Abstract
Assisted reproductive technology (ART) can benefit from the features of microfluidic technologies, such as the automation of time-consuming labor-intensive procedures, the possibility to mimic in vivo environments, and the miniaturization of the required equipment. To date, most of the proposed approaches are based on polydimethylsiloxane (PDMS) as platform substrate material due to its widespread use in academia, despite certain disadvantages, such as the elevated cost of mass production. Herein, we present a rapid fabrication process for a cyclic olefin copolymer (COC) monolithic microfluidic device combining hot embossing—using a low-temperature cofired ceramic (LTCC) master—and micromilling. The microfluidic device was suitable for trapping and maturation of bovine oocytes, which were further studied to determine their ability to be fertilized. Furthermore, another COC microfluidic device was fabricated to store sperm and assess its quality parameters over time. The study herein presented demonstrates a good biocompatibility of the COC when working with gametes, and it exhibits certain advantages, such as the nonabsorption of small molecules, gas impermeability, and low fabrication costs, all at the prototyping and mass production scale, thus taking a step further toward fully automated microfluidic devices in ART.
Keywords
Introduction
Assisted reproductive technologies (ARTs) have enabled millions of people in the world to have children who otherwise would not have been able to do so. The application of ART in animals, mainly by artificial insemination (AI), had a great impact in the improvement of the efficiency of animal production. Reproductive biotechnologies are intended to be used routinely to shorten generational intervals and to propagate genetic material among breeding animal populations. 1 To achieve this goal, reproductive technologies have been developed over the years, for instance, AI, embryo transfer, in vitro fertilization (IVF), and in vitro embryo production, as well as multiplication techniques (cloning) for the application of transgenesis. 1
Despite the remarkable progress made and the punctual relevance of some of the above-mentioned technologies, the efficiency of the processes is usually very low, with the exception of AI in cattle. Furthermore, ARTs such as oocyte maturation or IVF require labor-intensive, time-consuming procedures (including frequent pipetting of oocytes to be washed, media changes, etc., which have not significantly evolved since their establishment) and some critical steps that only highly specialized personnel are able to perform.
Microfluidics has emerged as a new tool for ART that could potentially solve these problems by automating the handling and preparation procedures, 2 thus minimizing the errors associated with manual operation and reducing manipulation stress on the cells. Microfluidics can also provide environments that mimic in vivo conditions since fabrication technologies enable the design of structures resembling biological environments,3–6 including those related to ART. 7 Furthermore, other inherent advantages of microfluidics, such as the low volumes required, the precise control of fluids, or the miniaturization and integration of different elements in portable devices, would also benefit ART.
The vast majority of microfluidic systems used in ART are made of polydimethylsiloxane (PDMS), a transparent and gas-permeable elastomer. This is the consequence of its widespread use in academia, which is due to several factors: cheap, rapid, easy prototyping, optical transparency, easy surface modification, elasticity, etc. 8 The latter also provides the grounds for the integration of actuation systems, such as pumping or valving. ART applications of PDMS devices range from sperm separation 9 to oocyte fertilization 10 or embryo culture, 11 among others. A comprehensive review of microfluidic systems applied in ART can be found in Swain et al. 12
However, certain limitations of PDMS have encouraged the use of other materials in microfluidics, such as thermoplastics. 13 Among these, polystyrene (PS) and cyclic olefin copolymers (COCs) have emerged as alternatives for the production of microfluidic devices for biological applications, overcoming the limitations of PDMS. On the one hand, PS has been the choice material for disposable cell culture labware since the 1960s, and recently has been used as a substrate material to produce microfluidic devices, 14 thus bringing together state-of-the-art microfluidics technology and a well-known material for biologists. However, the fabrication process is still challenging, mostly due to bonding issues. 15
On the other hand, COCs are a relatively new group of polymers with very promising properties for microfluidic applications, 16 such as chemical resistance (higher than most thermoplastics), excellent optical transparency (even in the near-ultraviolet [UV] region), low water absorption, and biological compatibility.17,18 Furthermore, COC has obtained the USP Class VI qualification 19 and is approved for use in medical devices. COC is a copolymer consisting of ethylene and norbornene, which offers several grade variations in terms of glass transition temperatures (Tg), ranging from 70 to 155 °C. 20 This is a big advantage in front of other thermoplastics, as it allows the bonding of different layers without channel deformation using grades with low Tg as sealants, 21 and avoiding processes like solvent bonding, which are potentially harmful for biology applications. COC also enables surface modification, 22 which can be useful for avoiding protein adsorption, for instance, in bioanalytical applications. Several fabrication techniques are available for the production of COC microfluidic devices, such as micromilling, hot embossing, and injection molding. The last two techniques enable the fabrication of the required microstructures to trap oocytes. However, the fabrication of the master is usually expensive and requires relatively complex manufacturing processes, such as photolithography.
Herein, we developed a new fabrication methodology combining computer numerically controlled (CNC) micromilling and hot embossing using low-temperature cofired ceramic (LTCC) masters, which enables fast prototyping, as well as the fabrication of microfluidic features in the 50–100 µm range. Furthermore, we studied for the first time the suitability of a COC monolithic microfluidic device for an ART application. We studied bovine oocyte maturation and sperm characteristics during the culture in a COC microfluidic device. We investigated whether oocytes maintain the same rates of in vitro maturation and their ability to be fertilized. We also evaluated spermatozoa-relevant parameters, such as viability, motility, and membrane integrity, after culture in the microfluidic device. This is, to the best of our knowledge, the first COC monolithic microfluidic device dedicated to an ART application, and from the obtained results, we can demonstrate the great potential of COC as a substrate material due to its biocompatibility and simple fabrication processes, at both the prototyping and mass production scales.
Materials and Methods
Reagents
All reagents were purchased from Sigma Chemical Co. (St. Louis, MO) unless otherwise stated. Plastic dishes, four-well plates, and tubes were obtained from Nunc (Roskilde, Denmark).
