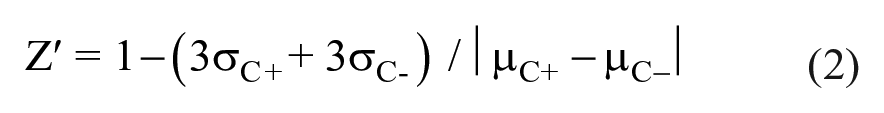Abstract
While G-protein-coupled receptors (GPCRs) represent the largest class of cell surface proteins, there are ≥100 orphan GPCRs whose endogenous ligands are unknown. Accordingly, these could prove to be potential therapeutic targets for the pharmaceutical intervention of various diseases. Constitutively active orphan GPCRs are activated without ligands; thus, inverse agonists may be very useful pharmacological tools for inhibiting constitutive activity. However, in general, inverse agonist screening is considered more difficult to perform with high quality than antagonist screening, particularly due to the narrow assay window. We developed a high-throughput screening (HTS)-compatible assay to identify inverse agonists of GPR3. GPR3 is expressed in the central nervous system (CNS) and is known to be related to Alzheimer’s disease and other CNS diseases. The GPR3 inducible cell line was established using T-REx 293 cells that stably expressed the tetracycline repressor protein, and the cAMP biosensor, GloSensor, was stably co-expressed. After optimization of the induction level of GPR3 and assay conditions, the GloSensor assay showed an approximately 20-fold signal-to-background ratio and high sensitivity. Using the HTS method, we successfully screened a library of hundreds of thousands of compounds for the inhibition of constitutive activity with good quality and excellent reproducibility. Finally, 35 compounds were identified as GPR3 selective inverse agonists. This inverse agonist screening approach using GloSensor in combination with the inducible expression of orphan GPCR indicates universal applicability to the search for inverse agonists of constitutively active orphan GPCRs.
Introduction
G-protein-coupled receptors (GPCRs) represent a large membrane protein family, encoding as many as 800 genes in the human genome.1,2 GPCRs regulate a wide variety of physiological functions, including those related to vision, heart rate, neurotransmission, and the immune system, through the binding of each ligand. 3 Indeed, the targets of approximately 30% of all Food and Drug Administration (FDA)-approved drugs involve the GPCR family, highlighting the fact that GPCRs are attractive targets for drug discovery. 4 Therefore, it is important to facilitate the functional analysis of those GPCRs that remain uncharacterized.
Traditionally, both academia and the pharmaceutical industry have attempted to screen natural or synthetic ligands for GPCRs, followed by the characterization of the receptors pharmacologically using these ligands. Nevertheless, approximately 150 GPCRs are still considered orphan receptors whose endogenous ligands have not yet been identified. 5 On the other hand, Costa and Herz have proposed the concept of constitutively active GPCRs by demonstrating that delta opioid receptors have an intrinsic activity in the absence of an agonist. 6 Some orphan GPCRs may exert their physiological function through constitutive activity without any endogenous ligand present. Recently, it has been demonstrated that GPR21, a constitutively active orphan GPCR, contributes to the development of insulin resistance and energy homeostasis. 7 This may be one example of a (patho)physiological function of an orphan GPCR without the presence of an endogenous ligand. Furthermore, it has been reported that increased constitutive activity caused by the overexpression or mutation of constitutively active GPCRs is associated with various pathologies. In fact, mutations linked to increased constitutive activity in the rhodopsin gene and in the thyrotropin receptor gene are known to cause retinitis pigmentosa 8 and hyperthyroidism, 9 respectively.
Inverse agonists, but not neutral antagonists, can inhibit the agonist-independent activities of these constitutively active GPCRs. For example, histamine H2 receptor ligands, cimetidine, ranitidine, and famotidine have been used as therapeutic drugs for duodenal or gastric ulcers and are categorized as neutral antagonists. 10 However, several studies have reported that the histamine H2 receptor possesses constitutive activity, and these compounds have currently been recognized as inverse agonists.11,12 These inverse agonists can convert constitutively active receptors into an inactive state. 13 Thus, inverse agonists may be beneficial tools for the detailed (patho)physiological analyses of constitutively active orphan GPCRs where there is very little known regarding their function. Ultimately, inverse agonists may have the potential to be therapeutic drugs.
Screening for inverse agonists that inhibit the function of constitutively active GPCRs is typically performed without agonists, whereas agonists are necessary for the screening of antagonists that inhibit GPCR responses elicited by agonists. 14 In general, inverse agonist screening is more difficult to carry out with high quality than antagonist screening since constitutive activity in the absence of an agonist is often lower than the agonist response. Therefore, the utilization of artificially mutated receptors is one strategy to augment the constitutive activity for screening. 14 However, the compounds obtained from the screening using artificially mutated GPCRs may not necessarily exhibit inverse agonist activity in native GPCRs. Thus, it is important to evaluate constitutive activity with high sensitivity in native GPCRs. GloSensor is a novel technology based on a luciferase-based biosensor that can detect real-time cAMP levels in live cells with high sensitivity. 15 Therefore, this methodology may be suitable for the screening of inverse agonists that inhibit constitutive activity. However, earlier studies on screening using GloSensor were limited to small-scale screening.16–18 To our knowledge, no comprehensive study has been described regarding a large-scale screening of inverse agonists.
