Abstract
With environmental and health issues being raised, researchers have shifted their study from fluorinated to fluorine-free waterproof agents. However, most of the reported fluorine-free water repellents exhibit a significant drawback in their poor stability and durability of water repellency. In addition, the use of higher concentrations in these repellents results in the fabric becoming stiff, which negatively impacts the hand of the treated fabrics. To address these issues, dendrimer-like waterborne polyurethane prepolymers were initially synthesized using isophorone diisocyanate, polytetrahydrofuran, N-methyldiethanolamine, and trimethylolpropane as the primary materials. In this synthesis, sorbitan tristearate served as the dendrimer-like blocking agent, while single-ended bis-hydroxypropyl silicone oil was employed as functional monomers to enhance the hand and water repellency of the prepolymers. Then, dendrimer-like waterborne polyurethane was finished on the cotton fabric using a pad–dry–cure procedure. The test results showed that the water contact angle of dendrimer-like waterborne polyurethane film increased from 104.28° to 107.00° with the content of single-ended bis-hydroxypropyl silicone oil increasing, and the water contact angle of dendrimer-like waterborne polyurethane-finished fabrics reached 148.6° alone with a standard spray test rating of 90–95. In addition, the water contact angle of dendrimer-like waterborne polyurethane-finished fabrics was still greater than 140° even after five standard washes. The single-ended bis-hydroxypropyl silicone oil addition facilitated similar hand to the original fabric in dendrimer-like waterborne polyurethane-finished fabrics with high concentrations, surpassing market water repellents. This work is of great significance for improving the stability and durability of water repellency, and the hand of waterborne polyurethane fluorine-free water repellents in the textile industry.
Keywords
Introduction
Because of their exceptionally low surface tension, fluorinated finishing agents are commonly employed in fabrics as waterproof finishing agents. However, there is a risk of carcinogenicity, and bioaccumulation associated with perfluorooctanoic acid (PFOA), perfluorooctanesulfonic acid (PFOS), and their related substances produced during the production and application of fluorinated finishing agents.1–4 International recognition of PFOA and PFOS as “permanent chemical substances” has been granted. 5 In recent years, there have been numerous reports on fluorine-free water repellents, which can be roughly categorized into polyacrylate, silicone, and polyurethane water repellents. 6 Polyacrylate has become the mainstream fluorine-free water-repellent due to its excellent processing performance and low cost. Following treatment with polyacrylate water repellent agents, fabrics exhibit superior water repellency but exhibit a stiff hand and prominent wrinkles. To mitigate these shortcomings, paraffin or silicone is typically compounded with the fabric. 7 On applying a silicone water repellent agent to the fabrics, the materials exhibit improved waterproofing and a softer touch. However, many silicones are primarily composed of two components, the technology involved in the processing is complex, and the emulsion easily demulsifies. 8 The waterborne polyurethane water repellent agent, a novel development that emerged in the 1960s, offers fabrics treated with it significant water repellency, enduring water resistance, and reduced wrinkling. In summary, it possesses outstanding comprehensive performance. In addition, the polyurethane material itself is characterized by its high strength, remarkable abrasion resistance, and excellent weatherability.9,10 In recent years, the application of and modification research on waterborne polyurethane have become research hotspots for related enterprises and university researchers. 11
Aliphatic compounds and silicone-containing compounds are commonly used as raw materials for the preparation of fluorine-free water repellents in the textile industry. 12 Zhong 13 used microwave heating to graft different aliphatic fatty chains onto cotton (cellulose) fibers to obtain the effect of water repellency. The results showed that the different fat chains could all lead to the acquisition of water repellency on the surface of cotton fabrics. But after 37 washes, the water contact angle (WCA) changed from 137° to 105°. Meanwhile, microwave heating made the yarn strength of cotton fabrics decrease by 50%. 14 An modified SiO2 with 3-methacryloyloxypropyl-trimethoxysilane (KH570), 2-(dimethylamino) ethyl methacrylate (DMAEMA), and 3-bromopropene in order. Then the modified SiO2, methyl methacrylate (MMA), butyl acrylate (BA), and octadecyl acrylate (SA) underwent a free radical polymerization reaction and could form a cationic SiO2/fluorine-free acrylate emulsion (SKD+/FFA). Fabrics finished with 12 g/L of SKD+/FFA reached a WCA of 147°, which only decreased by 5° after 50 washes. However, SiO2 nanoparticles are prone to agglomeration and thus destabilize the emulsion. This is a problem that needs to be addressed.