Fabrication of Microfluidic Devices
Two different microfluidic devices were fabricated on COC to separately study the maturation of oocytes and sperm storage. COC was obtained from TOPAS Advance Polymers GmbH (Florence, KY). TOPAS 5013 sheets (Tg 134 °C) were used in this study for the machining of microchannels, while 25 µm thick TOPAS 8007 foils (Tg 78 °C) were used as sealing substrate between layers using a temperature diffusion bonding technique, as described elsewhere. 23
Sperm Storage Microfluidic Device
The fabrication procedure of this device is based on a procedure previously reported in our research group. 24 Briefly, the device consists of three layers of TOPAS 5013 1 mm thick. The top and bottom layers were laminated with a 25 µm TOPAS 8007 (which acted later as glue to seal the device) 23 in a uniaxial hydraulic press (Talleres Francisco Camp, Granollers, Spain). The COC layers were machined using a micromilling CNC machine: Protomat S63 (LPKF Laser & Electronics, Garbsen, Germany). The fluidic channel was milled in the middle layer, by cutting the COC through. The top layer was drilled to create the inlets. Then, the three layers were aligned and laminated at 100 °C under a pressure of 6 bars. This approach prevents any surface roughness created by the milling machine when creating a bas-relief channel.
Oocyte-Trapping Microfluidic Device
The oocyte-trapping microfluidic device was fabricated using a combination of hot embossing and micromilling. The microchannels that retain the oocytes were fabricated by means of a hot embossing technique in order to obtain a “filter-like” structure with the appropriate dimensions to trap the oocytes—micromilling is not suitable for such dimensions—while the bigger features were machined using the CNC micromilling machine. Figure 1 shows a schematic representation of the fabrication process.
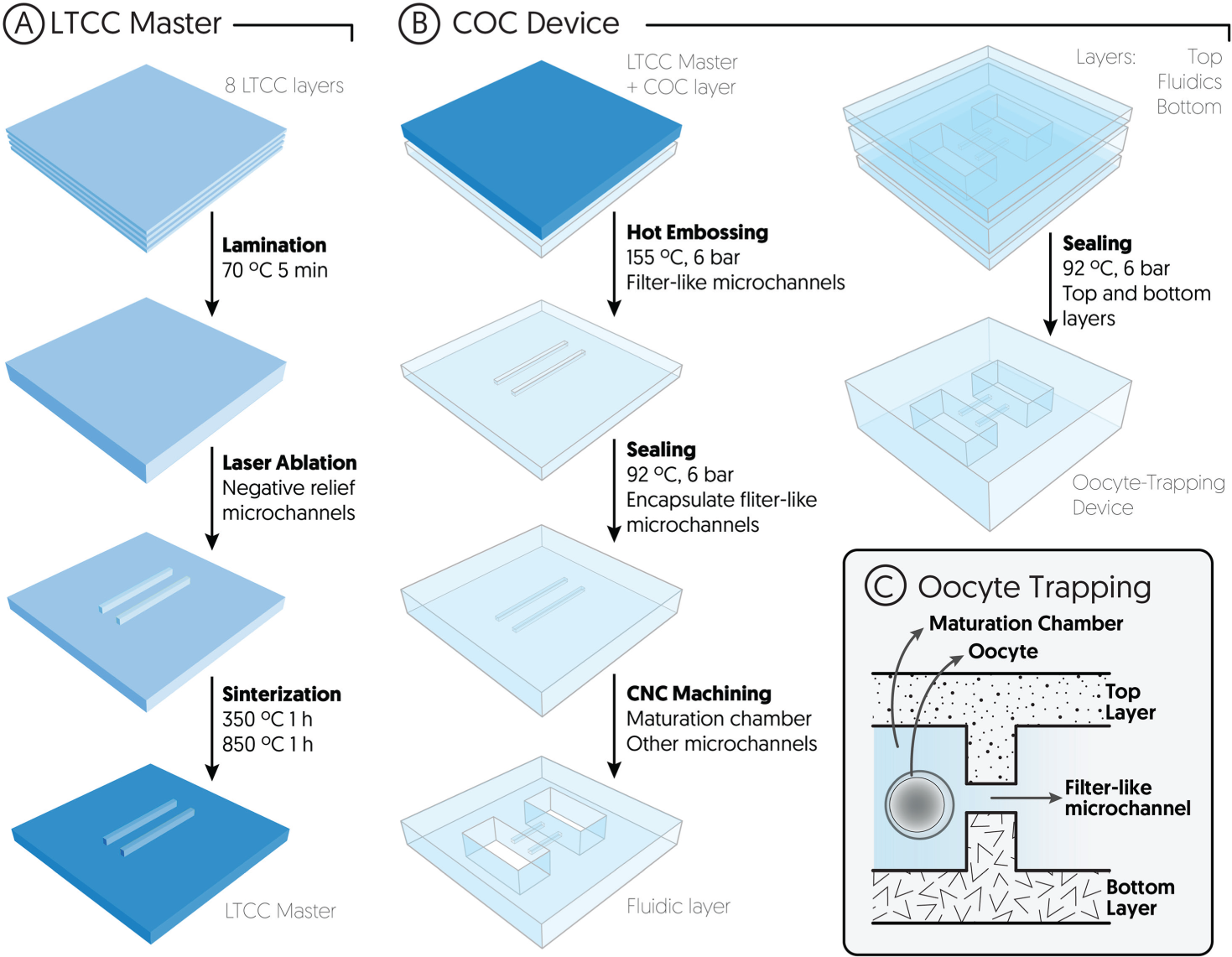
Schematic representation (not to scale) of the oocyte-trapping microfluidic device fabrication and working principle of the entrapment. (
First, a master was built using LTCC 951PX, 254 µm thick, from Dupont (Berlin, Germany). Eight LTCC layers were thermolaminated together in a uniaxial hydraulic press. Then, the ceramic block was etched by means of a Protolaser 200, from LPKF (Garbsen, Germany), and sintered in a programmable box furnace from Carbolite (CBCWF11/23P16, Afora, Spain). The master was designed to have four negative images of microchannels, in order to create a filter-like structure in the microfluidic device.
