Abstract
A novel halogen-free, formaldehyde-free, intumescent flame retardant, ammonium salt of erythritol phosphoric ester acid (AEPEA), was synthesized and coated onto cotton fabrics to increase the flame retardancy of cotton fabrics. Fourier-transform infrared, scanning electron microscopy, and X-ray diffraction were used to analyze cotton samples. The limiting oxygen index (LOI) of cotton treated with 300 g/L AEPEA reached 40.1% and the carbon length was 4.2 cm. After 50 laundering cycles, the LOI value could still reach 27.8%. Thermo-gravimetry and the cone calorimetry test were used to further investigate the flame retardancy and thermal stability of the treated cotton fabrics, and the results proved that the thermal stability of treated cotton was greatly improved. X-ray photoelectric spectrometry was used to analyze the carbon residue of flame-retardant cotton samples. In addition, the mechanical properties of the treated cotton only decreased slightly.
Introduction
Cotton fabrics are widely used in military, medical, clothing, and house decorative textiles, 1 because of their outstanding comfort, biocompatibility, and air permeability.2,3 However, the limiting oxygen index (LOI) value of cotton fabric is only 18%, which makes it a flammable textile. 4 Once there is a fire, cotton fabric will undergo rapid combustion, causing the spread of fire, endangering people’s lives and property,5,6 which also limits the applications of cotton fabric. Therefore, the flame-retardant modification of cotton fabrics can improve their safety and expand their application areas.
As the core of flame-retardant finishing, flame retardants have been most studied in recent years. Halogenated flame retardants have excellent flame retardancy and occupy a large proportion of flame retardants in the last decades, 7 which can play a role in both condensed phase and gas phase. In addition, the halogen-containing groups of halogen-based flame retardant can form covalent bonds with cellulose molecules, which will improve the durability of the treated cotton. However, halogenated flame retardants will release hydrogen halide during usage, which will endanger people’s lives and cause serious environmental pollution. 8 In recent years, some halogenated flame retardants have been banned by the European Union and the United States, so environmentally friendly flame retardants must be exploited to replace halogen-based flame retardants. 9
Phosphorus flame retardants have the advantages of low toxicity, environmental friendliness and high efficiency. During heating, phosphorus flame retardants will decompose into phosphoric acid and promote the formation of carbon layers on the surface of fabric, which can isolate the external heat and oxygen and play a role in both condensed and gas phases. So the phosphorus flame retardants has become a hotspot in recent years, and promising as substitutes for halogen flame retardants.10,11 The most widely used phosphorus-based flame retardants with the best flame-retardant effect are tetrahydroxy methyl phosphonium chloride and
To develop halogen-free, formaldehyde-free, and environmentally friendly flame retardants, some bio-based molecules have also been used for flame retardant in recent years, such as DNA, proteins, and chitosan. DNA contains phosphate groups which can be used as an acid source, deoxyribose units which can be used as a carbon source, and amino groups which can be used as a gas source. As a natural intumescent flame retardant, DNA has been used for flame retardant on cotton fabrics. And the limiting oxygen index of the treated cotton fabrics was increased from 18% to 28%.
14
However, the scarce sources and high cost of DNA greatly limit its application in flame retardant field. Protein contains P, N, and S elements, and can be used as a natural flame retardant. Bosco et al. coated whey protein on cotton fabric and the thermal stability of the treated cotton fabric was improved.
15
However, the protein molecule only contains a small content of flame-retardant elements and does not have good flame-retardant efficiency. Chitosan, as a natural polymer with
Erythritol (meso 1,2,3,4-butanetetraol) is prepared from starch with excellent stability when the temperature is lower than 200°C. 18 The polyhydroxyl groups in erythritol molecules can provide reactivity with many chemicals, so that erythritol can be widely used in food, medicine and chemical industry, etc. 19 In addition, When erythritol is introduced in flame retardants, the toxic smoke generated by combustion will be decreased. 20 In this work, a new environmentally friendly phosphorus–nitrogen based flame retardant was synthesized using phosphoric acid, erythritol and urea. The synthesized flame retardant has many reactive groups and can form P-O-C covalent bonds with cotton molecules, which give the treated cotton fabric good washing resistance. And the finishing process is simple, convenient, and easy to operate. The thermal stability and flame-retardant properties of the cotton fabric after finishing are significantly improved. Because the potential of hydrogen treatment process is nearly neutral, the mechanical properties of the fabric itself remained good.
Experimental
Materials
Cotton fabric (100%, 110 g/m2) was purchased from Zhejiang Guandong Textile Dyeing Garment (Zhejiang, China). Erythritol, phosphoric acid (85%), dicyandiamide and anhydrous ethanol were purchased from Sinopharm Chemical Reagent Co. All chemicals were used without further purification.
Synthesis of AEPEA
Erythritol (6.109 g, 0.05 mol) and phosphoric acid (23.05 g, 0.2 mol) were added into a 250 ml three-neck flask equipped with condensation reflux and magnetic stirring device. The temperature was raised to 120°C and stirred for 4 h. Then urea (24.024 g, 0.4 mol) was slowly added, the temperature was raised to 130°C and the mixture was stirred for 3 h. After cooling to room temperature, a white sticky solid was obtained. The crude product was purified with ethanol several times to remove the unreacted intermediate and urea. After drying in an oven at 70°C, the target product was obtained, and abbreviated as AEPEA. The synthesis of AEPEA is shown in Scheme 1 and analyzed by 1H NMR. The chemical shifts were as follows.
1H NMR (D2O, 400MHz) δ (ppm): 3.47(2CH2, H2, H3), 3.92(2CH2, H1, H4).
