Abstract
The Na+/taurocholate cotransporting polypeptide (NTCP) is located in the basolateral membrane of hepatocytes, where it transports bile acids from the portal blood back into hepatocytes. Furthermore, NTCP has a role for the hepatic transport of some drugs. Extrapolation of drug transport data from rodents to humans is not always possible, because species differences in the expression level, localization, affinity, and substrate selectivity of relevant transport proteins must be considered. In the present study, a functional comparison of human NTCP (hNTCP) and mouse Ntcp (mNtcp) showed similar Km values of 67 ± 10 µM and 104 ± 9 µM for the probe substrate estrone-3-sulfate as well as of 258 ± 42 µM and 199 ± 13 µM for the drug rosuvastatin, respectively. IC50 values for the probe inhibitor cyclosporine A were 3.1 ± 0.3 µM for hNTCP and 1.6 ± 0.4 µM for mNtcp. In a drug and pesticide inhibitory screening on both transporters, 4 of the 15 tested drugs (cyclosporine A, benzbromarone, MK571, and fluvastatin) showed high inhibitory potency, but only slight inhibition was observed for the 13 tested pesticides. Among these compounds, only four drugs and three pesticides showed significant differences in their inhibition pattern on hNTCP and mNtcp. Most pronounced was the difference for benzbromarone with a fivefold higher IC50 for mNtcp (27 ± 10 µM) than for hNTCP (5.5 ± 0.6 µM).
In conclusion, we found a strong correlation between the transport kinetics and inhibition pattern among hNTCP and mNtcp. However, specific compounds, such as benzbromarone, showed clear species differences. Such species differences have to be considered when pharmacokinetic data are transferred from rodent to humans.
Introduction
Bile acids efficiently circulate between the liver and gut. This is commonly referred to as enterohepatic circulation of bile acids. Within this process, the Na+/taurocholate cotransporting polypeptide (NTCP) is the most relevant hepatic bile acid uptake carrier.1,2 NTCP typically transports bile acids in a strictly sodium-dependent manner.3,4 In addition, members of the organic anion transporting polypeptide (OATP) family, such as OATP1B1 and OATP1B3, contribute to the hepatic uptake of bile acids. Impaired hepatic bile acid uptake and intestinal reabsorption affect the bile composition, and the consequence can be gallstone formation in the bile duct or gallbladder. 5 NTCP belongs to the Solute Carrier Family SLC10 and has orthologous genes in humans (gene symbol SLC10A1) and rodents (gene symbol Slc10a1). 2 The NTCP/Ntcp proteins in humans (hNTCP) and mice (mNtcp) share a high-amino-acid sequence identity of 77%, and both proteins are localized at the basolateral membrane of hepatocytes. Nevertheless, hNTCP (349 amino acids) is 13 amino acids shorter in its C-terminus than mNtcp (362 amino acids).3,6 NTCP/Ntcp mediates the uptake of several endogenous substrates like bile acids, sulfated steroid hormones (e.g., estrone-3-sulfate [E1S]), 2 and sulfated thyroid hormones (T3S, T4S), 7 as well as some drugs like rosuvastatin, pitavastatin, and fluvastatin. 8 Although the role of NTCP for hepatic drug transport is not so prominent as for the hepatic OATP carriers, drug-induced inhibition of hepatic bile acid transport via NTCP would increase plasma bile acid concentrations and might play a role for drug-induced liver injury.5,9 As an example, inhibition of the bile acid transport via NTCP by rifampicin or cyclosporine A can cause severe disruption of the bile homeostasis. Vaz et al. reported a hNTCP deficiency by homozygous point mutation at position 252 from arginine to histidine (R525H) in one patient, leading to a tremendous elevation (up to 100-fold) of total bile acid in plasma as well as impaired fat-soluble vitamins A, D, and K. As a consequence, decreased bone density and an elongated time of prothrombin formation can occur. 10
Transport data generated for laboratory animals cannot be uncritically transferred from rodent models to the situation in humans. Very often data are missing that directly compare the transport and inhibition characteristics of homologous carriers between humans and rodents. One striking difference between hNTCP and mNtcp is already known. Both proteins significantly differ in their roles as high-affinity hepatic entry receptors for the hepatitis B (HBV) and hepatitis D (HDV) viruses.11,12 Although both viruses can bind to hNTCP and thereby induce their cellular entry via endocytosis, mNtcp is only binding competent for the viral preS1-peptide of the HBV/HDV surface proteins it does not support HBV/HDV entry and infection. Thus, in contrast to hNTCP, mNtcp lacks the function as virus entry receptor. More detailed molecular analyses identified amino acids 84–87 of mNtcp to be responsible for this effect, and a mNtcp mutant bearing the human amino acids 84–87 at the corresponding positions fully supported in vitro HBV infection.13,14 In the course of identification of hNTCP as the high-affinity hepatic HBV/HDV entry receptor, this protein got additional importance as a novel drug target for virus entry inhibition. Several novel HBV/HDV entry inhibitors acting at hNTCP were recently described,15,16 making it even more important to better understand the inhibition pattern of this transporter/receptor protein.
The aim of the present study was to directly compare the substrate transport and xenobiotic inhibition patterns of hNTCP and mNtcp. In general, we found quite similar behaviors for both carriers, but also found some additional striking species differences.
Materials and Methods
Reagents
[3H]Estrone-3-sulfate ammonium salt (6,7- 3 H(N); further referred to as [3H]E1S) was purchased from American Radiolabeled Chemicals (50 Ci/mmol; St. Louis, MO). All nonlabeled chemicals were obtained from Sigma-Aldrich (Darmstadt, Germany).