Second, the LTCC master was used to fabricate the oocyte-trapping microfluidic device. A 500 µm thick layer of TOPAS 5013 was embossed with the master at 155 °C and 6 bars, thus obtaining four microchannels in the COC. Another 500 µm COC layer (previously laminated with a 25 µm thick TOPAS 8007 foil) was laminated with the replica, at 92 °C and 6 bars, to seal the microchannels and obtain the filter-like structure. The rest of the fluidic structures, namely, the maturation chamber and the inlet/outlet channels, were machined cutting through the whole COC block using the micromilling machine.
The COC block with the microfluidic structures was thermally laminated between two more layers of COC (top and bottom layers), thus obtaining the sealed microfluidic device. Both top and bottom layers had been prelaminated with a film of TOPAS 8007, which acted as gluing layer, as described elsewhere. 23 Inlet/outlet vias were drilled using the CNC micromilling machine.
Experimental Design and Setups
In order to evaluate the suitability and biocompatibility of COC as a substrate material for microfluidic devices used in ART, we designed two different microfluidic devices, one for sperm storage and another one for oocyte maturation. Three different sets of experiments were carried out.
First, bovine oocytes were randomly distributed between the microfluidic device and a four-well culture dish, and matured for 24 h. Maturation rates were analyzed following nuclear and cytoplasmic maturation (chromosome and cortical granule distribution, respectively) by means of staining. Second, sperm viability, acrosome abnormalities, and motility were assessed at 3, 6, 12, and 24 h in three different units, namely, the sperm storage microfluidic device, a four-well culture dish, and a 1.5 mL Eppendorf tube. Finally, bovine oocytes were matured either in the microfluidic device or in a four-well culture dish, and subsequently fertilized in vitro in four-well dishes to evaluate penetration rates. All the experiments were conducted in triplicate.
In order to carry out these sets of experiments, two different setups were designed. On the one hand, the experiments involving the sperm storage microfluidic device were carried out by simply using a pipette to inject or retrieve the sperm samples into or from the device.
On the other hand, the experiments involving the oocyte-trapping microfluidic device were carried out in a setup schematically depicted in Figure 2 . Inlet 1 was connected to a syringe pump (540060, TSE Systems, Bad Homburg, Germany) in combination with a 2.5 mL gastight glass syringe from Hamilton (Bonaduz, Switzerland) by means of polytetrafluoroethylene (PTFE, i.d. 0.8 mm) tubing. This syringe (S1) contained the maturation medium to fill the microfluidic channels. Inlet 2 contained a short fragment of flexible Tygon tube, which was used as an injection port for the polished glass capillary syringe (S2) containing the oocytes. Once the capillary was inserted in the injection port, the syringe was set on a pump in order to inject the oocytes into the microfluidic device. Port 3 was used as an auxiliary waste outlet during the process of filling the device with media in order to remove any air bubbles, and then was capped with a stopper for the rest of the experiment. Port 4 was linked to a three-way valve (161T031, NResearch, Northboro, MA) connecting the oocyte-trapping microfluidic device to either the waste (during oocyte loading) or a syringe (S4) with media (during oocyte collection after the experiment).
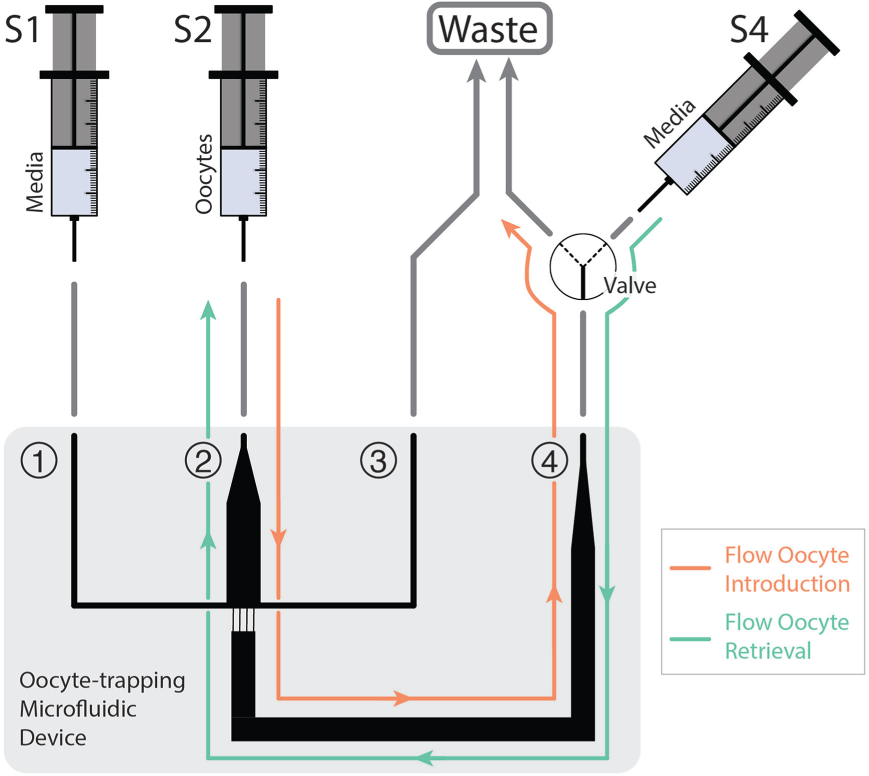
Schematic representation of the experimental setup for the oocyte maturation experiments on-chip. The oocyte-trapping microfluidic device consisted of four inlet/outlet ports. Ports 1 and 3 were simply used to fill the device with media and remove any air bubbles trapped next to the filter-like channels. Ports 2 and 4 were used for the oocyte injection/retrieval.