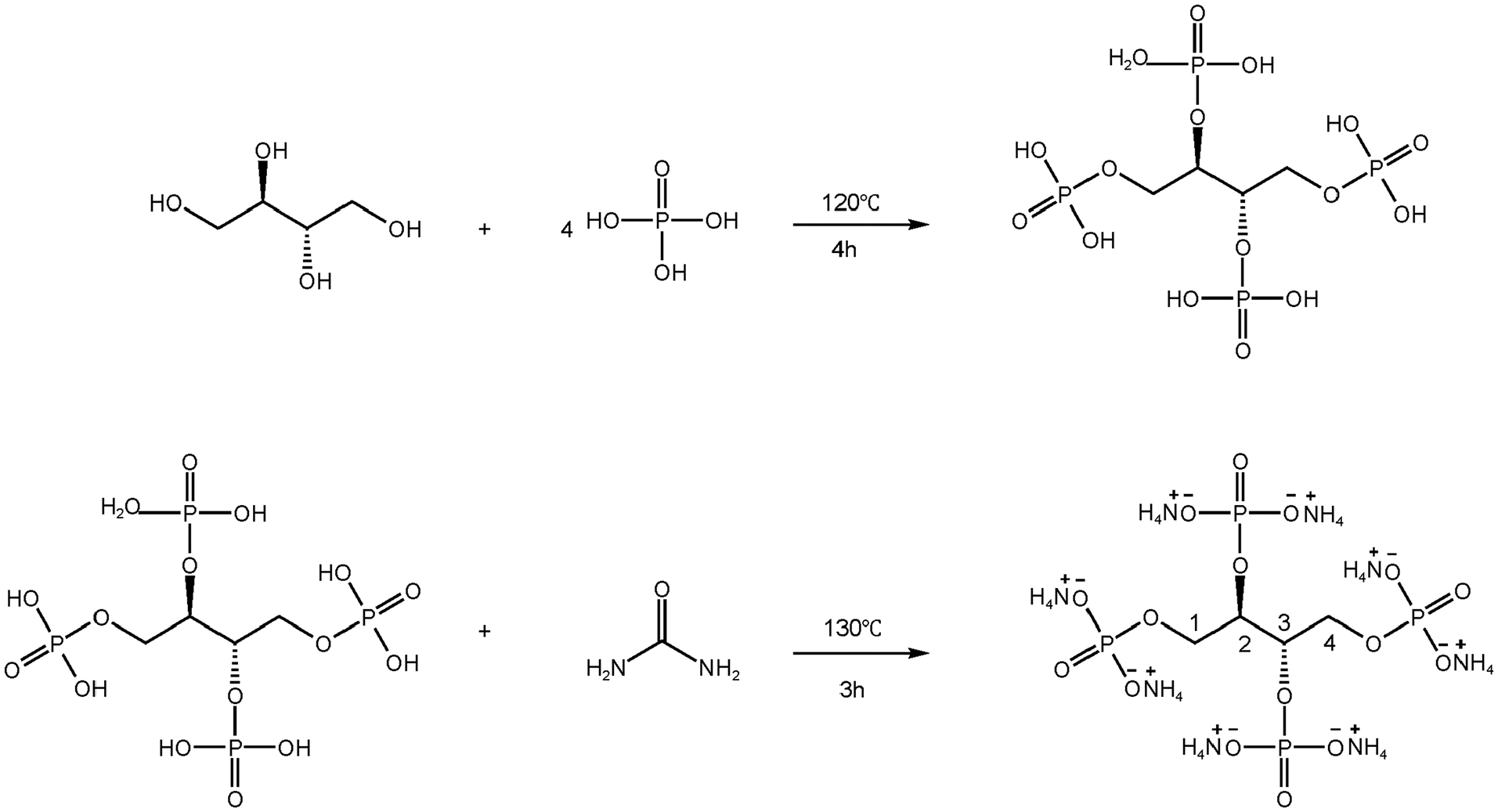
The synthesis route of AEPEA.
The treatment of cotton fabric by AEPEA
AEPEA was dissolved in deionized water to obtain finishing solutions (100, 200, and 300 g/L). Dicyandiamide (70 g/L) was added to the above solutions as catalyst. The cotton fabric was impregnated in the AEPEA solution for 30 min at 70°C with a bath ratio of 1:25. Then, the fabric was padded with 100% wet pick up. The operation was repeated two times. After that, the cotton fabric was dried in an oven at 80°C for 10 min, and then cured at 170°C for 2 min. The finished cotton fabric was washed with water to remove the unreacted AEPEA on the surface and then dried in an oven at 60°C. The treated cotton fabrics were named as FRC-10, FRC-20, and FRC-30. Scheme 2 shows the grafting reaction of AEPEA with cotton fabric.
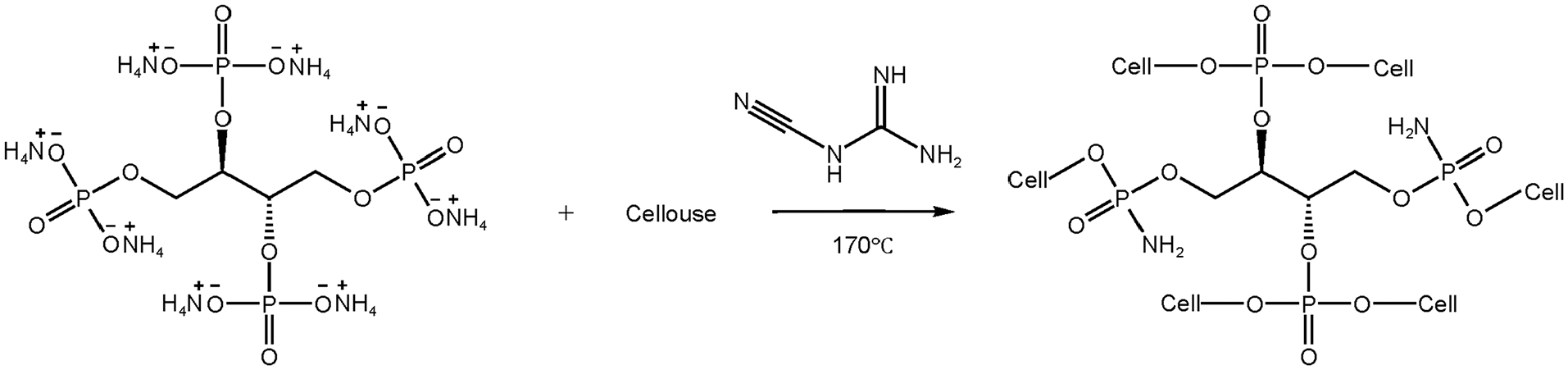
The grafting reaction of AEPEA with cotton fabric
The weight gain rate (WG) of the cotton fabric was calculated by equation (1).
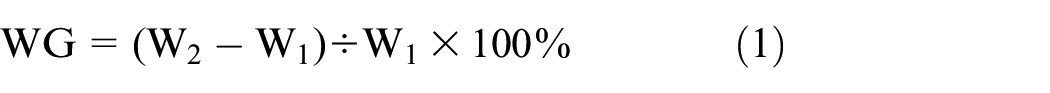
where
Characterizations
The cotton fabric before and after treatment was characterized using FTIR spectroscopy (NICOLET IS10, Thermo Scientific Company of the United States) with a scanning range of 4000–500 cm−1 and a resolution of 4 cm−1.
The crystalline structure of the cotton fabric before and after treatment was obtained by X-ray diffraction (XRD; D2 PHASER, Bruker Corporation, Germany) at 35 KW and 20 mA with a step size of 0.02°.
