Abstract
Celiac disease (CD) is a chronic inflammatory condition caused by the ingestion of gliadin-containing food in genetically susceptible individuals. Undigested peptides of gliadin exert various effects, including increased intestinal permeability and inflammation in the small intestine. Although many therapeutic approaches are in development, a gluten-free diet is the only effective treatment for CD. Affecting at least 1% of the population in industrialized countries, it is important to generate therapeutic options against CD. Here, we describe the establishment of a high-throughput screening (HTS) platform based on AlphaLISA and electrical cell–substrate impedance sensing (ECIS) technology for the identification of anti-inflammatory and barrier-protective compounds in human enterocytes after pepsin-trypsin-digested gliadin (PT-gliadin) treatment. Our results show that the combination of these HTS technologies enables fast, reliable, simple, and label-free screening of IgY antibodies against PT-gliadin. Using this platform, we have identified a new chicken anti-PT-gliadin IgY antibody as a potential anti-CD agent.
Introduction
Celiac disease (CD) is an autoimmune enteropathy where the ingestion of gluten damages the small intestine. There is a wide spectrum of clinical symptoms that may occur in the digestive system or other parts of the body. 1 Gluten is a protein that is found in wheat, rye, and barley, but it is also present in daily use products, such as lipsticks, beer, ice creams, and sweets.2,3 Because gluten is omnipresent, it is difficult to avoid for patients suffering from CD. 8
Among the gluten proteins, gliadin is the most abundant protein in wheat. Gliadin is rich in proline and glutamine, which contributes to resistance against proteolytic digestion. When passing the intestine, two main gliadin peptides, 33-mer (P55-87) and 25-mer (P31-55), remain undigested. Gliadin and gliadin-derived peptides exert various effects on cells and the integrity of the intestinal mucosa. These effects include increased intestinal permeability and a pro-inflammatory environment in the small intestine where both the innate and adaptive immune response are constitutively alerted.16,18 Because of the presence of gluten in many products, dietary and nondietary therapies and different strategies to neutralize the deleterious effects of gluten are under development.19–22 Recently, Gujral et al. demonstrated the neutralizing effect of anti-gliadin IgY antibodies on the pro-inflammatory and barrier-destructive effects induced by gliadin. 23
Several methods to measure intestinal barrier function, typically including cell culture monolayers grown on permeable membranes, have been reported. 6
These physiologically relevant models allow access to both basolateral and apical surfaces and mimic the characteristics of the in vivo barrier integrity. The most studied parameters quantifying the barrier function are the (1) penetrability to radio- or fluorescence-labeled macromolecular probes (e.g., albumin or dextran), (2) the permeability to water expressed as hydraulic conductivity, and (3) the permeability to small inorganic ions described by the transepithelial electrical resistance (TER).17,30 However, hydraulic conductivity is not easy to assess experimentally, and fluorescence-labeled probes for flux measurement are not sensitive enough to detect barrier function.17,24 Furthermore, the use of labeled tracer molecules can also affect the barrier integrity and may be capable of altering the tested cells so that they are unusable for further experiments. Therefore, noninvasive methods, such as the measurement of the TER, are best suited to monitor barrier function and monolayer integrity. TER was described as a very sensitive and reliable tool that allows the determination of cell confluency and the formation of cell–cell junctions. 30 The standard method of TER measurement is the use of simple handheld devices with chopstick-type electrodes that measure the electric resistance of barrier-forming cell cultures.
Recently, impedance measurement has been increasingly used to characterize epithelial or endothelial barrier function in vitro. The electrical cell–substrate impedance sensing (ECIS) device, a sophisticated impedance measurement system, was invented by I. Giaver and C. R. Keese (Applied BioPhysics, Inc., Troy, NY) to study cell behavior in real time.4,17,30 Therefore, cells are grown on gold electrodes (250 µm in diameter) embedded in the bottom of cell culture plates. A small-amplitude AC signal is imposed across a pair of electrodes onto which cells are placed. The resulting impedance is computed from the corresponding changes in voltage between electrodes. ECIS has proved to be a noninvasive, continuous monitoring, and label-free system capable of analyzing many physiological characters of living cells, including cell attachment, spreading, migration, cell–cell contacts, monolayer formation, transmigration, cell growth, and cell death, in vitro.4–7,30 Due to its broad applicability, ease of implementation, and availability in a 96-well format, ECIS has been described as a unique and reliable technology, with the greatest potential in drug testing, compared with other biosensing methods.7,17
In this article, we describe the development of a high-throughput screening (HTS) platform and its application for the identification of anti-gliadin IgY antibodies, which has the potential to reduce the pro-inflammatory and barrier-disintegrating effects of pepsin-trypsin-digested gliadin (PT-gliadin) in a Caco-2 cell line system. Therefore, we used the sensitive and reproducible ECIS technology for monitoring the barrier properties (permeability) of Caco-2 cell monolayers in a 96-well format and the AlphaLISA HTS system for the detection of the inflammatory marker, interleukin-8 (IL-8), after PT-gliadin treatment, and measured the influence of different anti-gliadin IgY antibodies. By implementing the combination of these HTS technologies, we identified a new anti-gliadin IgY fraction as a potential anti-CD agent.
Materials and Methods
Cell Culture
The colon epithelial cell line Caco-2 (ATCC) was grown in Dulbecco’s modified Eagle’s medium (DMEM) (high glucose) supplemented with 10% fetal calf serum (FCS), 2 mM
Microscopy
Caco-2 cells (1 × 105) were plated on 24-well plates and allowed to grow for 24 h. Cells were treated with PT-gliadin (1 mg/mL) only or with combinations of PT-gliadin and specific IgY antibody, unspecific IgY antibody, or bovine serum albumin (BSA) (3.5 mg/mL each) for 24 h. Phase contrast pictures were taken in 100× magnification with a Leica SP2 microscope.