Bovine Oocyte Collection and In Vitro Maturation
Bovine ovaries were collected from recent culled dairy heifers at local slaughterhouses and immediately transported to the laboratory. They were then washed three times in warm saline solution (38 °C). Subsequently, follicles with a diameter of 2–6 mm were aspirated using an 18-gauge needle. Only unexpanded cumulus–oocyte complexes surrounded by five or more cumulus cell layers and with homogeneous cytoplasm were matured in vitro and cultured at 38.5 °C in a humidified atmosphere of 5% of CO2 for 24 h. The maturation medium contained TCM-199 medium supplemented with 10% (v/v) fetal calf serum, 10 ng/mL epidermal growth factor, and 50 µg/mL gentamycin. 25
Assessment of Sperm Parameters
Commercial frozen bull semen of proven fertility was used. Semen straws were thawed in a water bath at 38.5 °C for 30 s, and then spermatozoa were immediately centrifuged at room temperature in a top-layer solution of a discontinuous gradient (BoviPure, Nidacon International AB, Göthenborg, Sweden) for 10 min and 100g. The supernatant was removed, and the pellet was resuspended in 3 mL of BoviPure wash solution and centrifuged again for 5 min at 100g. The sperm concentration of the pellet was determined using a hemocytometer chamber (Neubauer chamber) and adjusted to a final concentration of 1 × 106 spz/mL with fertilization medium (Tyrode’s medium supplemented with 25 mM sodium bicarbonate, 22 mM sodium lactate, 1 mM sodium pyruvate, 6 mg/mL fatty acid–free bovine serum albumin [BSA], and 10 mg/mL heparin sodium salt [Calbiochem, Darmstadt, Germany]), and cultured at 38.5 °C in a humidified atmosphere of 5% of CO2 for 24 h.
The sperm cells’ viability and acrosome abnormalities were assessed by the nigrosine–eosin stain method. 26 Ten microliters of sperm sample and 10 µL of the dye solution were mixed and smeared onto a glass slide and allowed to air-dry. Then, the slide was covered with mounting medium and a cover glass. Slides were analyzed using an optical microscope (Motic BA210, Barcelona, Spain) at 1000× magnification under immersion oil. As many as 200 cells were counted on each slide, and the percentages of sperm viability and spermatozoa with acrosome abnormalities were calculated.
The motility characteristics of the frozen–thawed spermatozoa were determined using a computer-assisted sperm analysis (CASA) system (Integrated Sperm Analysis System V1.2; Proiser SL, Valencia, Spain). The CASA system is based on the analysis of 25 consecutive digital images taken from a single field at 100× magnification in a dark background in a time lapse of 1 s. A sample drop of 5 µL was placed on a prewarmed slide and viewed in a phase contrast microscope equipped with a warmer stage at 37 °C. At least five separate fields were taken of each sample, and a minimum of 200 cells per sample were examined. The motility descriptors obtained after CASA were progressive motility (percentage of spermatozoa that showed an average path velocity [VAP] above 50 µm/s and 70% of straightness coefficient) and total motility (percentage of spermatozoa that showed a VAP above 10 µm/s).
Oocytes and Sperm Handling On-Chip
The microfluidic devices were washed with 70% ethanol and rinsed twice with Milli-Q water. After drying, they were exposed to UV light for 30 min for sterilization. Prior to introduction of either oocytes or sperm, the microfluidic devices were respectively filled in with maturation or fertilization medium equilibrated at 38.5 °C in a 5% CO2 incubator.
Oocytes were loaded into the microfluidic device through a flexible Tygon tube (Port 2, Fig. 2 ), which acted as an injection port, where the tip of the polished glass capillary syringe (S2) was inserted. Then, the syringe was coupled to a pump and a flow rate of 50 μL/min was applied until the oocytes reached the maturation chamber. To achieve that, Port 3 was capped and therefore the flow was directed from Port 2 to Port 4 through the filter-like microchannels, and then into the waste ( Fig. 2 ). The oocytes were thus retained in the filter-like structure since their size is bigger than the microchannel dimensions. In order to retrieve the oocytes after the maturation experiments, the flow direction was inverted: the syringe in Port 2 was unplugged—leaving only a small fragment of tube—and the valve in Port 4 was switched, connecting the device to the syringe (S4). Then, a 50 μL/min flow rate was applied in the direction from Port 4 toward Port 2 ( Fig. 2 ). Oocytes were thus collected in four-well plates.
Sperm samples were injected into the sperm storage microfluidic device directly with a pipette. Immediately after injection, both inlet and outlet were sealed using an adhesive film AB-1170 from Thermo Scientific (Schwerte, Germany), and the microfluidic device was incubated at 38.5 °C for the corresponding period of time. After the incubation, the film was removed and the sperm sample was collected with a pipette. The sperm samples were then analyzed to assess sperm quality parameters after exposure to the COC.
In Vitro Fertilization
After maturation, the cumulus–oocyte complexes were washed twice in phosphate-buffered saline (PBS) solution and then transferred to fertilization medium. Frozen–thawed bull spermatozoa were counted in a Neubauer chamber and diluted in an appropriate volume of fertilization medium to give a final concentration of 106 spermatozoa/mL. All cumulus–oocyte complexes were coincubated with spermatozoa for 20 h at 38.5 °C in fertilization medium in a humidified 5% CO2 incubator.