Transfection with small amounts of GPR3 expression plasmid DNA elicited a significant increase in intracellular cAMP levels. 19 Furthermore, knockdown of endogenous GPR3 inhibited its functions, including neurite outgrowth in neurons. 20 Thus, GPR3 has been recognized as a constitutively active Gs-coupled orphan GPCR that results in elevated intracellular cAMP levels in a wide variety of cells;19–21 however, we cannot completely exclude the possibility that unidentified endogenous agonists activate GPR3. GPR3 is highly expressed in the central nervous system (CNS), particularly in the hippocampus, hypothalamus, cerebral cortex, and cerebellum.20,22 Recent studies have suggested the involvement of GPR3 in neurological disorders, such as Alzheimer’s disease, 23 anxiety, and depression, based on studies using GPR3 knockout mice. 24 In particular, GPR3 inverse agonists may prove to be therapeutic drugs for Alzheimer’s disease since GPR3 facilitates amyloid-beta production. However, very few GPR3 selective inverse agonists have been reported. 25
In this report, we demonstrated that the GloSensor assay is applicable for a large-scale HTS campaign to identify inverse agonists of GPR3. The receptor activity of constitutively active GPCRs is correlated with its gene expression. Thus, in this study, an inducible expression system was utilized to develop and verify the assay systems for the GPR3 GloSensor assay and the counterassay. To our knowledge, this is the first comprehensive report of a large-scale HTS campaign for inverse agonists toward constitutively active orphan GPCRs using GloSensor in combination with an inducible expression system. Moreover, multiple GPR3 inverse agonists were identified through this HTS campaign.
Materials and Methods
Cells
T-REx 293 cells stably expressing the tetracycline repressor protein (Thermo Fisher Scientific, Waltham, MA) were used to develop a stable cell line expressing human GPR3 (T-REx-293/hGPR3). The pT-REx-DEST30/hGPR3 plasmid was constructed by recombining hGPR3 complementary DNA with an amino-terminal HA tag cloned in the pENTR/D-TOPO vector into the pT-REx-DEST30 (mammalian expression vector with the cytomegalovirus (CMV) promoter/enhancer and a tetracycline repressor operator sequence) using LR clonase II (Thermo Fisher Scientific). T-REx 293 cells were then transfected with the pT-REx-DEST30/hGPR3 plasmid using Lipofectamine 2000 reagent (Thermo Fisher Scientific) and further selected in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum, 100 units/mL penicillin, 100 units/mL streptomycin, 5 μg/mL blasticidin S, and 250 μg/mL G-418 at 37 °C with 5% CO2 in a humidified atmosphere. Cell clones resistant to G-418 were isolated and selected by their ability to induce hGPR3 expression following the addition of tetracycline. The selected cell line, T-REx-293/hGPR3, was then transfected with the pGloSensor-22F cAMP plasmid (Promega Corporation, Fitchburg, WI) and cultured in the above-described medium supplemented with 300 μg/mL hygromycin B. Resistant clones were isolated and selected based on luminescence increased by 3-isobutyl-1-methylxanthine (IBMX) in the GloSensor assay following tetracycline addition. The selected clone was referred to as T-REx-293/hGPR3-GS22F.
HEK 293 cells (American Type Culture Collection, Manassas, VA) were used to develop a stable cell line expressing the human melanocortin 4 receptor (hMC4R) and GloSensor 20F (HEK293/hMC4R-GS20F). The pcDNA3.1(+)-hMC4R plasmid was constructed by ligating hMC4R complementary DNA into the multiple cloning sites of the mammalian expression vector pcDNA3.1(+) (Thermo Fisher Scientific). HEK 293 cells were transfected with the pcDNA3.1(+)-hMC4R and pGloSensor-20F cAMP plasmids (Promega Corporation), transfected using Lipofectamine 2000 reagent, and selected in DMEM supplemented with 10% fetal bovine serum, 100 units/mL penicillin, 100 units/mL streptomycin, 500 μg/mL G-418, and 300 μg/mL hygromycin B at 37 °C with 5% CO2 in a humidified atmosphere. Resistant clones were isolated and selected by constitutive activity and responsiveness to an hMC4R inverse agonist, agouti-related protein (AGRP; Peptide Institute Inc., Osaka, Japan), in the GloSensor assay. The selected clone was referred to as HEK293/hMC4R-GS20F.
In the present study, cryopreserved cell stocks were prepared for the HTS campaign. T-REx293/hGPR3-GS22F cells were pretreated overnight with 0.3 μg/mL tetracycline and collected using phosphate-buffered saline (PBS)-based Cell Dissociation Buffer (Thermo Fisher Scientific). Collected cells were suspended in a serum-free cell cryopreservation medium, CELLBANKER 2 (Nippon Zenyaku Kogyo Co. Ltd., Koriyama, Japan) and stored at −80 °C for the hGPR3 GloSensor assay. T-REx293/hGPR3-GS22F cells without pretreatment of tetracycline and HEK293/hMC4R-GS20F cells were cryopreserved in the same manner for the counterassay and the hMC4R GloSensor assay, respectively.