ELISA and Western Blot for Anti-PT-Gliadin Chicken IgY Antibodies
PT-gliadin, PT-gelatin as a control, and chicken anti-PT-gliadin IgY were prepared as described previously by Stadlmann et al. 29 Specificity of the anti-PT-gliadin IgY was confirmed by enzyme-linked immunosorbent assay (ELISA) and Western blot analysis. For the ELISA, 96-well plates (MaxiSorp, Nunc) were coated overnight at 4 °C with decreasing amounts of PT-gliadin, PT-gelatin, and BSA (5–0.5 µg/100 µL coating buffer [0.1 M NaHCO3, pH 8.6] per well). After blocking with 3% milk (Cell Signaling Technology [CST], Danvers, MA) in coating buffer for 1 h at room temperature (RT) and washing with phosphate-buffered saline (PBS)-T (0.05% Tween-20; Sigma, St. Louis, MO), anti-PT-gliadin IgY (1:200 in 1% milk in coating buffer) was added and incubated overnight at 4 °C. Wells were washed extensively, horseradish peroxidase (HRP)–coupled goat anti-IgY (1:4000 in 1% milk in coating buffer, Sigma) was added, and the wells were incubated at RT for 2 h. After final washing steps, ELISAs were developed by adding 100 µL of 1-Step Ultra TMB-ELISA Substrate Solution (Thermo Fisher Scientific, Waltham, MA). After 15 min incubation at RT, the reaction was stopped by the addition of 100 µL of 2 M sulfuric acid. For the anti-gliadin Western blot, indicated amounts of PT-gliadin, PT-gelatin, and BSA were separated on 4%–15% Mini-PROTEAN TGX Precast Protein Gels (Bio-Rad Laboratories, Hercules, CA) to a nitrocellulose membrane. The membrane was blocked in Tris-buffered saline (TBS) supplemented with 5% milk powder overnight at 4 °C, washed, and probed with either anti-PT-gliadin IgY or a corresponding unspecific IgY (~1 µg/mL in TBS supplemented with 1% milk) for 2 h. After washing, blots were incubated in goat anti-chicken IgY (H+L)-HRP antibody (Southern Biotech, Birmingham, AL) at a 1:10,000 dilution in TBS with 1% milk powder for 1 h. Bands were detected using 1-Step TMB-Blotting Substrate Solution (Thermo Fischer Scientific).
Cell Viability Assays
Caco-2 cells (2 × 104) were plated on 96-well plates and allowed to grow for 24 h. Cells were treated with different concentrations of PT-gliadin, PT-gelatin (0.008–3 mg/mL), specific and unspecific IgY (10–0.625 mg/mL), or combinations thereof for different time periods (8, 24, 48, or 96 h). Subsequently, cells were incubated for 2 h with 10 vol% of CellTiter-Blue (Promega, Madison, WI), and cell survival was analyzed at an extinction wavelength of 530 nm and an emission wavelength of 590 nm using the SpectraMax Paradigm Modular Multi-Mode Reader (Molecular Devices, Sunnyvale, CA), and normalizing to untreated cells.
Western Blot for NFκB Phosphorylation
For Western blot analysis of phospho-NFκB, 3 × 105 cells were plated in 3 mL of medium in cell culture dishes (6 cm; Nunc). After 3 days of incubation, a confluent monolayer was obtained and the medium was replaced with 1 mL of fresh medium containing PT-gliadin only (1 mg/mL), PT-gliadin in combination with an anti-PT-gliadin IgY (3.5 mg/mL), or anti-PT-gliadin IgY only. After 45 min of incubation, medium was removed, and the cells were washed with ice-cold PBS and harvested by scraping in 1 mL of ice-cold PBS. Cells were further pelleted by centrifugation (200g for 5 min at 4 °C), and resuspended in 300 µL of lysis buffer containing 20 mM HEPES (pH 7.2), 1% Nonidet P-40, 10% glycerol, 50 mM NaF, 1 mM Na3VO4, and a protease inhibitor cocktail (Sigma). Following a further incubation of 10 min on ice, insoluble cell debris was removed by centrifugation (maximum speed, 4 °C, 10 min). The protein concentration in the cleared supernatant was determined using Bradford Assay Reagent (Bio-Rad) with BSA as a standard. The samples were diluted in lysis buffer, and 4× Sample Buffer (Bio-Rad) was added, resulting in a final protein concentration of 1 µg/µL. Samples were denatured at 95 °C for 5 min, and 20 µg protein per lane was separated on 10% Mini-PROTEAN TGX Precast Gels (Bio-Rad). The gels were blotted to nitrocellulose membranes (Trans-Blot Turbo Mini Nitrocellulose Transfer Packs, Bio-Rad), and the membranes blocked afterward with 5% BSA in TBS-T (50 mM Tris [pH 7.5], 150 mM NaCl, and 0.1% Tween-20) and probed with the primary antibodies anti-NFκB p65 (sc-8008, Santa Cruz), anti-phospho-NFκB p65 (Ser536; 3033S, CST), and anti-GAPDH (2118S, CST) as control. All primary antibodies were diluted 1:1000 in TBS-T, 1% (w/v) BSA. For detection with the Odyssey CLX device (LI-COR, Lincoln, NE), membranes were incubated with IRDye 800CW labeled anti-rabbit or anti-mouse IgG antibodies (92632210 or 92632211, respectively; both LI-COR) diluted 1:20,000 in TBS-T with 1% (w/v) BSA. To enable an optimal quantification of the bands, Image Studio Software Odyssey CLx (LICOR) was used.