The thermal properties of cotton before and after treatment were analyzed using TGA 2 (METTLER Company, US) analyzer under a nitrogen atmosphere with a temperature range of 30–600°C and a temperature rise rate of 10°C/min.
Vertical flame test of cotton samples (80 mm × 300 mm) was performed on a vertical fabric flame resistance tester (YG(B)815D-1, Da Rong spinning instrument company, China) according to GB/T5455-2014. The ignition time and height were 4 s and 40 mm.
The LOI of cotton fabric samples (60 mm × 80 mm) was measured with a JF-3 type A limiting oxygen index meter (Beijing Xinsheng Zhuorui Technology Co., Ltd, China) according to GB/T5455-2014.
The surface morphology of control cotton fabric and treated cotton fabric before and after burning was observed using a scanning electron microscope (SEM) (Regulus 8100, Hitachi High-Technologies, Ltd., Japan) with an operating voltage of 15 kV.
Elemental analysis of control cotton, treated cotton, treated cotton after laundering cycles (LCs) and burning was performed using an SEM equipped with an energy-dispersive X-ray spectroscope (EDS) module with an operating voltage of 15 kV.
To test the durability of treated cotton fabrics, the cotton samples was washed in accordance with AATCC61-20062A standard. The washing time was 45 min, the temperature was 49 °C, and 50 steel balls were added. One wash was equivalent to five times of household washing.
The breaking strength of the control cotton fabric and treated cotton fabric were tested in accordance with the ASTMD-5034 standard using YG(B)026H-250 electronic fabric tester (Da Rong spinning instrument company, China); the size of the samples was 5 cm × 20 cm. Each sample was tested five times, and the average value was calculated.
According to the ISO 5600 standard, the burning behavior of control cotton and treated cotton (100 mm × 100 mm) was tested using cone calorimeters (FTT0007 UK) with an irradiative heat flux of 35 kW/m2.
The elemental composition of the treated cotton samples after the burning test was tested using a K-Alpha at 1486.6 eV (XPS).
The whiteness of samples was tested using Datacolor spectravision (Datacolor USA) according to standard AATCC 110–2000.
Results and Discussion
Structure of Cotton Fabrics
The FT-IR spectra of control cotton fabric and FRC-30 are presented in Figure 1(a). The control cotton and FRC-30 have a strong absorption peak at 3332 cm−1, which is the characteristic adsorption of the -OH group. The C-H bond absorption peak is located at 2904 cm−1. The finished cotton fabric has a C=O absorption peak at 1701 cm−1, which is formed when the cotton fabric is treated at a high temperature. 21 The new absorption peaks at 1231 and 834 cm−1 belong to P=O 22 and P-O-C bond. 23 The test result proves that AEPEA can form a P-O-C bond with the fabric.
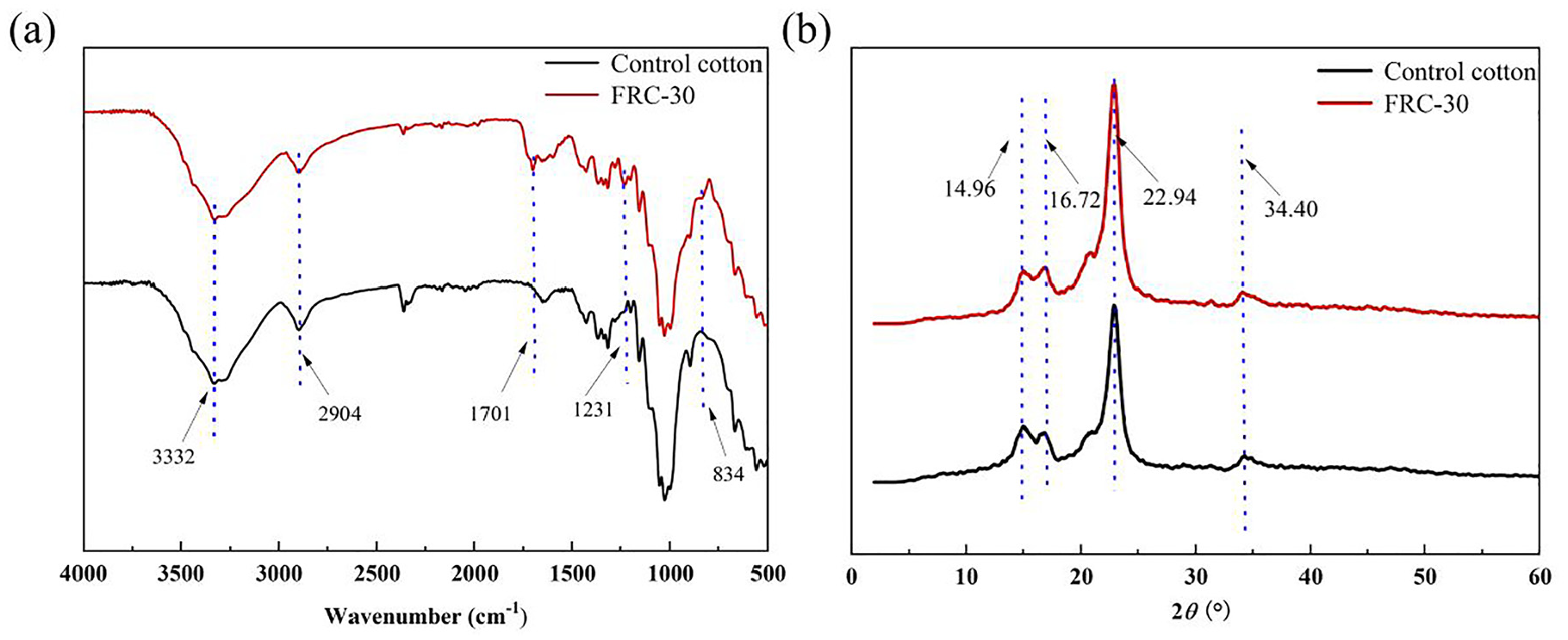
FTIR (a) and XRD (b) of cotton fabrics.