Oocyte Evaluation and Classification
After 24 h of in vitro maturation, oocytes were totally denuded of cumulus cells by gentle pipetting in PBS. In order to evaluate nuclear stage and cortical granule distribution after in vitro maturation, oocyte samples were fixed in a solution of 4% (w/v) of formaldehyde and PBS at 38.5 °C for 30 min and permeabilized in Triton X-100 2.5% (v/v) in PBS for 15 min. Then, oocytes were immunostained for cortical granule detection with fluorescein isothiocyanate–labeled Lens culinaris agglutinin (FITC-LCA). Fixed and stained oocytes were mounted on poly-
Cultured oocytes were checked to have reached the metaphase II (MII) stage. Oocytes that reached the MII stage after maturation were classified into two categories: (1) normal MII, uniform alignment of the chromosomes on the spindle, and (2) anomalous MII, nuclear content changed into chromatin-like structure forming condensed aggregates, forming aberrantly distributed chromosomes or the absence of chromosomes.
Translocation of cortical granules to the oolema was used as an indicator of cytoplasmic maturation. 27 The criteria used to define the cortical granule distribution of oocytes was classified into two categories: (1) normal cytoplasmic maturation, cortical granules were distributed adjacent to the plasma membrane and positioned such that they formed a continuous layer, and (2) anomalous cytoplasmic maturation, cortical granules appeared aggregated in clusters, or cortical granules were distributed in the cortical area (not limited to the vicinity of the plasma membrane), or there was an absence of cortical granules.
Evaluation of Sperm Penetration
At 20 h postinsemination, the presumptive zygotes were pipetted to remove excess sperm and cumulus cells, washed three times in PBS, fixed in 4% (v/v) paraformaldehyde in PBS at 38.5 °C for 30 min, stained with Vectashield containing DAPI, and mounted on glass slides. The number of pronuclei was assessed under an epifluorescence microscope (Axioscop 40FL, Carl Zeiss, Göttingen, Germany). Once stained, slides were examined and parameters including penetration, monospermic penetration, male pronucleus formation, and polyspermic penetration were recorded. Penetration was determined by the presence of one or more swollen sperm heads and/or male pronuclei. The presence of three or more pronuclei was designated as polyspermic.
Statistical Analysis
All statistical analyses were performed using the R program (version 2.15.0; R Development Core Team, 2009). Data of on-chip oocyte maturation were analyzed using contingency tables and Pearson’s chi-squared statistical test. The analysis of differences among the different tested devices for sperm viability and motility parameters was carried out using the Kruskal–Wallis test. All data are expressed as mean ± SD. In all cases, differences between groups with p < 0.05 were considered significant.
Results and Discussion
Microfluidic Design and Fabrication
Two different microfluidic devices were designed in order to study the oocytes and the sperm samples independently. The microfluidic device for sperm consisted of a simple channel to store the sperm for different periods of time and then compare the quality of the samples with the control experiments. Four independent microfluidic units were fabricated on the same substrate to study the sperm samples at 3, 6, 12, and 24 h, respectively. The dimensions of the channel were chosen to fit the required volume for the motility and viability tests, plus excess in case it might be needed (total volume ca. 90 μL). Figure 3 shows the microfluidic device used in the study of the sperm samples.

Microfluidic device for sperm storage. The device features four independent fluidic units to analyze a sample at different times. For clarity purposes, the channels were filled with blue dye.
The oocyte-trapping microfluidic device consisted of a maturation chamber with a filter-like structure to trap the oocytes and four inlet/outlet ports ( Fig. 4 ). Ports 1 and 3 were used to fill the entire device with the maturation medium and remove any air bubble prior to oocyte introduction, and then they were not used anymore during the experiments. Ports 2 and 4 were used to inject/retrieve the oocytes into/from the device by applying a flow rate in one or another direction. The features of the oocyte microfluidic device were more challenging than those of the sperm microfluidic device, from the fabrication perspective. Microfluidic channels with a smaller size than the oocyte diameter were required to trap them, and common micromilling equipment, usually employed for thermoplastics, is not able to achieve such dimensions. 28 Therefore, filter-like channels were hot embossed on the COC plates.

(
Several materials can be found in the literature for the fabrication of masters, for example, micromilled brass, 29 photolithographycally patterned silicon, 30 or epoxy. 18 However, the fabrication of these type of masters requires time-consuming and expensive processes. Therefore, the master used in this work was fabricated using LTCC technology, due to its simple and fast iteration from design to prototype (around 6 h) in an inexpensive way. 31 Furthermore, no clean room facilities were required for its fabrication. Several COC replicas were fabricated without noticeable degradation of the master. The simple prototyping enabled by the LTCC technology enhances the optimization process of the master features required by the desired application.
The different layers of COC were laminated together using the thermal diffusion bonding technique. TOPAS 8007 foils, 25 μm thick, were used as a “gluing” layer. The lower Tg of this substrate enables the lamination of the layers with minimal deformation. In this case, this is particularly important in order to avoid occlusion due to the reduced dimensions of the filter-like microchannels. The availability of a number of COC grades with different thermal properties is one of its assets in front of other thermoplastics like PS for several reasons: the fabrication process becomes simpler, smaller features can be implemented in monolithic devices, and surface modification/activation or solvents are not required for bonding. The latter fact is very important in biological applications, since solvents and other reagents used for surface modification could potentially harm cells.
The filter-like microchannels obtained after sealing the microfluidic device had a trapezoidal shape, with dimensions of approximately 100 and 50 μm, respectively, for the bases, and 55 μm high ( Fig. 4C ). The oocyte-trapping microfluidic device enabled the injection, maturation, and removal of the oocytes ( Fig. 5 ).