ECIS for Barrier Function Analysis
A Caco-2 monolayer breakdown assay was performed using ECIS model 9600Z (Applied BioPhysics, Troy, NY). The measurement system consists of a 96-well cell culture dish (96W10E) with 10 active electrodes. A small-amplitude AC signal is imposed across the electrodes, onto which cells are deposited. The resulting impedance is calculated by the ECIS device. For the measurement of the impedance, the ECIS 96-well plates were coated with laminin (10 µg/mL in PBS; Sigma) for 1 h at 37 °C and 5% CO2. Then 3 × 104 Caco-2 cells were plated per well in a final volume of 100 µL and incubated for 2 days, resulting in a confluent monolayer. The medium was changed and the baseline at 4000 Hz was determined for 1 h. Then the cells were treated with PT-gliadin (1 mg/mL) and/or anti-PT-gliadin IgY (3.5 mg/mL), and the change in impedance was recorded over the following 12 h.
IL-8 AlphaLISA Assay
For the IL-8 AlphaLISA assay, 105 cells were plated in a final volume of 100 µL per well in Nunc Delta 96-Well MicroWell plates and incubated overnight. For the treatment, half the medium was removed and replaced by 50 µL of fresh medium containing PT-gliadin only (final concentrations ranging from 6.125 µg/mL to 1.5 mg/mL), PT-gliadin (1 mg/mL) in combination with an anti-PT-gliadin IgY (10–20 mg/mL), or the anti-PT-gliadin IgY (20 mg/mL) only. For the combined treatment, PT-gliadin and the anti-PT-gliadin IgY were preincubated for 1 h, at 37 °C shaking moderately. After 6 h of treatment, supernatants were harvested and stored at –80 °C until further analysis.
AlphaLISA detection of IL-8 in the supernatants of treated cells was performed in white wall 96-well plates (Brand GmbH) in a final volume of 25 µL containing 10 µg/mL AlphaLISA acceptor beads, 1 nM anti-IL-8 antibody, and 5 µL of undiluted supernatant. After 1 h of incubation at RT in the dark, 25 µL of streptavidin-coupled donor beads were added to a final concentration of 40 µg/mL. The reaction was incubated for another 30 min at RT in the dark, and finally measured using the SpectraMax Paradigm Modular Multi-Mode Reader (Molecular Devices) equipped with the AlphaScreen detection cartridge (excitation at 680 nm and emission at 570 nm).
Additionally, to assess the quality of the assay and its suitability for HTS, the Z′ factor was investigated. Therefore, 105 Caco-2 cells were plated in a final volume of 100 µL per well in Nunc Delta 96-Well MicroWell plates and incubated overnight. After treatment with 1 mg/mL PT-gliadin or fresh medium for 6 h and subsequent IL-8 AlphaLISA assay, the Z′ factor was calculated.
Results
Purified IgY Antibody Specifically Recognizes Toxic PT-Gliadin
A substantial number of novel anti-gliadin IgY antibodies obtained from chickens immunized with PT-gliadin were generated, purified, and analyzed for their specificity using ELISA (data not shown). The antibody with the desired binding affinity was further tested for its specificity against PT-gliadin and potential background binding. Therefore, the obtained specific anti-PT-gliadin IgY antibody was compared with an unspecific IgY fraction by using Western blot analysis and ELISA. PT-gliadin (15 and 30 µg), PT-gelatin (30 µg), and BSA (30 µg) were separated via gel electrophoresis and transferred to nitrocellulose membranes. The membranes were probed with either anti-PT-gliadin IgY or unspecific IgY in equitable concentrations. Upon detection with a HRP-coupled goat anti-IgY IgG, a difference in the binding spectrum of the primary antibodies could be observed with clearly distinguishable bands at 22 and 30 kDa for the specific antibody ( Fig. 1A ). Additionally, a first estimation of the background binding to BSA and PT-gelatin supported the specificity of the isolated antibody fraction. To determine the cross-reactivity of the anti-PT-gliadin IgY antibody with another method, a quantitative ELISA was performed. Therefore, plates were coated with 0.5-5 µg of PT-gliadin, PT-gelatin, and BSA and detected using anti-PT-gliadin IgY and HRP-coupled goat anti-IgY IgG as primary and secondary antibodies, respectively. After development with Ultra TMB, the absorbance was measured at 405 nm. As depicted in Figure 1B , the anti-PT-gliadin IgY antibody showed a concentration-dependent increase in PT-gliadin- but not in PT-gelatin- or BSA-coated plates, indicating high specificity of this antibody.
