Abstract
Quality control monitoring of cell lines utilized in biomedical research is of utmost importance and is critical for the reproducibility of data. Two key pitfalls in tissue culture are 1) cell line authenticity and 2) Mycoplasma contamination. As a collaborative research institute, the National Center for Advancing Translational Sciences (NCATS) receives cell lines from a range of commercial and academic sources, which are adapted for high-throughput screening. Here, we describe the implementation of routine NCATS-wide Mycoplasma testing and short tandem repeat (STR) testing for cell lines. Initial testing identified a >10% Mycoplasma contamination rate. While the implementation of systematic testing has not fully suppressed Mycoplasma contamination rates, clearly defined protocols that include the immediate destruction of contaminated cell lines wherever possible has enabled rapid intervention and removal of compromised cell lines. Data for >2000 cell line samples tested over 3 years, and case studies are provided. STR testing of 186 cell lines with established STR profiles revealed only five misidentified cell lines, all of which were received from external labs. The data collected over the 3 years since implementation of this systematic testing demonstrate the importance of continual vigilance for rapid identification of “problem” cell lines, for ensuring reproducible data in translational science research.
Introduction
The reproducibility and quality of scientific data are influenced by a wide range of factors, and require clear experimental methodologies to be reported, as well as the use of equivalent experimental reagents where possible. Reproducibility also requires the characterization of reagents to ensure that they are unsullied. Cell culture (often referred to as tissue culture) is a cornerstone of modern biomedical research. Multiple quality control measures are critical for ensuring that reported data are reproducible, including cell line authentication and confirmation that cell lines are not contaminated.
Cell line authentication is essential to ensure that cell lines have not been accidentally mislabeled or cross-contaminated, leading to inaccurate interpretations based on disease models.1,2 Perhaps the most notorious example of cell line misidentification is that of the HeLa human adenocarcinoma cell line, which has cross-contaminated many other cell lines. 3 The high doubling rate of HeLa cells means that a small number of contaminating cells can outgrow and overtake a cell line it has contaminated. One of the better-known examples of this is the KB cell line. It was derived by Harry Eagle as an epidermoid carcinoma in the 1950s and shown a decade later to be a HeLa contaminant line. 4 In another instance of cell line cross-contamination, MCF7 human breast cancer cells selected for resistance to the chemotherapeutic adriamycin (originally named MCF-7/AdrR) were later found to be derived from the human ovarian carcinoma cell line OVCAR-8. The resistant line was one of the seminal NCI60 cell line panel and has been renamed NCI/ADR-RES. 5 In the instance of the rare cancer adenoid cystic carcinoma, the few cell lines available to the scientific community were shown to be misidentified, in fact including rat and mouse cell lines. 6 The International Cell Line Authentication Committee (ICLAC) maintains a valuable resource in their register of misidentified cell lines (https://iclac.org/databases/cross-contaminations/). 7 To avoid future misidentifications, DNA profiling of human cell lines by short tandem repeat (STR) analysis upon receipt, and periodically during culture, can verify the identity of a cell line and mitigate severe and costly repercussions.8,9
Mycoplasma contamination has similarly plagued the practice of cell culture. Mycoplasma are single-celled prokaryotes that are devoid of a cell wall. Contamination of in vitro cultures by Mycoplasma was first reported in 1956 10 and has continued to present a challenge to routine culture ever since. The primary source of Mycoplasma in a continuous cell culture is believed to originate from the human oral cavity (i.e., laboratory cell culture operators, speaking, and breathing11,12). This hypothesis was supported by one author upon testing and confirming the presence of Mycoplasma in their own saliva. More recently, there has been an increasing awareness of the impact of Mycoplasma contamination on reproducibility in the scientific literature. Screening of cell culture collections has produced estimates that 15%–35% of cell lines are Mycoplasma contaminated13,14 (and higher numbers have been reported in individual collections). The recognition of the ease with which tissue cultures can be contaminated and the implications for industries such as biotherapeutic production have stimulated the development of off-the-shelf Mycoplasma detection kits to allow routine laboratory screening for Mycoplasma contamination. Guidelines for systematic testing of cell cultures have been previously published. 15
