Abstract
Neurodegeneration and neuroinflammation are key features in a range of chronic central nervous system (CNS) diseases such as Alzheimer’s and Parkinson’s disease, as well as acute conditions like stroke and traumatic brain injury, for which there remains significant unmet clinical need. It is now well recognized that current cell culture methodologies are limited in their ability to recapitulate the cellular environment that is present in vivo, and there is a growing body of evidence to show that three-dimensional (3D) culture systems represent a more physiologically accurate model than traditional two-dimensional (2D) cultures. Given the complexity of the environment from which cells originate, and their various cell–cell and cell–matrix interactions, it is important to develop models that can be controlled and reproducible for drug discovery. 3D cell models have now been developed for almost all CNS cell types, including neurons, astrocytes, microglia, and oligodendrocyte cells. This review will highlight a number of current and emerging techniques for the culture of astrocytes and microglia, glial cell types with a critical role in neurodegenerative and neuroinflammatory conditions. We describe recent advances in glial cell culture using electrospun polymers and hydrogel macromolecules, and highlight how these novel culture environments influence astrocyte and microglial phenotypes in vitro, as compared to traditional 2D systems. These models will be explored to illuminate current trends in the techniques used to create 3D environments for application in research and drug discovery focused on astrocytes and microglial cells.
The CNS in Three Dimensions
Life as we know it exists in three dimensions within space. From
Recent technological advancements, particularly high-throughput sequencing of genomic DNA and expressed RNA, have enabled an unparalleled opportunity for studies ranging from single-cell phenotyping all the way up to large-scale population genetics. These advances have uncovered a hitherto unappreciated plethora of roles for glial cells in health and disease. Both astrocytes4–9 and microglia10,11 have now been shown to exhibit a great range of developmental, regional, and species-dependent specializations in gene expression profiles and cellular phenotypes. Historically, as with basic neuroscience, drug discovery efforts into human neurodegenerative conditions have also been dominated by neuronal biology. Recent years have, however, seen a great resurgence in interest and research into glial cells within the context of human disease. The association between glia and human CNS disease and damage has been known for many decades. Astrocyte and microglial “activation” and inflammatory responses have long been observed to accompany CNS damage in both chronic neurodegenerative states and acute brain trauma.12–15 A classic and well-known example of this is the presence of activated microglia and astrocytes around amyloid plaques in Alzheimer’s disease. Similarly, the “glial scar,” which is largely composed of reactive astrocytes, is known to arise in response to acute and chronic CNS disease or damage, where it can inhibit effective functional regeneration.
16
The role of glia as drivers of human neurodegenerative diseases, as opposed to being simply responders that have the potential to exacerbate damage, has been an area of intense debate for decades.
17
The aforementioned advances in sequencing technology during the past decade have opened the door for increasingly large and reliable genome-wide association studies (GWAS) of human neurodegenerative diseases, such as for Alzheimer’s disease (AD),18,19 Parkinson’s disease (PD),
20
multiple sclerosis (MS),
21
amyotrophic lateral sclerosis (ALS),
22
and frontotemporal dementia (FTD).23,24 These GWAS have identified numerous genes with enriched or specific expression in glial cells that associate with neurodegeneration in populations. These include genes such as apolipoprotein E (ApoE) in astrocytes and triggering receptor expressed on myeloid cells 2 (TREM2) in microglia, which are linked with AD risk.25,26 Notably, genes with roles in glial inflammation pathways, are particularly highly represented in neurodegeneration GWAS data for AD, PD, ALS, and FTD.23,26,27 Thus, the concept of human CNS disease being not simply exclusively
The complex 3D organization of cells and tissues in vivo contrasts with the vast majority of research in cell biology that has been undertaken throughout many decades to understand it. For practical and technical reasons, the majority of reductionist cell biology—with neuroscience being no exception—has been undertaken in “traditional” 2D cell culture systems. Such culture systems typically use rigid and flat, plastic or glass tissue cultureware. Although traditional 2D tissue culture has played an absolutely pivotal role in building knowledge and understanding of cell biology and disease mechanisms, we are now reaching the limits of its physiological relevance when considering more complex cell and organ systems, developmental biology, and candidate compound or drug effects. 34 CNS drug candidates traditionally have a high attrition rate, and a component of this problem may be attributable to early discovery efforts using inappropriate and poorly representative CNS in vitro 2D models. Recently, preclinical drug discovery has somewhat seen a transition back toward using phenotypic screening approaches, rather than target-based discovery methods, in which candidate molecules and targets are screened in assay systems that aim to closely recapitulate the complex native cellular environment found in disease states. Historic data suggests that phenotypic screening approaches are highly valuable for the successful discovery of novel medical entities and have greater potential to replace animal screens. Indeed, it has been demonstrated that the contribution of phenotypic screening to the discovery of first-in-class small-molecule drugs in recent years exceeds that of target-based approaches. 35 Adoption of phenotypic assays for CNS and other drug discovery efforts is, however, limited by advances in recapitulating the native 3D in vivo environment in multiwell plates, the format required for medium- to high-throughput pharmaceutical compound screening.
There is now little doubt that 3D in vitro models for practically all cell types are more in vivo–like and thus more biologically relevant than 2D models. 36 The shortcomings in 2D biological systems become even more apparent as the complexity of the tissue model increases, such as for biological barriers (gut, blood–brain barrier, or skin), multicellular systems (e.g., immunobiology), complex organs such as the brain, or a complex disease biology with variable environmental conditions (e.g., differing oxygen gradients within tumors).37–41 This review will now focus on describing current efforts to culture and characterize astrocytes and microglia in 3D formats, with an emphasis on those approaches that are likely to be most amenable and applicable to drug discovery in the near term. We will briefly describe traditional 2D methods of culturing glial cells, outline the features of the 3D formats that may be most translatable to facilitating high-throughput drug discovery efforts using glial cells, summarize key efforts in the field to date, and describe how they may affect the development of new therapies for devastating neurodegenerative and neuroinflammatory human diseases of the CNS.
Traditional 2D Glial Cell Culture
Glial cells are traditionally cultured, at least as a starting point, as
Although 2D culture systems have proved to be powerful workhorses for CNS research, they have caveats to be considered. Astrocytes and microglia are the cellular “first responders” in CNS disease and damage, and they will mount inflammatory and adaptive responses to changes in homeostasis. Clearly then, it should come as no surprise that such cells are able to sense that they are in an abnormal environment when cultured in 2D, on rigid plastic, and in the altered gas composition of tissue-culture incubators. Through their dimensionality and stiffness, classic 2D systems may contribute to an artificial in vitro glial reactivity, and shift the normal homeostatic morphology and function of astrocytes and microglia away from that found in vivo. Indeed, many experimental observations from previous decades have indicated this to be true. Astrocytes cultured in 2D show increased expression of the same reactivity markers that are upregulated on disease and traumatic injury in vivo and that accompany phenotypic changes which attempt to contain and counteract damage. This classically includes intermediate filament proteins such as glial fibrillary acid protein (GFAP) and vimentin.5,9,53 Similarly, microglial cells in 2D culture also show expression of numerous genes that are associated with activation and proinflammatory responses, including cytokines, chemokines, complement cascade proteins, and cell-surface molecules that facilitate immunological processes.54–57 These cells are not fully activated in 2D, however, because further activation and proinflammatory responses can clearly be seen following treatments with proinflammatory stimuli or toxins. Astrocytosis, for example, has been described as a graded process rather than an all-or-nothing event. 58 A more “fully” activated astrocyte phenotype can be induced in vitro by transforming growth factor-beta (TGFβ) or cyclic adenosine monophosphate (cAMP) analogs, 59 and proinflammatory responses in astrocytes and microglia can be induced from very low basal states in vitro by molecules such as bacterial lipopolysaccharide (LPS) or cytokines like tumor necrosis factor-alpha (TNFα).
