Abstract
The vascular endothelium plays a critical role in the health and disease of the cardiovascular system. Importantly, biomechanical stimuli generated by blood flow and sensed by the endothelium constitute important local inputs that are translated into transcriptional programs and functional endothelial phenotypes. Pulsatile, laminar flow, characteristic of regions in the vasculature that are resistant to atherosclerosis, evokes an atheroprotective endothelial phenotype. This atheroprotective phenotype is integrated by the transcription factor Kruppel-like factor-2 (KLF2), and therefore the expression of KLF2 can be used as a proxy for endothelial atheroprotection. Here, we report the generation and characterization of a cellular KLF2 reporter system, based on green fluorescence protein (GFP) expression driven by the human KLF2 promoter. This reporter is induced selectively by an atheroprotective shear stress waveform in human endothelial cells, is regulated by endogenous signaling events, and is activated by the pharmacological inducer of KLF2, simvastatin, in a dose-dependent manner. This reporter system can now be used to probe KLF2 signaling and for the discovery of a novel chemical-biological space capable of acting as the “pharmacomimetics of atheroprotective flow” on the vascular endothelium.
Introduction
The vascular endothelium, located at the interface of blood and the vessel wall, plays a critical role in maintaining homeostasis of the cardiovascular system. 1 Importantly, endothelial cells acquire functional phenotypes in response to a variety of humoral and biomechanical stimuli. For example, pulsatile blood flow generates a range of hemodynamic forces through the vasculature that act as local modifiers of endothelial cell structure and function.2,3 In particular, arterial branch points and curvatures are characterized by the presence of disturbed flow patterns, consisting of a high oscillatory index, and low wall shear stress. These areas are highly susceptible to the development of atherosclerotic lesions. In contrast, straight segments of the vasculature are exposed to pulsatile laminar shear stress and are resistant to atherosclerotic lesion development.2,4 –7 Previous work in our laboratory defined two prototypic arterial waveforms, atheroprone and atheroprotective, that represent the wall shear stresses characteristic of regions that are susceptible or resistant, respectively, to atherosclerosis in the human carotid system. Importantly, when these shear stress waveforms are applied to cultured human endothelial cells, they evoke distinct patterns of gene expression leading to an atheroprone or atheroprotective endothelial cell phenotype. Among the differentially expressed genes, selectively upregulated in endothelial cells exposed to the atheroprotective shear stress waveform, is the transcription factor Kruppel-like factor-2 (KLF2). 8 Several studies have established KLF2 as a critical transcriptional integrator of endothelial mechano-activated programs8,9 and have documented a critical role in endothelial homeostasis 10 and atheroprotection. 11 Collectively, these findings support the concept of endothelial expression of KLF2 being used as a proxy for flow-dependent endothelial atheroprotection.
Considering the central role that biomechanical forces have for endothelial atheroprotection, experimental tools are needed to monitor the expression of mechano-activated genes and signaling pathways. Ideally, these tools should be scalable and can be deployed in combination with high-throughput technologies. Recent efforts have been directed toward developing cell-based molecular sensors of fluid shear stress. One such approach exploited a shear stress response element in the promoter region of a transcription factor, early growth response protein-1 (EGR1). Red fluorescent protein (RFP) was placed under the control of a portion of the EGR1 promoter in fibroblasts, which responded to various shear stress levels. 12 Others used a modified form of the mechanosensitive G-protein-coupled receptor (GPCR) bradykinin receptor B2, which activated enhanced green fluorescence protein (eGFP) transcription through a linker module, accurately reporting shear stress magnitude. 13
Given the unique atheroprotective flow-dependent expression of KLF2 and its function as a transcriptional integrator of endothelial atheroprotection, a KLF2 reporter is uniquely suited to function as a molecular sensor of not only atheroprotective flow but also, more broadly, an atheroprotective endothelial phenotype. Here, we report the generation and characterization of a cellular KLF2 reporter system based on green fluorescence protein (GFP) expression driven by the proximal human KLF2 promoter. This reporter system constitutes a novel tool that can be used in high-throughput screening (HTS) to dissect signaling pathways regulating KLF2 expression and to identify novel pharmacological regulators of KLF2 expression.