XRD tests were performed on the control cotton and the treated cotton, and the results are shown in Figure 1(b). The peaks of the original cotton fabric are located at 15.02°, 16.89°, 23.14°, and 34.94° corresponding to the crystalline structure of the cellulose I in the regions of (1–10), (110), (200), and (004). 24 And the peaks of the cotton fabric treated with AEPEA are the same as those the original cotton fabric, with only a slight decrease in the intensity of the peaks. This may be because AEPEA molecules and dicyandiamines enter inside fiber molecules during the impregnation process and react with the cotton fabric under high temperatures. 25
Flame Retardancy and its Durability
The flame retardancy and durability of samples were evaluated by LOI test and vertical flame test, and the results are shown in Figure 2 and Table 1. The LOI of cotton fabric before treatment is only 17.8% while the LOIs of the FRC-10, FRC-20, FRC-30 are 30.1%, 37.8%, and 40.1%, respectively. The weight gain rate of treated cotton fabrics is not high. With the increase in number of laundry cycles, the LOI value decreases. The LOI value of FRC-20 can still reach 28.5% after 30 LCs and the LOI value of FRC-30 can reach 27.8% after 50 LCs, which is higher than the international flame-retardant standard (26.0−28.0%). 26 So, FRC-30 can be regarded as durable flame-retardant cotton fabric. Since AEPEA is not simply physical coated on the cotton, it can form P-O-C covalent bonds with cellulose molecules under the catalysis of dicyandiamide during finishing. That is why the finished cotton fabric has excellent flame retardancy and durability. 27
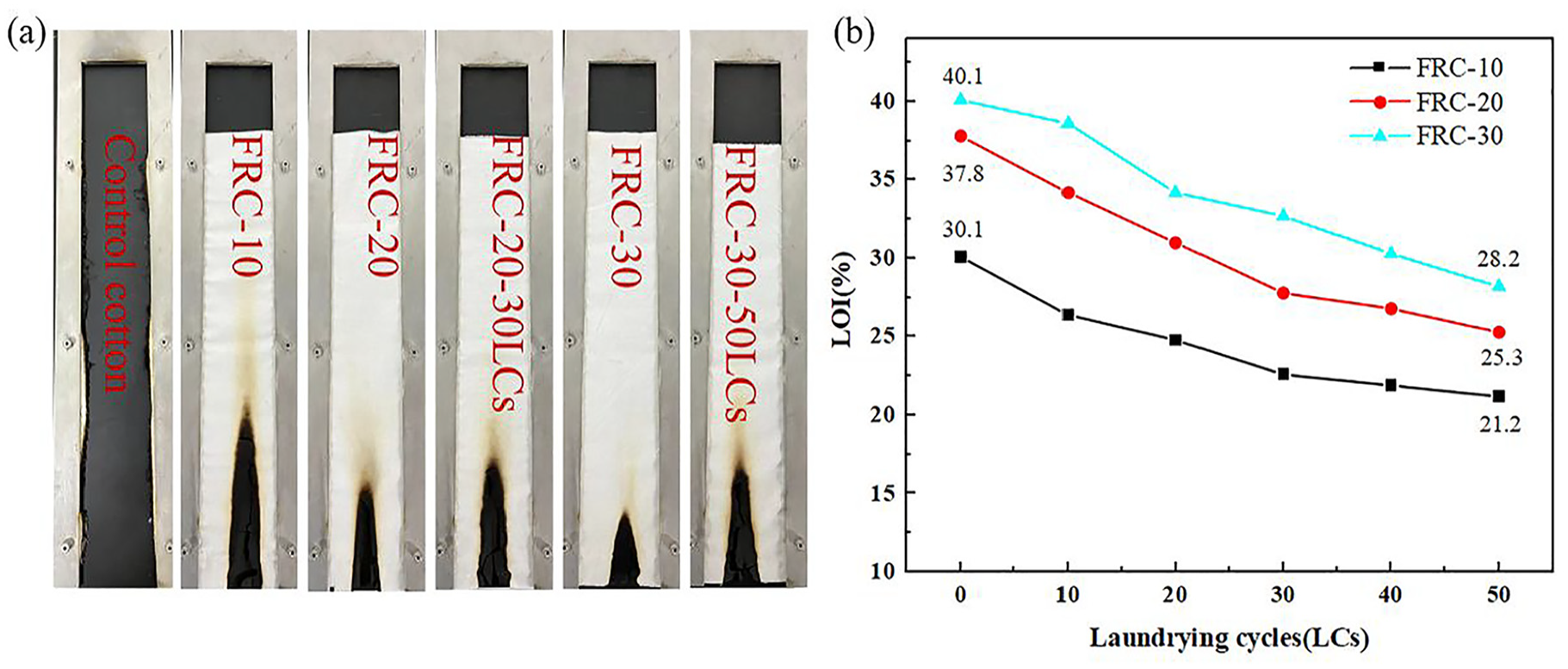
Cotton samples after vertical flame test (a); LOI of treated cotton fabrics after LCS (b).
LOI value and vertical flame test result of cotton samples.
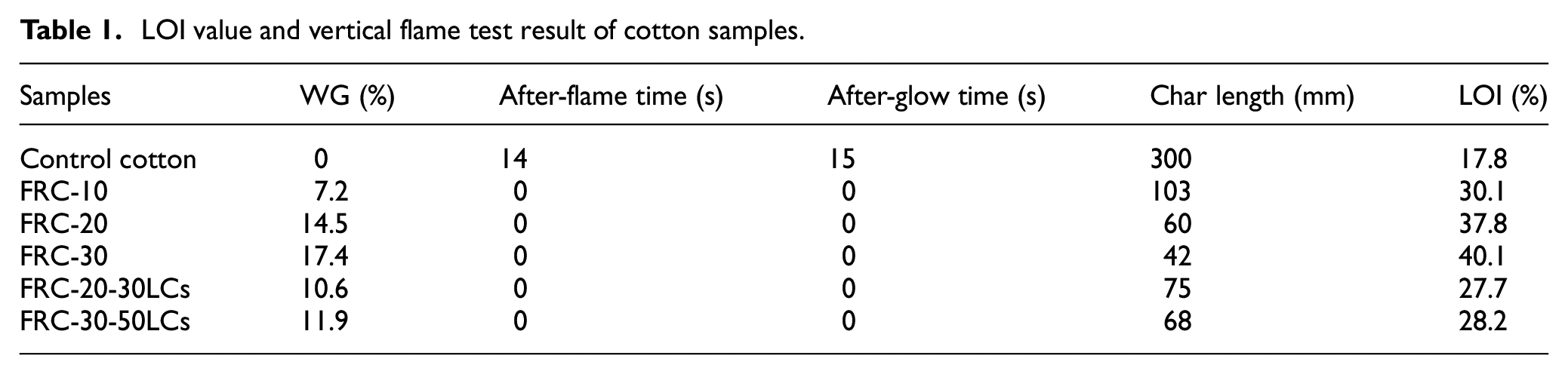
The cotton fabric is burned rapidly after ignition, with a post-flame time of 14 s and an after-glow time of 15 s. And there is almost no carbon residue after combustion. After removal of the flame, the treated cotton fabrics can self-extinguish immediately, and the burned area will form a complete carbon layer. The flame retardancy of treated cotton is further improved with the increase of AEPEA concentration. The char lengths of FRC-10, FRC-20, FRC-30 are 103, 60, and 42 mm, respectively. After laundering cycles, the treatment cotton samples still can self-extinguish, with no post-flame time and after-glow time. The test result proved that the treated cotton samples have excellent durability.