While Mycoplasma infection can easily go unnoticed given that infected cells often do not present with visible or morphological symptoms, Mycoplasma contamination of cell lines can broadly impact cellular biology. These perturbations can alter DNA, RNA, protein synthesis, metabolics, and general cellular processes, though few examples of systematic studies of the impact of Mycoplasma have been reported. 16 Microarray analyses on contaminated human cells in culture have reported that Mycoplasma can affect the expression of hundreds of genes, including those encoding ion channels, receptors, growth factors, and oncogenes.17,18
A number of phenotypes that are reported to be due to Mycoplasma contamination can be misinterpreted as impacting the underlying human biology and can produce plausible but irreproducible data that impede translational science. For cancer experimental therapeutics, Mycoplasma has been shown to affect the response of cancer cell lines to chemotherapy. For example, Liu and colleagues noted increased sensitivity to cisplatin, gemcitabine, and mitoxantrone in HCC97L human hepatocarcinoma cells infected with Mycoplasma hyorhinis compared with uninfected cells. 19 Mycoplasma-contaminated HCT-116 colon cancer cells were found to be 5- to 100-fold more resistant to 5-fluorouracil and 5-fluorodeoxyuridine, respectively, than cells that were “cured” of infection with antibiotics. 20 One of the authors has published a report that tiopronin (thiola) selectively kills multidrug-resistant (MDR) cancer cell lines, 21 but subsequent to publication it was found that the MDR cells were Mycoplasma contaminated. 22 Treating the cells with plasmocin to remove Mycoplasma contamination reversed the sensitivity of the cells to tiopronin, and resistant cell lines were sensitized to tiopronin when intentionally contaminated with Mycoplasma. As a consequence, the authors elected to retract the publication and correct the record. While the data were legitimate, the scientific conclusions were not valid (“tiopronin kills Mycoplasma-contaminated drug-resistant cell lines”). 22 For each individual example above, there are likely thousands of unrecognized examples in the literature.
Mycoplasma Testing Experience at NCATS
At the NIH National Center for Advancing Translational Sciences (NCATS), there is a strong scientific focus on collaborating with the scientific community. Using assay development and high-throughput screening (HTS), and advanced technologies such as induced pluripotent stem cells (iPSC) and tissue printing, we are working to further our understanding of rare and neglected diseases, novel targets, and expand basic biological understanding of the “undrugged” genome. This is accomplished through a team science approach that often includes assay development and automated quantitative high-throughput screening (qHTS) with a small-molecule library to identify active hits in biochemical or cell-based assays. 23 Hits may progress to medicinal chemistry to develop a small-molecule probe, guided by orthogonal and cell-based counterassays. In such a collaborative environment, NCATS routinely receives cell lines from partnering labs and a range of commercial vendors.
Given the phenotypic impact of Mycoplasma contamination, executing an HTS of hundreds of thousands of compounds with such a cell line would be costly and wasteful. Accepting the reality that Mycoplasma-contaminated cell lines may regularly be received from collaborators, or that contamination may arise during culture in labs at NCATS, we established a routine weekly Mycoplasma testing system. A central location was established for NCATS scientists to deliver a sample of expended culture media, and samples were tested each Friday with the MycoAlert assay by Lonza (Morristown, NJ), with results emailed to those submitting samples. The MycoAlert assay couples the production of ATP (from a provided substrate) by an endogenous Mycoplasma enzyme with a luciferase enzyme to produce light (chemiluminescence). Implementation was achieved with minimal burden: the assay is affordable and can be accomplished in the laboratory within 1 h. A weakness of the MycoAlert assay is that it can only detect a limited number of Mycoplasma species, while PCR-based methods of Mycoplasma detection are highly sensitive due to species-specific primers. PCR-based Mycoplasma kits may offer greater sensitivity than enzymatic assays; 24 however, PCR-based detection can be more time-consuming and costly.
A set of policies were implemented for use of cell lines for discovery purposes in the NCATS biology and HTS labs in tandem with this weekly testing protocol:
All cell lines must be certified Mycoplasma-free prior to receipt. As part of establishing a collaboration, any cell lines developed by the collaborating laboratory that are to be shipped to NCATS must be affirmed as Mycoplasma-free by the collaborator.
All cell lines are tested at NCATS upon receipt. When received cell lines are thawed into culture for the first time, they are tested to confirm that they are Mycoplasma-free.