The drive to develop better in vitro models for investigating glial biology, and for application to drug discovery, thus has multiple facets. First, there is the recognition of the apparently artificial phenotype and activation status of in vitro cultured astrocytes and microglia that clearly indicates that better, more in vivo–like models are required. In addition, the occurrence of glial scarring as a pathological mechanism has also influenced the field of in vitro 3D glial culturing, not only from the point of view of modeling the glial scar itself, but also within the field of biomaterials and neural regeneration using implanted devices. Many groups are working toward using biomaterials and scaffolds to implant devices into the brain and spinal cord to serve as regenerative units for repair or replacement of damaged regions of the CNS. Implanted biomaterial scaffolds may be able to act as conduits to promote neuronal outgrowth through a region of damage to facilitate functional recovery, for example. The implantation of many devices, however, has been demonstrated in animal models to trigger astrocyte and microglial invasion, proinflammatory responses, and glial scarring, which can act to block neuronal regeneration. Thus, research is ongoing to investigate glial reactivity in these devices and minimize their activation and proinflammatory responses to facilitate device integration and neuronal repair in vivo. This is conceptually the same goal as trying to develop in vitro models that mimic normal, in vivo–like glial phenotypes for investigational studies and drug discovery. Promoting more quiescent and homeostatic glial phenotypes in vitro will provide a valuable platform for basic biology and drug discovery research, allowing investigation of the transformation of astrocytes and microglia from homeostasis to disease-responsive, or even disease-causing, states. Although the majority of the technical developments in 3D in vitro glial studies have thus far been conducted in low-throughput formats, they are most certainly amenable to miniaturization into HTS plate-based modes, where they could subsequently be applied to drug screening for compounds that may modulate neurodegenerative and neuroinflammatory mechanisms, in a more relevant and in vivo–like model system.
Key Features of Frequently Used Biomaterials for Investigating Glial Cell Biology in 3D Formats In Vitro
Hydrogels
Hydrogels are networks of polymers or proteins that may be cross-linked to create surface coatings for cell attachment in vitro. Hydrogels may be either natural (purified) or synthetically made, and they can be used in a range of tissue culture formats for 3D cell culture. 60 Primarily, hydrogels are used as coatings for any type of surface to facilitate improved cell attachment and viability. In certain applications, however, they may be used to encapsulate cells and subsequently be layered onto a matrix or surface.61,62 Natural hydrogels are typically made up of ECM proteins, such as collagen, laminin, fibronectin, or hyaluronic acid. Being endogenous cell-surface molecules, they are naturally compatible and effective in promoting a range of cellular functions, such as attachment, proliferation, and long-term cell viability. Hydrogel use for cell culture is well validated in the literature and widely used in cell biology, with the ease of handling and coating onto surfaces, such as on microwell plates, being an obvious attraction for their adoption and continued use.63,64 Although natural hydrogels present an opportunity for improvement of the cell culture models, there are also a number of inherent considerations for their use. Being naturally derived (and often purified from animal sources), these components have an inevitable variability between preparations and can also contain contaminants, such as growth factors, that may interact and interfere not only with cellular responses in assays but also with the assay reagents themselves. To overcome some of these challenges, synthetic hydrogels have been developed based on nonnatural, biologically inert polymers that include polyethylene glycol (PEG), 65 poly(vinyl alcohol), 66 and alginate. 67 These synthetic hydrogels are highly reproducible and simple to manufacture, making them an attractive proposition. Although they may not offer the intrinsic biocompatibility of the natural polymers, they can be modified and tuned to change the mechanical properties. This is becoming an increasingly important factor in the use of stem cells in 3D culture models. 68
Scaffolds and Solid Platforms
The use of solid scaffolds for the culture of cells in a 3D microenvironment has grown rapidly in recent years. The aim of a scaffold or solid platform (synthetic or natural) is to reproduce structural features that are found naturally within the ECM and required for a cell to adhere to and function normally. Developments in this area mean there are now a range of scaffold substrate materials available, including metal, plastics, and polymers. The ability to control and optimize many scaffold parameters, including molecular composition, structure, stiffness, and porosity, has stimulated significant development and interest in the use of polymer-based scaffold technology. Current 3D scaffold technologies are largely based on the use of polystyrene or biodegradable polymers such as polycaprolactone (PCL), polylactide (PLA), and polyglycolide (PGL).69–72 The use of a scaffold-based approach allows cultured cells to easily exchange nutrients, gases, and waste products, and it can prevent the development of a necrotic core and hypoxic microenvironment, as is often seen with spheroid-based models. The use of biomaterial scaffolds thus provides a significant opportunity for areas such as regenerative medicine and tissue-engineering applications. There are a number of clinical scenarios in which a functioning implant would require a biodegradable scaffold that, after implantation, is remodeled by the body and replaced by native tissue to restore original function.73,74 The scaffold must provide the implanted cells with support for growth and allow them to remain viable without initiating an immunological response, an important consideration for clinical product development. From an in vitro perspective, scaffold-based applications have increased significantly through development of a range of different fabrication techniques. Stereolithography (bioprinting), microextrusion, microarray, and electrospinning are now all used to produce a 3D scaffold to support cell growth in vitro.75–78 Compared to other 3D technologies, scaffold-based approaches are rapidly advancing in terms of their integration into HTS or compound toxicity studies. Scaffolds are now commercially available for applications using 6-well up to 384-well microplate formats and thus can now be more easily integrated into HTS workflows and technologies. There still remain challenges with scaffold technology—particularly in terms of compatibility with imaging systems and recovery of cells from the matrix—but these are likely to be readily surmountable given the current pace of technological developments.
Modeling Astrocyte Biology in 3D Culture
The astrocytes of the brain and spinal cord are belatedly becoming recognized as the unsung heroes of the CNS.79,80 During the past century, and in particular in the past 30 years, the fundamentally critical roles that astrocytes play in regulating brain homeostasis have increasingly become more apparent. Far from being space-filling cells, research in the preceding decades has uncovered an astoundingly broad range of critical CNS functions for astrocytes, including the regulation of the brain’s ionic homeostasis and extracellular environment, regulation of neurotransmitter levels, establishment and maintenance of the blood–brain barrier, and now even their own signaling networks that run in parallel to neuronal network activity.58,80 Reactive astrocytes are a key pathological feature of most neurodegenerative diseases, including AD, PD, and ALS, and in brain trauma. In the case of brain tissue damage through disease and trauma, astrocytes form the key component of the glial scar.13,14,16,81 This is a complex, 3D tissue structure consisting mainly of highly reactive astrocytes and deposited ECM molecules, particularly chondroitin sulfate proteoglycans (CSPGs). Reactive astrocytes and glial scarring may have some neurotrophic and regenerative role, such as through the production of growth factors like brain-derived neurotrophic factor (BDNF), but they also constitute an inhibitory environment for neuronal regeneration and can severely impede functional recovery from CNS damage. Due to its complex 3D nature, the glial scar has also been very difficult to accurately model in standard 2D systems. In vivo, astrocytes are regionally, morphologically, and likely functionally heterogeneous, but they can be broadly categorized as being protoplasmic or fibrous.4,6,7 They assume a highly complex morphology, particularly in the human brain, with intricate branched processes that interact with neurons and other cell types.82–84 In vitro, astrocytes in mixed glial culture tend to be large, flat, and polygonal. Cultured astrocytes have been classified as Type 1 (polygonal) or Type 2 (branched), but the relationship of these phenotypes to fibrous and protoplasmic astrocytes in vivo is still unclear, and they may represent, at least partially, artefactual features of being cultured in rigid 2D systems.