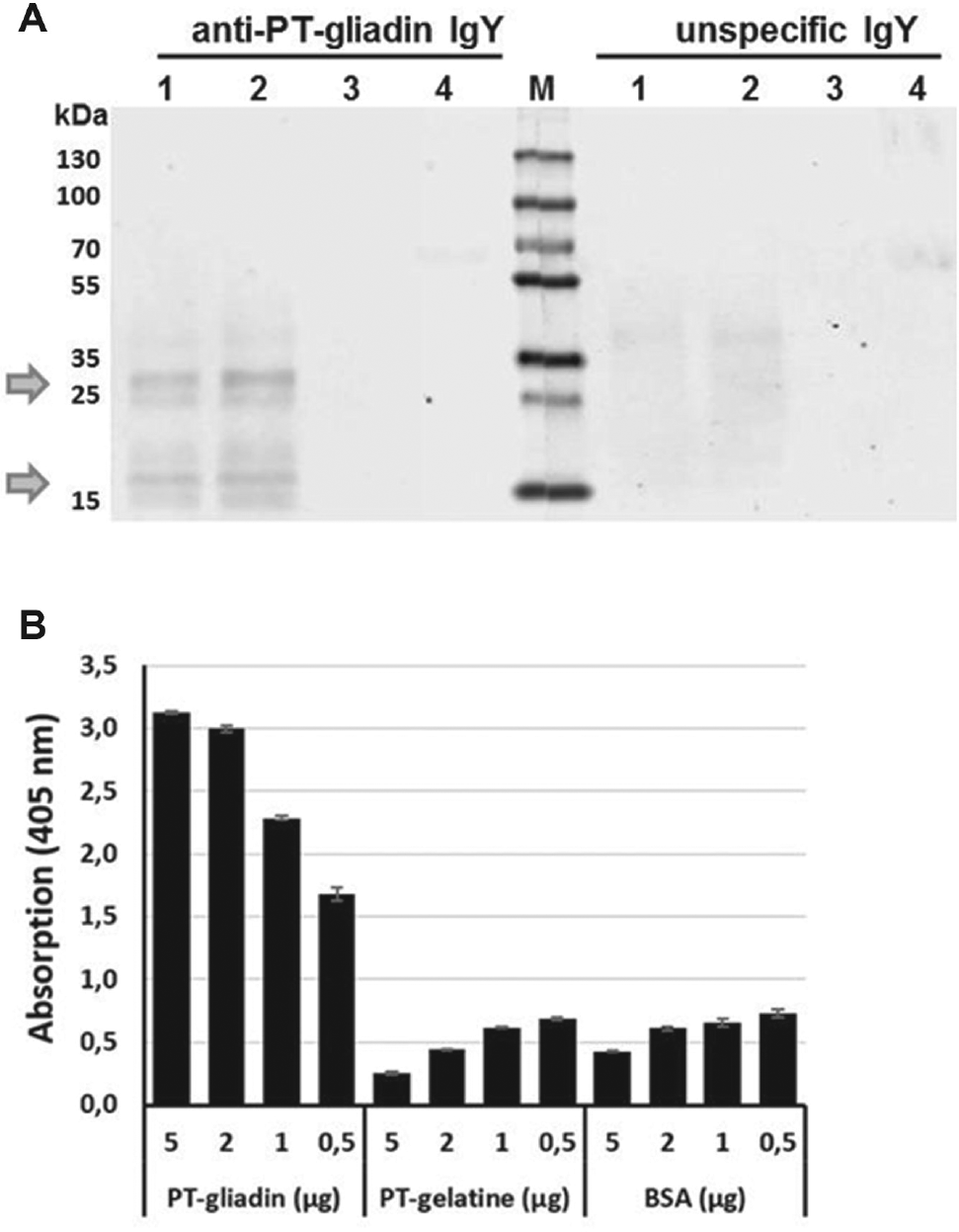
Chicken anti-PT-gliadin IgY specificity. (
Treatment of Caco-2 Cells with PT-Gliadin or Anti-PT-Gliadin IgY Does Not Significantly Influence Cell Viability
In order to investigate the cytotoxic potential of PT-gliadin, PT-gelatin, and anti-PT-gliadin IgY antibody, a cell viability assay (CellTiter-Blue, Promega) was performed. Caco-2 cells (2 × 104 per well in a 96-well plate) were allowed to attach and grow to a monolayer for 24 h. Afterwards, the cells were treated with 0.1 µg/mL to 10 mg/mL PT-gliadin or PT-gelatin for a further 24 h before the PrestoBlue assay was performed ( Fig. 2A ). No significant decrease in viable cells could be observed, indicating that PT-gliadin and PT-gelatin did not interfere with growth and viability even at the highest concentration (10 mg/mL). Interestingly, the only noticeable effect was the formation of holes in the monolayer ( Fig. 3 ). Even though the neutralizing potential of the antibody fractions could not be evaluated, they still were subjected to the test system. In order to assess the applicability of the antibody fraction and for proper evaluation of further test results, the toxicity of the isolated antibody fractions is of high importance. Therefore, Caco-2 monolayers were treated with 10–0.62 mg/mL anti-PT-gliadin IgY for 24, 48, or 96 h and the assay was performed as described above. Compared with the negative control, only high concentrations (10 mg/mL) and long exposure (96 h) reduced cell metabolism for about 20%. These findings indicate that the tested substances showed no significant effect on cell metabolism, proliferation, or cell survival.