All cell lines in regular culture must be tested at least once a month, or when thawed from cryovial stocks.
All cell lines must be tested immediately prior to HTS execution. As part of planning for an automated HTS, cell culture scale-up must include retesting for Mycoplasma. The negative result must be presented as part of the assay protocol handover to the automation team.
Contaminated (positive) cell lines should be destroyed immediately and backup frozen stock cultured and assessed for Mycoplasma status. All stock should be destroyed if other cryovials prove to be positive. In exceptional circumstances, contaminated cells can be quarantined in a dedicated incubator outside the tissue culture room, and plasmocin treatment can be used to destroy culture (an example situation is described below).
Cell lines that receive ambiguous results should be quarantined and retested the following week. Two ambiguous tests should be considered contaminated and dealt with as outlined in step 5.
Occasionally, a Mycoplasma-contaminated cell line was treated with the anti-Mycoplasma compound plasmocin to remove the contamination. 25 Plasmocin is successful at removing contamination 25 in most but not all cases, and studies with contaminated immortalized monocytes 26 and human pluripotent stem cells demonstrated that plasmocin treatment did not alter cellular phenotypes (including stemness and pluripotency) compared with the original cell line. 27 Resourcing of cell lines is preferred, but not always an option.
By way of example (anonymized to protect the guilty), NCATS scientists sourced a tumor cell line from the laboratory that created it. The cell line harbored a somatic mutation and was one of the few cell lines available to scientists at the time. However, on receiving the cell line, it was found to be highly contaminated and the originating lab was alerted. Discussions revealed that the lab had never tested for Mycoplasma and had shared the cell line with many other labs (none of which had raised the alarm), and that backup stock was likely also contaminated. The NCATS scientists treated the cells with plasmocin over multiple weeks and then cultured them plasmocin-free for 4 weeks prior to testing and confirming that the cells were Mycoplasma-free. The cell line was then shipped back to the originating lab as the new noncontaminated cell line. By increasing awareness of the issue of Mycoplasma contamination, we hope to encourage routine testing and provide suggestions for responding to contamination to lower the percentage of contaminated cell lines used in research laboratories.
STR Testing for Cell Line Identity
In parallel, we established a routine practice for STR profiling for authentication of cell lines. STR profiling of human cell lines utilizes a PCR-based assay to determine the number of repeated tandem DNA sequences at specific loci throughout the human genome. 8 The location of these sequences, or markers, is consistent for all humans; however, each human has a differential number of STR sequences at each marker, which are inherited parentally. By assessing multiple STR markers for each person, one can build a profile that can be compared with others to determine if the samples are from related or unrelated individuals.
Standard methods for STR profiling involve using Promega (Madison, WI) kits that amplify 10, 16, or 18 markers per sample. Since each STR marker is present on both copies of each chromosome, each STR marker is reported as two numbers corresponding to the number of STRs at that marker.
If a cell line has a known STR profile (reference profile), then any sample purported to match it should be compared with the reference profile to ensure the sample is authentic. ICLAC standards state that related samples have greater than 80% of their STR alleles matching using the algorithm below:
If a cell line does not have a known STR profile or is a newly generated cell line, it is important to establish the STR profile early on in order to track potential misidentification. A supplementary method that can be employed for rapid testing of cells is PCR-based identification of the species from which the cell line was derived. 28
STR profiling is utilized by NCATS scientists to confirm the identity of human cell lines being used in our research laboratories, authenticate cell lines received from external collaborators (compared against reference STR profiles), or establish an STR profile for newly created cell lines.
Materials and Methods
Mycoplasma Testing
A MycoAlert (Lonza) assay was adapted for 96-well plates and completed as specified by the manufacturer. In brief, a 50 µL sample and reagent were combined and incubated, and analyzed for a primary signal, and then 50 µL substrate was added and incubated, followed by a secondary read. Assay plates were read on a ViewLux microplate imager (PerkinElmer, Waltham, MA) to quantify luminescence over a 30 s exposure. If the ratio of secondary to primary luminescence was >1.2, the sample was marked positive for Mycoplasma (<0.9 was negative and 0.9 ≤ 1.2 indicated an ambiguous result).