Materials and Methods
Generation of the KLF2 Reporter
A ~1.2 kb region of the proximal KLF2 promoter spanning roughly −1.1 kb to +50 bp, including the TATA box, was amplified from human genomic DNA with the following primers incorporating XhoI and HindIII sites at the 5′ and 3′ ends, respectively: (1) CGCTCGAGCAGCCACCT and (2) CCAAGCTTACGGGGACA. The Xho1–HindIII PCR product was phosphatase-treated and blunt-end ligated into the PCR–Blunt II–TOPO vector (Invitrogen, Carlsbad, CA) so that the promoter insert is flanked by EcoRI sites on both ends, resulting in pKPROM3. pTRH1–mCMV–dscGFP (System Biosciences, Mountain View, CA) is a lentiviral expression construct encoding a GFP variant driven by the minimal cytomegalovirus (mCMV) promoter. To obtain our final lentiviral reporter construct, pTRH1–KLF2–dscGFP, the mCMV promoter of pTRH–mCMV–dscGFP was replaced with the EcoRI insert from pKPROM3. Lentivirus was produced in HEK293FT cells, then titered using a QuickTiter Lentivirus Titer Kit (Cell Biolabs, San Diego, CA). Human umbilical vein endothelial cells (HUVEC) were transduced with the lentiviral construct at a multiplicity of infection (MOI) of 2.5 for 24 h. Cells were passaged once, and approximately 2.4 × 107 cells were used for sorting. Cells were trypsinized, washed with phosphate-buffered saline (PBS), and resuspended in fluorescence-activated cell sorter (FACS) buffer (PBS containing 5% fetal bovine serum [FBS] and 2 mM EDTA). Using a FACS Aria sorter (BD Biosciences, San Jose, CA), GFP negative (GFP−) and GFP high expressers (GFPhigh) were excluded, whereas GFP low expressers (GFPlow) were sorted (KLF2–GFP endothelial cells [EC]). Sorted cells were kept in culture until used for subsequent experiments.
Cell Culture and Flow Experiments
HUVEC were isolated and cultured at our core facility as previously described. 14 HUVEC were cultured in endothelial growth medium EGM-2 (CC-3162, Lonza, Walkersville, MD). For flow experiments, KLF2–GFP EC were seeded on 0.1% gelatin-coated (gelatin from BD, Sparks, MD) plastic disks (custom-made product, QC Precision Machining, Salem, NH) at a density of 6 × 104 cells/cm2 (1.7 × 105 cells/well) and maintained at 37 °C, 5% CO2 for 24 h. After 24 h, medium was replaced with 7.5 mL of flow medium (EGM-2 medium with 1.7% dextran [D5376-100G, Sigma, St. Louis, MO]). After 1 h, KLF2–GFP EC were exposed to atheroprotective or atheroprone waveforms using our dynamic flow system15,16 for 48 h. The system is composed of a cone and plate device, it is equipped with a computer-controlled user interface that generates shear stress, and the system maintains cultured cells at 37 °C, 5% CO2. For simvastatin experiments, KLF2–GFP EC were seeded in 0.1% gelatin-coated wells of a six-well plate at a density of 6 × 104 cells/cm2 (5.7 × 105 cells/well) in 2 mL of EGM-2 medium. After 24 h, cells were treated with simvastatin (567021, EMD Millipore, Billerica, MA) at concentrations of 1 µM and 5 µM for 36 h. For MEK5 inhibitor experiments, BIX 0218917 (4842, Tocris, Minneapolis, MN) was added to the flow medium at a concentration of 5 µM. KLF2–GFP EC were incubated with the inhibitor or DMSO for 2 h, followed by 48 h of atheroprotective flow in the presence of the inhibitor or DMSO.
RNA Processing and Real-Time TaqMan PCR
Total RNA was isolated and purified using the Qiagen RNeasy Micro kit (Qiagen, Hilden, Germany), according to the manufacturer’s protocol. RNA was reverse transcribed to complementary DNA (cDNA) using a MultiScribe-based reaction (Applied Biosystems, Foster City, CA). cDNA templates were amplified by real-time TaqMan PCR on an ABI Prism 7900HT Fast Detection System (Applied Biosystems). Expression of KLF2 and GAPDH was analyzed using predesigned gene expression assays (Applied Biosystems), and gene expression was normalized against GAPDH endogenous controls. PCR reactions were performed in duplicate, and nuclease-free water was used as no-template controls.
