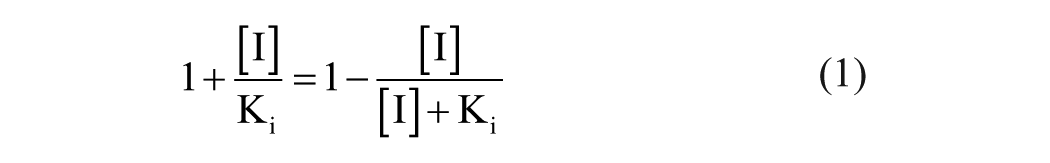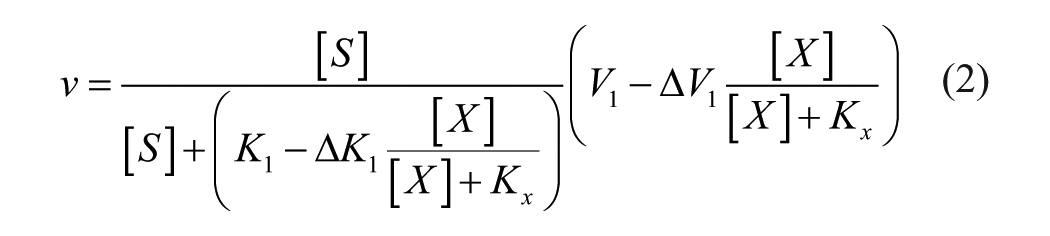Abstract

Having come across this article, 1 I was quite disappointed in its one-sided biased endorsement of classical inhibition models.
Whereas it may be inconceivable to many biochemists practicing in the field today that there is controversy surrounding the classical models of enzyme inhibition, one only needs to look at the propagation of subsequent inhibition models over the years to realize that the classical way of modeling segregates interactions into very strict predefined limitations. For example, traditional competitive inhibitors only decrease substrate affinity by linearly increasing the value of the KM with increasing inhibitor concentration. However, a mathematical model does not indicate mechanism; rather, it provides support for a hypothesis, which is why you may have allosteric effects that present as competitive, as outlined by the authors. Given that these equations do not really define specific interactions, there should be no point in advocating their use if there is a single equation that can model the data as well as or, in most cases, better than they can. The omission of this point from the article greatly reduces the overall usefulness of a review.
By recognizing that the apparent inhibition term in the classical inhibition equations is an inversion of the inhibitor binding isotherm (eq 1), 2 one can directly relate changes in enzymatic activity to the fraction of the enzymatic population bound. Consequently, changes in enzymatic activity can be described through observation rather than strictly defined limits imposed by the classical equations (eq 2). 3
This equation has been tested against the classical equations with real data and has been found to allow for an equivalent or improved fit in all cases.3–5 The flexibility of this approach also allows the equation to be used to describe activators in addition to inhibitors. Any researchers can also quickly and easily test this approach with their own data and evaluate the fit against the classical models using a freely available Excel template. 4 Therefore, it is a disservice to the research community for the authors to recommend constraining mechanistic studies to classical inhibition equations with clear mathematical limitations based on mechanistic models the authors concede are not valid.