STR Testing
Frozen samples of cell lines were deposited in a common area with an associated request form specifying the number of STR markers to be profiled and the name of the cell line within the sample and passage number. A regular monthly shipment of samples was sent to a commercial provider of STR testing, primarily the Genetic Core Resources Facility at Johns Hopkins University. STR profiling reports including the full profile as well as the results of profile searches within the American Type Culture Collection (ATCC) and Deutsche Sammlung von Mikroorganismen und Zellkulturen (DMSZ; a German facility that provides comprehensive scientific cell culture services) databases were distributed to scientists.
Results
Mycoplasma Testing
Since routine Mycoplasma testing was implemented at NCATS, just over 2000 samples have been tested. In the three full calendar years of testing to date (2016, 2017, and 2018), an average of 500 samples were tested per year ( Table 1 ). In the first year of testing, 13% of the cell line samples were positive ( Table 1 ). A month-by-month breakdown of historical data ( Fig. 1 ) shows a 19% positive return rate in the first month. These data are consistent with other reports on contamination rate,13,14 and perhaps unsurprising given that routine testing had not been implemented previously. Subsequent to the first year of testing, 6%–8% of cell lines tested positive ( Table 1 ), with occasional “spikes” (e.g., see November 2018 and April 2019 in Fig. 1 ) that were associated with testing and retesting of a number of cell lines received from external laboratories.
Mycoplasma Testing Results.
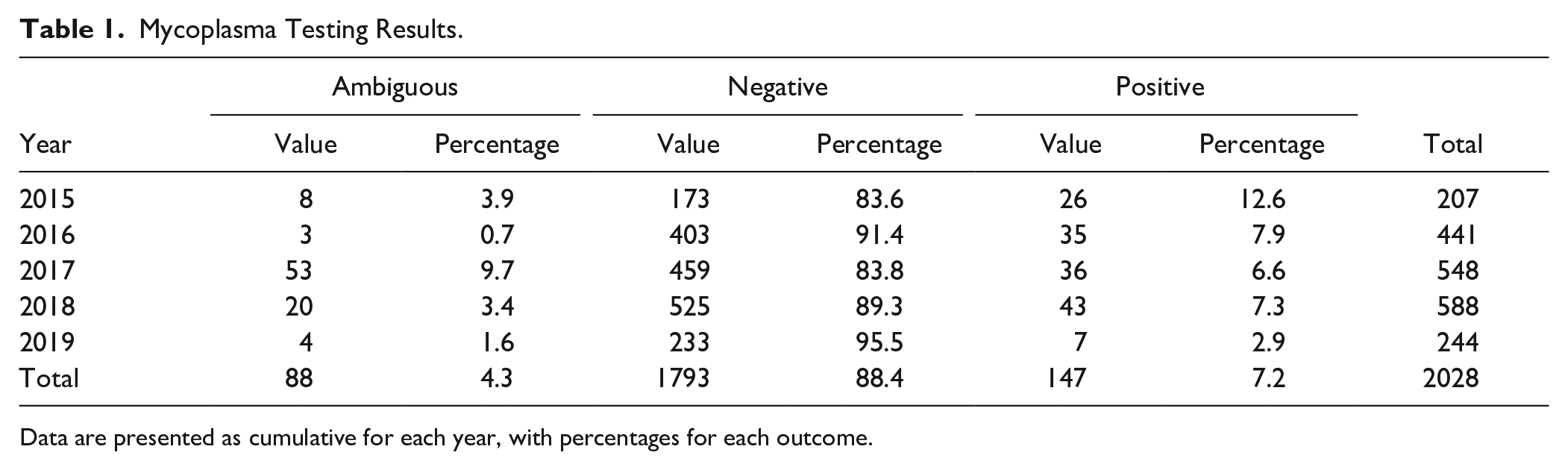
Data are presented as cumulative for each year, with percentages for each outcome.