The development and application of novel 3D culture systems can be expected to advance the modeling of in vivo astrocyte phenotypes and functions significantly. Recent work investigating the in vitro culture, and phenotypic properties, of astrocytes in 3D formats has broadly focused on low-throughput matrices of hydrogels or nanofibrillar scaffolds. Much of the emphasis in these studies has been placed on the investigation of reactivity status and glial scarring. Although these various hydrogel and nanofiber scaffolds are significantly different in terms of their composition and structure, many common features have arisen from research using them for astrocyte culture in 3D, including effects on cell viability, shape, motility, and the expression of genes associated with in vivo responses to damage and disease, such as GFAP.
Morphological Aspects of Astrocytes Cultured in 3D Collagen Hydrogels
Several groups have now reported that primary rodent astrocytes can be directly seeded into collagen gels, where they successfully establish and subsequently adopt more in vivo–like branched, stellate, or ramified morphologies that are far more reminiscent of their normal appearance in the CNS compared to the flat, polygonal cells that grow in 2D culture85–89 ( Table 1 ). Astrocytes in collagen gels are reported not to proliferate to a significant degree, which may reflect a less astrogliotic phenotype. 87 Although apparently dead cells are present in the 3D matrix with this culture method, these may represent those already dying and damaged from the initial cell isolation, and the seeded astrocytes are otherwise healthy and apparently nonreactive. 87 Astrocytes in 3D collagen gels are more motile than their 2D counterparts on collagen, and they are able to extend filopodia over distances of some 400 µm. 87 Recent investigations have further extended observations into how 3D collagen hydrogel matrices can influence astrocyte morphology, and may even recapitulate some of the developmental and morphological heterogeneity that is observed in vivo. A recent study demonstrated that, when growing primary astrocytes on collagen in 2D or in a 3D collagen hydrogel, different astrocyte morphologies can be observed throughout time in culture. 85 After 4 days in vitro, a variety of morphologies can be observed, including round, bipolar, and stellate astrocytes. GFAP+ve cells in 2D are large and flat, with those in 3D being smaller and more spherical. Moreover, a greater proportion of cells in 3D collagen hydrogels assume in vivo–like bipolar or stellate morphology. Fascinatingly, the bipolar cells morphologically resemble the radial glial cells that are present in the developing brain, whereas the stellate cells more closely resemble mature in vivo astrocytes. Prolonging culture time in 3D hydrogels to 10 days reveals an additional morphological phenotype that resembles in vivo perivascular astrocytes, showing characteristics such as apparent end-feet. 85 Gene expression analysis of the cells indicated that the “mature” stellate and perivascular-type astrocytes in 3D expressed GFAP and S100B, but not markers of immature glial cells such as nestin. 85 The bipolar, radial glia-like cells in 3D culture, however, did co-express markers of developing cells, for example nestin, further supporting the notion that they represent a developmental astrocytic phenotype. 85 This data raises the fascinating prospect that by growing astrocytes in 3D, in collagen hydrogels at least, dimensionality may to some degree recapitulate the developmental ontogeny seen in vivo, with spatial orientation having a more dominant effect on cell morphology and proliferation than other factors such as serum. 85 Primary astrocytes thus seed into and survive well in naturally derived hydrogel matrices and assume more realistic cell morphologies, with greater ramification and smaller, more spherical volumetric shapes, when compared to those in 2D, which are extremely flat with few projections.
Summary of published literature reporting astrocyte and microglial cell types cultured in different 3D in vitro systems.
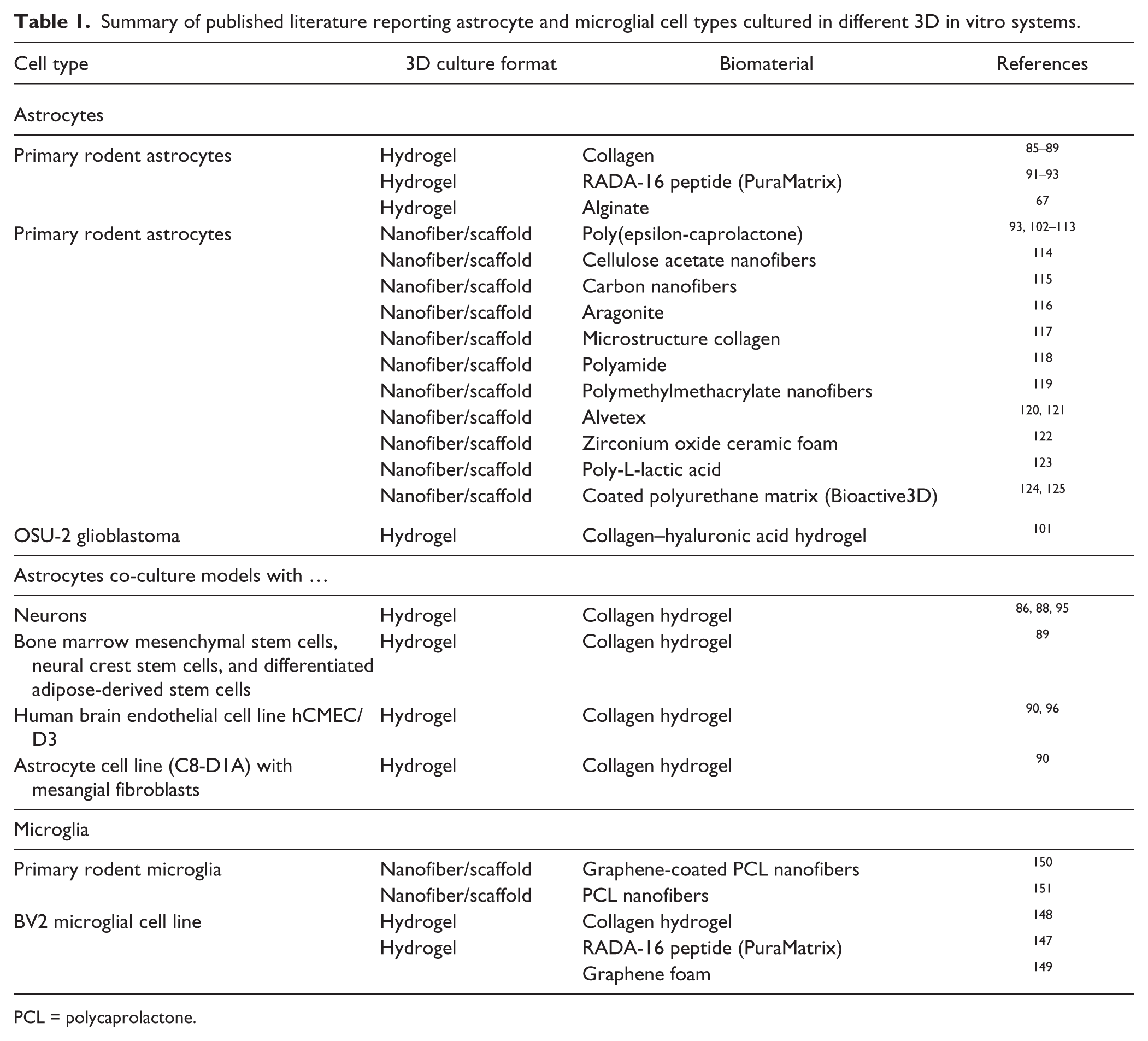
PCL = polycaprolactone.
