expanded access case, a patient with metastatic castration-resistant prostate cancer (mCRPC) was treated with ZEN-369489,90 (input 1) in combination with enzalutamide 91 (input 2). Prior to engaging CURATE.AI, the patient was managed with a standard of care approach—the drugs were given at maximum tolerated doses. The desired outcome was to suppress the prostate-specific antigen 91 (PSA; output) level toward its elimination. The individualized CURATE.AI profile was generated based on six data triples (input 1, input 2 : output) exclusively from the patient’s data. Each subsequent data triple was incorporated into the patient’s profile to ensure that the dose recommendation was relevant to the changes in the patient’s condition. The initial CURATE.AI profile recommended sustaining enzalutamide at a constant dose of 80 mg and reducing ZEN-3694 by 33%, from 36 mg to 24 mg. The CURATE.AI outcome prediction for this dose combination was a PSA level of 0.65 ng mL−1. The clinician followed the recommendation and over 10 weeks the PSA dropped from 0.91 ng mL−1 to 0.68 ng mL−1, striking close to the CURATE.AI prediction, while also decreasing undesired side effects. A further dose reduction of ZEN-3694 to 12 mg initiated by the clinician resulted in a PSA increase to 1.60 ng mL−1 over 12 weeks. This value was in a close approximation to the CURATE.AI profile prediction (1.52 ng mL−1) for that dose combination (12 mg of ZEN-3694 and 80 mg of enzalutamide). Subsequently, the ZEN-3694 dose was increased back to 24 mg, which led to a PSA level of less than 1 ng mL−1. The remaining course of treatment adhered to the evolving CURATE.AI recommendations and resulted in a durable PSA suppression. The desired results were confirmed with radiographic assessment—over 24 weeks, the single-baseline target lesion decreased from 6 to 4 mm and no new lesions were detected. This study demonstrates the potential of CURATE.AI to pinpoint the right dose combination that is an alternative to the widely used approach in oncology of dosing maximum tolerable doses. CURATE.AI recommended using markedly reduced ZEN-3694 doses compared with the starting dose and managed an mCRPC patient’s combination treatment in a sustained and prolonged fashion.
CURATE.AI application outside of a clinical setting was demonstrated by Kee et al. for creating a cognitive profile. 86 Five individuals (two individuals were later excluded from the analysis) underwent cognitive training with the Multi-Attribute Task Battery—a flight simulator released by the Air Force (AF-MATB).92,93 The individual’s performance score (combined performance at two tasks before a training block) was used as input 1, the training block intensity (low, medium, or high) was used as input 2, and performance improvement (performance score difference before and after the training block) was used as the output. Those data triples were used to generate individualized CURATE.AI profiles for each individual with an aim to recommend the next training intensity toward maximized performance improvement, such that each individual can follow his or her own personalized learning strategy. This study sets a foundation for the application of CURATE.AI in personalized learning, digital therapeutics, and the prevention of cognitive decline. 86