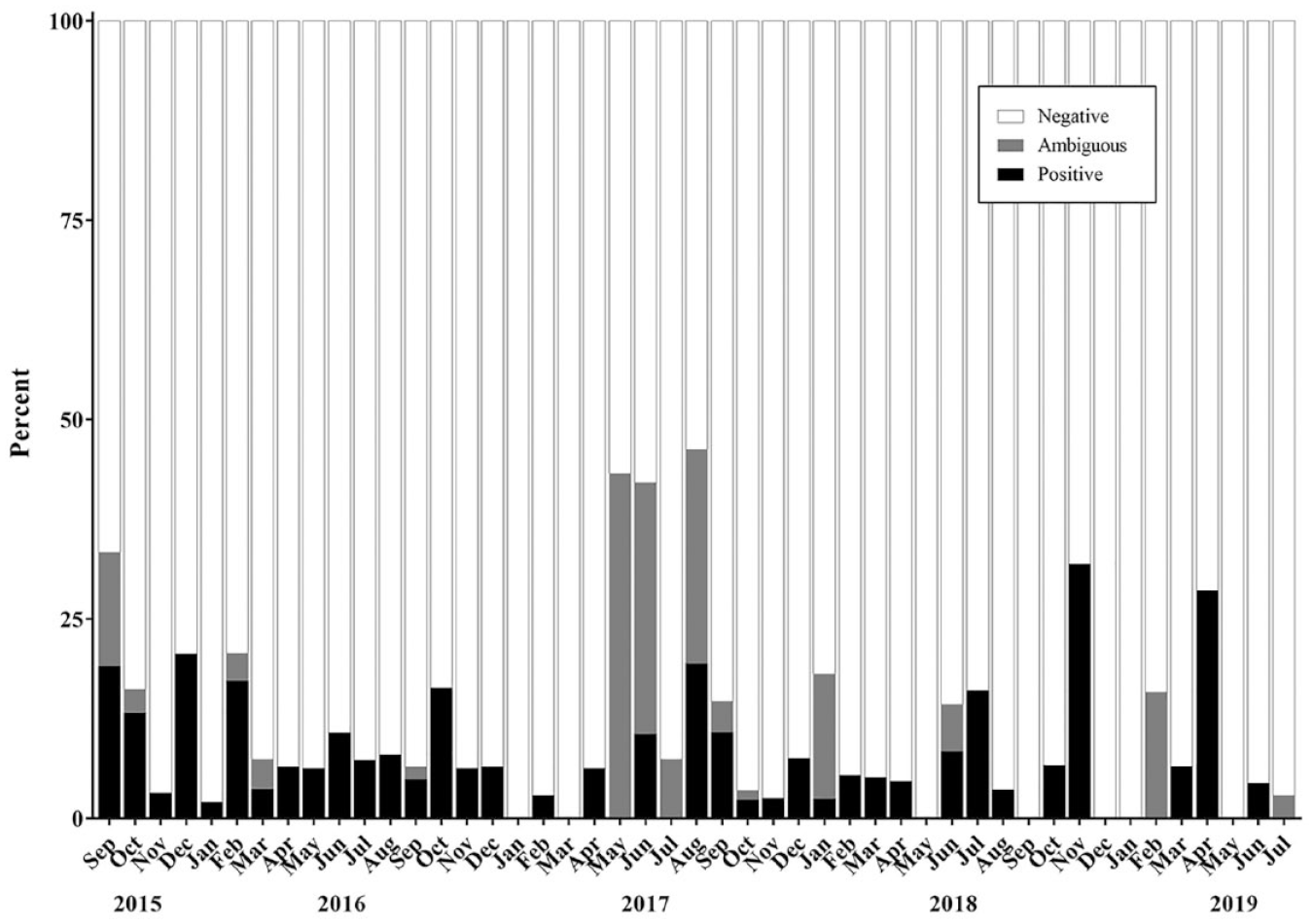
Mycoplasma testing results by month from September 2015 through July 2019.
While cell culture can occasionally suffer from fungal or bacterial contamination across tissue culture flasks that is visually evident in culture medium, 29 our anecdotal evidence is that no such “outbreak” of Mycoplasma contamination was observed over the almost 4-year period. This work did not consider the difference between primary and established cell lines with respect to prevalence of mycoplasma infection; however, the following paper reports that across a large dataset, only cultured cells and not primary tissue were Mycoplasma contaminated—which suggests user contamination as the source of infection. 30
For the final 12 months of data collection (beginning in July 2018), samples were further annotated with regard to their origin: “NCATS Internal” or “Collaborator” ( Fig. 2 ). Any cell line in culture or brought into culture from frozen stock after being cultured at NCATS was designated “NCATS Internal.” In a 12-month period, a larger proportion of collaborator samples (13%) than internal samples (5%) tested positive for Mycoplasma.