Reactivity and Activation Status of Astrocytes Cultured in 3D Collagen Hydrogels
Astrocytes cultured in collagen hydrogels show significantly reduced levels of gliosis and reactivity markers, with reduced expression of GFAP, vimentin, aquaporin-4 (AQ4), and CSPGs. 87 When TGFβ is applied to astrocytes grown in 3D collagen hydrogels for 15 days, to model triggering of gliosis following CNS damage, astrocytes become more ramified and produce more inhibitory CSPG molecules. 87 Perimeters of vimentin and AQ4 staining increase significantly with TFGβ treatment, along with release of the proinflammatory cytokine interleukin-6 (IL6), indicating astrocyte hypertrophy and reactivity. 87 An astrocyte cell line (C8-D1A) is also observed to proliferate in collagen gels, 90 unlike some observations with primary astrocytes, 87 although GFAP expression is reduced compared to 2D formats in both studies using this cell line.87,90 Application of TGFβ increases GFAP expression in C8-D1A cells, indicating enhanced astrocyte reactivity in response to this growth factor in the 3D gel. This enhanced reactive state induces a decrease in the expression of a key tight-junction protein, claudin-5, in a co-cultured endothelial cell line, demonstrating a gliosis-like response that is reminiscent of pathological processes that induce breakdown of the blood–brain barrier in vivo. 90
Culture of Astrocytes in Other Hydrogel Matrices
In addition to collagen-based hydrogel matrices, some laboratories have also investigated alternative materials, such as those derived from RADA-16 or alginate. RADA-16 (also known as Puramatrix) is a self-assembling ionic peptide with four repeats of the RADA (arginine–alanine–aspartic acid–alanine) amino acid sequence. The RADA-16 peptides have a high propensity to self-assemble into hydrogels composed of superstructured β-sheets. RADA-16 peptide hydrogels have been particularly applied in the field of nervous tissue regeneration, where they support the survival of neural stem cells and subsequently differentiated astrocytes.91–93 Other groups have successfully seeded astrocytes into alginate hydrogels and used these in co-culture models with neurons and meningeal fibroblasts. 94
Co-cultured 3D Hydrogel Models
Models of astrocyte culture in 3D collagen matrices have been extended further to more complex co-culture systems alongside neurons86,88,95 and stem cells. 89 Astrocytes that have been aligned in 3D by compression into collagen gels provide a more permissive environment for allowing primary dorsal root ganglion neuron (DRG) neurites to project, in a model that could have potential applications as a conduit for spinal cord regeneration. 86 A complex co-culture interaction system, in which astrocytes are cultured in a gel format adjacent to gel-bound primary DRG cells, allowed the investigation of reciprocal astrocyte–neuron interactions in a 3D environment, through a physically contiguous association between the two cell types. 88 An additional elegant 3D model to investigate the cellular responses of astrocytes to stem cells, developed to investigate potential in vivo responses to stem cell therapies, has also been described; it involves seeding astrocytes into 3D collagen gels and then layering bone marrow, mesenchymal or adipose-derived stem cells on top of the astrocyte-filled gel. 89 A similar technique has recently also been applied to generate a 3D model of the blood–brain barrier, seeding a human brain endothelial cell line (hCMEC/D3) on top of an astrocyte-filled collagen gel.90,96 Primary astrocytes can also migrate into 3D collagen gels, and this migration is significantly potentiated if the gel is infused with fibroblast growth factor-2 (FGF2). 97 Three-dimensional collagen gels have also been shown to be permissive for the differentiation of primary rodent neural stem cells (NSCs) into astrocytes.98–100 Finally, in an attempt to further mimic the protein–glycosaminoglycan ECM environment of the brain extracellular space, normal human astrocytes have been seeded into collagen–hyaluronic acid hydrogels, where they showed a spherical morphology, in contrast to a spindle-like morphology from transformed glioblastoma cells in the same environment. 101
Culture of Astrocytes in Nanofiber Matrices
Significant progress has recently been made on the culture of astrocytes in a variety of synthetic, natural, and hybrid nanofiber matrix systems ( Table 1 ). These studies used 2D biofilms of the nanofibrillar scaffold material for comparison with 3D nanofibrillar formats, in either random or aligned orientations. Investigations of astrocyte culture and biology using nanofiber scaffolds have predominantly used PCL and its derivatives.93,102–113 Other matrices such as cellulose acetate, 114 carbon nanofiber, 115 aragonite, 116 microstructured collagen, 117 polyamide, 118 polymethylmethacrylate, 119 Alvetex,120,121 zirconium oxide ceramic foam, 122 poly-L-lactic acid, 123 and a coated polyurethane matrix termed Bioactive3D124,125 have also been used. These investigations have seeded astrocytes on nanofibers with a range of diameters, with studies variously using 0.8–3.6 µm; 102 0.665 µm; 103 1, 5, and 8 µm; 104 0.5 µm; 108 and 2.5 µm 123 nm fibers.
Astrocyte Adherence, Viability, and Proliferation on Nanofiber Scaffolds
Several groups have now described that primary astrocytes will readily seed into nanofibers of PCL and its derivatives.108–113 PCL itself is not toxic to astrocytes, and cell culture media “conditioned” by incubation with this biomaterial does not induce any toxic effects. 111 Although some groups report some evidence of dead and dying cells when seeding into nanofiber scaffolds, these may have carried over from the initial brain homogenization to isolate cells, and others have described the viable culture of astrocytes on nanofibers for several weeks in vitro. 108 When PCL is modified by oxygen plasma treatment to give oxygenated surface chemistry, astrocytes are observed to have slightly higher viability. 109 PCL-containing extract of the blue-green algae spirulina, either infused into the nanofiber matrix or incorporated into the nanofibers themselves, showed concentration-dependent effects on astrocyte growth, with lower concentrations promoting viability when grown on nanofibers.106,110 Overall, nanofiber topography has the dominant effect on the astrocytes, not the spirulina extract. 110 On PCL–ethylethylene phosphate (EEP) nanofibers, astrocytes seed but show reduced proliferation throughout 7 days in culture compared to those on PCL–EEP 2D films. 103 The astrocytes in 3D PCL–EEP showed reduced proliferation, as judged by incorporation of 5-ethynyl-2′-deoxyuridine (EdU) into DNA, and also higher apoptosis appears to occur. 103 This data suggests that it is the nanofiber topography and not cellular confluence that influences proliferation rate. 103 Hippocampal astrocytes seeded into collagen-coated PCL nanofiber mats also survive, albeit with viability slightly lower than on 2D plastic, for 1–14 days in culture. 102 Alvetex is a commercially available, highly porous polystyrene scaffold with pore sizes in the region of 40 µm. It is a substrate that is easy to prepare and physiologically inert. Alvetex has recently been applied to the study of astrocyte reactivity and interactions with neurons. Astrocytes can be successfully seeded in Alvetex, where they will adhere and survive.120,121 Astrocytes on Alvetex showed lower mitochondrial activity as judged by MTT assays, indicating either lower levels of cell proliferation or higher levels of cell death. 