Transfection and Cell Culture
The respective cDNAs of hNTCP (GeneBank accession no. NM_003049.3) and mNtcp (GeneBank accession no. NM_001177561.1) have been cloned into the expression vector pcDNA5/FRT/TO. Human embryonic kidney cells (T-REx HEK 293 [Invitrogen, Darmstadt, Germany]; further referred to as HEK cells) were transfected using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s protocol. Twenty-four hours after transfection, 175 µg/mL hygromycin B was added to the medium to select stable clones. After 2 or 3 weeks, single colonies were picked and expanded in Dulbecco’s modified Eagle’s medium (DMEM; high glucose) supplemented with 10% fetal bovine serum (Biochrom, Berlin, Germany) and 1% penicillin (10,000 U/mL)/streptomycin (10 mg/mL). Cell lines were grown in a humidified atmosphere containing 5% CO2 at 37 °C.
Transporter-Mediated Uptake of Radiolabeled Substrates
For uptake assays, 2 × 105 cells in 0.5 mL of growth medium per well were seeded into 24-well plates, coated with poly-
For time-dependent experiments, cells were incubated at increasing time points (1–30 min) at 37 °C in the presence of labeled [3H]E1S (10 nM) filled up to 10 µM with unlabeled E1S. To determine the affinity (Km) of E1S as a substrate of NTCP, saturation experiments at an initial rate period were performed as determined in time dependency experiments. NTCP-transfected HEK and empty vector-HEK cells were incubated for 3 min with 10 nM [3H]E1S and increasing concentrations (1–500 µM) of nonlabeled E1S. Experiments were conducted on two separate days as triplicates.
Inhibition experiments for IC50 determination were performed for 3 min with the known inhibitor of NTCP, cyclosporine A. At 5 µM E1S (containing 10 nM [3H]E1S), the uptake was cis-inhibited by increasing concentrations of cyclosporine A (0.5–100 µM).
For screening experiments, cis-inhibition was carried out in duplicate by measuring the uptake of the labeled probe substrate in the absence and presence of 10 or 100 µM of the respective pesticide or drug inhibitor. Transporter- and vector-transfected HEK cells were incubated with 10 nM [3H]E1S. Inhibitory effects in percent were calculated from the net uptake (mean NTCP-specific transport rates).
Transporter-Mediated Uptake Measured by LC-MS
To quantify the NTCP-mediated accumulation of statins in HEK cells, 24-well plates seeded with stable transfected hNTCP as well as mNtcp cells were prepared as described above. After culturing the cells for 3 days, 10 and 100 µM of statins (atorvastatin, fluvastatin, pravastatin, and rosuvastatin) in HBSS buffer were incubated for 1 min. The uptake was terminated by three times washing with ice-cold PBS. After removal of the washing buffer, the cells were dissolved in acetonitrile/water (3:1 ratio).
NTCP-mediated uptake of statins and the accumulation in HEK cells were quantified using liquid chromatography tandem mass spectrometry (LC-MS/MS) analysis. The samples for LC-MS/MS detection were lysed in 500 µL of lysis buffer containing 75% acetonitrile with the appropriate internal standard (5 ng/mL cerivastatin). The cell lysate (150 µL) was mixed with 300 µL of acetonitrile/methanol (9:1), and so cell debris was removed by centrifugation at 16,500g for 15 min. The supernatants were transferred to a 96-well collection plate. Ten microliters of the samples was used for LC-MS/MS analyses with fluid 75% acetonitrile/25% water/0.3% formic acid. High-performance liquid chromatography (HPLC) was performed using Chromatography Series 200 (PerkinElmer, Massachusetts) coupled with an API 4000 tandem mass spectrometer (Life Technology). For separation, a Brownlee SPP RP-Amide column (4.6 × 100 mm inner dimeter, 2.7 µm particle size, PerkinElmer) with a SecurityGuard C18pre-columm (4 × 2 mm, Phenomenex) was used. The substrate peak was detected using substrate-specific mass transition and retention times. The area under the peak was used for quantification based on the standard curve conducted in the same experiment. 18
Determination of Protein Concentrations
The cellular protein amount was determined using a method described by Bradford. 19 Six wells per cell line were analyzed in parallel to the transport experiments in an additional 24-well plate. Therefore, cell monolayers were washed three times with 0.5 mL of incubation buffer and afterward stored at –20 °C. After thawing, the cells were lysed 30–60 min in 100 µL 1× lyses buffer (Promega, Mannheim, Germany), and then the cell lysates were filled up with ddH2O to 1 mL per well and mixed thoroughly. For the protein determination in 96-well plates (flat bottom; Sarstedt, Nümbrecht, Germany), bovine serum albumin (BSA) was used as the standard for a calibration curve ranging from 50 to 300 µg/mL. A total of 20 µL of BSA standards or 20 µL of sample (1:1 diluted in ddH2O) was mixed with 200 µL of 1× Bradford reagent (Carl Roth) per well. After 10–20 min of incubation at room temperature, absorption was measured at 595 nm (microplate reader: Wallac Victor2; PerkinElmer, Rodgau, Germany).
Data Analysis
For the Km calculation of E1S, the transporter-mediated uptake (pmol/mg protein/min) was plotted against E1S concentrations. The Km and Vmax values were obtained using SigmaPlot version 13 by fitting the Michaelis–Menten equation V = Vmax*[S]/(Km + [S]), where V refers to the rate of substrate transport, Vmax refers to the maximum rate of substrate transport, [S] refers to the concentration of substrate, and Km is defined as the concentration of substrate at the half-maximal transport rate. The inhibitory effect I (%) was calculated according to the formula I (%) = 100 – (Vwith inhibitor*100/Vw/o inhibitor), and for the IC50 calculation of the inhibitor, the inhibitory effect I (%) was plotted against inhibitor concentrations and fitted using a three-parameter Hill equation using SigmaPlot version 13.