Thermal Stability
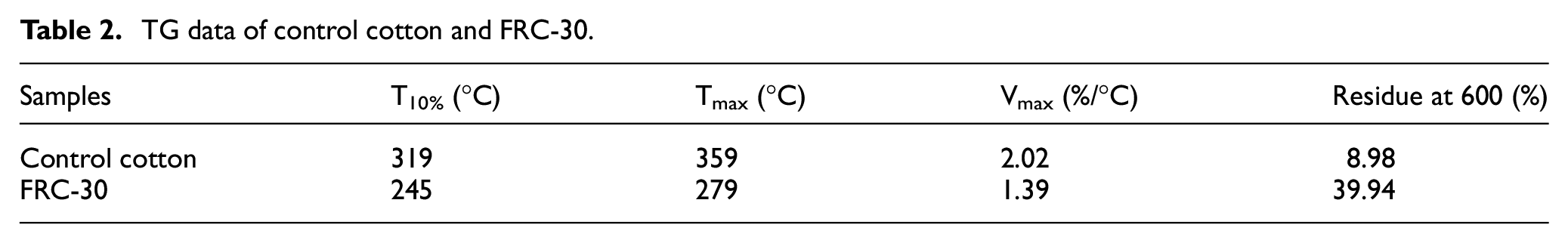
The TG and DTG curves of cotton fabric and FRC-30 are shown in Figure 3, which is used to analyze the thermal stability of samples, and the data is shown in Table 2. The T10% (temperature of 10% weight loss) of raw cotton fabric is 319°C, and the residual carbon at 600°C is only 9.48%. There is about 80% weight loss at 300–400°C. There are two main decomposition paths for cotton fabrics at this stage: one is the dehydration of sugar-based units, and the other is the formation of volatile levoglucosane-based sugar derivatives. 28 The T10% of FRC-30 is 245°C, which is significantly lower than that of original cotton fabric, but the carbon residual at 600°C reaches 39.94%. The reason is that AEPEA molecule decompose before the cotton fabric molecules during the heating process. AEPEA molecules decompose into phosphoric acid and poly phosphoric. The formed acids can promote the carbon formation of cotton fabric. And the formed carbon layer on the surface of the fabric can prevent the further decomposition of the fabric.
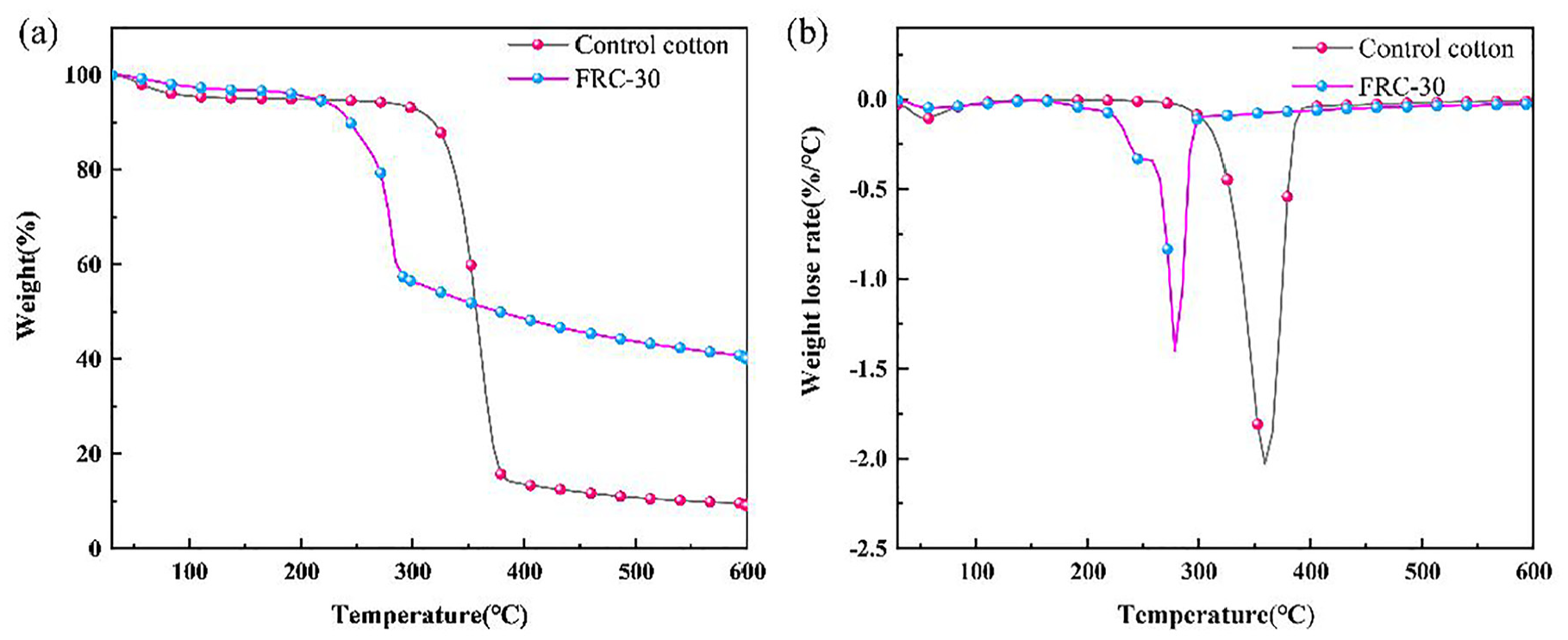
TG (a) and DTG (b) curves of control cotton and FRC-30.
TG data of control cotton and FRC-30.