121 Postnatal astrocytes have also been co-cultured in Alvetex matrix with neurons, and electrophysiological recordings have been made through culturing on multiple electrode arrays (MEAs). Co-cultures in Alvetex on MEAs show lower levels of spike frequency but nonetheless have spontaneously active firing networks and active glutamatergic and GABAergic transmission. 120 A further iteration of a nanofiber format termed Bioactive3D was developed using polyurethane nanofibers sequentially coated with poly-D-lysine (PDL) and then the ECM protein laminin.124,125 Astrocytes have poor survival on polyurethane nanofibers, which is greatly improved by PDL–laminin. 124 Proliferation of astrocytes in Bioactive3D was also found to be lower when compared with 2D, as assessed by immunostaining for the nuclear cell proliferation marker antigen Ki-67 or for incorporation of the nucleoside analog EdU into DNA. 124 In addition to the nanofiber scaffold formats discussed here, astrocytes have also been described in 3D culture on carbon, 115 poly-L-lactic acid (PLLA), 123 and cellulose acetate 114 nanofibers. Astrocytes cultured on carbon nanofibers adhere preferentially to fibers with the lowest surface energy and on fibers with a diameter greater than 100 nm. 115 Astrocytes are also found to better adhere to PLLA nanofibers coated with fibronectin, rather than laminin or collagen. 123 Finally, astrocytes have been shown to be adherent and viable for up to 3 weeks on cellulose acetate nanofibers. 114
Astrocyte Morphology on Nanofiber Scaffolds
Culturing complex, branched cells such as astrocytes on nanofiber scaffolds immediately provides the opportunity for promoting more in vivo–like morphological phenotypes, because the fibers themselves can act as both a stimulus and guide for process formation. Indeed, although hydrogels have many advantageous properties for promoting more natural cell morphologies, they can be quite dense and may even inhibit process formation. Numerous groups have now reported similar observations that describe that both random and aligned nanofiber scaffolds promote a more stellate and in vivo–like astrocyte morphology, which is greatly different from the flat, polygonal cells that are observed in 2D culture on rigid surfaces.102,103,108,110–113,121,123,124 Astrocytes plated out onto 3D PCL nanofibers are stellate and branched compared to their flat, polygonal 2D counterparts on PCL films.110,111,113 On PCL–EEP films, astrocytes are flat with a large surface area and a crisscrossed GFAP staining pattern, whereas on nanofibers of the same substrate, cells were elongated and ramified, and exhibited condensed GFAP staining. 103 Indeed, the surface area of astrocytes on PCL–EEP 2D films was some 2–3-fold higher than that of cells growing on nanofibers of the same material. 103 On randomly oriented nanofibers, tight colonies are observed at 4 days in culture that expanded to fill the scaffold by 12 days. 108 On the aligned fibers, rectangular colonies aligned with the fiber orientation are observed. 108 Astrocytes on aligned nanofibers extend large processes greater than 300 µm in length, compared with 50–75 µm on random fibers. 108 GFAP-positive astrocyte branches are observed to penetrate distances of some 10 µm in both random and aligned nanofiber matrices. 108 Other groups have made similar observations indicating that astrocytes grow along the lengths of the nanofibers and adopt an elongated shape that is more in vivo–like. 102 Astrocytes have also been observed to extend process “bridges” across fibers and to contact adjacent cells. 102 Directly inducing changes in astrocyte morphology, through altering their reactivity status with compounds, has also uncovered differences in morphology between 2D and 3D matrices. In 2D cultures, the cAMP analog dibutyryl cyclic adenosine monophosphate (dBcAMP) or Rho-associated protein kinase (ROCK) inhibitors induce a stellate morphology with more cellular processes when compared to polygonal control cells; in 3D on PCL nanofibers, however, the same compounds induce tight clusters of ramified cells on randomly orientated nanofibers and astrocytes with elongated processes of 100–250 µm on aligned nanofibers. 112 More in vivo–like astrocyte morphologies have also been described on other nanofibrillar scaffolds. Astrocyte morphology on PLLA nanofibers in aligned and random formats has also been investigated and compared to 2D films of the same material. 123 Astrocytes form confluent cell layers on all PLLA topography formats coated with fibronectin, with random alignment of cells on the randomly organized nanofiber matrix and cells aligning along the aligned nanofibers. 123 On randomly oriented PLLA nanofibers, astrocytes extend processes of ~50 µm and up to 200 µm, whereas astrocytes on aligned PLLA nanofibers extend processes of ~200 µm and up to 400 µm long, in the direction of single fibers. 123 PLLA fibers were ~2.5 µm in diameter for both the aligned and random formats, indicating that fiber topography is the key determinant of differences in astrocyte growth between the two formats. 123 Indeed, cytoskeletal alignment with fiber direction was observed for astrocytes on PLLA aligned fibers, but not on randomly oriented or 2D films of the material. 123 In Alvetex matrix, both embryonically and postnatally derived astrocytes will extend intricate and extensive processes, exhibiting a morphology similar to that of cells in live brain slices.120,121 Differences in morphology from the different aged tissues are evident also; postnatally derived astrocytes appear more protoplasmic in appearance, whereas cells derived from embryos have smaller cell bodies. 121 Astrocytes on PDL–laminin-coated polyurethane Bioactive3D nanofibers also show a more in vivo–like morphology with long, complex cellular extensions, compared to flat cells cultured on similar substrates in 2D. 124 No differences were observed in astrocyte cell volume when quantitatively assessed using enhanced green fluorescent protein (EGFP) under the GFAP promoter. 124 Astrocytes in vivo are known to signal to each through gap junctions that contain connexin-43. A 59% reduction in expression of connexin-43 was observed in 3D culture with Bioactive3D compared to 2D. 124 Here, astrocytes in Bioactive3D only contacted each other via their fine processes, when compared to the far greater surface area for contact in 2D monolayer culture.
Astrocyte Migration on Nanofiber Scaffolds
Aside from within the developing brain, astrocytes and microglia tend to undergo migratory processes under conditions of CNS pathology, in which they will migrate to sites of damage or neurodegeneration. In a normal brain or spinal cord, both astrocytes and microglia keep their cell bodies generally in the same location; however, their multiple branches are motile and will move around to sample their local environment, maintain CNS homoeostasis, and detect disturbances.126,127 Establishing astrocyte motility assays in 3D could thus provide a useful model for investigating their normal brain surveillance mechanisms and migratory functions during CNS disease and damage. Astrocytes have been observed to infiltrate collagen–PCL scaffolds, but only when the nanofibers are micron size and not submicron. 102 Cell migration on fibronectin-coated PLLA nanofibers has also been investigated by establishing discrete astrocyte colonies using cloning rings. 123 Astrocytes on aligned fibronectin-coated PLLA nanofibers elongate and migrate along the direction of, but not perpendicular to, the nanofibers (~750 µm), but little migration is observed in 2D fibronectin–PLLA or randomly oriented nanofibers. 123 Using time-lapse microscopy, astrocyte extensions in 3DBioactive with PDL–laminin-coated polyurethane are found to be more motile, extending and withdrawing quicker than filopodia in 2D, 124 in a manner that appears to be more akin to their normal homeostatic function in the CNS.