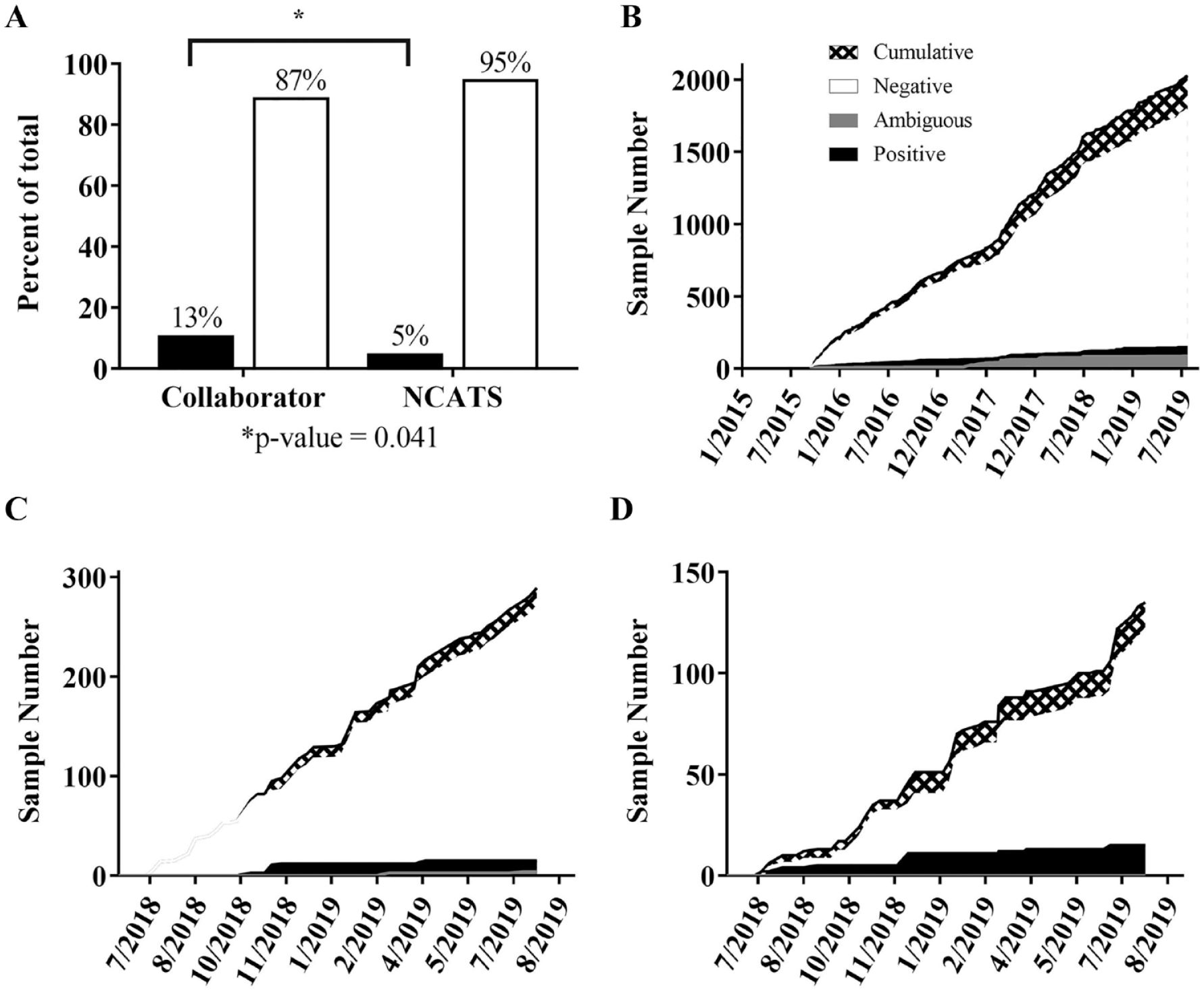
(
STR Analysis
To date, NCATS has tested 306 cell line samples for STR profiling, representing 274 unique cell lines. Of these samples, 89 were primary and iPSC cell lines, where the goal was to ensure that subsequent clones had the same STR profiles as the original clone. Thirty-one samples had no published STR profile for reference, even though the cell lines were published in the literature. The remaining samples were cell lines with a known reference STR profile to compare against. Five cell lines did not match the known STR profile for that cell line (one example is described below), including a case where two cell lines being used by a scientist had been inadvertently switched. The remaining cell lines showed >80% match (accepted threshold) to the available reference STR profile and were considered to be matches ( Fig. 3 ).