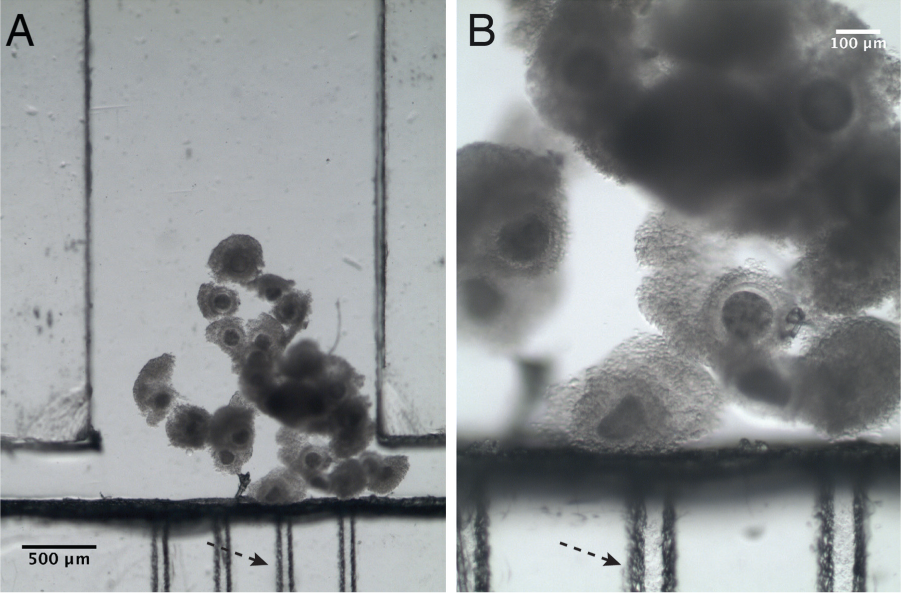
Microscope pictures of the oocytes trapped in the microfluidic device. The dashed arrow indicates the position of the capillary channels that act as a filter for oocytes. (
On-Chip Oocyte Maturation
The oocytes were injected into the microfluidic device at an optimal flow rate of 50 µL/min (experimentally determined). Using smaller flow rates would involve longer times for oocyte loading/retrieval. No oocyte deformation was observed using this flow rate. Higher flow rates would cause deformation and/or denudation of the oocytes in the filter-like channels (channel constriction). Other studies in the literature take advantage of this phenomenon for cumulus removal. 32 However, we were not interested in it due to the important role of cumulus cells in oocyte maturation. 33 The oocytes were retained in the vicinity of the filter-like microchannels of the maturation chamber, as seen in Figure 5 .
Figure 6A shows the results obtained from the oocytes in vitro matured into the microfluidic device compared with the four-well culture dish. Figure 6B , C shows examples of oocytes correctly matured. Although oocytes matured in the microfluidic device showed a low-grade cumulus cell expansion, no significant differences (p > 0.05) were found in the percentage of oocytes progressing to normal MII and cytoplasmic maturation between both devices. These results are in agreement with others found in the literature, 34 in which pig oocytes matured in PDMS microchannels did not show significant differences (p > 0.05) in the percentage of maturation rates compared with oocytes matured in conventional 500 µL drops or in 8 µL drops (volume control). In addition, Walters et al. 34 also observed a low grade of cumulus cell expansion of the cumulus–oocyte complexes matured in the PDMS device. The molecular processes involved in cumulus cell expansion need to be evaluated to check presumptive implications on processes related to fertilization and embryo development.
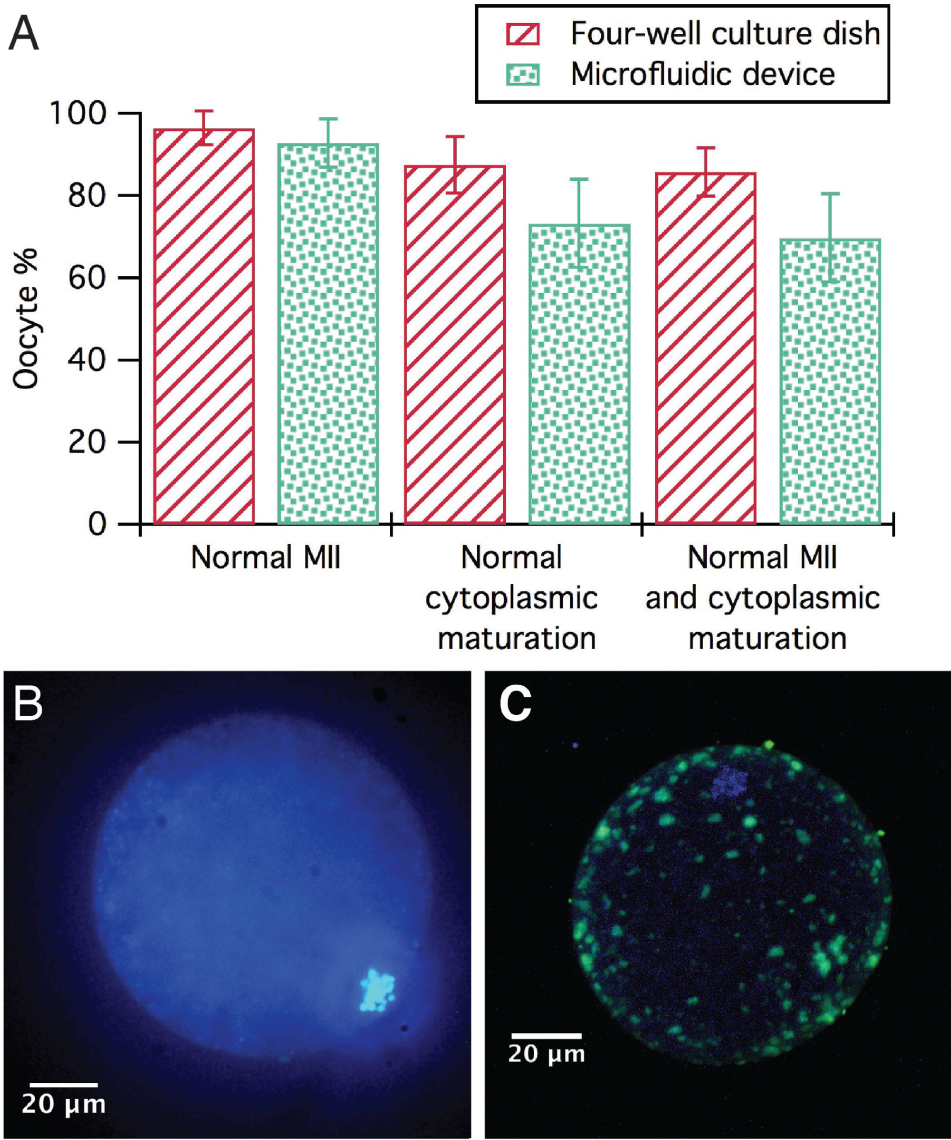
(
The small volumes used in microfluidics may lead to a rapid depletion of factors and/or a pH shift in the surroundings of the oocytes. The pH in the microfluidic device is not buffered with the CO2 atmosphere of the incubator because it is airtight—unlike the four-well dish—and this could cause the lower cumulus cell expansion, since oocytes appear to lack porters to regulate their intracellular pH,35,36 which is greatly affected by CO2 and O2 concentrations. 37 Although this could be overcome by employing dynamic culture, that is, imparting flow and thus renewing the media, in this study we evaluated the static culture, as it can be more easily compared with the traditional four-well dish method.