GloSensor cAMP Assay Using Cultured Cells
T-REx-293/hGPR3-GS22F cells were seeded at 5000 cells/well in a 384-well white-well/clear-bottom plate (Corning Inc., Corning, NY) and cultured overnight in the presence of 0.1 μg/mL tetracycline unless stated otherwise, at 37 °C with 5% CO2 in a humidified atmosphere, in order to induce hGPR3 expression. The culture media were then removed and replaced with 20 μL of CO2-independent medium (Thermo Fisher Scientific) containing 10% fetal bovine serum and 2% GloSensor cAMP Stock Reagent (Promega Corporation). The cells were then incubated for 2–3 h at room temperature under light-shielded conditions. The luminescence of each well was determined at 30 min after the addition of the test compound solution (5 μL) with a SpectraMax Paradigm Multi-Mode Microplate Reader (Molecular Devices, San Jose, CA) unless stated otherwise.
GloSensor cAMP Assay Using Cryopreserved Cells
Cryopreserved cells were thawed at 37 °C and suspended in CO2-independent medium containing 10% fetal bovine serum and 2% GloSensor cAMP Stock Reagent. The cells were dispensed at 5000 cells/20 μL/well in white, tissue-cultured, 384-well plates with MultiDrop Combi (Thermo Fisher Scientific). Cells were incubated for more than 3 h at room temperature under light-shielded conditions. The luminescence of each well was determined at 30 min after the addition of the test compound solution (5 μL) using an EnVision 2104 Multimode Plate Reader (PerkinElmer, Waltham, MA) unless stated otherwise.
HTS Campaign
Primary screening for human GPR3 inverse agonists was conducted using cryopreserved stocks of T-REx-293/hGPR3-GS22F pretreated with 0.3 μg/mL tetracycline, as described above. Briefly, 5 μL of the compound solution was added to 20 μL of cell suspension using EDR-384UX (Biotech Co. Ltd., Tokyo, Japan). We evaluated the inhibitory effect of a test compound at 10 μM on the constitutive activity of hGPR3 in the presence of 0.5 mM IBMX. Luminescence was measured at 60 min after the addition of the compound solution using an EnVision 2104 Multimode Plate Reader. The inverse agonist activity of each test compound was expressed as inhibitory activity toward the cAMP response (percent inhibition). Luminescent values in the presence and absence of 0.5 mM IBMX were defined as 0% and 