Constitutively Active MEK5 (MEK5-CA) Expression
For MEK5-CA experiments, KLF2–GFP EC were seeded in 0.1% gelatin-coated wells of a six-well plate at a density of 6 × 104 cells/cm2 (5.7 × 105 cells/well) in 2 mL of EGM-2 medium. After 24 h, cells were infected with empty vector adenovirus (ad-CTRL) or MEK5-CA (ad-MEK5-CA) at an MOI of 100 as described previously. 8 Twenty-four hours after infection, cells were rinsed with PBS and kept in culture for an additional 24 h.
Fluorescence Microscopy
KLF2-GFP EC were washed with EGM-2 medium and visualized using a GFP fluorescence filter cube (Nikon GFP HQ) on a Nikon Eclipse TE2000-U microscope and a metal halide arc lamp-based fluorescence illumination system. Images were captured using MetaMorph software (Molecular Devices, Sunnyvale, CA). Brightness and contrast settings were auto-adjusted and standardized for all images with ImageJ (National Institutes of Health [NIH], Bethesda, MD).
Transfection of Small Interfering RNA (siRNA)
KLF2-GFP EC were seeded in 0.1% gelatin-coated wells of a six-well plate at a density of 5 × 104 cells/cm2 (4.75 × 105 cells/well) in 2.5 mL of EGM-2 medium 16 h prior to transfection. Negative control siRNA (4390843, Thermo Fisher Scientific, Waltham, MA) or MEKK3 siRNA (s8672, Thermo Fisher Scientific) were transfected at 10 nM by adding 1.5 µL siRNA, 5 µL of Lipofectamine RNAiMAX (Invitrogen), and 500 µL of Opti-MEM (Life Technologies, Grand Island, NY) per well, in a total volume of 3 mL per well. Four hours after transfection, cells were washed with PBS, trypsinized, and seeded on 0.1% gelatin-coated plastic disks suitable for use on the dynamic flow system at a density of 6 × 104 cells/cm2 (1.7 × 105 cells/well). Twenty-four hours after seeding, cells were exposed to atheroprotective flow for 48 h or maintained as static cultures.
Flow Cytometric Analysis
KLF2-GFP EC were trypsinized, washed with PBS, and resuspended in FACS buffer (PBS containing 5% FBS and 2 mM EDTA). DAPI was added for dead cell exclusion (1 µg/mL). Cells were processed using a FACS Canto II apparatus and FACSDiva software (BD Biosciences), and they were analyzed with FlowJo software (FlowJo LLC, Ashland, OR).
384-Well Plate Assays
KLF2-GFP EC were seeded in 0.1% gelatin-coated wells of a 384-well plate (Greiner Bio-One, Kremsmunster, Austria) at a density of 6 × 104 cells/cm2 (3.3 × 103 cells/well) in 40 µL per well. After 24 h, simvastatin (0.1, 0.25, 0.5, 1, 2.5, 5, and 10 µM in DMSO) was added at the indicated concentrations in four wells per condition. After 24 h of incubation, the plate was analyzed using a Phenix Operetta plate reader (PerkinElmer, Waltham, MA). The excitation wavelength was set at 488 nm, and the 500–550 nm filter set was used to read emissions. Captured images were processed using Harmony cellular image analysis software (PerkinElmer). For experiments designed to determine the Z’ factor, simvastatin was added at a concentration of 5 µM to half of the plate (192 wells), and DMSO was added to the other half (192 wells). After 24 h of incubation, the plate was analyzed as described above. The Z’ factor was determined as previously described. 18
Dose–Response Analysis Using FACS
For dose–response experiments using FACS analysis, KLF2-GFP EC were seeded in 0.1% gelatin-coated wells of a 96-well plate at a density of 6 × 104 cells/cm2 (1.92 × 104 cells/well) in 200 µL per well. Twenty-four hours after seeding, simvastatin (0.1, 0.25, 0.5, 1, 2.5, 5, and 10 µM in DMSO) was added at the indicated concentrations in four wells per condition. After incubation for 24 h, cells were trypsinized, and a single cell suspension was prepared in 200 µL FACS buffer (PBS containing 5% FBS and 2 mM EDTA) per sample. The plate was read using automated acquisition on an LSR II system (BD Biosciences), and mean GFP fluorescence intensity values were obtained. FACSDiva software (BD Biosciences) was used for cell acquisition, and FlowJo software was used for analysis.