Results
Functional Characterization of Human and mNTCP
To elucidate the functional characteristics of hNTCP and mNtcp, initially, time-dependent experiments were performed using 10 µM E1S as substrate. The uptake into NTCP-HEK cells was measured over a period of 1–30 min as indicated in
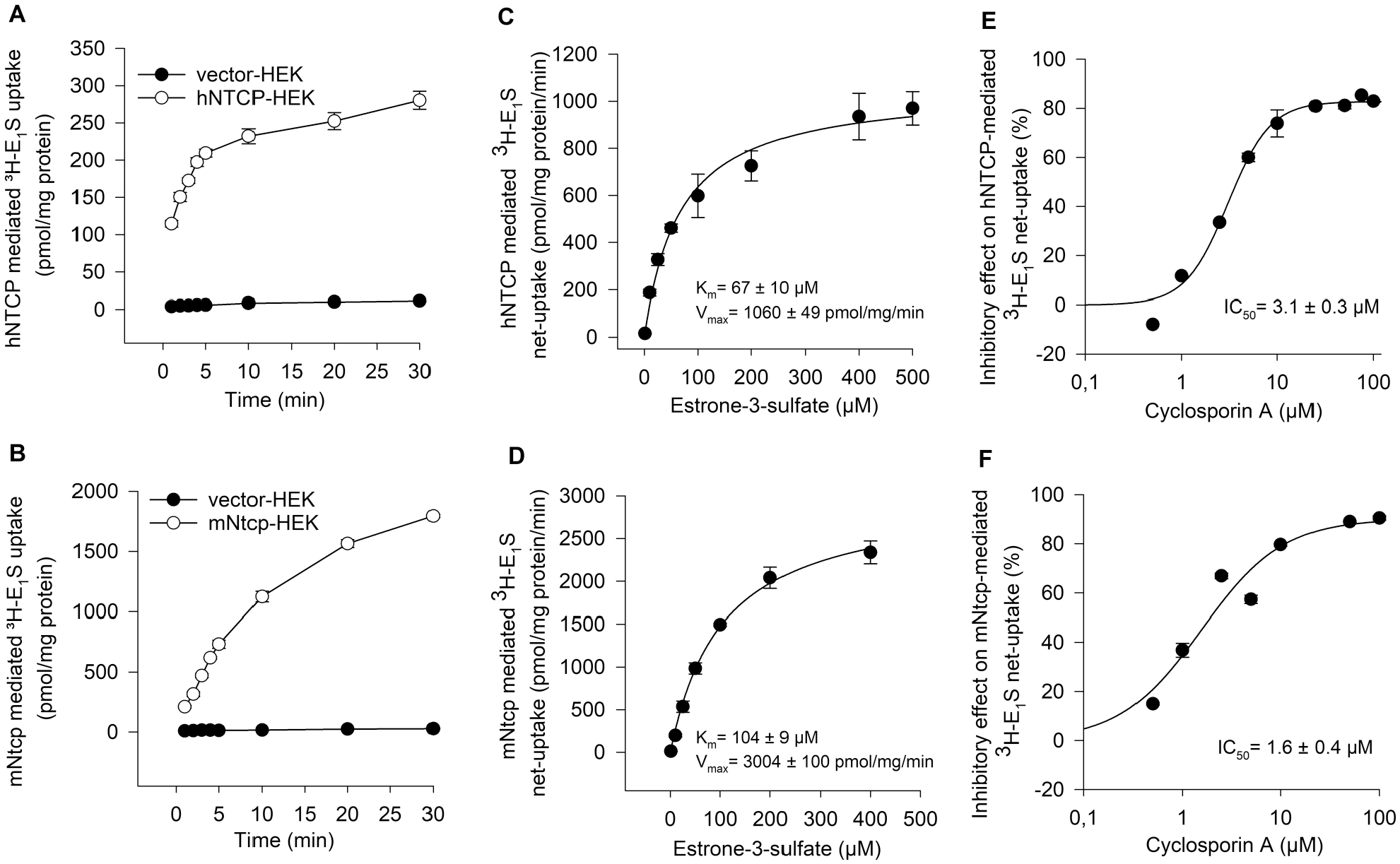
Functional comparison of hNTCP and mNtcp. Time-dependent uptake of [3H]E1S in (
Although the inhibitory potential of cyclosporine A on hNTCP was demonstrated in several previous publications, a systematic comparison of mNtcp and hNTCP was not yet reported, to our knowledge. Therefore, for further functional comparison, the E1S uptake of mNtcp and hNTCP was inhibited by increasing concentrations of cyclosporine A, resulting in IC50 values of 3.1 ± 0.3 µM for hNTCP and 1.6 ± 0.4 for mNtcp (
Interaction of hNTCP and mNtcp with Drugs and Pesticides