Gene Expression Changes in Astrocytes Cultured on 3D Nanofiber Scaffolds
As with the previous models describing the biology of astrocytes seeded into hydrogels, investigation of astrocyte reactivity in nanofiber scaffolds is a key area of 3D model development for establishing an in vivo–like culture environment that more accurately represents cell behavior in both health and disease. Indeed, nanofiber scaffolds have numerous advantageous features that allow the fine elucidation of reactive astrocyte phenotypes in 3D culture, including amenability to imaging with greater clarity than hydrogel formats allow, and allowing the effective extraction of RNA and protein from cells without contaminating the samples with molecules from the hydrogel matrix. Several groups have investigated the comparative expression of panels of genes in astrocytes, with particular emphasis on genes related to reactivity and core astrocyte functions in vivo, such as antioxidant systems and handling of the excitatory amino acid glutamate.108,113,114,121,123–125 Glutamate is a neurotransmitter but also an excitotoxic molecule that kills neurons during neurodegenerative processes, and astrocytes play a key role in regulating extracellular levels of this amino acid in the CNS. On PCL nanofibers, genes for cell motility, chemokines, TGFβ-induced migration, glutamate transport, antioxidant defense, and the neurotrophin BDNF are increased in 3D culture compared to 2D. 108 Although 2D PCL also induced some gene expression changes, these were observed to be more pronounced in 3D. 108 Astrocytes cultured on PCL nanofibers, and incubated with the blue-green algae antioxidant C-phycocyanin after hydrogen peroxide treatment to induce oxidative stress, show enhanced expression of antioxidant enzymes such as CuZn superoxide dismutase and catalase compared to those cultured in 2D. 113 GFAP, synemin, vimentin, and nestin are all found to be lower in astrocytes cultured on Bioactive3D when compared to 2D, and these expression levels are closer to those in astrocytes freshly isolated from tissue. 124 Using Affymetrix gene chip profiling of astrocytes that were immuno-panned using glutamate–aspartate transporter (GLAST) antibodies, distinct global gene expression profiles are found between 2D and 3D cultures on Bioactive3D, although gene expression levels for the astrocyte markers GLAST, glutamate transporter-1 (GLT1), connexin-30 (cx30), fibroblast growth receptor-3 (FGFR3), and AQ4 are similar between both conditions. 124 Both 2D and 3D cultures show different overall expression profiles to freshly isolated astrocytes, however. 124 KEGG (Kyoto Encyclopedia of Genes and Genomes) analysis indicates that astrocytes cultured on 2D versus 3D substrates show differential expression for genes that modulate cellular proliferation, shape, and motility. This is consistent with the observed morphological and phenotypic data, although, notably, the gene expression profiles for both 2D and 3D do not match that of mature brain astrocytes. 124 Differences in the expression of genes between embryonically or postnatally derived astrocytes are observed on the Alvetex 3D matrix. Embryonically derived astrocytes exhibit lower gene expression of astrocytosis markers GFAP, lipocalin-2 (LCN2), Serpin3n, pentraxin-3 (PTX3), and connexin-43 (cx43); glutamate system transporters GLT-1 and GLAST; and the CSPGs known as structural maintenance of chromosomes-3 (SMC3) and chondroitin sulfate proteoglycan-4 (CSPG4/NG2), by RNA and protein. 121 Surprisingly, however, and in contrast to reports of 2D versus 3D culture on other hydrogel or nanofiber substrates, postnatally derived astrocytes do not show major changes in gene expression in 3D culture on Alvetex, with only minor changes in expression observed, and GFAP levels remaining essentially unchanged. 121 Heparin-binding epidermal growth factor–like protein (HB-EGF) is a growth factor that supports astrocyte survival in 2D, serum-free culture but does not alter their phenotype. 44 HB-EGF induces a strong proliferative response in astrocytes grown on Bioactive3D, and genes induced by this growth factor differ depending on whether the cells are cultured in 2D or 3D. 125 Astrocytes on Bioactive3D also show reduced expression of epidermal growth factor receptor (EGFR), further suggesting a less reactive phenotype is obtained. 125 Critically, although astrocytes grown in Bioactive3D appeared significantly less reactive, they respond equally in 2D culture to adenosine triphosphate (ATP) stimulation, as assessed by ERK phosphorylation, following 4 h of serum starvation. Astrocytes grown on Bioactive3D are thus minimally reactive, but fully responsive to physiological stimuli such as ATP and HB-EGF.124,125 Expression and protein levels of key genes for astrocyte function show a trend of upregulation on fibronectin–PLLA nanofibers, with glutamate transport and metabolism genes (GLT-1 and GLAST) and glutamine transporters [system N glutamine transporters-1 and -2 (SN1, SN2), and glutamate-ammonia ligase 1 (GLN1)] increasing, although glutamate–ammonia ligase-2 (GLN2) is decreased. 123 Accompanying this increase in glutamate transporter expression, functional glutamate uptake is increased by 35–50% in astrocytes grown on 3D fibronectin–PLLA nanofiber formats compared to 2D films. 123 This observation supports the concept of a less reactive, more homeostatic glial phenotype, whereby astrocytes have normal or increased ability to handle glutamate in 3D. Other groups have performed estimates of glutamate uptake in 2D versus 3D models using [ 3 H]-aspartate; however, no difference is reported between the different dimensional formats. 108
Astrocyte Activation and Functional Reactivity Status on Nanofiber Scaffolds
Astrocytes grown in 2D in vitro display phenotypic characteristics that resemble those seen in vivo during CNS trauma or neurodegenerative disease, such as changes in cell morphology as a result of increased expression of intermediate filament proteins like GFAP, vimentin, and nestin. Similarly, astrocytes in 2D may also increase expression of proinflammatory genes. These apparently “reactive” features in 2D culture are likely due, at least in part, to the unnatural constraints and stress placed on the cell when growing it in two dimensions on a rigid plastic substrate. The addition of serum to cell culture media may also trigger reactivity; glial cells would not normally encounter serum proteins in the CNS and thus may react to them in a manner similar to an in vivo breach of the blood–brain barrier by disease or trauma. Expression of GFAP in particular has been used as a marker of astrocyte reactivity in vitro. Astrocytes have long been demonstrated to express GFAP and CSPGs in culture, and these may be further upregulated in vitro by both molecular and mechanical induction that mimics in vivo diseased or damaged environments. 128 Numerous studies using nanofiber scaffolds composed of various biomaterials have now demonstrated that, compared to 2D culture on the same substrate, astrocytes on 3D nanofibers show reduced expression of GFAP and other intermediate filament proteins.103,108–114,118,124,125,129 These observations have been interpreted as a reduction in the activation status of astrocytes grown in 3D on nanofiber scaffolds, with decreased cytoskeletal stress responses as a component of this.
Astrocytes on PCL nanofibers, for example, show reduced cytoskeletal stress in 3D environments and have lower GFAP expression and increased G-actin, accompanied by stellate morphology and extensive arborization. 108 In these cultures, GFAP expression in astrocytes grown on random and aligned nanofibers is reduced by up to 80% compared to 2D cultures, and it is also accompanied by increased expression of genes with neuroprotective functions, such as BDNF and the glutamate transporter EAAT2. 108 ROCK inhibitors have been found to be additionally beneficial in reducing reactivity and promoting a cytotrophic astrocyte phenotype on PCL nanofibers. 112 ROCK inhibitors or cAMP analog compounds cause a reduction in F-actin and an increase in G-actin, indicative of stress fiber disassembly and the promotion of a less migratory, cytotrophic phenotype with increased cellular arborization. 112 Cyclic AMP analogs such as dBcAMP, which is known to promote a “reactive” astrocyte phenotype in vitro, 130 are found to induce a significant increase in process length on several 2D substrates, including poly-L-lysine (PLL), Aclar, and PLL-functionalized Aclar but not 3D nanofibers. 129 In addition, in these same cultures, GFAP and tubulin levels are lowest on nanofibers compared to the other substrates. 129 A lower induction of CSPGs is also observed following dBcAMP treatment, and ras homolog gene family member A (RhoA) signaling is depressed relative to cell division control protein-42 (Cdc42) only on 3D nanofibers, compared with 2D substrates. 129 Nanofibers, in this case polyamide, thus uniquely influence the differentiation and reactivity of dBcAMP-treated astrocytes. Untreated astrocytes on polyamide nanofibers show stellation via RhoA depression, which does not change significantly with dBcAMP treatment, and both conditions induce an in vivo–like morphology. 129 Association between GFAP and tubulin is also reduced on nanofibers, indicating a lower reactivity state. 129 Thus, morphological and biochemical differences in astrocytes responding to the “reactive” dBcAMP stimulus are modulated by surface topography. These studies indicate that 2D versus 3D culture can influence astrocyte phenotype and behavior via the RhoA–ROCK pathway. Indeed, when culturing astrocytes in 3D on polyamide nanofibers, preferential activation of Cdc42 over RhoA is observed, whereas RhoA is dominant over Cdc42 in 2D on glass. 118 This data is interpreted as showing a greater astrocyte reactivity in 2D on glass, associated with the presence of cytoskeletal stress. On nanofibers, however, astrocyte morphology is stellate and accompanied by long process formation, as opposed to lamellapodia and filopodia formation on glass. 118 The astrocytes on nanofibers show increased cell–cell interaction via their stellate processes, associated with depressed RhoA signaling. 118 Surface topography and material roughness thus influence preferential activation of the RhoA GTPase family, with demonstrable morphological consequences in astrocytes. 118 The nanofibers that promoted this phenotype were, in fact, the least rough and elastic. These observations are interesting in regard to the fact that astrocytes have been demonstrated by other research groups to be able to sense and respond to substrate stiffness. 131 In line with such observations, the response of astrocytes to substrates of differing stiffness has recently been investigated using the natural polysaccharide polymer cellulose acetate. Here, applying heat treatment to the biomaterial provides matrices of differing stiffness, and GFAP expression is reduced on 3D cellulose acetate nanofibers compared to 2D cultures. 114 Cellulose acetate nanofibers that were subject to heat treatment to stiffen them showed slightly higher astrocyte GFAP expression and increased synthesis of ECM components such as collagen and glycosaminoglycans. These observations indicate that fiber stiffness can affect astrocyte phenotype irrespective of nanofiber surface chemistry, topography, and density. 114 Such observations further support the hypothesis that matrix stiffness contributes to features of in vitro astrocyte reactivity such as intermediate filament organization and ECM deposition, and these observations may have implications for understanding and modeling glial scarring. It has been demonstrated that the astrocyte phenotype can be modulated by 3D culture even after a period of culture in standard 2D conditions. Transferring astrocytes cultured for 7 days in 2D culture to Bioactive3D reversed the high levels of intermediate protein expression back to a negligible baseline in only 1.5 days. 124 An additional cell stress marker, heat shock protein-70 (Hsp70), is also significantly downregulated on transfer from 2D to Bioactive3D. 124 This study further emphasizes the importance of culture topography on astrocyte reactivity. Experiments were carried out in the presence of serum and thus indicate that matrix topography, not reactivity to serum proteins, is the main driver of the astrocyte stress phenotype in vitro. 124 Astrocyte phenotypes in vitro thus appear to be malleable and able to switch, depending on the environment in which they find themselves. This observation may reflect how the cells alter their phenotype in vivo when circumstances change from normal and healthy to diseased or damaged states.