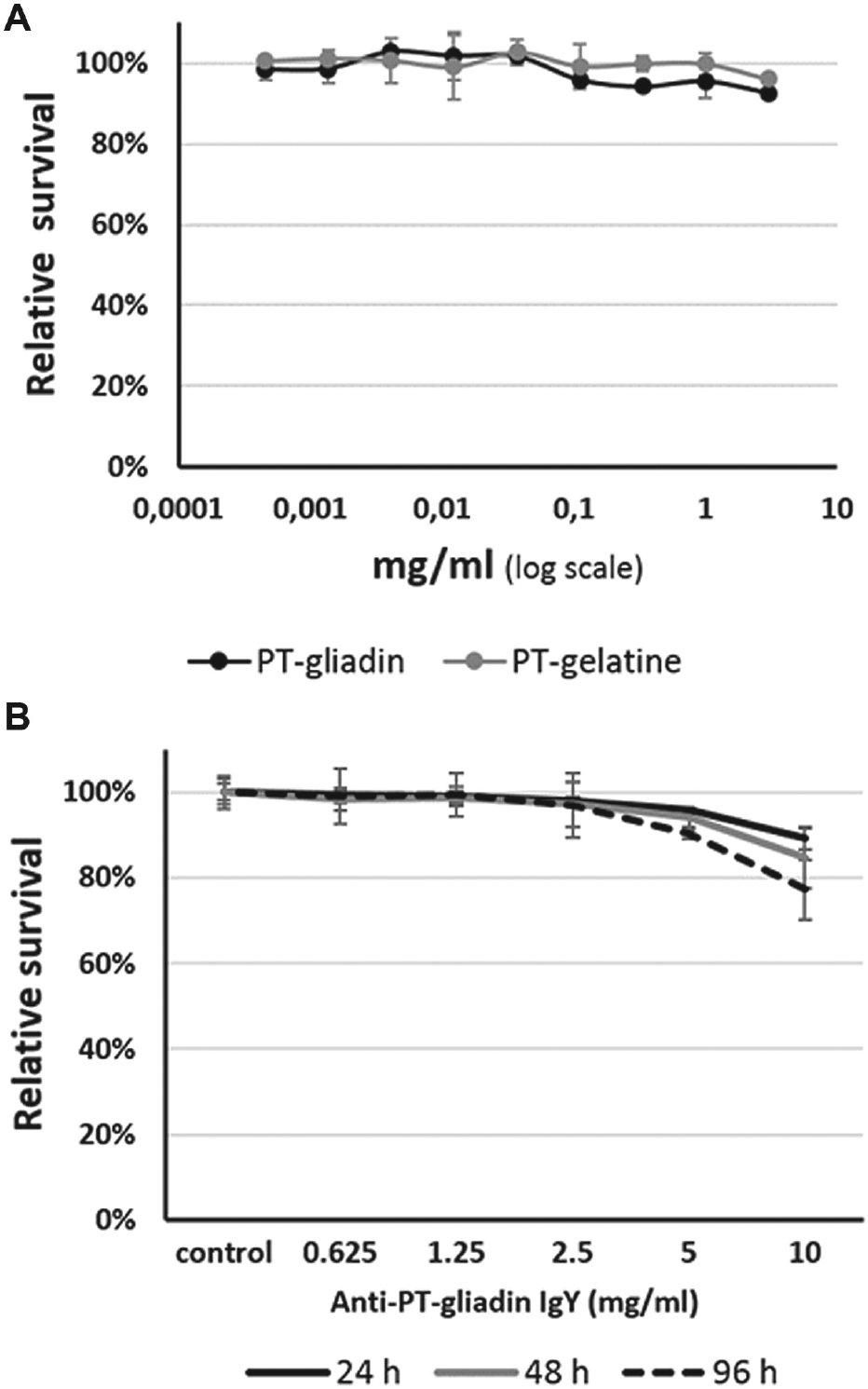
Influence of PT-gliadin on cell viability. (
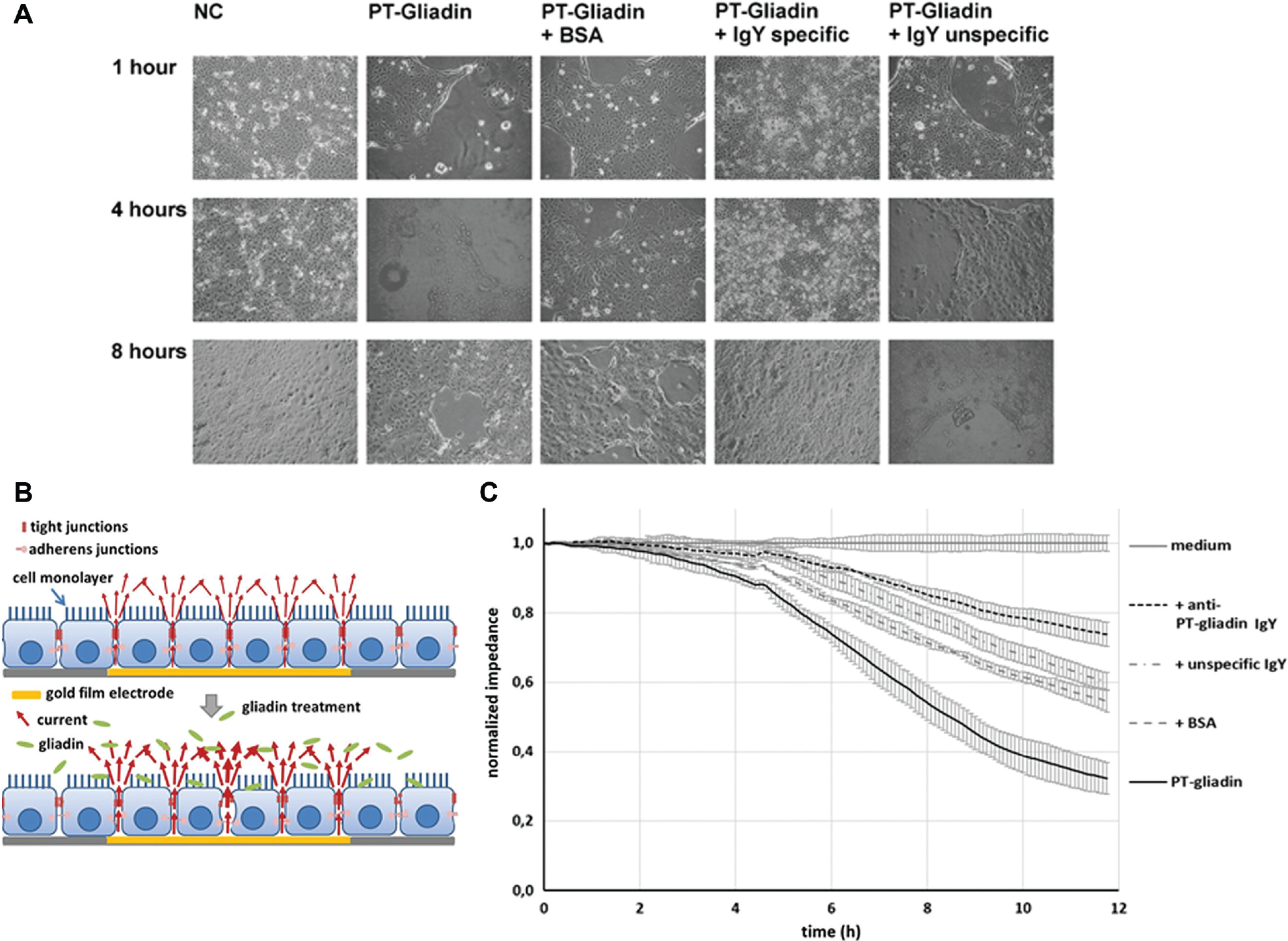
Impact of PT-gliadin on cell–cell contacts. Caco-2 monolayers were left untreated, or challenged with PT-gliadin (1 mg/mL) preincubated without or with BSA, specific anti-PT-gliadin IgY, or unspecific IgY (3.5 mg/mL each). (
Anti-PT-Gliadin IgY Diminishes Barrier-Disruptive Activities of PT-Gliadin on Caco-2 Monolayers
Based on our observation that PT-gliadin triggered monolayer disruption and the known fact that gliadin increases intestinal permeability via zonulin release, experiments were designed to assess the antibody’s potential to counteract the barrier-disruptive activities. 25 First, we investigated microscopically the effect of PT-gliadin alone or preincubated with BSA or specific or unspecific anti-PT-gliadin antibodies on Caco-2 monolayers for 1, 4, or 8 h. As indicated in Figure 3A , the disruption of intestinal barrier function by PT-gliadin clearly decreased when gliadin was preincubated with the specific anti-PT-gliadin IgY antibody, whereas BSA or unspecific IgY displayed no significant effect. To verify the inhibitory effect of the specific anti-PT-gliadin antibody with a high-throughput system (HTS), ECIS technology was applied. Caco-2 monolayer disruption can thus be monitored in real time by means of measuring the decrease in impedance over time. As such, gliadin, which causes monolayer disruption, can be detected, as it will lead to a changed resistance of the monolayer ( Fig. 3B ). Caco-2 cells were cultured onto 96-well ECIS arrays (pretreated with laminin) and formed a confluent monolayer within 48 h. The monolayer was challenged with PT-gliadin alone (positive control) or PT-gliadin together with BSA, specific IgY, or unspecific IgY antibody, or with medium as negative control. Caco-2 monolayer disruption was subsequently assessed by continuous resistance measurements. PT-gliadin-mediated monolayer breakdown was decreased most after treatment with specific anti-PT-gliadin IgY fractions, followed by unspecific IgY antibodies, BSA, and nontreated cells ( Fig. 3C ), indicating that the specific IgY antibody is well suited to rescue cell monolayer integrity.
Diminution of Inflammatory Transcription Factor Activation upon Preincubation with Neutralizing Anti-PT-Gliadin Antibody Fraction