Western Blotting
KLF2-GFP EC were lysed using radioimmunoprecipitation assay (RIPA) buffer (Sigma), and supplemented with Complete Mini Protease inhibitor (Roche, Basel, Switzerland) and phosphatase inhibitors (Sigma). Equal amounts of protein were run on a 4–20% sodium dodecyl sulfate–polyacrylamide gel and transferred to a nitrocellulose membrane. After transfer, the membrane was blocked for 1 h with 10 mL of blocking buffer (Tris-buffered saline, containing 0.1% Tween 20 and 5% [wt/vol] nonfat dry milk), and incubated with primary antibody rabbit anti-MEKK3 diluted 1:1000 (5727S, Cell Signaling, Danvers, MA) and rabbit anti-GAPDH 1:3000 (sc-25778, Santa Cruz Biotechnologies, Dallas, TX) in 5 mL of blocking buffer for 1 h at room temperature. After washing, the membrane was incubated with antirabbit horseradish peroxidase–conjugated secondary antibody, diluted 1:20,000 (Jackson Immunoresearch Laboratories, West Grove, PA) in 5 mL of blocking buffer.
Statistical Analysis
Statistical analyses and curve fitting were performed with GraphPad Prism software (GraphPad Software, La Jolla, CA). Statistical significance was determined using the two-tailed t-test, or one-way ANOVA followed by the Holm–Sidak multiple comparison test when appropriate. p-Values smaller than 0.05 were considered significant.