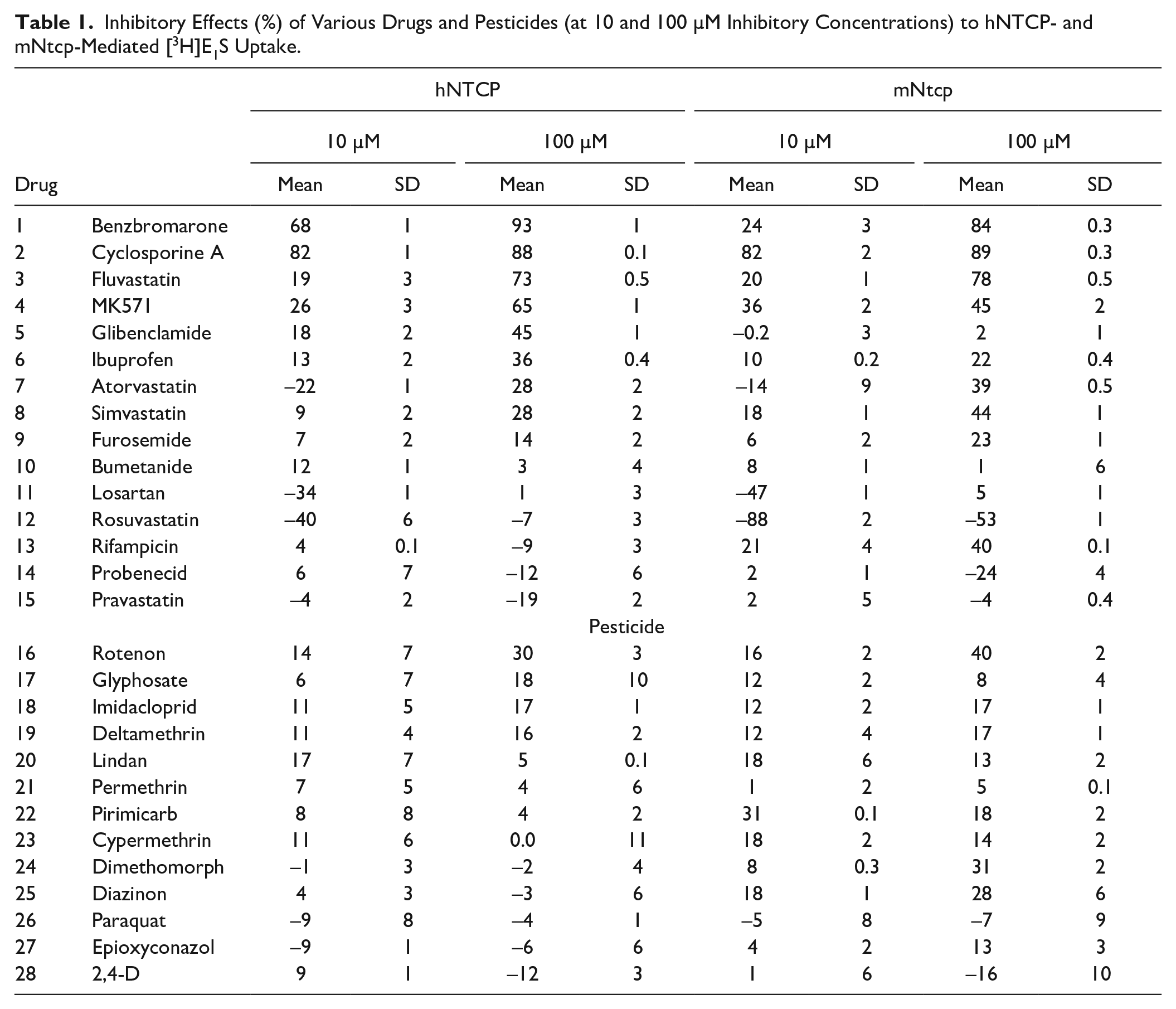
After completion of the functional characterization and validation, we evaluated the inhibitory potential of 13 pesticides as well as 15 Food and Drug Administration (FDA)-approved ones to hNTCP and mNtcp transport activity. The herbicides glyphosate, paraquat, and 2,4-dichlorophenoxyacetic acid (2,4-D), and the insecticides lindan, diazinon, pirimicarb, cypermethrin, deltamethrin, permethrin, imidacloprid, and rotenon, as well as the fungicides epoxyconazole and dimethomorph, were examined regarding their potential to interact with hNTCP and mNtcp. For each pesticide, 10 or 100 µM inhibitor concentrations were used to evaluate the inhibitory effect on the mNtcp- and hNTCP-mediated E1S uptake (
Inhibitory Effects (%) of Various Drugs and Pesticides (at 10 and 100 µM Inhibitory Concentrations) to hNTCP- and mNtcp-Mediated [3H]E1S Uptake.