Having demonstrated that the specific anti-PT-gliadin antibody attenuates intestinal barrier disruption caused by PT-gliadin ( Fig. 3 ), we next examined whether the specific IgY antibody is able to diminish the expression of inflammatory markers such as NF-κB, which play an important role in the common chronic intestinal inflammatory CD. Therefore, we challenged monolayers of Caco-2 cells with PT-gliadin (1 mg/mL), or with preincubated solutions of PT-gliadin and anti-PT-gliadin IgY (3.5 mg/mL), and used whole cell lysates for sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis and subsequent Western blot analysis. To enable an optimal quantification of the bands, infrared labeled secondary antibodies and the Image Studio Software Odyssey CLx (LICOR) were used. As indicated in Figure 4A , B , NF-κB phosphorylation was significantly (p < 0.05) reduced upon treatment with specific anti-PT-gliadin IgY, whereas the total NF-κB levels showed no significant effect. GAPDH was used as loading control.
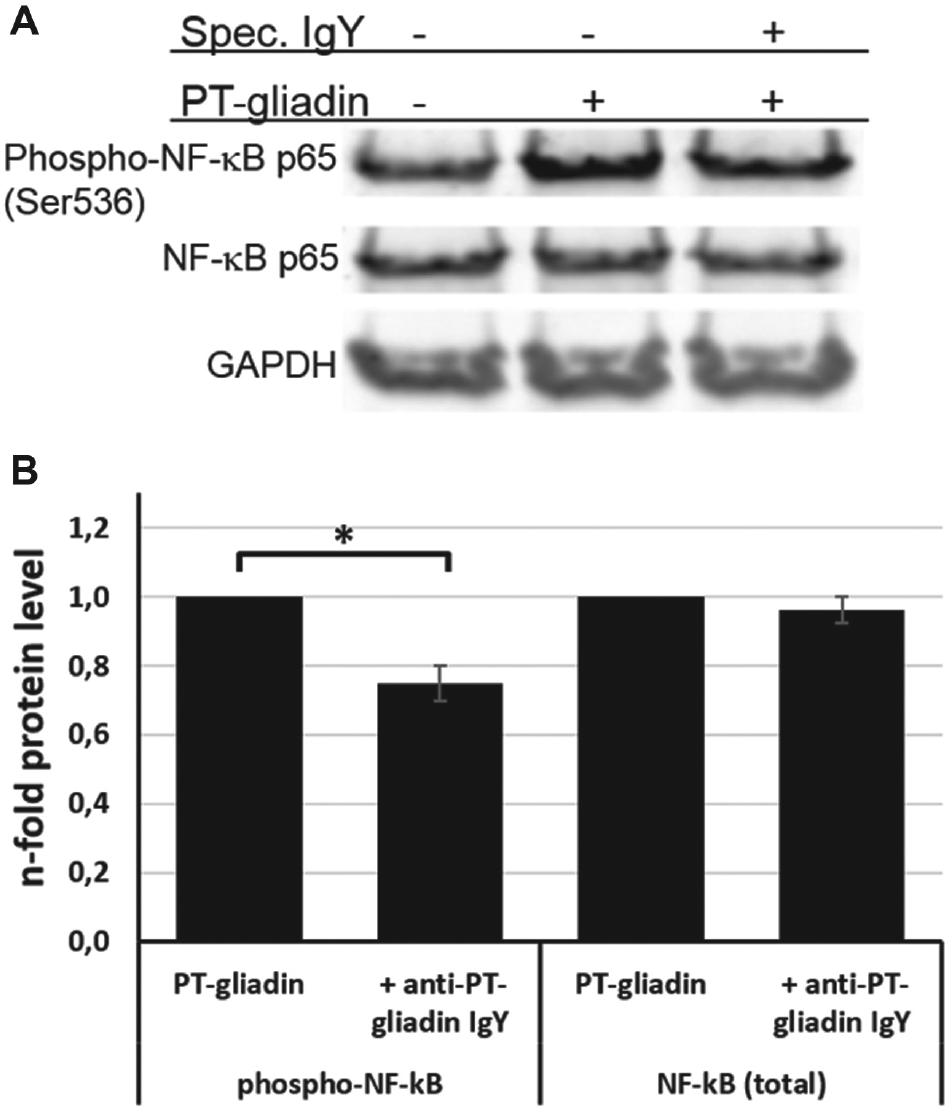
NF-κB activation by PT-gliadin. Caco-2 monolayers were challenged with PT-gliadin (1 mg/mL) or with preincubated solutions of PT-gliadin and anti-PT-gliadin IgY (3.5 mg/mL), or were left untreated for 45 min. (
Anti-PT-Gliadin IgY-Mediated Downregulation of IL-8 Secretion in Response to PT-Gliadin
To further substantiate our findings, we next applied the AlphaLISA HTS system for the detection of the inflammatory marker, IL-8. The AlphaLISA system eliminates all cell lysis steps, since only the supernatant of the stimulated cells is used. The principle of this technology is shown in Figure 5A . In a first step, the robustness of the assay was determined to evaluate whether the assay would be useful in HTS. This was done by treating Caco-2 monolayers with either PT-gliadin or plain DMEM for 6 h. As depicted in Figure 5B , the Z′ factor calculated from the generated data is 0.71, which indicates an excellent assay and its applicability in HTS. Subsequently, Caco-2 cells were challenged with PT-gliadin ( Fig. 5C ), PT-gelatin ( Fig. 5D ) in decreasing concentrations, or preincubated solutions of PT-gliadin (1 mg/mL) and decreasing amounts of anti-PT-gliadin IgY ( Fig. 5E ). These results demonstrate that the anti-PT-gliadin IgY antibody is able to downregulate PT-gliadin-mediated IL-8 expression in a concentration-dependent manner. In addition, IL-8 downregulation was further determined by testing diverse combinations of PT-gliadin with anti-PT-gliadin IgY, unspecific IgY, and recombinant human tumor necrosis factor alpha (TNF-α) without or with anti-PT-gliadin IgY. Taken together, these data confirm significantly (p < 0.01) that the observed downregulation of IL-8 is caused by the specific PT-gliadin neutralizing IgY antibody.