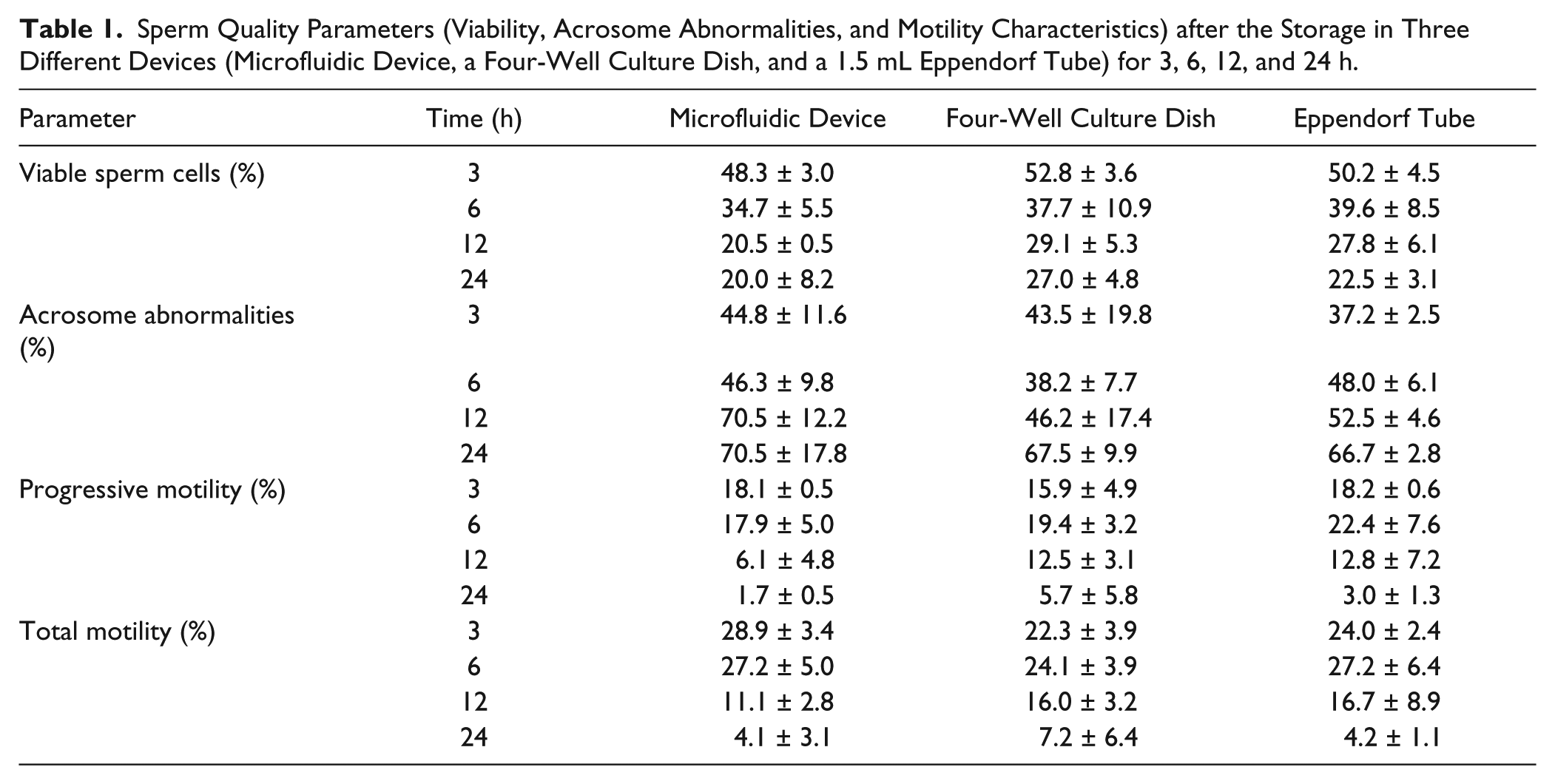
Time-Dependent On-Chip Sperm Evaluation
Results obtained from the analysis of the sperm storage into the microfluidic device, a four-well culture dish, and a 1.5 mL Eppendorf tube are shown in Table 1 . Data show the expected decrease of the percentage of viability and motility, and an increase of the percentage of acrosome abnormalities over time, with no significant differences among the three devices (p > 0.05). This suggests that COC is an innocuous and biocompatible material regarding sperm quality parameters, and it could be used for the development of microfluidic systems. Previous studies have tested the biocompatibility of materials such as PS (standard petri dish material), polymides, silicons, and PDMS, assessing mice embryo development and pig sperm motility parameters.38–40 However, to the authors’ best knowledge, this is the first study that has tested the biocompatibility and absence of the toxicity of COC on bovine gametes.