Species Differences in the Inhibition Pattern between hNTCP and mNtcp
To search for species-specific interactions among 15 FDA-approved drugs and bioactive compounds, cyclosporine A, probenecid, glibenclamide, bumetanide, furosemide, ibuprofen, losartan, rifampicin, benzbromarone, MK571, rosuvastatin, fluvastatin, pravastatin, atorvastatin, and simvastatin were used at 10 or 100 µM concentrations for inhibition assays toward hNTCP and mNtcp. Probenecid, pravastatin, losartan, and rosuvastatin stimulated the E1S uptake via NTCPs by up to 88%, while glibenclamide, ibuprofen, atorvastatin, simvastatin, furosemide, bumetanide, and rifampicin inhibited the uptake by less than 50%. MK571 showed inhibitory effects at 100 µM for hNTCP and mNtcp of 65% and 45%, respectively. High inhibitory effects were also observed for benzbromarone, cyclosporine A, and fluvastatin. These compounds inhibited the transport activity of hNTCP and mNtcp at 100 µM concentrations to 7% to 27%. All three abovementioned drugs showed high and comparable inhibitory effects to hNTCP and mNtcp. Differential species-dependent effects were observed for rosuvastatin, rifampicin, and glibenclamide. In detail, rosuvastatin at 10 and 100 µM increased the mNtcp transport capacity by 88% and 53%, respectively, while only 40% and 7% stimulation of hNTCP was observed, respectively. Rifampicin showed negligible inhibitory or stimulatory effects of 4% or 9%, respectively, for hNTCP, but in contrast decreased the mNtcp activity at 10 and 100 µM down to 79% and 60%, respectively. Glibenclamide did not show any inhibition of mNtcp, but it inhibited the hNTCP-mediated transport of E1S at 10 µM up to 82% and at 100 µM up to 55%. Hence, rifampicin and glibenclamide demonstrated clear species-specific inhibition patterns at the 100 µM inhibitory concentration. While rifampicin preferentially inhibited mNtcp, the contrary was true for glibenclamide (
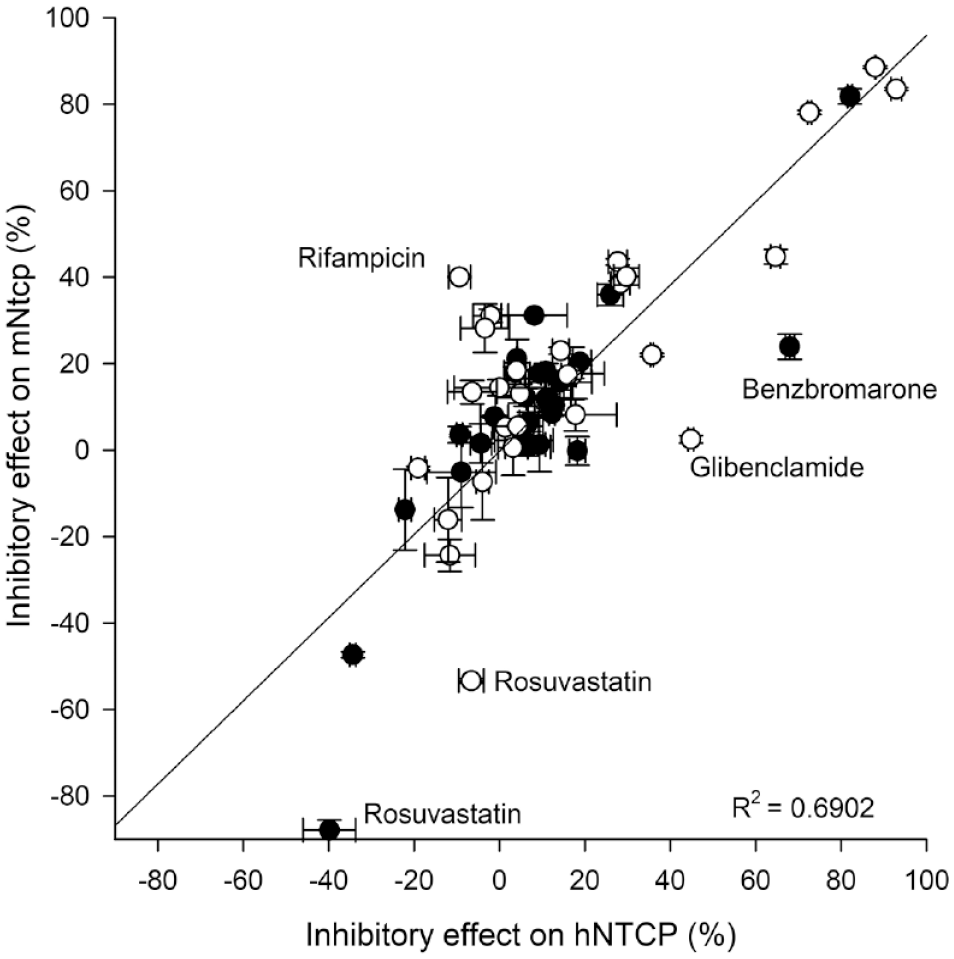
Scatterplot analysis correlating inhibitory and stimulatory effects of different drugs and pesticides at 10 µM (filled symbols) and 100 µM (open symbols) inhibitory concentrations on hNTCP- and mNtcp-mediated [3H]E1S uptake. The mean values and standard deviations of one individual experiment are presented in detail in
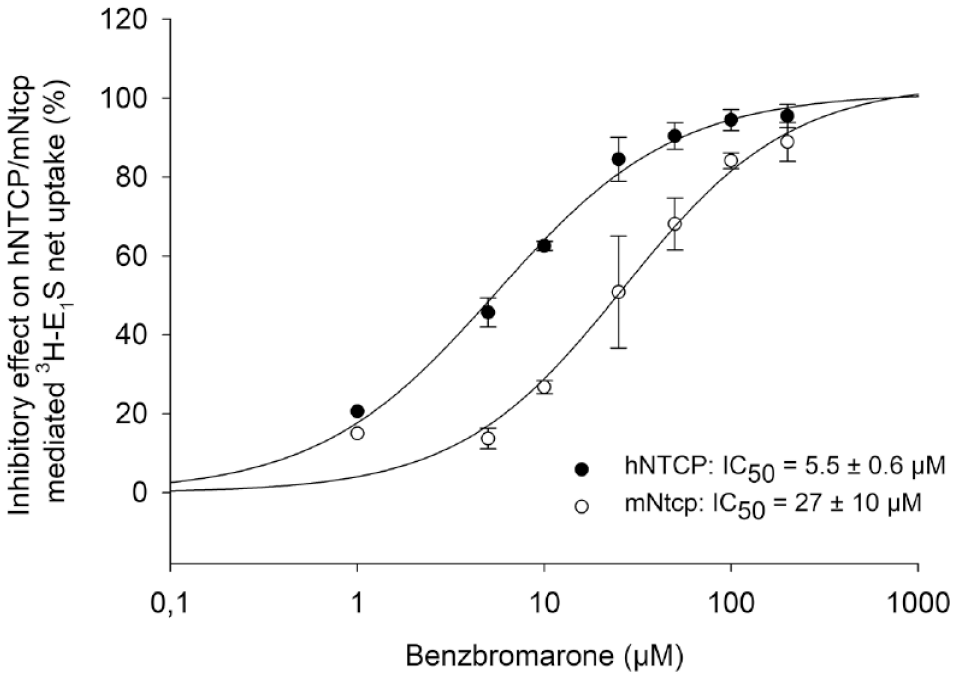
Inhibitory effects of increasing concentrations of benzbromarone (1–200 µM) on hNTCP- and mNtcp-mediated [3H]E1S uptake (10 nM). Data points represent the mean inhibitory effect (%) calculated from the net uptake (mean NTCP-specific transport) of two independent experiments ± standard deviation.