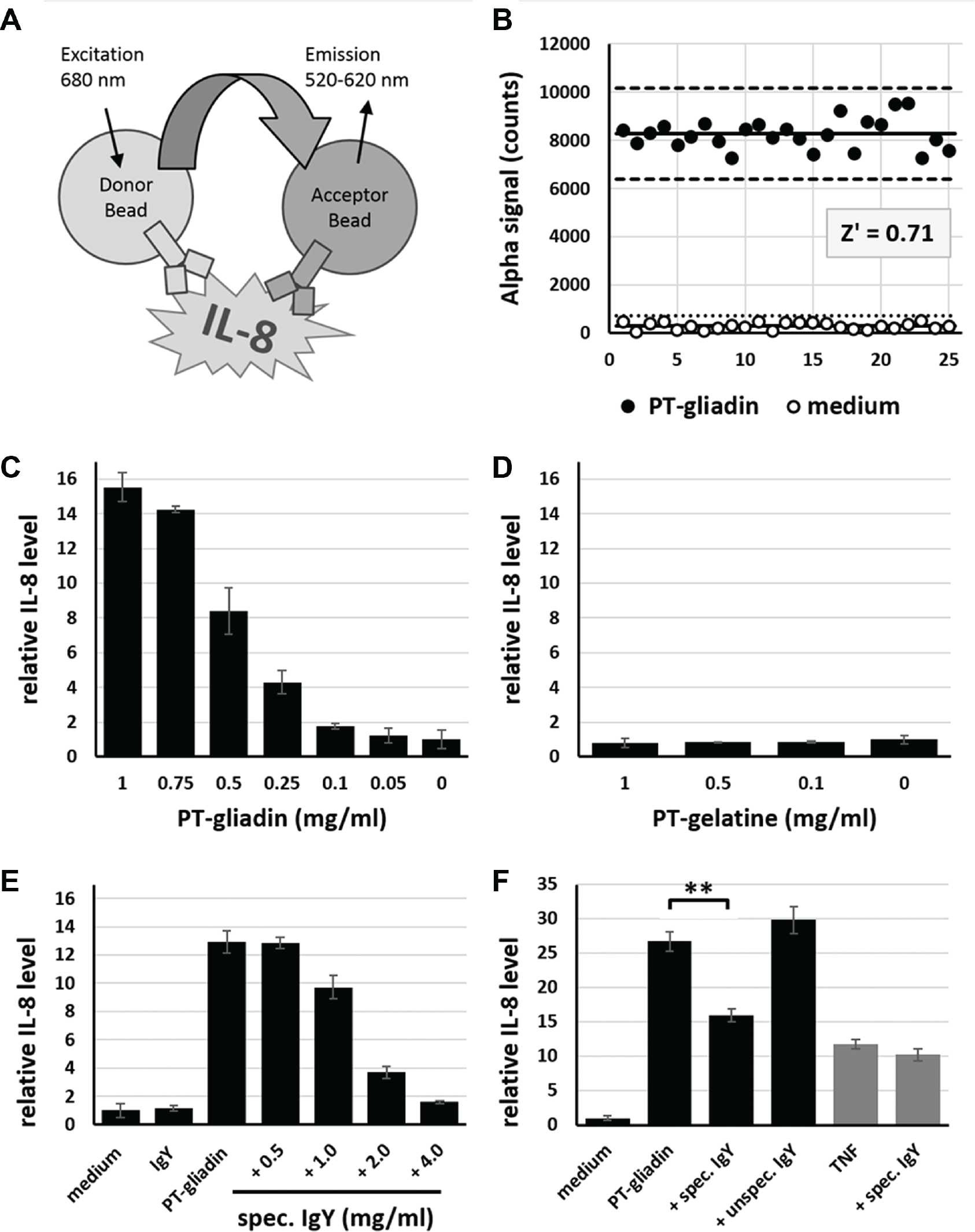
IL-8 secretion in response to PT-gliadin. (
Discussion
CD constitutes one of the most common chronic intestinal inflammatory disorders worldwide, affecting 1%–3% of the population. 26 Dietary uptake of gluten, more precisely, the response upon encountering its immunogenic fraction gliadin, triggers an increased intestinal permeability via the upregulation of zonulin and T-cell activation. Gliadin induces a MyD88-dependent zonulin release by binding to the CXCR3 receptor, leading to PKC-mediated cytoskeletal rearrangement, loss of occluding-ZO1 protein–protein interaction, increased monolayer permeability, and augmented cytokine production in intestinal cells. 16 As the intestinal permeability increases, gliadin-derived peptides are transported via paracellular crossing to the lamina propria and induce a strong immune response, leading to characteristic intestinal lesions.9–15 Currently, the sole therapy available to patients suffering from CD is a lifelong gluten-free diet. 2 An effective neutralizing antibody would constitute a major improvement not only for the individuals affected by CD, but also for the population suffering from nonceliac gluten sensitivity, which is estimated to constitute 0.63%–6% worldwide.25,28 Several studies have examined the use of IgY against gliadin, and there are several benefits over polyclonal antibodies, including low costs of animal maintenance, availability of large quantities of eggs and ease of using eggs, and the high antibody content. 27
Therefore, we used layers to produce specific chicken yolk antibodies (IgY) against PT-gliadin. Of all the anti-gliadin IgY antibody fractions tested, one of these indicated the capacity to reduce PT-gliadin-mediated monolayer breakdown and decreased production of pro-inflammatory cytokines like IL-8 and TNF-α in Caco-2 cells. This can be attributed mainly to the ability of the IgY antibody to bind specifically to PT-gliadin and neutralize the toxic effect by inhibiting gliadin absorption. The efficacy may be amplified by the fact that IgY is polyclonal and can therefore bind to multiple epitopes on various fractions of PT-gliadin. Hence, the novel anti-PT-gliadin IgY fraction produced in this study has an ideal prerequisite to be used for preventative or therapeutic purposes, especially for the treatment of CD.