Modeling Microglial Biology in 3D Culture
Microglia are best known as the resident macrophages of the CNS. They survey their local environment for signs of injured or dying cells, proliferate and migrate to the site of damage, and initiate an innate immune response resulting in clearance of damaged cells and tissue remodeling. Beyond this classic role, microglia are now recognized to have additional functions that are broader than previously appreciated. 132 For example, microglia have now been demonstrated to have an important developmental role in synaptic pruning of weak neuronal synapses, and this role may extend to disease states in the adult brain.133–135 Activated microglia are a long-recognized feature of human neurodegenerative disease, and they are the major players in neuroinflammatory responses in many CNS conditions such as neurodegeneration, glioma, and stroke.15,136 Microglial phenotype and morphology in vivo differ substantially from 2D cultures of primary cells. 137 A major caveat of studying microglia in vitro is thus their clear propensity to lose many of their innate morphological and phenotypic characteristics when removed from their natural CNS environment. Activated microglia in vivo go through a striking morphological change as they participate in neuroinflammatory responses in the diseased or damaged CNS. 137 Microglia in the normal, healthy brain are highly ramified with many fine processes extending from their cell body in three dimensions, surveying their local surrounding tissue for perturbations. On activation during an inflammatory response, the microglial cell body undergoes hypertrophy, the fine ramifications retract, and eventually the cell becomes a round, amoeboid, macrophage-like cell. 137 Purified isolated cultures of microglia tend to display a more activated, amoeboid morphology than those in vivo, which have a more ramified morphology. 137 Several studies have claimed more in vivo–like, ramified microglial cells when grown in 2D, but these descriptions are still likely to be some way from being true recapitulations of normal morphology.138–143 Microglial morphology is thus a good, basic marker of activation status, although this phenomenon status is far subtler than just cell shape. Several studies have also indicated that the gene expression profile of cultured microglia differs substantially from that of cells freshly isolated from in vivo, with noticeable expression of activation markers such as proinflammatory and motility genes in 2D culture.54–56,144–146 Thus, as with astrocytes, a further advancement in the in vitro modeling of microglial biology and neuroinflammatory responses is to provide an environment in which a true, quiescent microglial phenotype can be attained, whereby the cells extend processes in all directions, interact with other CNS cell types, and receive and respond to stimuli as they would normally in the brain. Several groups have now begun to investigate these models; however, there are fewer reports focused specifically on in vitro microglial biology in 3D when compared with astrocyte studies ( Table 1 ).
Investigations of Microglia on 3D Hydrogel and Nanofiber Matrices In Vitro
A commonly used amoeboid microglial cell line, BV2, has been applied to 3D culture in PuraMatrix peptide hydrogel. In PuraMatrix, BV2 cells lose the characteristic amoeboid morphology seen in 2D cultures and instead display multiple ramifications. 147 Addition of ECM proteins to the scaffold results in reduced ramifications in this system. This was the first study to show that microglia cells grown in a 3D culture system more closely resemble microglial in vivo morphology. Aside from improving morphology characteristics, studies have also sought to ensure that the scaffold itself does not affect microglial cell viability, and crucially does not affect the inflammatory status of the cell. The response of BV2 cells to the proinflammatory toll-like receptor 4 (TLR4) agonist lipopolysaccharide (LPS) in a 3D culture system has been studied using a 3D collagen matrix. 148 BV2 cell viability is similar in both 2D and 3D cultures, even in the presence of LPS. The inflammatory response induced by LPS was measured by an increase in nitric oxide, the presence of CD40-positive cells, and levels of proinflammatory cytokines, all of which were similar in both 2D and 3D cultures, indicating that BV2 microglia cells grown in 3D cultures retained similar levels of cell viability and response to proinflammatory stimulus. 148 Graphene has emerged as innovative material on which to grow neural cells because of its electrical conductance properties, although questions remain as to whether the material may trigger neuroinflammatory responses. These potential issues have been investigated in a study that analyzed the neuroinflammatory properties of 3D graphene foam on BV2 microglia. 149 Although basal levels of proinflammatory cytokines were not significantly changed on exposure to 3D graphene, cytokine secretion was inhibited in LPS-stimulated BV2 cells. 149 A follow-up study used a sophisticated technique involving coating graphene onto electrospun PCL nanofibers. 150 This study reported the effects of microglia infiltration and inflammatory response on glial scarring around the scaffold. It was observed that although there is initially more microglial infiltration into graphene-coated scaffolds, microglial numbers significantly decreased throughout time in graphene-coated scaffolds versus uncoated scaffolds. Furthermore, the morphology of infiltrated microglia in the graphene-coated scaffold was ramified and nonphagocytic, indicating a resting or surveying microglia phenotype. 150 Primary microglia cultured on poly(trimethylene carbonate-co-1-caprolactone) [P(TMCCL)] electrospun fibers are smaller and more branched than their counterparts on films of this biomaterial, which remained round and amoeboid. 151 There were no significant differences in cytokine release, or myelin basic protein phagocytosis, on 2D versus 3D formats of this biomaterial in this study. 151 Finally, in a highly exciting recent development, the recent identification of a robust protocol for the differentiation of hiPSC microglial cells demonstrated that in vivo–like, ramified morphologies can be obtained when culturing the cells in 3D organotypic-like formats compared to 2D ones. 52
Application of 3D Astrocyte and Microglial Models for HTS Campaigns
A recognition of the higher validity of 3D cultures for recapitulating in vivo cellular environments in vitro is now well beyond doubt.34,36 Although more time-consuming, costly, and technically demanding, such techniques are now becoming far more routinely used in both academic laboratories and industrial drug discovery settings. The disadvantages of 3D culture versus standard 2D are, however, traded off against the more representative and, crucially for drug discovery, potential predictive nature of these models. Although relatively in its infancy, research into 3D culture models using glial cells, which are highly relevant to both normal CNS function and responses to neurodegeneration and trauma, is progressing and gathering pace. As outlined above, early investigations culturing astrocytes and microglia in 3D clearly show consistent features indicating that these in vitro environments can promote more in vivo–like phenotypes (summarized in Table 2 ). Astrocytes and microglia cultured in hydrogels and on nanofibers look morphologically more similar to their in vivo counterparts, and crucially they show more limited basal reactivity when compared with glial cells plated into classic 2D systems. Although these research efforts demonstrate a significant advancement in the in vitro modeling of glial cells, to meaningfully affect human CNS diseases they must be transferred into HTS platforms and used for successful compound screens. Furthermore, these screens must then show differential hit compound outputs, with better efficacy in, and predictivity for, human diseases when compared with 2D efforts. Such comparative screens have been reported for other, non-CNS cell types, such as within the field of oncology.152,153 Such side-to-side 2D versus 3D screens have shown that 3D cultures are able to identify different compounds between the two formats, with those identified in 3D likely to have greater relevance to the human clinical situation. There are, however, surprisingly few reported “traditional” HTS screens, even in 2D, that have been directly focused on glial cells such as astrocytes and microglia. This is likely a further reflection of the bias within CNS drug discovery toward neurons. Recent reports have described HTS screens conducted in 2D to identify compounds that could ameliorate oxidative stress 154 and edema 155 using astrocytes as the target cell type. Other efforts have used microglial cells and cell lines to screen for compounds in 2D that may counteract inflammatory and neurotoxic processes.156–158 Thus far, there are no reports of screening in 3D using astrocytes and microglia as the target cell type, although several groups have now reported 3D HTS screening platforms for neuronal systems, including human hiPSC cortical neurons 159 and neural stem cells.160,161 It is highly likely that such screening efforts using astrocytes and microglia will be applied to human CNS disease-relevant systems in the near future, given the excellent recent developments made in the field as outlined above.