Statin Transport by hNTCP and mNtcp
In the inhibition assay, we examined four statins, which showed strongly different results. While 100 µM fluvastatin inhibited hNTCP and mNtcp with inhibitory effects of 73% and 78%, respectively (
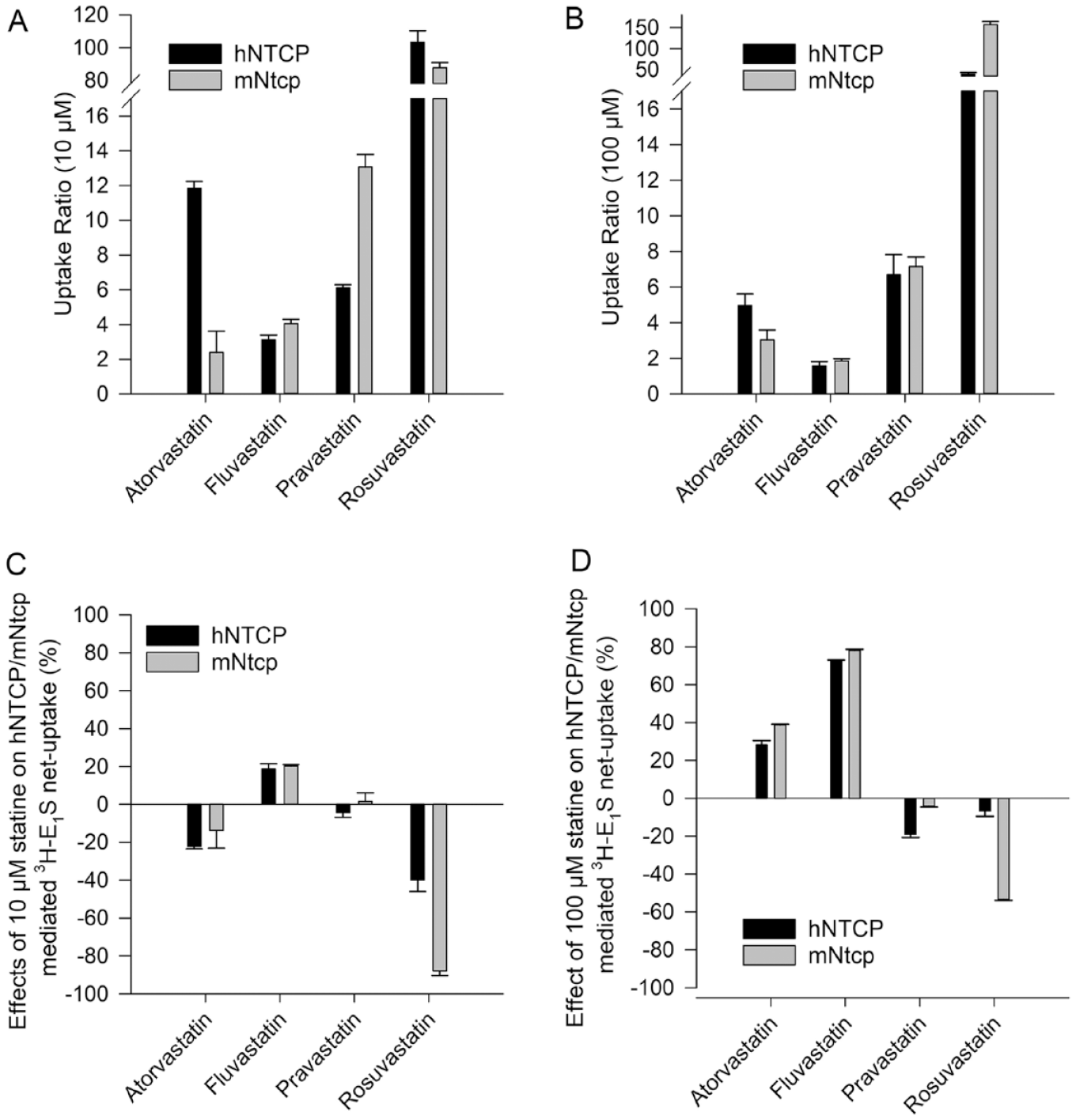
Transport and inhibition patterns of statins at hNTCP and mNtcp. The uptakes of (
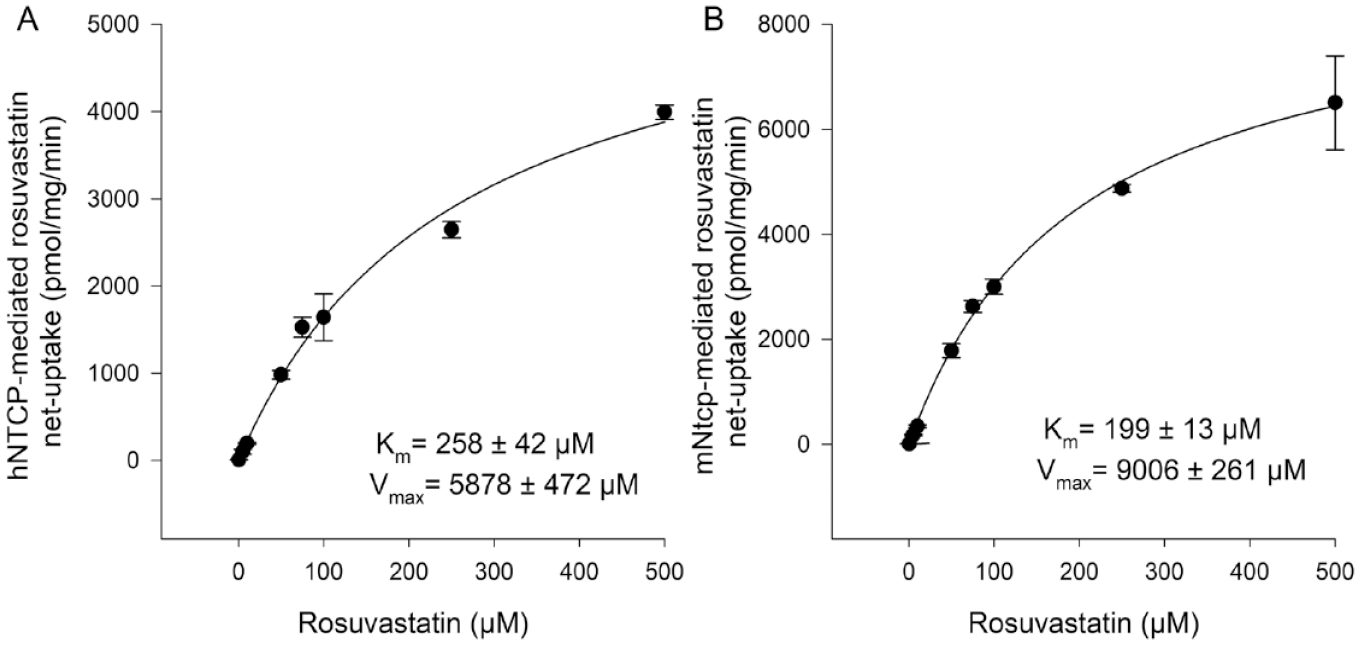
Kinetics of (
Discussion
Cellular uptake and secretion processes of endogenous metabolites like bile acid derivatives as well as exogenous molecules like nutrients, drugs and their metabolites, and toxins are dependent on the specific tissue differential expression of certain membrane transporters. One of the substrate-selective and highly tissue-specific transporters is NTCP. It belongs to the SLC10 family and is exclusively expressed on the basolateral membrane of hepatocytes, where it mediates the uptake of bile acids.1,2 Furthermore, NTCP facilitates the transport of several drugs and therefore is a potential site for drug–drug interaction in the liver. From 72 FDA-approved drugs, 31 were identified as NTCP inhibitors by in vitro drug screening. 20 Therefore, substantial inhibition of NTCP by drugs could result in impaired uptake of bile acids and impairment of their enterohepatic circulation, but it could also affect the hepatobiliary clearance and overall pharmacokinetics of NTCP-transported drugs, such as statins. Furthermore, hNTCP represents an attractive novel drug target for HBV/HDV entry inhibitors. 10
To elucidate drug pharmacokinetics in early development, preclinical studies are often performed in rodents. However, unexpected interspecies differences in the hepatobiliary excretion of drugs and endogenous metabolites can significantly change pharmacokinetic parameters and bile acid secretion. As an example, the human nonrenal clearance of napsagatran was overestimated by 7-fold 21 and that of the sulfasalazine analog susalimod by 20-fold 22 according to allometric scaling based on rat bile clearance data.