The maximum thermal decomposition temperature of the original cotton fabric is 352°C, while the maximum thermal decomposition temperature of FRC-30 is 279°C, which is significantly lower than that of the original cotton fabric. Because the phosphoric acid and polyphosphoric acid produced by the decomposition of AEPEA molecules can catalyze the cotton fabric to carbonize it, the treated cotton fabric can quickly form a carbon layer to protect the internal structure, isolate the heat and achieve flame retardancy. 29
SEM-EDS
SEM was conducted to determine the morphological changes of cotton fabric before and after treatment with AEPEA as well as before and after burning, and the results are shown in Figure 4. The surface of the original cotton fabric is smooth, as shown in Figure 4(a1, a2), and the cotton fabric after treatment with AEPEA (Figure 4b1, b2) is almost the same as the original cotton fabric. 30 It is obvious that the cotton fabric after burning retains the original basic structure of the fabric (Figure 4d1, d2). AEPEA will decompose into phosphoric acid and polyphosphoric acid, which can promote the cotton forming a dense carbon layer. And there are some bubbles on the surface, which are due to the gases produced by the decomposition of AEPEA. 31

SEM image of control cotton (a1, a2), FRC-30 before (b1, b2) and after 50 LCs (c1, c2), FRC-30 after LOI test (d1, d2). EDS images of control cotton (a3), FRC-30treated cotton before (b3) and after 50LCs (c3), FRC-30 after LOI test (d3).
The elemental composition and content of the original cotton and FRC-30 were analyzed by EDX, and the relevant data and images are shown in Figure 4(a3, b3, c3, d3). It can be found that carbon, oxygen, phosphorus, and nitrogen are detected on the cotton fabric after treatment with AEPEA. After 50 washing cycles, phosphorus and nitrogen elements can still be detected with slight decreases. After burning, the content of phosphorus elements on cotton fabric increases significantly, which indicates that P element mainly plays a role in the condensed phase. And the content of carbon elements also increases significantly, which means that more carbon residue is kept in the degradation process. The experimental data further confirms the mechanism of AEPEA is promoting the formation of carbon layers in cotton fabrics to isolate heat and prevent combustion.
Cone Calorimetry Test
To further investigate the combustion behavior of cotton fabrics, the cone calorimetry test (CCT) was performed to simulate the combustion behavior of the fabrics in a real fire. The heat release rate (HRR), total heat release (THR), total smoke production (TSP) and residue weight curves are shown in Figure 5, and the related data are listed in Table 3. Raw cotton fabric is ignited at 20 s, while FRC-30 is not ignited. After the test, the residual amount of the original cotton fabric is only 2.98%, while the residual amount of FRC-30 reaches 23.74%, which is much higher than that of raw cotton fabric. The peak heat release rate of FRC-30 is 36.99 kW/m2, which is much lower than that of the original cotton fabric (321.28 kW/m2), indicating that AEPEA can significantly slow down the oxidative decomposition of cotton fabric. The increase of TPHRR (time of PHRR) also indicates that AEPEA can slow down the decomposition of cotton fabric. The total heat release of FRC-30 (8.19 MJ/m2) is lower than that of the original cotton fabric (18.88 MJ/m2). CO2 and CO are the two main gases released from the combustion of cotton fabric, the lower ratio of CO2/CO is, the slower the degradation rate of cotton fabric, indicating the better flame retardancy. 32 The CO2/CO ratio of FRC-30 is only 10.58, while the value for the original cotton fabric is 138.73, which is much higher than that of FRC-30. This is mainly due to the decomposition of AEPEA into phosphoric acid and poly phosphoric acid, which promotes the formation of a carbon layer on the surface of the fabric, isolates oxygen and heat, and further prevents combustion of the fabric. The TSP value of FRC-30 (0.58 m2) is higher than that of the control cotton (0.09 m2), which is due to the formed carbon layer preventing the further decomposition of cotton fabric. 33 The value of flame spread rate (FGR) is used to assess the fire safety factor of the cotton fabric by equation (2); a lower FGR means higher safety.

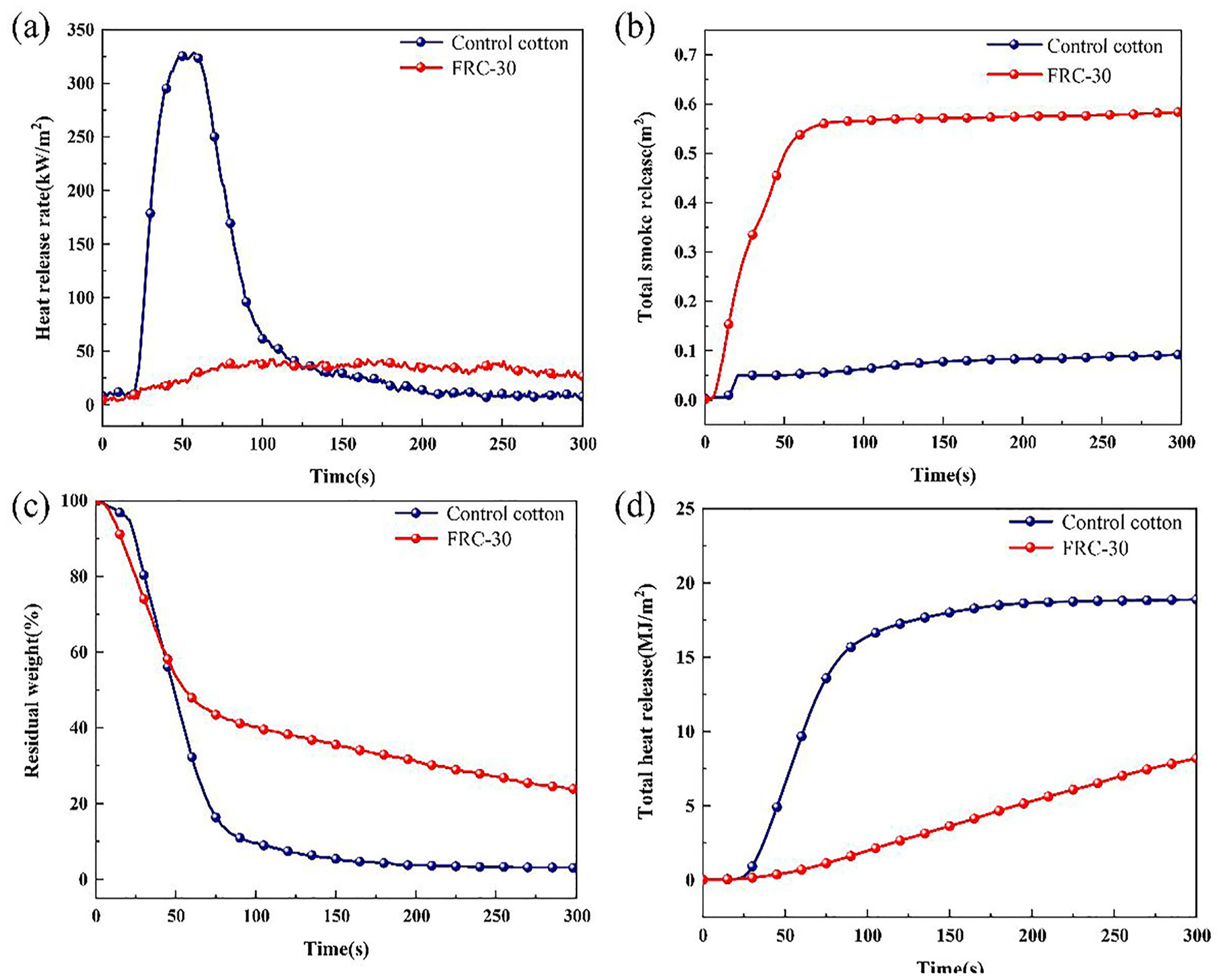
HRR (a), TSP (b), residue curves (c), and THR (d) of control cotton and FRC-30.