CURATE.AI is a technology platform that is based on polynomial correlation between inputs of varying intensity and the phenotypic output. The generation of an individualized CURATE.AI profile requires collecting a small dataset exclusively from that individual. Due to its simplicity and versatility, it is applicable to multiple indications. The extent of CURATE.AI applicability is only being realized. Its implementation is expected to guide intervention strategies for a wide variety of indications, within healthcare and beyond.
Implementation: Challenges and Opportunities
Indication Selection
CURATE.AI applicability is assessed case by case for each indication (
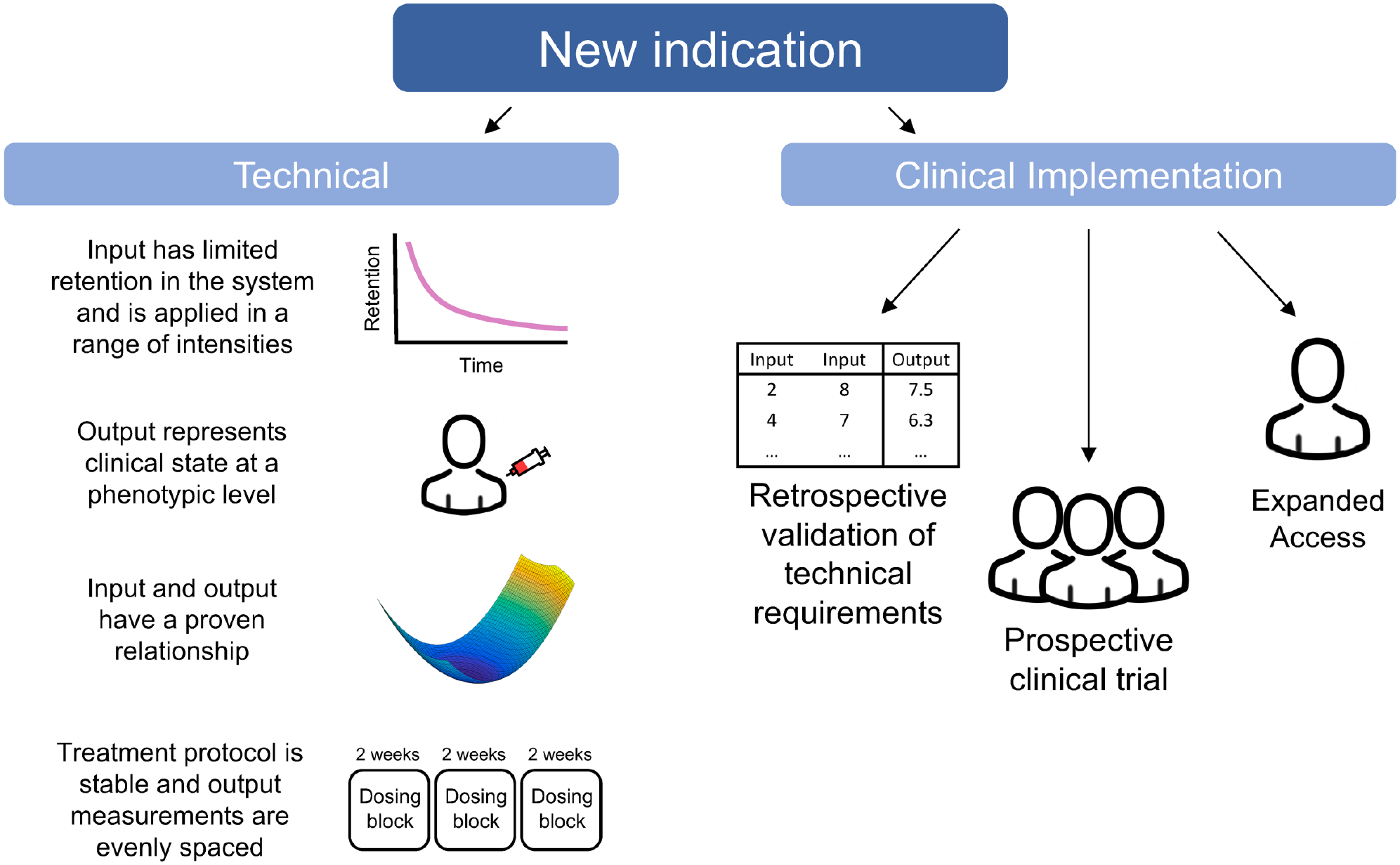
An outline of determining the applicability of CURATE.AI to a new indication. Several technical factors are required to perform CURATE.AI analysis. Importantly, the input should have limited retention in the system in order to minimize the carryover effect. The input must be deliverable in a range of intensities, and the treatment protocol must allow for the dynamic modulation of input intensities. The output should be representative of clinical activity, and there must be a proven relationship between the input and output. The treatment protocol should also be stable as to limit frequent regimen changes, and the output should be measured in evenly spaced intervals to reduce time variability. For clinical implementation of CURATE.AI, retrospective analysis is used to validate the technical requirements. Prospective application of CURATE.AI is done in the form of a formal clinical trial. For patients that do not qualify under the strict inclusion criteria of clinical trials, expanded access provides access to CURATE.AI for treatment purposes.
The input choice follows several guidelines. At least one of the inputs has to be deliverable in a range of intensities, with the minimum number of the intensities being three. The higher the resolution of the input intensities, the bigger the potential of recommending a dose toward an ultimate optimum of the output. The continuous intensity range of the input is the most desirable, albeit practically impossible in a clinical setting where step dosing is practiced, for example, multiplication of the contents of a single pill. Additionally, during the profile calibration, the treatment protocol should allow spreading the input across the dosing range. This is to minimize interpolation and maximize accuracy of the generated CURATE.AI profile within the specified dosing range. Similar to the input, the output also has to fulfill several requirements. It has to be indicative of the clinical state at a phenotypic level. In practice, established, quantifiable markers are preferred. As CURATE.AI dynamically manages an individual’s treatment throughout the course of the treatment and may require a higher number and/or higher frequency of the output measurements, the output collection should be safe, fast, and cost-effective. As of now, the most common outputs used in CURATE.AI are blood markers, as their measurements can be obtained frequently, with minimal clinical intrusion and minimal additional risk experienced by the patient. Additionally, blood tests are accessible and well characterized and the results are returned within days or even hours. However, for multiple indications blood markers are not suitable or are nonexistent. CURATE.AI has the potential to modulate a variety of outputs: tumor size, genetic profile, blood pressure, or cognitive performance, among many others. Special caution has to be applied to using measures that are vulnerable to bias from one dosing block to another, for example, questionnaires, and to measures with a limited sensitivity, specificity, or accuracy.