In the present study, the functional and pharmacological characteristics of hNTCP and mNtcp were directly compared under the same experimental conditions. We obtained the following basic information about the behavior of the two carriers. (1) The transport kinetics for the substrates E1S and rosuvastatin were quite similar for both carriers. This particularly applies for the respective Km values, while the Vmax values for both substrates were somewhat higher for mNtcp. This is possibly caused by higher protein expression levels that could not be exactly determined in the present study. For standardization of the cell amount, we seeded the same number of cells in each well; on the day of the uptake experiment, the whole protein concentration was determined and included in the calculation of the uptake rat. (2) Regarding pharmacological inhibition of hNTCP/mNtcp, three classes of inhibitors/modulators could be identified: cyclosporine A as an equipotent inhibitor (IC50 = 3.1 µM for hNTCP and 1.6 µM for mNtcp), benzbromarone as a differently active inhibitor (IC50 = 5.5 µM for hNTCP and 27 µM for mNtcp), and rosuvastatin as a stimulatory compound. The latter compound is most interesting for several reasons. Rosuvastatin is a direct transport substrate of both carriers hNTCP and mNtcp, and its transport rates are the highest among the group of several statin drugs (atorvastatin, fluvastatin, and pravastatin). Based on this, it was expected that rosuvastatin would inhibit the transport of another substrate such as E1S. However, rosuvastatin did not inhibit the E1S transport but instead stimulated it, and this stimulatory effect was somewhat higher for mNtcp compared with hNTCP. Therefore, rosuvastatin can be classified as a mixed substrate/allosteric stimulator with a higher modulator activity at mNtcp. Such stimulatory effects on membrane transporters were reported before. As an example, caffeine, bupivacaine, lidocaine, and quinidine stimulated the taurocholate uptake via hNTCP in a previous study. 23 However, in this study it was not analyzed if these compounds are also transported by hNTCP. Differences between hNTCP and mNtcp also occurred with some compounds that only inhibited one of the carriers. While glibenclamide inhibited only mNtcp, rifampicin solely showed an inhibitory effect on hNTCP. So, the inhibitory potential of both drugs to the respective transporter was species specific.