Cone calorimetry data of control cotton and FRC-30.
The FGR of FRC-30 is only 0.39, which is significantly lower than that of the original cotton fabric (5.6), indicating that the flame propagation of the cotton fabric treated with AEPEA is greatly slowed down and safe in practice. 34
XPS of Carbon Residue
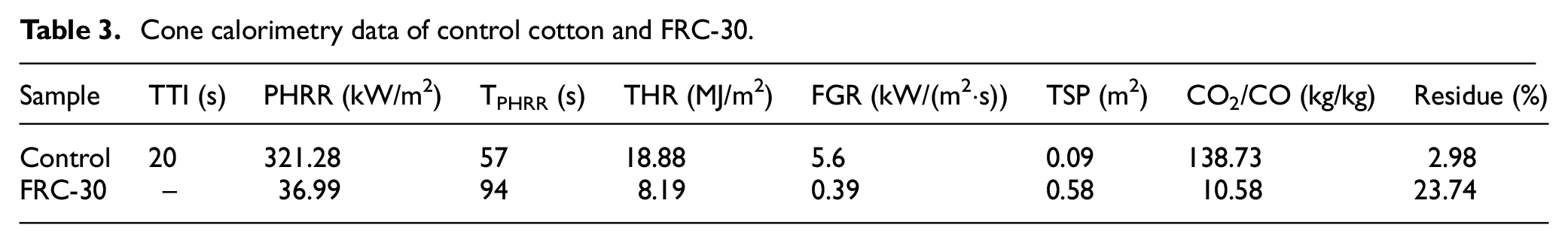
To further verify the flame-retardant mechanism of AEPEA, an XPS test was conducted on the vertical burning samples to analyze the elemental composition and bonding of the treated cotton fabric after burning. The test result is shown in Figure 6. The carbon residue of FRC-30 has phosphorus and nitrogen elements after combustion. The strongest peak at 284.8 eV corresponds to C-C and C-H, 35 which also indicates the main bonding mode of C element in cotton fabric. For the C1S spectrum, there are two characteristic peaks, C-O-C and C-O-P at 285.6 eV, and a weak peak at 288.4 eV, C=O, which is caused by the high temperature treatment of the fabric and corresponds to the FTIR test results. 36 As shown in Figure 6(c), the O1S image after burning of the FRC-30 has two absorption peaks, the absorption peak of P=O at 531.3 eV and the characteristic peaks of C-O and P-O at 532.9 eV. 37 As shown in Figure 6(d), the P2P image of the finished cotton fabric has two absorption peaks, the characteristic peak of P-O-C at 133.9 eV and the characteristic peak of P=O at 134.7 eV. Based the XPS results, AEPEA can catalyze the carbon formation of cotton fabric. 38
XPS spectra of FRC-30 carbon residue (a), and XPS spectra of treated carbon residue C1S (b), O1S (c), and P2P (d).
Flame Retardant Mechanism
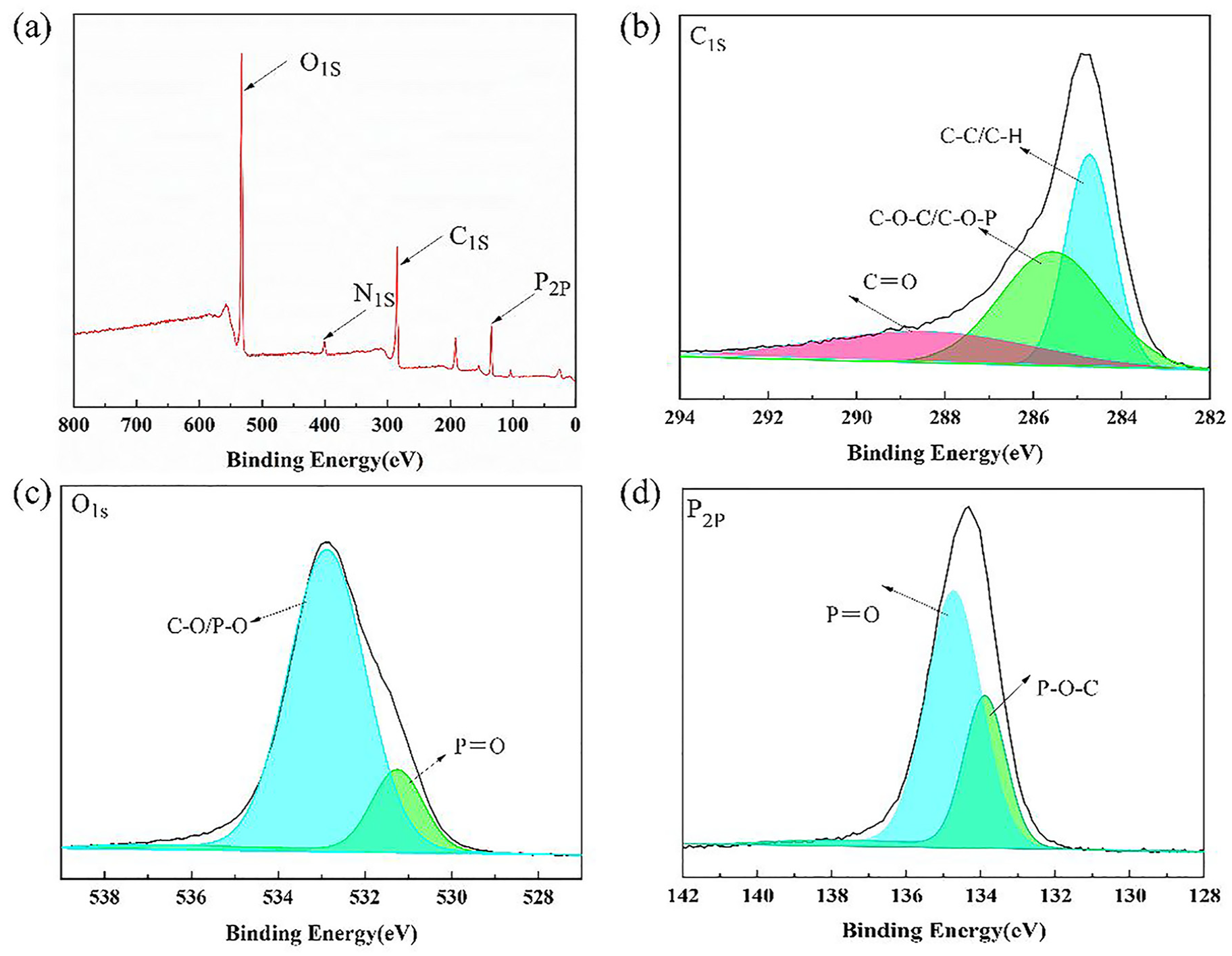
Based on the above analysis of the treated cotton fabrics, the possible flame- retardant mechanism is speculated and shown in Figure 7. AEPEA can play a role in both the gas phase and the condensed phase. When the cotton fabric is heated, AEPEA molecules preferentially decompose and degrade to produce phosphoric acid and poly phosphoric acid, which can promote the formation of a dense carbon layer to isolate oxygen and heat, preventing further oxidation and decomposition of the fabric. 39 AEPEA molecules will also produce free radicals (PO), which can capture the (H) and (OH) free radicals generated by cotton fabrics, reducing the concentration of combustible gases to slow down the degradation of cotton fabrics. 40 In addition, CO2, H2O, and NH3 will be produced during degradation, 41 which can reduce the concentration of oxygen and slow down the oxidation and decomposition and play the flame retardant in gas-phase.