100% inhibition, respectively, in the GloSensor assay for hGPR3. The cryopreserved stocks of T-REx-293/hGPR3-GS22F without pretreatment of tetracycline were used for the counterassay. We evaluated the inhibitory effect of a test compound at 10 μM on the cAMP response stimulated by 0.5 μM forskolin in T-REx-293/hGPR3-GS22F without induction of hGPR3 expression in order to exclude nonspecific compounds. Luminescent values in the presence of 0.5 mM IBMX with 0.5 μM forskolin and 0.5 mM IBMX without forskolin were defined as 0% and 100% inhibition, respectively, in the counterassay.
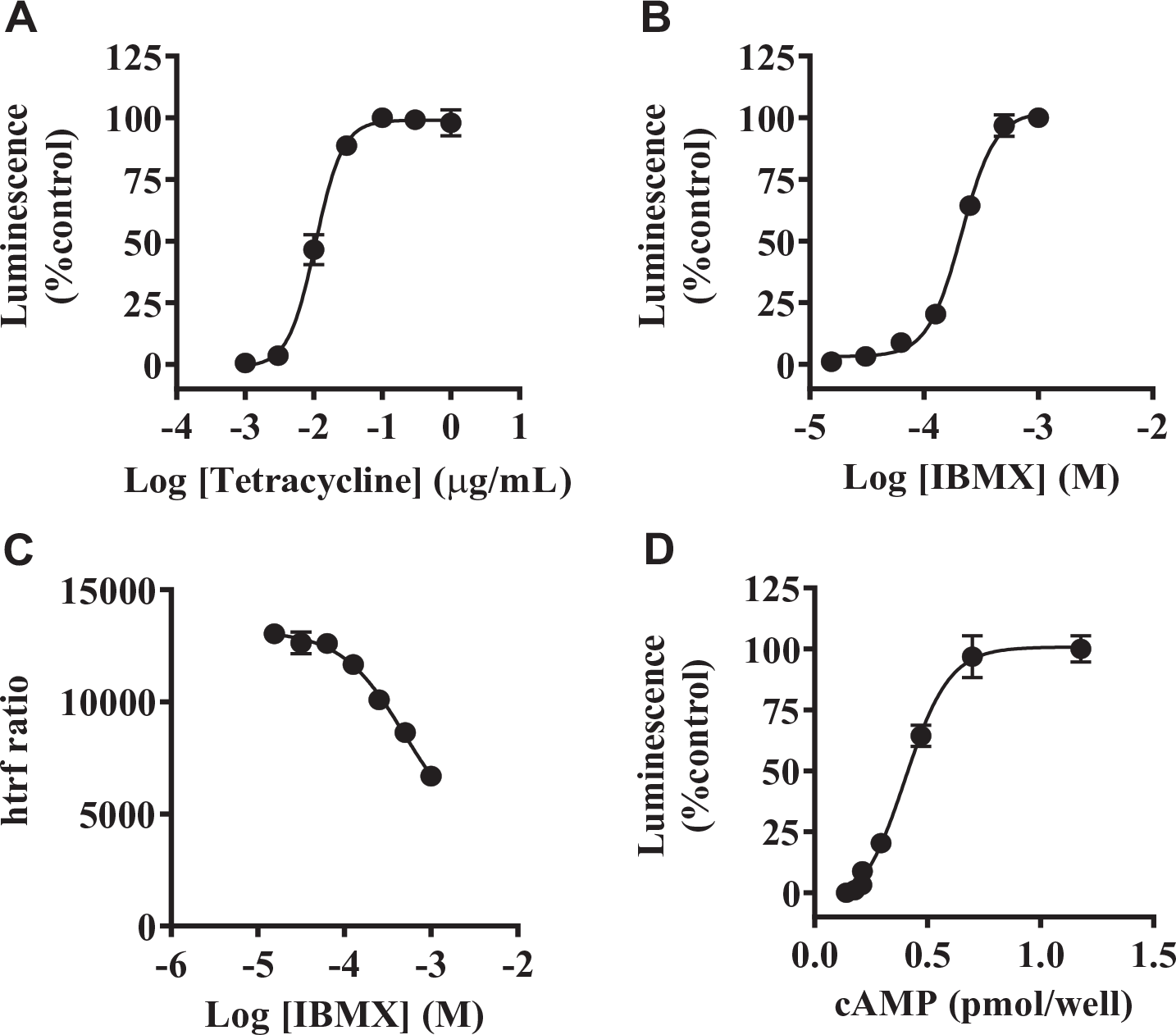
Concentration–response curves for both hGPR3 and hMC4R were generated using the cryopreserved stocks of T-REx-293/hGPR3-GS22F and HEK293/hMC4R-GS20F with different concentrations of test compounds in the same manner as the primary screen. Luminescent values in the presence of 0.5 mM IBMX and 0.5 mM IBMX with 1 μM AGRP, which has been reported as a full inverse agonist for hMC4R,26,27 were defined as 0% and 100% inhibition, respectively, in the GloSensor assay for hMC4R.
Homogeneous Time-Resolved Fluorescence cAMP Assay Using Cultured Cells
T-REx-293/hGPR3-GS22F cells were seeded and cultured overnight in the presence of 0.1 μg/mL tetracycline in the same manner as those for the GloSensor assay using cultured cells. cAMP levels were determined with a cAMP HiRange Kit (Cisbio Bioassays, Codolet, France) according to the manufacturer’s protocol. Briefly, the culture media were replaced with Hanks’ Balanced Salt Solution containing 20 mM HEPES (pH 7.4), cAMP-d2, and test compound, and incubated at room temperature for 30 min. Lysis buffer containing anti-cAMP cryptate was added to each well and incubated for 1 h. The homogeneous time-resolved fluorescence (HTRF) ratio was measured for fluorescence emission at 616 nm for the donor and at 665 nm for the acceptor using a SpectraMax Paradigm Multi-Mode Microplate Reader.
Data Analyses and Statistics
Data analyses were performed using GraphPad Prism (version 8.1.1; GraphPad Software, San Diego, CA) and TIBCO Spotfire Analyst (version 7.11.1; TIBCO, Palo Alto, CA). Concentration–response curves and standard curves for cAMP were fitted using nonlinear regression analysis using the following equation:
The Z′ factor, 28 the index for assay quality assessment in assay development and optimization, was determined using the following equation:
where µC+ and µC– are the means of positive control signal (0.5 mM IBMX) and negative control signal (DMSO), respectively, and σC+ and σC– are the standard deviations of the corresponding signals. For the counterassay, positive and negative controls are defined as 0.5 mM IBMX with 0.5 μM forskolin and 0.5 mM IBMX without forskolin, respectively.
Results
GPR3 is known as a constitutively active Gs-coupled orphan GPCR. Thus, the expression of GPR3 without an agonist could stimulate adenylate cyclase via Gs, resulting in increased intracellular cAMP levels.19–21 T-REx-293/hGPR3-GS22F is a cell line stably expressing GloSensor 22F and it inducibly expresses hGPR3 under the control of the CMV promoter/enhancer with a tetracycline repressor operator sequence. This cell line was used to evaluate the usefulness of the GloSensor assay in the HTS for GPR3 inverse agonists. Induction of hGPR3 expression in T-REx-293/hGPR3-GS22F cells with pretreatment of tetracycline increased the luminescence in the presence of 1 mM IBMX in the GloSensor assay, suggesting that hGPR3 expression elicits a cAMP response without the presence of an agonist, which is consistent with previous studies (
GloSensor cAMP assay using T-REx-293/hGPR3-GS22F. (
We evaluated the relationship between intracellular cAMP concentrations and luminescence of the GloSensor assay using the conventional HTRF assay. To this end, the GloSensor assay and the HTRF assay were conducted at the same time, and intracellular cAMP levels in the cells were determined in the presence of different concentrations of IBMX. IBMX increased luminescence in the GloSensor assay using T-REx-293/hGPR3-GS22F cells pretreated with 0.1 µg/mL tetracycline (
T-REx-293/hGPR3-GS22F cells were cryopreserved after pretreatment with tetracycline. Cryopreserved cells were thawed and used for the GloSensor assay immediately after thawing without cell culture. In the GloSensor assay using cryopreserved T-REx-293/hGPR3-GS22F cells, IBMX increased intracellular cAMP levels in a concentration-dependent manner (
GloSensor cAMP assay using cryopreserved cells. (