CURATE.AI has been already proven as a powerful clinical tool for multiple indications. Retrospective data analysis is invaluable in an ongoing effort to explore and realize the full potential of CURATE.AI via initial validation of new candidate indications. It also holds a promise to provide means to leverage some of the listed above requirements for new candidate indications, and further increase the applicability of CURATE.AI within a clinical practice.
Clinical Implementation
For CURATE.AI to be implemented in the clinical setting and practice, regulatory standards and qualifications must be satisfied similar to those applied to traditionally defined drugs and devices, with recent additions such as combination products95–97 and now medical software.98–100 To address the newly emergent field of medical software, the International Medical Device Regulatory Forum (IMDRF) and Food and Drug Administration (FDA) have broadly classified such software as Software as Medical Device (SaMD) and Clinical Decision Support Software (CDSS).101,102 Importantly, in 2017, a new draft guidance ruled CDSS exempt from FDA regulation if the software intended to enable healthcare professionals to independently review the basis for the software’s recommendations, such that clinical decisions are based primarily on the judgment of the healthcare professional. 103 Within this framework, CURATE.AI can be potentially classified as a CDSS and be excluded from FDA oversight toward entering the market in the United States, since publicly accessible literature exists to explain its methodology, and since its quadratic surface correlation is understandable on a clinician level. On an international level, however, CURATE.AI is subject to the more conservative IMDRF guidelines, which heavily regulate software under a comprehensive risk-based framework to prevent premature market approval under unbacked claims. 104
The claims for medical devices and SaMDs are indication specific and performance driven. These claims are substantiated and supported through randomized clinical trials, similar to the ongoing clinical trials with CURATE.AI for multiple myeloma (NCT03759093) and immunosuppression management (NCT03527238). For patients that do not qualify under the strict inclusion criteria of clinical trials, expanded access provides an accessible pathway to innovative technologies like CURATE.AI for treatment purposes. In addition to randomized clinical trials, the use of real-world data (RWD) and the real-world evidence (RWE) derived from these data is becoming increasingly important in the evidentiary standards of medical devices. 105 For conventional AI-based platforms, retrospective and comparative observational studies play a pivotal role in providing RWE due to the large datasets they can provide. 106 Analyses of retrospective RWD from acute lymphoblastic leukemia and liver transplant patients have provided RWE to validate the use of CURATE.AI to optimize drug dosing in these indications.83,84
Currently, prospective patient management under CURATE.AI is carried out under two main schemes: a single trial (e.g., NCT02711956) or as a series of N-of-1 trials (NCT03759093 and NCT03527238). The N-of-1 trial has been recognized to be among the most relevant study designs for evidence-based clinical practice in an individual patient’s care.107–109 The Oxford Centre for Evidence-Based Medicine classified the N-of-1 trial as the highest-ranked evidence for identifying intervention benefits and harms for the individual (single N-of-1 trial) and overall (systematic review of N-of-1 trials). 110 N-of-1-enabled personalized care can be easily integrated into the mobile space paving a way toward personalized digital therapeutics.86,111,112 A series of N-of-1 trials, in addition to personalized care, informs about the overall group performance and can help identify trends and subgroups for understanding the mechanisms and intervention. 113 While the N-of-1 trial design has shown applicability for the validation of CURATE.AI in case-by-case scenarios, several other trial designs have been emerging that may potentially allow indication categorization and more robust collection of RWE for supporting the regulatory approval of the clinical implementation of such software 114 toward downstream widespread market adoption.
QPOP: Drug Selection
Identifying the most effective combination treatment requires the selection of both drugs and their doses. As a result, the possible combination space reaches an impractical size to test one by one—for example, five possible drugs at five possible doses provide more than 3000 combinations. Traditionally, the combination treatment parameters are selected in two steps: (1) drugs at single doses are selected relying on molecular mechanisms and predetermined drug interaction information, and (2) doses are readjusted if necessary. By disregarding the dose-dependent nature of drug–drug interactions in the first step, this process considers a very narrow scope of the possible combination space, omits understudied combinations and dose ratios, and is highly vulnerable to interpatient variability. As a result, applied combination treatment is often suboptimal. QPOP aims to address these challenges and identify the most efficacious drug combinations and their initial optimal doses in the presence of genomic and biological variance between patients.
QPOP operates by correlating phenotypic outputs with drug combinations at multiple dosing levels as inputs. Similar to CURATE.AI, QPOP fits the relationship between the inputs and their outputs to a smooth, second-order polynomial surface. QPOP identifies which features of the second-order equation are the most impactful to the system and ranks the combinations from the least to the most effective.