The possible flame-retardant mechanism of AEPEA.
Physical Properties
To assess the effect of the treatment process on the cotton fabrics, the whiteness and tensile strength of the cotton samples were measured. The results of the tensile strength and whiteness of the cotton fabric before and after treated with AEPEA are shown in Figure 8. With the increase of the amount of AEPEA, the tensile strength and whiteness of the cotton fabrics are decreased. The loss of whiteness and tensile strength may be caused by the reaction between AEPEA and cellulose macro-molecule. The warp tensile strength of FRC-30 is decreased to 505 N and the weft tensile strength is decreased to 230 N, respectively, and above 75% of the strength is maintained compared with the original cotton fabric. The results show that after the treatment, the tensile strength and whiteness of cotton fabric are reduced, but are within the acceptable range and do not affect the practical usage of cotton fabric.
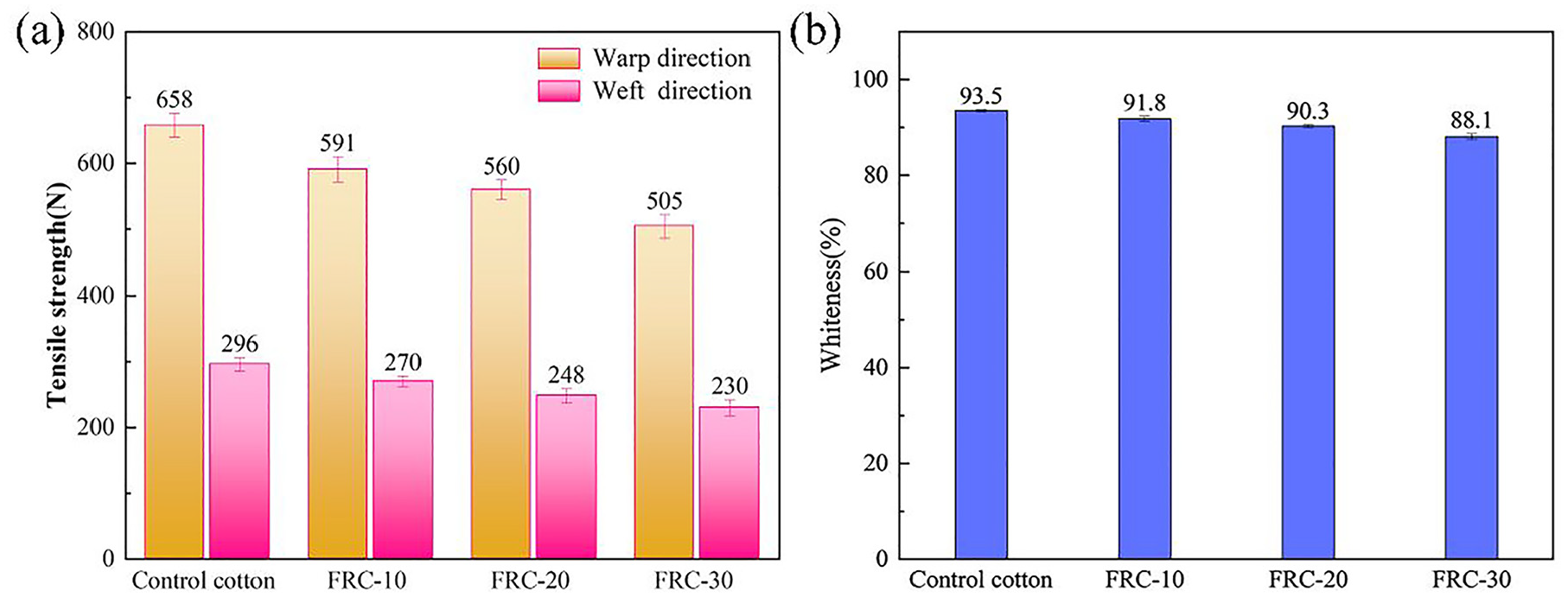
Tensile strength (a) and whiteness (b) of samples.
Conclusions
An environmentally friendly intumescent flame retardant was successfully synthesized and used in the finishing of cotton fabrics. The flame retardancy of cotton fabric was significantly improved after treatment, and the LOI value of FRC-30 could reach 40.1%. After 50 LCs, the LOI value could still reach 27.8%, indicating excellent durability of the treated cotton fabric. XRD and SEM results indicated that the flame-retardant finishing has almost no effect on the surface morphology and crystallinity of cotton fabric. The carbon formation ability of the fabric after treatment was significantly improved, and the carbon residue amount under nitrogen atmosphere was increased from 8.98% to 39.94%. The cone calorimetry test results showed that the THR and PHRR of the treated cotton fabric decreased by 56.62% and 88.49% respectively, and the carbon residue amount was significantly improved, indicating that the safety of the fabric was greatly improved. The cotton fabric was also able to maintain the original structure after burning. The tensile strength of the cotton fabric after finishing did not decrease too much and this would not affect usage.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Natural Science Foundation of China (no. 21706093).