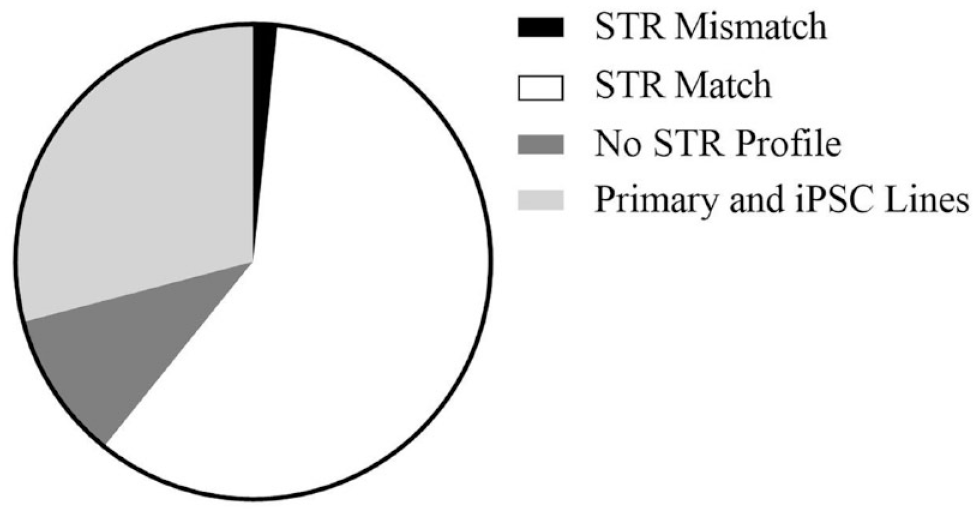
STR profiling results from 306 tested samples; 186 samples had existing STR profiles, of which 181 matched and 5 were a mismatch to the expected profile. A match of >80% to the available STR profile was accepted as a match. There were no published STR profiles for 31 samples, and 89 were primary and iPSC lines without STR profiles.
Discussion
Benefits of Regular Cell Line Testing
The benefit of a routine STR screening program can be made by the following example (as above, anonymized to protect the guilty). A collaboration was initiated with the goal of screening drug libraries against an isogenic pair of cell lines: a commonly used pancreatic cancer line BxPC3, and the same cell line with a stable knockdown of a specific gene. The aim was to identify approved drugs that may demonstrate synthetic lethal activity against the knockout line compared with the wildtype, with the long-term goal of being able to take a validated active drug to the clinic. Upon receipt of the two cell lines, STR testing was performed, and it was found that the knockdown cell line was in fact another pancreatic cancer cell line (MiaPaCa-2). The collaborator had no earlier passage cells to utilize. An HTS using this mismatched cell line pair could have been wasteful, with any bioactive hits from the screen leading to false conclusions regarding the translational potential of hits.
Given the high (>15%) tissue culture Mycoplasma contamination rates reported in the literature, it is likely that a significant number of published phenotypes may be affected by Mycoplasma contamination. At NCATS, systematic testing for Mycoplasma and STR profiles has prevented misidentified cell lines and contaminated cells from being used in HTS. This improves the quality of research and the likelihood of reproducibility of our data.
It is hoped that increasing the awareness of the benefits of regular Mycoplasma testing will reduce the number of positive cultures, particularly given that some cell lines provided from collaborator labs may have been utilized in research for many years. To ensure that findings are robust, we suggest that phenotypes studied in long-term laboratory cell cultures be reconfirmed using the same cell line resourced from a cell line repository or originating laboratory.
Footnotes
Acknowledgements
We would like to thank NCATS summer interns Shelley Austin, Madison Gamma, and Aster Samuel for contributing to Mycoplasama analyses. The authors prepared this work within the scope of their employment with the National Center for Advancing Translational Sciences, National Institutes of Health.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the intramural research program of the National Center for Advancing Translational Sciences (NCATS).