115
An optimal combination of the most synergistic and effective drugs is reached by iteratively discarding lower-ranking combinations and subsequently performing QPOP analyses on the remaining top-ranking combinations. Importantly, like CURATE.AI, QPOP only analyzes input and output data. Thus, by treating the biological system as a black box, it is mechanism independent and implicitly accounts for system-specific characteristics. QPOP (and its alternative form, PRS [parabolic response surface]) has been successfully used to identify optimal drug combinations from libraries of 10+ drugs115–117 (
QPOP-Based Studies for Personalized Treatment.
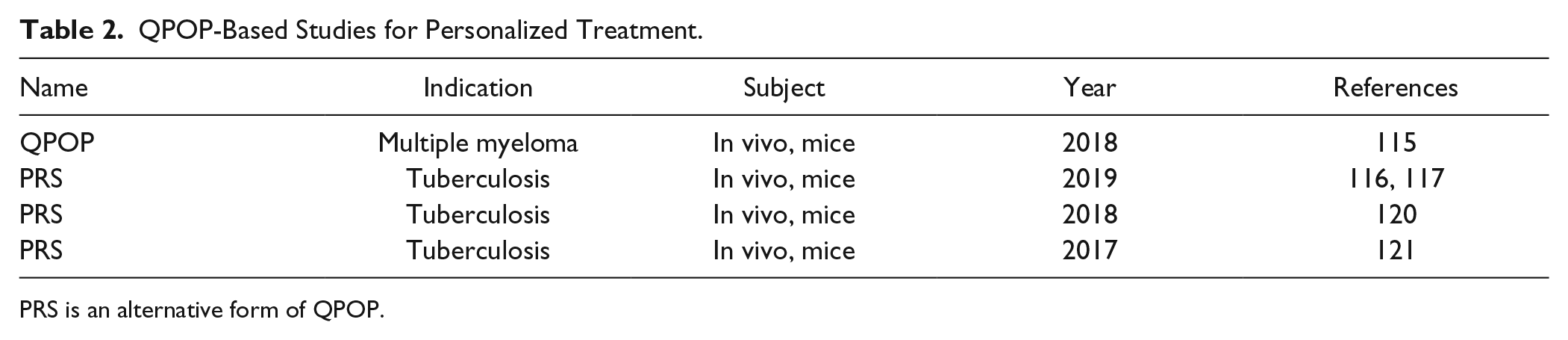
PRS is an alternative form of QPOP.
Conclusions
In the past few years, AI has established itself as a paradigm-shifting technology in healthcare, especially within diagnostics and the identification of prophylactic measures.118,119 PPM seeks to expand the capabilities and utilization of AI to the interventional domain, providing guidance for identifying the appropriate treatment for the individual. Specifically, QPOP identifies personalized drug combinations and initial doses, and CURATE.AI calibrates adaptable patient-specific profiles that identify optimal doses throughout the entire course of treatment. As mechanism-independent approaches, QPOP and CURATE.AI function in the absence of pharmacokinetic and drug–drug interaction information, even capable of generating new system-specific knowledge.
In a fully realized clinical implementation of PPM, a patient will have QPOP applied to a sample isolated from blood or a resected tumor and have identified an efficacious and personalized combination; subsequently, CURATE.AI will guide the dose modulation in the same individual. Thus, the patient’s care will be personalized from start to finish—from selecting the drugs and their initial doses to managing the dosing throughout the course of treatment. It is a highly plausible scenario once the two approaches are validated separately for each specific indication.
CURATE.AI has been clinically validated in a prospective pilot study for mCRPC, demonstrating the capability for modulating combination therapy for individuals. This study has paved the path to clinical implementation and expanded CURATE.AI-enabled dose optimization for prospective treatment of multiple myeloma (NCT03759093) and immunosuppression (NCT03527238). Similarly, QPOP has been validated in numerous in vivo studies for a multitude of indications, including oncology, tuberculosis, and multiple myeloma. With growing RWE collected from additional clinical trials, CURATE.AI and QPOP will be validated in new indications and move closer toward the FDA and international regulatory approval required for widespread clinical adoption.
When applied at a broad scale, PPM may have far-reaching implications even on a scale of the whole society, for example, in terms of healthcare burden. With the validated impact on patients’ lives, PPM technologies—CURATE.AI and QPOP—have already proven to have a transformative potential. We are still to see what the full realization of their impact is on a broad scale.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.K. and A.B. are listed as co-inventors in intellectual property pertaining to CURATE.AI technology
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge support from the National University of Singapore for the following grants: NUS Start Up Funds (R-397-000-310-133) and N.1 Core Operations (C-719-000-001-001). The authors acknowledge support from the Ministry of Education of Singapore for its funding in the form of the MOE Tier 1 FRC Grant (R-397-000-333-114). A.B., J.K., and T.K. co-wrote the manuscript.