In the present study, we defined GPR3 inverse agonist activity as inhibitory activity of test compounds on the increase in intracellular cAMP levels in the presence of 0.5 mM IBMX in T-REx-293/hGPR3-GS22F cells with the induction of hGPR3 expression. The counterassay was designed to discriminate between compounds with inverse agonist activity toward hGPR3 and nonspecific compounds reducing intracellular cAMP levels regardless of the constitutive activity of hGPR3. For the counterassay, the forskolin-stimulated cAMP response was evaluated in T-REx-293/hGPR3-GS22F cells without induction of hGPR3 expression in the same assay format. Forskolin in the presence of 0.5 mM IBMX increased intracellular cAMP levels in the GloSensor assay using cryopreserved T-REx-293/hGPR3-GS22F cells without induction of hGPR3 expression (
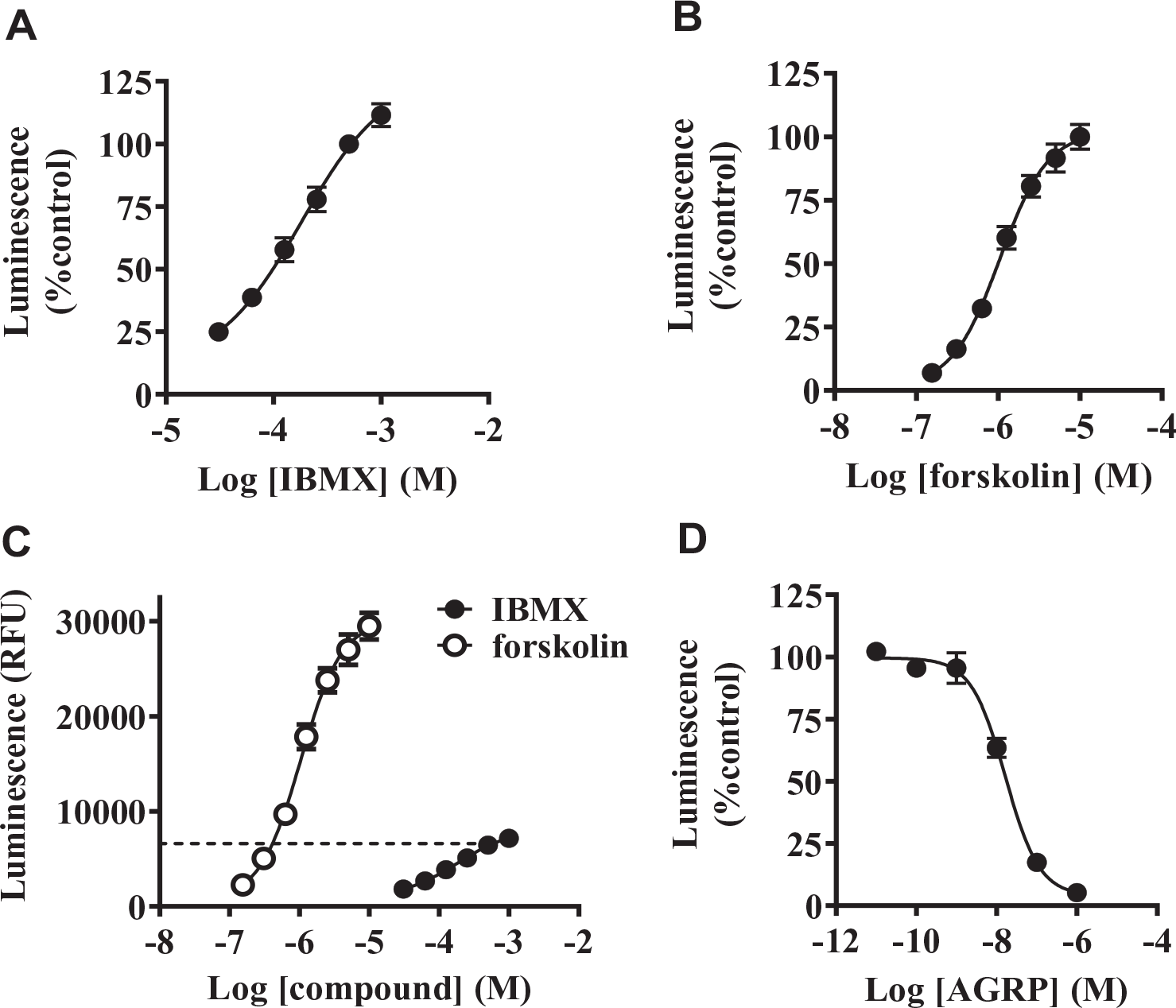
Luminescence in the hGPR3 GloSensor assay was sequentially monitored until 300 min after the addition of IBMX (
Time course of luminescence in the hGPR3 GloSensor cAMP assay. (
Before the initiation of a whole HTS campaign, we carried out a pilot screen for the 4218 test compounds at final concentrations of 10 µM with 0.5% DMSO. In the pilot screen, the hGPR3 GloSensor assay and the counterassay were conducted in triplicate. Z′ factors in the hGPR3 GloSensor assay and the counterassay were 0.84 ± 0.04 and 0.85 ± 0.03 (mean ± SD), respectively. The S/B ratios in the hGPR3 GloSensor assay and the counterassay were 25.0 ± 2.2 and 37.6 ± 2.4 (mean ± SD), respectively. hGPR3 inverse agonist activity of the tested compounds was expressed as the percent inhibition in the hGPR3 GloSensor assay. The activity of the compounds was calculated as the mean percent inhibition of triplicates in the hGPR3 GloSensor assay and in the counterassay, respectively. The calculated mean + 3 SD of the percent inhibition in the hGPR3 GloSensor assay among all tested compounds was 27.8% (
In the whole HTS campaign, more than 300,000 compounds were tested at a final concentration of 10 µM with 0.5% DMSO for the hGPR3 GloSensor assay. About 900 plates were evaluated in 10 days. The Z′ factor was 0.84 ± 0.04 (mean ± SD) with an S/B ratio of 19.7 ± 2.7 (mean ± SD) (
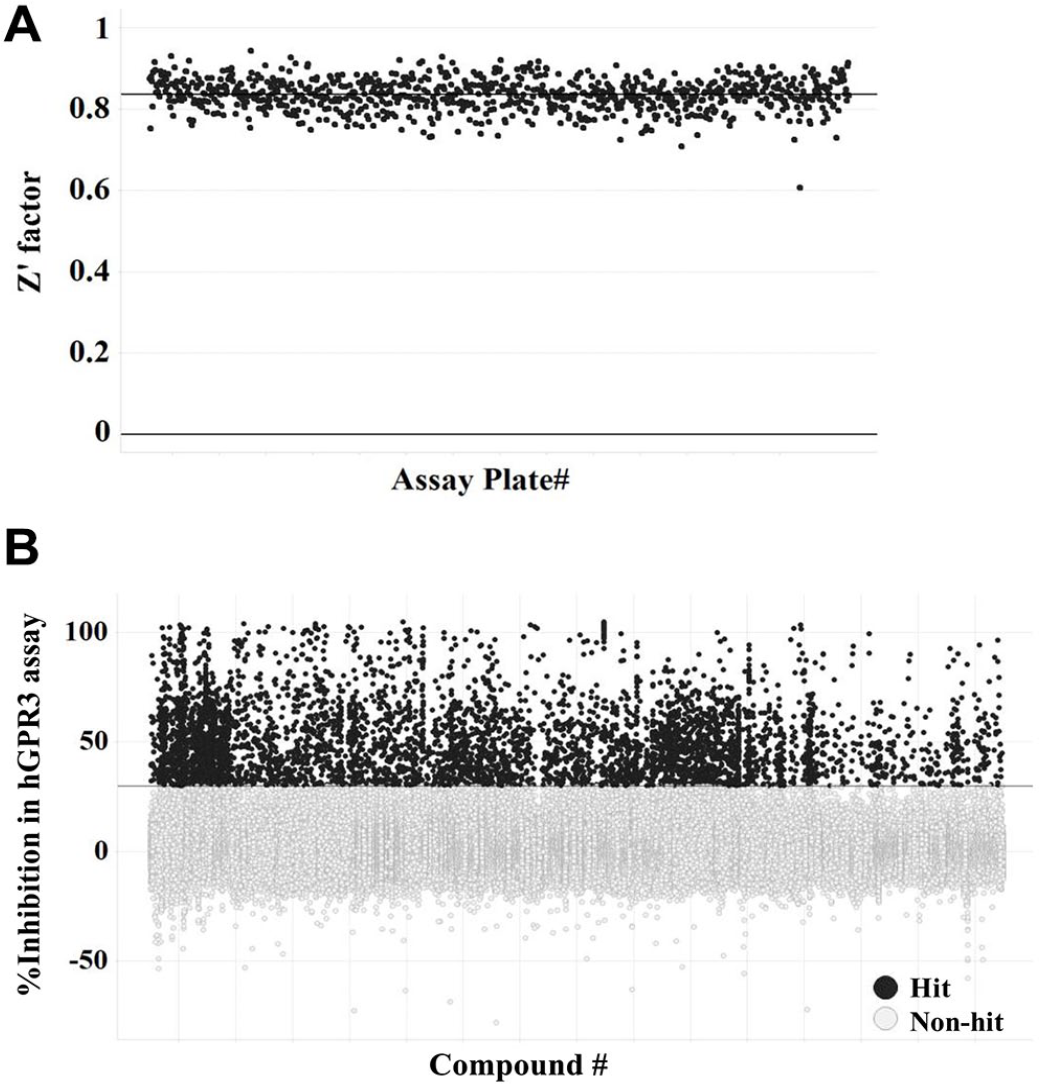
Results of the primary screen. More than 300,000 compounds were screened at the final concentration of 10 μM using the hGPR3 GloSensor assay. (