Sperm Quality Parameters (Viability, Acrosome Abnormalities, and Motility Characteristics) after the Storage in Three Different Devices (Microfluidic Device, a Four-Well Culture Dish, and a 1.5 mL Eppendorf Tube) for 3, 6, 12, and 24 h.
In Vitro Fertilization after On-Chip Oocyte Maturation
Table 2 shows the significantly higher percentages of sperm penetration observed in the four-well culture dish when compared with the microfluidic device at 18 h after IVF (61.44% vs. 35.63%, respectively; p < 0.05). However, in terms of normal fertilization with male pronuclear formation, no significant differences were observed between both devices (p > 0.05). These data suggest that when an oocyte is penetrated by a sperm cell, fertilization is comparable and efficient in both maturation systems. Thus, the detected reduction in the fertilization rate after on-chip oocyte maturation seems to be a consequence of the quality of the matured oocyte rather than due to deficiencies in the process of fertilization. The oocyte maturation system adequately supported nuclear maturation but probably failed to produce oocytes with a complete cytoplasmic competency, additionally to correct cortical granule migration and maturation. Cytoplasmic maturation encompasses a wide array of metabolic and structural modifications, including events that ensure the occurrence of normal fertilization, meiotic-to-mitotic cell cycle progression, and activation of pathways required for genetic and epigenetic programs of preimplantation embryonic development. 41 Additionally, it is known that cumulus cell expansion is an important marker for oocyte maturation. 42 Indeed, in cattle, inhibition of cumulus cell expansion was shown to be independent from nuclear maturation, but essential for fertilization and subsequent cleavage and blastocyst development. 43 Thus, it might be thought that the low grade of cumulus cell expansion observed after on-chip maturation could also be related to a poorer maturation of the zona pellucida, necessary for a proper fertilization of the oocyte. However, and as stated before, employing a dynamic culture with the on-chip oocyte maturation system could help to improve the overall oocyte maturation.
Effects of Devices on the In Vitro Fertilization Rates of In Vitro Matured Bovine Oocytes.

Values within a column differ significantly, p < 0.05.
The differences between the microfluidic device and the four-well culture dish can also be explained by the fact that the COC microfluidic device is airtight and does not allow the gas exchange, thus impeding the buffering of the media by the CO2 contained inside the incubator. Similar maturation studies in PDMS microfluidic devices (gas permeable) show a significantly higher percentage of porcine embryo cleavage rates (67% vs. 49%; p < 0.05). 44 However, the gas permeability of PDMS implies that the microfluidic device needs to be in an incubator with controlled atmosphere. In contrast, COC microfluidic devices, in combination with a suitable heater,45,46 would not require a control of the atmosphere, and therefore minimize the required equipment. Moreover, the problem of the depletion of factors around the oocytes and the media buffering could be overcome by using a dynamic culture, that is, renewing the media by pumping a fresh one into the maturation chamber.
Conclusions
This work describes a novel rapid prototyping process for COC microfluidic devices, combining a hot embossing step—using an LTCC master—for the smaller features required and a micromilling step for the rest of the fluidic structures. We have demonstrated that the COC microfluidic device enables the functional and automated maturation of oocytes, and that it is nontoxic for gametes. The microfluidic device allows trapping the oocytes by means of a filter-like structure and their maturation. No significant differences were observed in oocytes reaching normal MII and cytoplasmic maturation in terms of cortical granule migration, compared with the control experiments. Lower penetration rates were observed for oocytes matured in the microfluidic device, which could be related to the lower grade of cumulus cell expansion of the cumulus–oocyte complexes or an incomplete cytoplasmic maturation of the oocytes. However, this issue could be addressed by improving the factor availability and pH buffering capacity in the surroundings of the oocytes, for instance, by dynamic culture.
The properties of COC open new possible applications for microfluidic systems in ART due to the simple fabrication processes at both the prototyping and mass production scales, taking advantage of the different material grades commercially available. COC is not hindered by mass production limitations, absorption of molecules, or gas permeability, thus avoiding a strict control of the exterior atmosphere conditions, and provides simpler fabrication methods than other thermoplastics, especially regarding bonding.
Current efforts focus, on the one hand, on the development of a temperature control system to avoid the use of big, heavy, and expensive equipment such as incubators, and on the other hand, on the control of the factors and the buffering of the media in the surroundings of the oocytes. These two features would enable the construction of an independent automated microfluidic platform using COC for different ART applications.
Footnotes
Acknowledgements
The authors gratefully acknowledge the financial support of the Ministerio de Economía y Competitividad and FEDER (projects CTQ2012-36165 and AGL2013-46769-P) and by the government of Catalonia (SGR 2014-837). M. Berenguel-Alonso was supported by the government of Catalonia with a scholarship (FI-DGR 2012, cofunded by the ESF). M. Sabés-Alsina was supported by a predoctoral research fellowship (PIF) from the Universitat Autònoma de Barcelona, Bellaterra, Spain. R. Morató was supported by a postdoctoral research fellowship (“Juan de la Cierva” Scheme; Ministry of Economy and Competitiveness, Spain). Finally, the authors want to thank A. Pujol for the photography work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support of the Ministerio de Economía y Competitividad and FEDER (projects CTQ2012-36165 and AGL2013-46769-P) and by the government of Catalonia (SGR 2014-837). M. Berenguel-Alonso was supported by the government of Catalonia with a scholarship (FI-DGR 2012, cofunded by the ESF). M. Sabés-Alsina was supported by a predoctoral research fellowship (PIF) from the Universitat Autònoma de Barcelona, Bellaterra, Spain. R. Morató was supported by a postdoctoral research fellowship (“Juan de la Cierva” Scheme; Ministry of Economy and Competitiveness, Spain).