Summary of the major phenotypic differences between astrocytes and microglial cells as they exist in their normal in vivo state and when cultured in vitro in 2D and 3D formats.
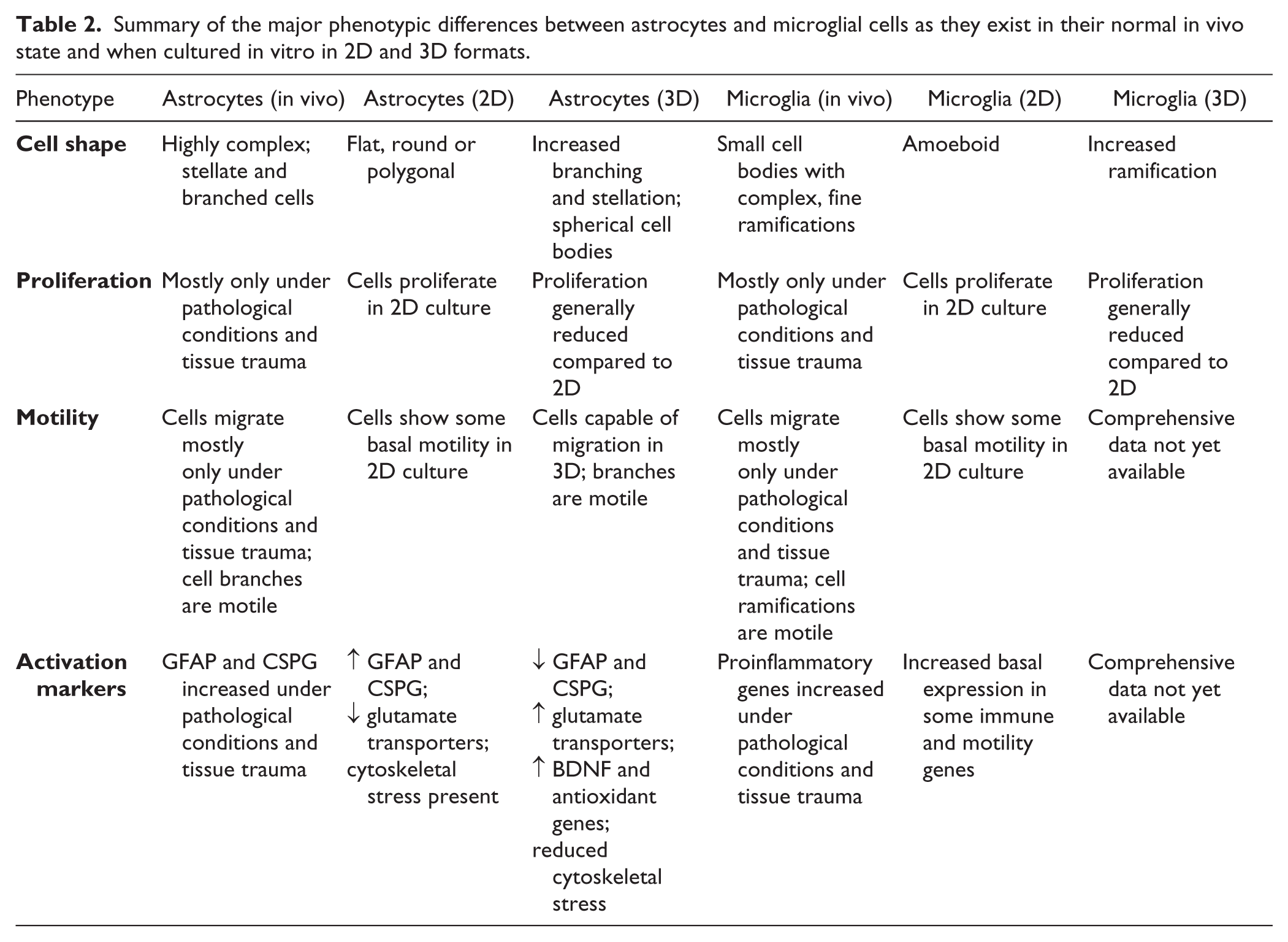
Indeed, several groups have now begun to use 3D culture systems to model neurodegenerative diseases such as AD.159,162–166 These models typically use disease-relevant cells derived from human stem cells and can exhibit cardinal features of AD that are poorly represented in 2D models, such as amyloid plaque–like structures and hyperphosphorylated tau. 167 Recently, descriptions of novel 3D models relevant to PD168,169 and FTD 170 have also been reported. Although focused on neuronal biology, introducing glial cells into these models will further increase and strengthen their validity. Beyond the currently used hydrogel and nanofiber scaffold systems described above, increasingly sophisticated methods for modeling the CNS, such as brain organoids,168,169,171 brains-on-chips, 172 modular bioengineered tissue construction, 173 and 3D-printed brain-like tissues, 174 are being developed and reported. Although these are technical triumphs and possess significant intrinsic potential for both basic biology and drug discovery, challenges remain for the application of these more advanced CNS models to drug discovery. In the near term, these models are most likely to be more suitable for profiling smaller numbers of lead compounds rather than for screening hundreds of thousands of molecules. Easily manufactured multiwell plate-based formats, such as electrospun nanofibers or hydrogels that are better able to model the 3D in vivo CNS environment while also being amenable to translation into HTS plate-based formats for screening large compound libraries, will likely drive more predictive CNS drug discovery efforts in the near future. For phenotypic screening, these 3D models should hopefully also be able to mimic disease states more closely and accurately.
Summary
As described here, the need for better 3D glial research models has been recognized elsewhere and should thus be a key area of focus for future research in basic biology and drug discovery for CNS diseases. 175 It is beyond doubt that 3D is better. To make an impact on the development of new therapeutics, however, 3D systems must also be translatable into “drug discovery modes” and amenable to HTS. Several groups have now performed HTS and high content imaging (HCI) screens in various 3D culture formats, largely in non-CNS contexts.176–183 It is now possible to clearly envision taking similar approaches to identify new compounds and targets for neurodegeneration and neuroinflammation. We are thus now in the exciting early stages of being able to screen disease-relevant 3D CNS systems. Indeed, HTS screens in 3D and using patient-relevant neuronal systems, such as hiPSC, are now being reported and demonstrate the feasibility of such an approach for complex CNS drug discovery. 159 Given recent descriptions of the previously underappreciated complexities of astrocyte and microglial biology in the context of normal CNS function and in disease, the continued development of in vitro methods to better model these fascinating cells in 3D will surely unlock the door and open new vistas for fresh drug discovery efforts to help patients with devastating neurological diseases.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