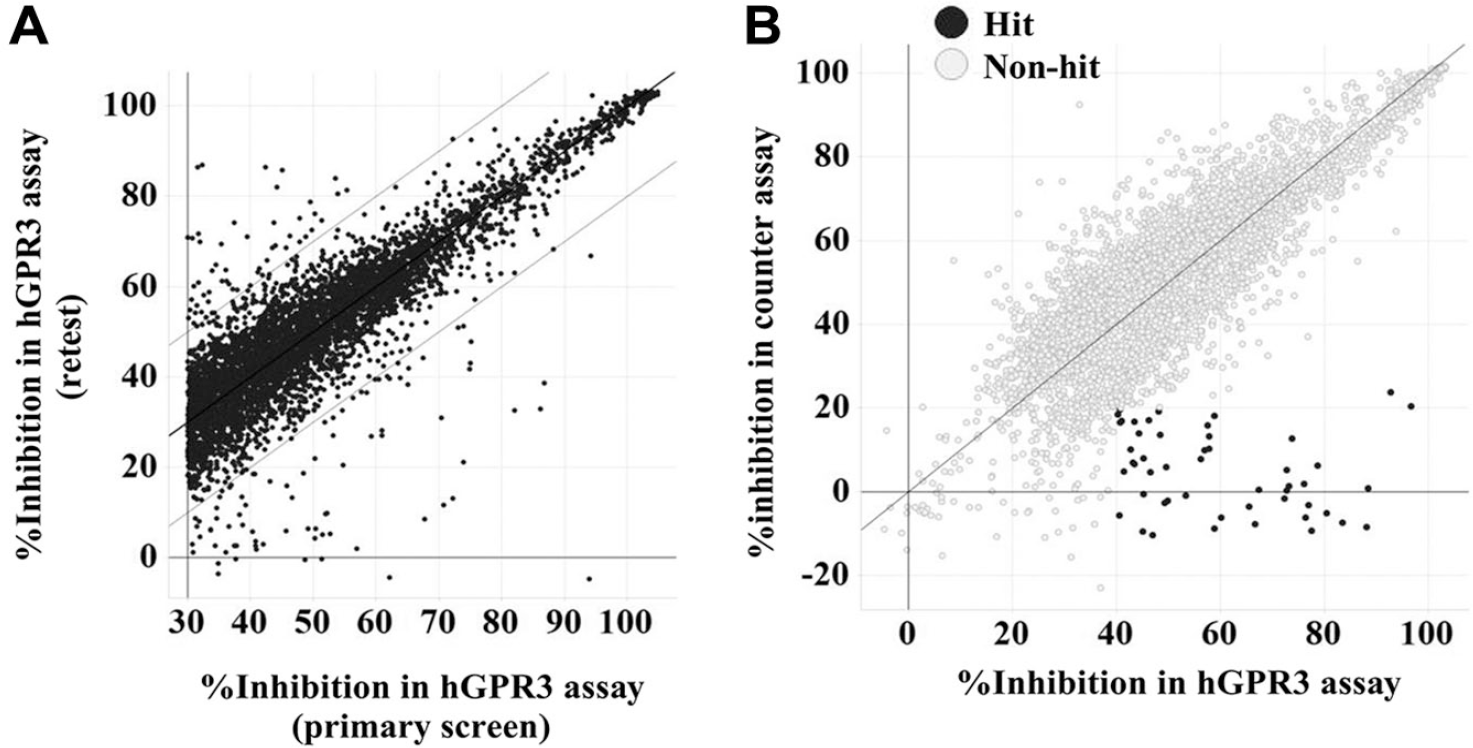
Results of the confirmation assay. Compounds (5124) were tested at a final concentration of 10 μM using the hGPR3 GloSensor assay and the counterassay. (
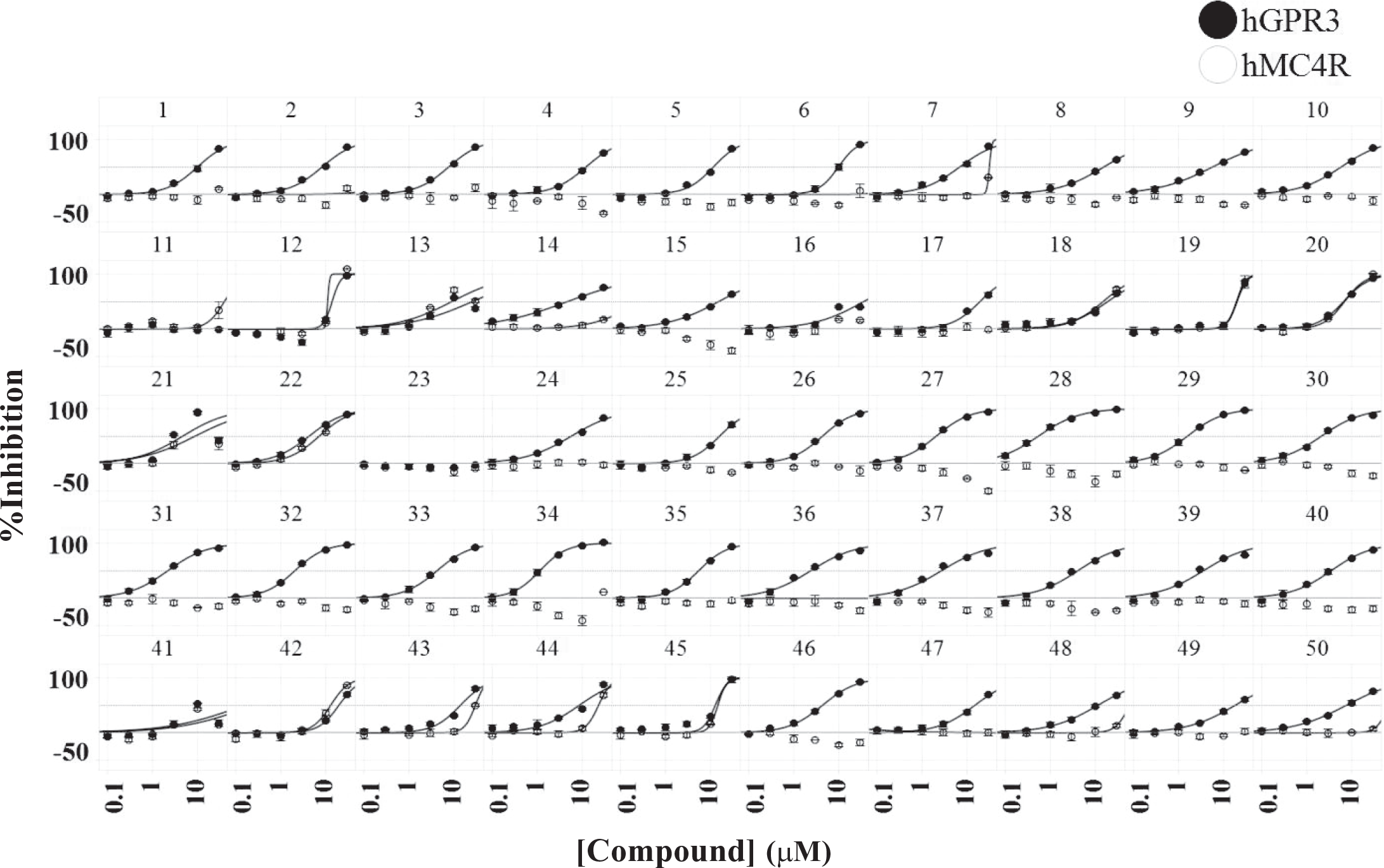
Concentration–response curves of 50 selected compounds. The hGPR3 GloSensor assay and the hMC4R GloSensor assay were conducted in the presence of different concentrations ranging from 0.1 to 30 µM. Closed circles and open circles represent percent inhibition in the hGPR3 GloSensor assay and percent inhibition in the hMC4R GloSensor assay, respectively. Data are means ± SD of quadruplicate experiments.
Discussion
cAMP is an important second messenger molecule produced by adenylyl cyclase that is mainly regulated via activation of Gs and Gi proteins. 29 Several methods for quantifying intracellular cAMP levels have been developed, including the radioimmunoassay (RIA), enzyme-linked immunosorbent assay (ELISA), scintillation proximity assay (SPA), cAMP response element-luciferase reporter assay (CRE-Luc assay), and time-resolved fluorescence resonance energy transfer (TR-FRET) assay, such as HTRF.30–35 Among these, RIA has historically been used but needs a radioisotope, and ELISA is a well-established method as an alternative of RIA; however, both are considered low-throughput assays because of the laborious steps involved, such as multiple washes. SPA technology is based on a homogeneous assay without wash steps prior to detection, but this method is relatively expensive for screening a large number of compounds and still requires a radioisotope. The CRE-Luc assay is relatively easy to set up and allows for the conduction of cell-based HTS. However, this assay is characterized by high false-positive hit rates since it can detect compounds interfering with the downstream components of the cAMP pathway and gene expression. Furthermore, longer incubations of the compounds in the assay tend to detect compounds with cellular toxicity as false positives. On the other hand, the features of HTRF include a simple mix-and-measure protocol, and it is a safer nonradioisotope method in comparison with RIA, ELISA, and SPA. In addition, it provides more direct detection of cAMP and shorter compound incubation in comparison with the CRE-Luc assay. For these reasons, a TR-FRET assay, such as HTRF, is currently widely used in HTS. 36