Results and Discussion
The KLF2-GFP Reporter Is Activated in Endothelial Cells Exposed to Atheroprotective Flow, but Not Atheroprone Flow
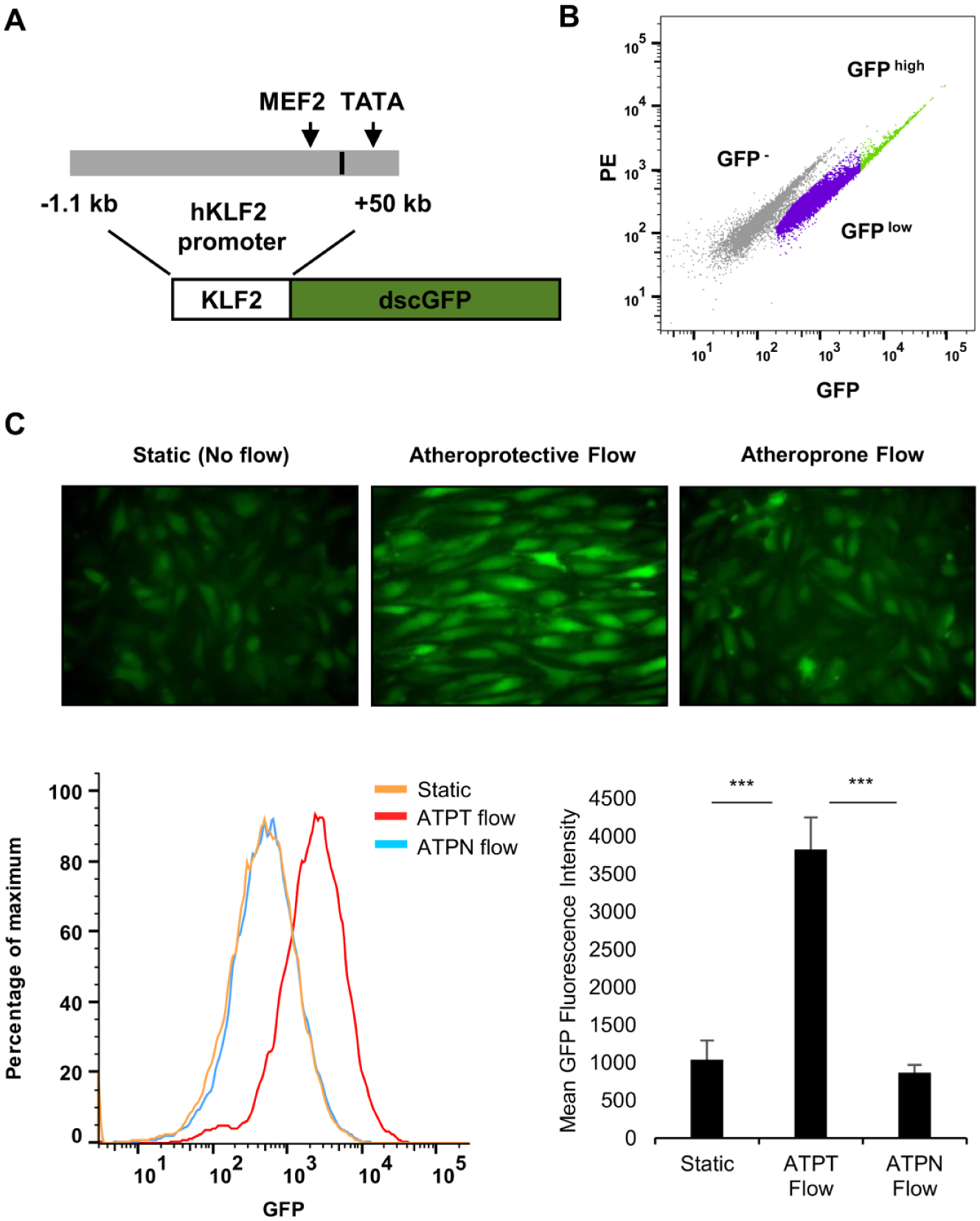
To develop a transcriptional KLF2 reporter system, a ~1.2 kb segment of the proximal promoter of the human KLF2 gene was cloned from human cDNA and coupled to a destabilized form of the green fluorescence protein (dscGFP) using the pTRH lentiviral vector ( Fig. 1A ). This promoter segment has been shown previously to be important for the basal 19 and flow-dependent expression of KLF2. 20
(
HUVEC were infected with lentivirus encoding the KLF2-GFP construct, and they were sorted based on the level of GFP expression. A homogeneous GFP-expressing population (GFPlow) was selected by exclusion of GFP− cells and the brightest expressers (GFP− and GFPhigh;
Fig. 1B
). As seen in
Figure 1C
, the basal activity of the reporter in KLF2-GFP EC cultured under static (no flow) conditions was low. When the KLF2-GFP EC were exposed to an atheroprotective shear stress waveform for 48 h, however, the expression of GFP was strongly increased. In contrast, the reporter system was not activated beyond basal levels in cells exposed to an atheroprone shear stress waveform for the same period of time. Importantly, this pattern follows the endogenous expression profile of KLF2 in EC exposed to these in vitro flow conditions, as described before