A limitation of the present study is that we only analyzed one isoform of mNtcp. Cattori et al. first described that mNtcp can be expressed in two isoforms of 362 amino acids (mNtcp1) or 317 amino acids (mNtcp2), which occur by alternative splicing. The shorter mNtcp2 showed a Km value of 14 µM for the taurocholate transport, which was sixfold lower compared with the mNtcp1 isoform. 6 Based on this, it cannot be excluded that mNtcp2 would have shown a different transport and inhibition behavior with the substrates and inhibitors used in the present study.
Another part of this study was the examination of 13 pesticides with regard to their hNTCP and mNtcp interaction. The intake of pesticides with nutrition is one of the major problems in the modern agricultural production of foodstuffs. Unfortunately, pesticides became a part of our nutrition, so the responsible agencies worldwide try to protect consumers by setting the maximum residue level (MRL). However, the MRL level is often exceeded accidentally or intentionally.24,25 People are continually exposed to pesticides by neighborhoods being close to farms that are intensively treated with pesticides, which leads to an intake of pesticides through inhalation as well as through the skin by contaminated air, but primarily through residues in foodstuffs. 26 Food safety reports (2014) of the German Federal Office of Consumer Protection and Food Safety, as well as the commission of the European Community for monitoring of pesticide residues in plant products, confirmed that pesticide residues were found to different extents in several foods (vegetables). Glyphosate is the most used pesticide worldwide, and 4000 exposures are reported by the U.S. Poison Control Center each year. Almost 10% of these cases are intentional (suicide) ingestions. 27 Thousands of accidental and intentional deaths by ingestion of paraquat are also observed. A plasma concentration of 734 µg/mL was determined in a patient who intentionally ingested glyphosate. 27
Pesticide-induced liver damage was reported, for, example for rotenone, glyphosate, paraquat, and deltamethrin,28–31 even though the contribution of NTCP to the accumulation of these pesticides in the liver is unknown. The interaction of several pesticides with NTCP, OCT1, OATP, MDR1, MRP2, and BCRP was examined by Bucher et al. in human HepaRG cells. 32 None of the eight pesticides—4.4 DDT, 2.4 DDT, endosulfan, chlordane, heptachlor dieldrin, lindane, and chordecone—showed a significant inhibition of NTCP-mediated uptake of taurocholate. In contrast, endosulfan, chlordane, heptachlor, dieldrin, and lindane demonstrated a significant inhibition of OCT1-mediated TEA uptake. 32 We observed the highest (about 40%) species-independent inhibitory effect on NTCP activity by rotenone. If NTCP facilitates the uptake and accumulation of rotenone in the liver, it could be involved in the rotenone-induced liver toxicity described by Jiang et al. 28 Pirimicarb, dimethomorph, and diazinon inhibited the E1S uptake by mNtcp down to 70% remaining activity; in contrast, hNTCP does not interact with these pesticides. Although on a low inhibition level, pirimicarb, dimethomorph, and diazinon demonstrated a species-dependent inhibition of mNtcp-mediated E1S uptake.
In conclusion, the interaction of drugs and pesticides with hNTCP and mNtcp showed principally a good correlation; therefore, in the case of Ntcp/NTCP, for most substances a transfer of data from the mouse model to humans seems to be possible. Nevertheless, for several drugs and pesticides like glibenclamide, rifampicin, pirimicarb, dimethomorph, and diazinon, a differential species-dependent interaction as well as altered affinity for benzbromarone was observed. Since it is currently impossible to predict which substances will interact differently between species, interaction studies of new drugs and also pesticides with transporter proteins from different species are still important to avoid unnecessary overestimation as well as underestimation of data.
Supplemental Material
sj-docx-1-jbx-10.1177_24725552211017500 – Supplemental material for Functional and Pharmacological Comparison of Human and Mouse Na+/Taurocholate Cotransporting Polypeptide (NTCP)
Supplemental material, sj-docx-1-jbx-10.1177_24725552211017500 for Functional and Pharmacological Comparison of Human and Mouse Na+/Taurocholate Cotransporting Polypeptide (NTCP) by Saskia Floerl, Annett Kuehne, Joachim Geyer, Juergen Brockmoeller, Mladen V. Tzvetkov and Yohannes Hagos in SLAS Discovery
Footnotes
Acknowledgements
For excellent technical support Anja Herdlitschke, Rovena Halpape and Cornelia Willnow. For funding the German Federal Ministry of Education and Research (BMBF).
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.F., A.K., and Y.H. are employed by PortaCellTec Biosciences GmbH, and their research and authorship of this article was completed within the scope of their employment with PortaCellTec Biosciences GmbH.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the German Federal Ministry of Education and Research (BMBF).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