In this article, we investigated potential methods for the HTS of antibody fractions for the neutralization of PT-gliadin in vitro. Since the main focus was to enable screening of large amounts of antibody fractions, we concentrated on methods like, for instance, the ECIS technology, which allows quantitative cell analysis (cellomics) in an automated manner in a 96-well format and the AlphaLISA technology for the simple and rapid quantification of biomarkers in biological samples.15,17 The ECIS-based HTS method used here has been shown to be a suitable, rapid, and very sensitive method to determine the barrier-protective effects of anti-gliadin IgY antibodies by measuring living Caco-2 monolayers. Compared with other fluorescence-based high-content screening assays, which need preparation time and postwashing steps, ECIS was able to measure cell monolayer integrity in real time without any cell culture modifications. Bischoff et al. analyzed the impact of different pharmacological tools on endothelial barrier function by comparing transwell tracer assays and the two impedance devices xCELLigence and ECIS. 17 The two impedance techniques provided similar results, whereas macromolecular permeability readings were found to be partly inconsistent with impedance. They recommend a complementary combination of both approaches to overcome the restrictions of each assay. In order to achieve accurate and meaningful results, we have screened a substantial number of novel anti-PT-gliadin IgY antibodies by measuring the barrier function and IL-8 secretion in HTS by means of combining both ECIS and AlphaLISA methods. The data were verified with standard methods like ELISA, Western blotting, and phase contrast microscopy; however, for future use these methods can be omitted after successful establishment of the screening platform. Together, the combination of these technologies has proved to be an excellent platform for HTS that enables a fast and reliable detection of IgY antibodies against CD.
Footnotes
Declaration of Conflicting Interests
Sciotec Diagnostic Technologies holds a patent for the use of IgY and fragments thereof in DC 30 therapy, which relates to this work. A.M. is the patent inventor, as well as the founder and former chief scientific officer of Sciotec Diagnostic Technologies GmbH. When this study was conducted, A.M. was the associate and external consultant of the company. V S. was employed as a PhD student and F.F. was employed as the head of R&D at Sciotec Diagnostic Technologies when this study was conducted. The other authors have no competing interests to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Austrian FFG (Forschungs Förderungs Gesellschaft) Project COIN, Nr. 286415 to Christoph Wiesner and Project BRIDGE, Nr. 2647553 to Harald Hundsberger.