8
and as shown in
Activation of the KLF2 Reporter Is Regulated by Endogenous Signaling Events
The atheroprotective flow-mediated expression of KLF2 depends on the activation of a MAPK signaling cascade (MEKK3/MEK5/ERK5/MEF2) (
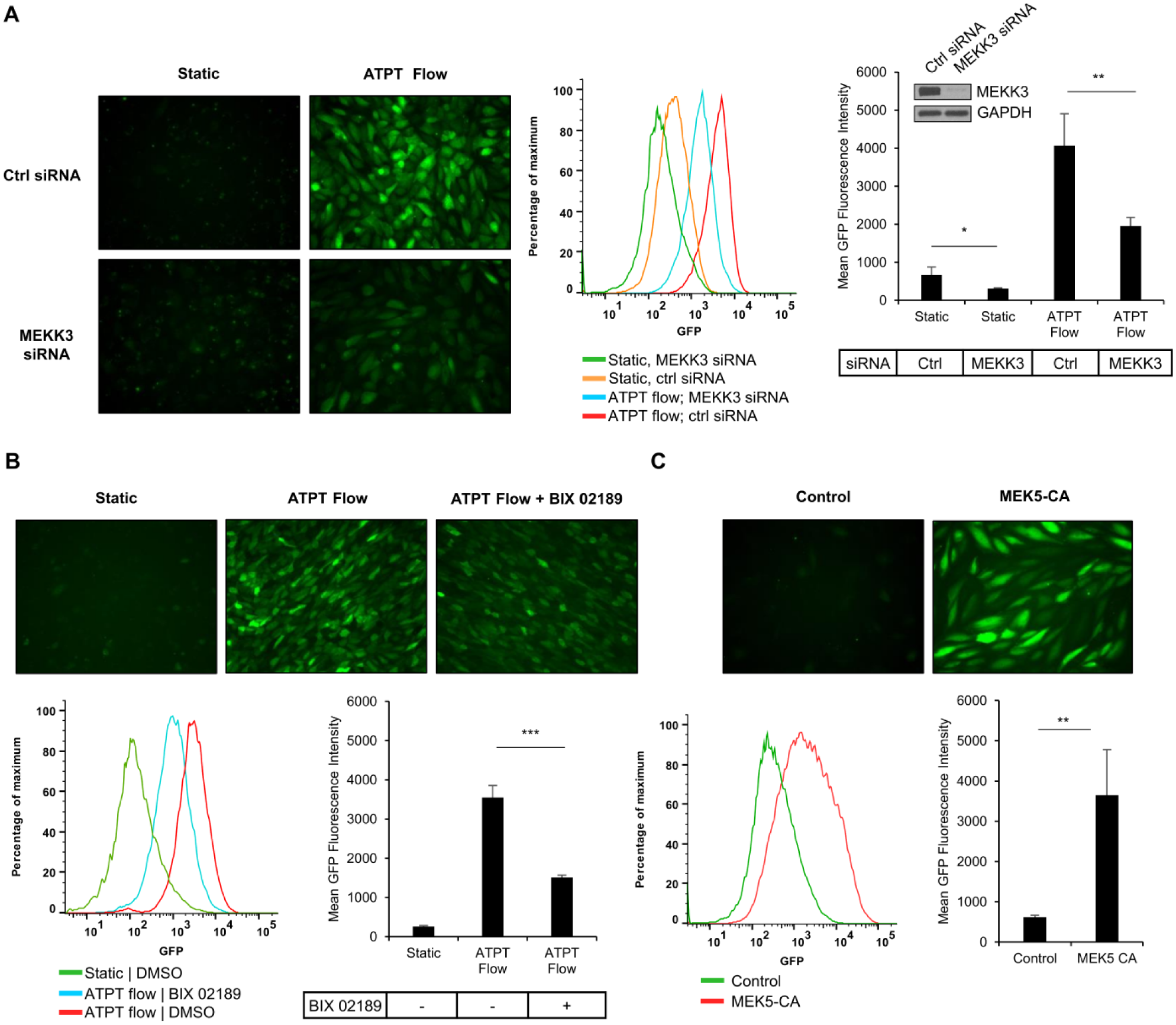
(
Simvastatin Induces the KLF2 Reporter
The HMG-CoA reductase inhibitor statins are a well-known class of activators of KLF2 expression.21,22 To assess if the KLF2 reporter is responsive to activation by statins, we exposed KLF2-GFP EC cells to simvastatin at different concentrations (1 µM and 5 µM) for 36 h. As seen in Figure 3 , GFP expression was induced in a dose-dependent manner as measured by fluorescence microscopy (top panel) and FACS (bottom panel).
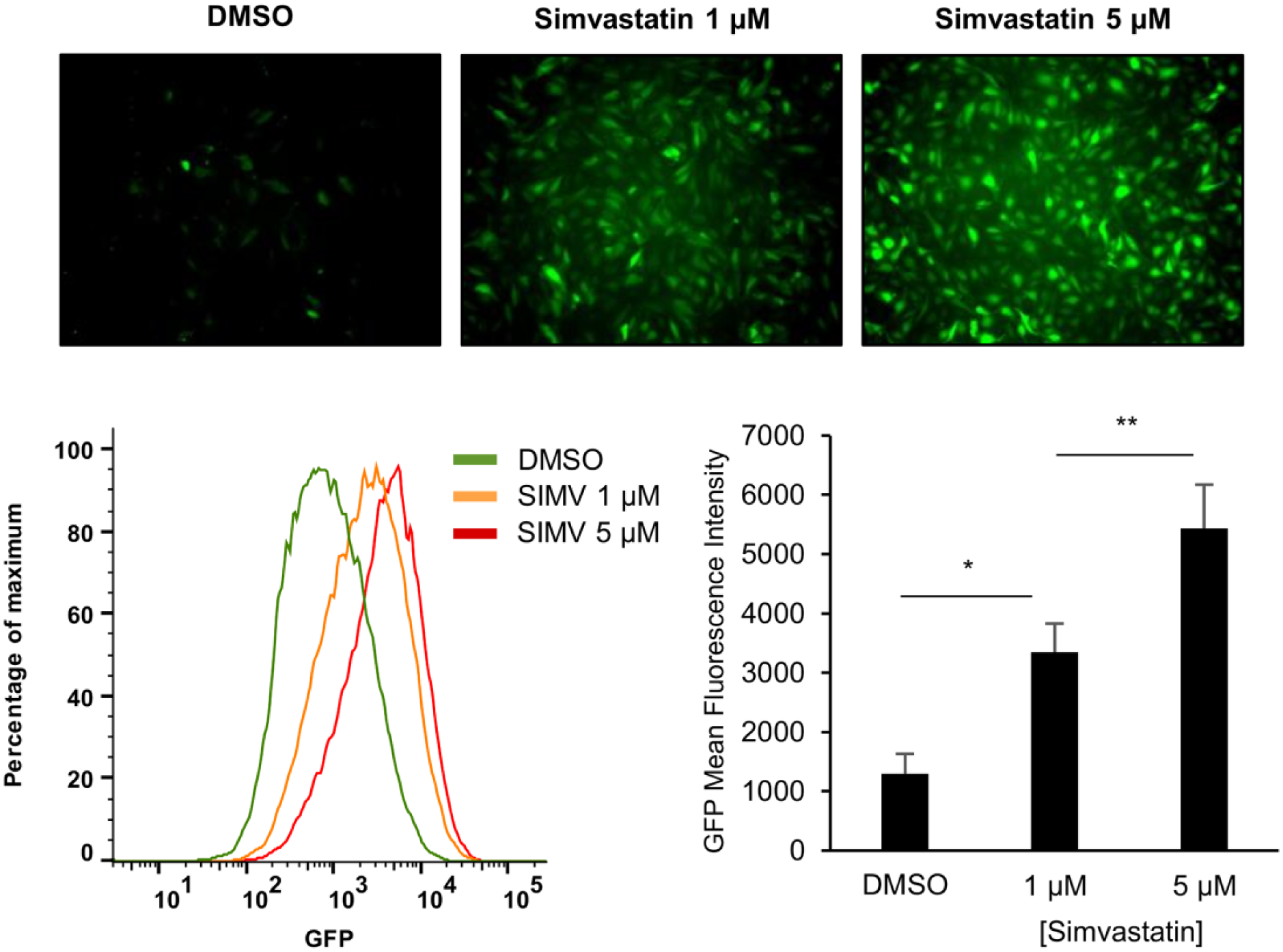
Kruppel-like factor-2 green fluorescence protein (KLF2-GFP) endothelial cells (EC) were cultured in a six-well plate in the presence of DMSO or simvastatin (1 µM and 5 µM) for 36 h. GFP expression was visualized by fluorescence microscopy (top panel), and GFP mean fluorescence intensities were measured by fluorescence-activated cell sorter (FACS) analysis (bottom panel). Bar graphs represent mean ±SD (n = 3) of three independent experiments. *p < 0.05; **p < 0.01; ANOVA with Holm–Sidak multiple comparison test.