GloSensor is a relatively new luciferase-based biosensor that detects intracellular cAMP levels and can easily measure the kinetics of live cells with a wide dynamic range.
15
These characteristics provide many advantages over other methods for measuring intracellular cAMP levels. In fact, we monitored luminescence sequentially until 300 min and were able to easily find an optimum condition for evaluating compounds (
The activity of the constitutively active orphan GPCR such as GPR3 correlates with the expression level on the cell surface.
38
The T-REx system was utilized as an inducible expression system in the present study. The inducible expression system was useful for the development and validation of the assay systems for GPR3 since activity can be easily modulated by the simple addition of tetracycline without GPCR ligands (
Among the 35 compounds with selectivity against hMC4R, 18 compounds were found to be derivatives of triazolopyrimidine that have already been reported as a GPR3 inverse agonist (
In conclusion, we demonstrated the feasibility of the GloSensor in large-scale screening for inverse agonists of constitutively active orphan GPCR in combination with an inducible expression system. The GloSensor assay can be set up easily and fast with high sensitivity, large assay windows, and remarkable reproducibility. Thus, it could facilitate the screening for constitutively active GPCR screening and provide an opportunity for further understanding of lesser known GPCRs.
Supplemental Material
DS_DISC875101 – Supplemental material for Development of a High-Throughput Screening-Compatible Assay for Discovery of GPR3 Inverse Agonists Using a cAMP Biosensor
Supplemental material, DS_DISC875101 for Development of a High-Throughput Screening-Compatible Assay for Discovery of GPR3 Inverse Agonists Using a cAMP Biosensor by Kumiko Ayukawa, Chie Suzuki, Hiroyuki Ogasawara, Tomomi Kinoshita, Masahiro Furuno and Gentaroh Suzuki in SLAS Discovery
Footnotes
Acknowledgements
We would like to thank Dr. Makoto Tsuda (professor in the Department of Life Innovation, Graduate School of Pharmaceutical Sciences, Kyushu University) for helpful discussion and advice.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.