Suitability of the KLF2-GFP Reporter for High-Throughput Screening Assays
Next, we evaluated the suitability of the KLF2-GFP reporter for use in HTS assays by assessing the dynamic range and the 384-well plate assay performance. To define the dynamic range of the reporter system and its dose-responsiveness, we generated a dose–response curve using a 384-well plate assay on a plate reader used for HTS. KLF2-GFP EC were exposed to a range of concentrations of simvastatin (0.1, 0.25, 0.5, 1, 2.5, 5, and 10 µM) for 24 h. Representative fluorescence images for each of the doses are shown in
Figure 4A
(top panel). A dose–response curve was established based on GFP fluorescence readings, demonstrating a typical dose–response sigmoid curve (
Fig. 4A
, bottom panel). The curve was fitted (R2 = 0.88), and the EC50 concentration was calculated and determined at 0.79 µM (0.55–1.26; 95% confidence interval [CI]). This assay was also performed on a FACS device capable of high-throughput sampling using a 96-well plate assay, yielding comparable results (
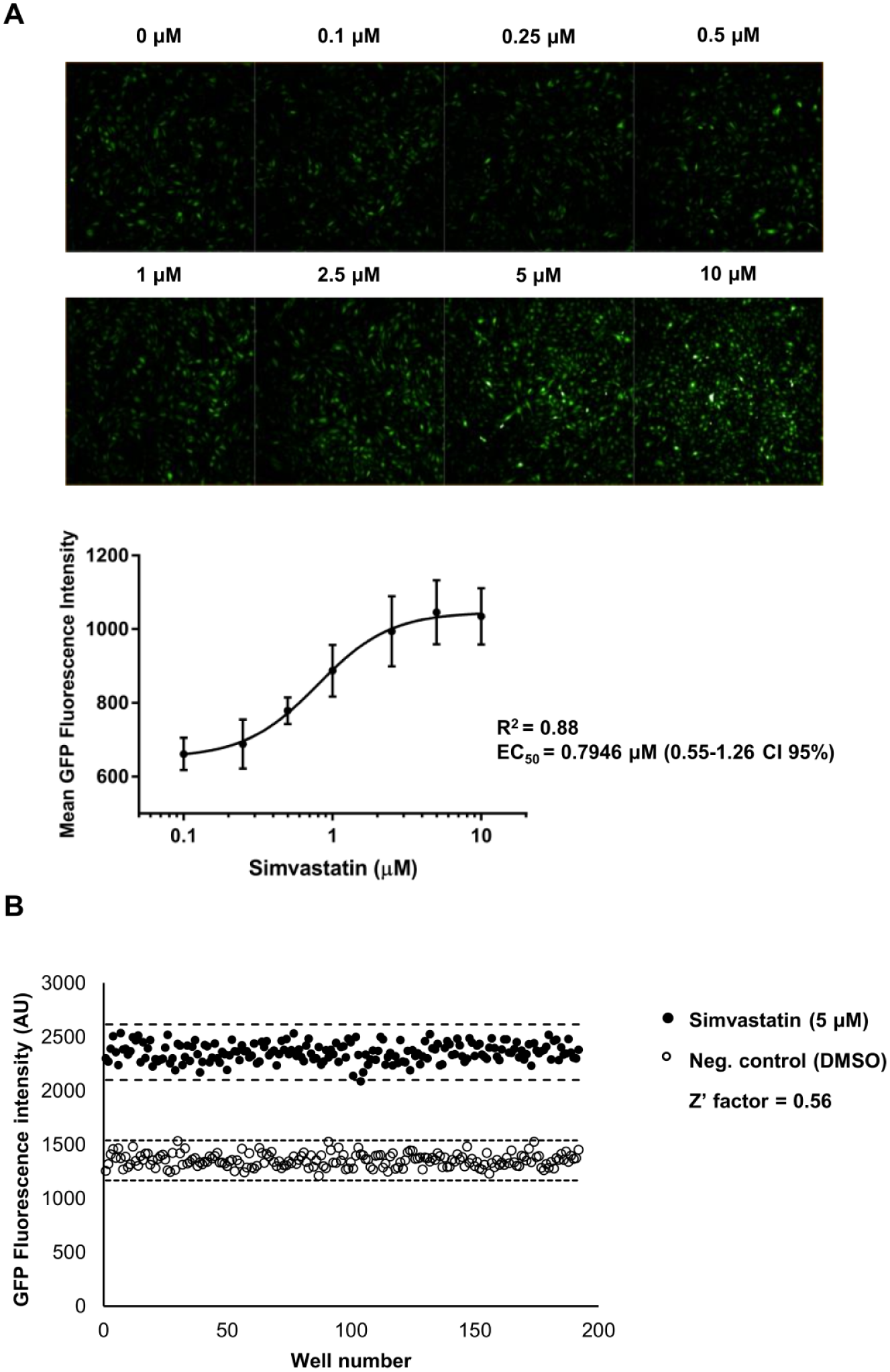
(
In summary, in this article, we report the generation and characterization of a reporter system for the expression of the human KLF2 gene, and thus by extension an atheroprotective endothelial phenotype. Collectively, our data demonstrate the KLF2 reporter system is uniquely capable of distinguishing distinct shear stress waveforms, because it is activated selectively by atheroprotective flow and not atheroprone flow, and it is regulated by the signaling events that regulate endogenous KLF2 expression. Moreover, statins are capable of regulating KLF2 reporter activity, and this regulation is dose dependent. This KLF2 reporter system can now be used in multimodal screens to characterize KLF2 signaling, and to discover a novel chemical space directed to establish or restore the atheroprotective phenotype of the vascular endothelium.
Footnotes
Acknowledgements
We thank Kay Case for isolation and culture of endothelial cells, Kate Hartland and Maria Alimova (Broad Institute of MIT and Harvard) for assistance with the 384-well assay performance experiments, and Adam Chicoine (Human Immunology Center Flow Cytometry Core) for cell-sorting assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the NIH (RO1 HL130624) and a Director’s Transformative Award from the Brigham and Women’s Hospital Research Institute to G. García-Cardeña.