Silicone compounds are also used as fluorine-free water repellents because of their low surface energy, biocompatibility, and self-cleaning properties.15,16 Liu 17 created a self-crosslinkable polydimethylsiloxane (PDMS) compound for the superhydrophobic treatment of cotton fabrics, which demonstrated adequate water-repellency (a score of 90 according to AATCC Test Method 22-2010) and reasonable washing durability. Liu 18 prepared a glyceryl monostearate (GMS)-modified waterborne polyurethane (GWPU) fluorine-free water repellent with PDMS as the main chain and GMS as the branched structure, and investigated the performance of GWPUs with different GMS contents. The results showed that the combination of silicone chain segments and long-chain alkanes produced a synergistic water repellency effect. However, the silicone chain, as the main chain segment, could not be effectively enriched on the fabric surface to achieve a better water-repellency effect and give the fabric a smooth hand.
In this study, dendritic waterborne polyurethane (DWPU) fluorine-free water repellents were prepared using single-ended bis-hydroxypropyl silicone oil (SBHS) and sorbitan tri-stearate (ST) as the functional monomers. Fourier transform infrared spectroscopy (FTIR), particle size distribution analysis, zeta potential analysis, water resistance analysis, static contact angle, surface energy analysis, and X-ray photoelectron spectroscopy (XPS) were used to study the structure and properties of the prepared water repellents. In addition, we used a spray test, PhabroMeter, and fiber friction coefficient apparatus to study the water repellency and hand of the DWPU-finished fabrics.
Experimental
Materials
Isophorone diisocyanate (IPDI, AR (Analytical Reagent), 93 wt% purity), polytetrahydrofuran (PTMG, Mn = 1000 g/mol, AR, 98 wt% purity), sorbitan tri-stearate (ST, AR, 85 wt% purity), N-methyl diethanolamine (N-MDEA, AR, 99 wt% purity), dibutyltin dilaurate (DBTDL, AR, 95 wt% purity), trimethylolpropane (TMP, AR, 99 wt% purity), and 4-methyl-2-pentanone (MIBK, AR, 99.5 wt% purity) were purchased from Shanghai Titan Scientific Co., Ltd. The SBHS oil (Mn = 2000 g/mol, CP (chemical pure), 95 wt% purity) was supplied by Shanghai Huiyan New Materials Co., Ltd. Acetic acid (HAc, AR, 98 wt% purity) was purchased from Sinopharm Chemical Reagent Beijing Co., Ltd. The comparative sample X-9 of the DWPU water-repellent agent was provided by Fibrochem Advanced Materials (Shanghai) Co., Ltd.
Preparation of DWPU
The DWPU was synthesized in our laboratory on a laboratory scale. Prior to the synthesis, the required quantities of PTMG, TMP, and MIBK were accurately measured and placed in a four-neck flask equipped with a mechanical stirrer, thermometer, a spherical reflux condenser with a drying tube, and a dropping funnel. The mixture was dehydrated under vacuum at 110°C. Subsequently, the mixture was dissolved at 60°C with stirring, followed by the addition of IPDI. The mixture was then reacted at 90°C with stirring for 1 h to yield an –NCO-terminated prepolymer. SBHS was then introduced into the prepolymer, and after thorough mixing, three drops of DBTDL were added. The reaction was further continued at 90°C with stirring for 4 h to obtain an SBHS-grafted polyurethane prepolymer. Once the temperature of the reaction system was reduced to 70°C, an MIBK solution of N-MDEA (8.1% by weight of the total DWPU) was added to the prepolymer, with an MIBK-to-N-MDEA weight ratio of 2:1. The reaction was carried out at 70°C for an additional 0.5 h. After warming the reaction system to 90°C, ST was added as a capping agent for the isocyanate groups. The capping process was conducted for 4 h after adding two drops of DBTDL. On cooling the polyurethane to room temperature, HAc (80 wt% of the N-MDEA) was introduced into the system, and the neutralization reaction was performed for 15 min to yield the dendritic aqueous polyurethane. Gradually, an adequate amount of distilled water was poured into the polyurethane to form an emulsion. This emulsion was then dispersed using a high-shear emulsion machine for 2 min. Finally, MIBK was removed in a rotary evaporator to obtain the DWPU emulsion, with a solid content ranging from 20 to 25 wt%. The molar ratio of –NCO/–OH in the reactants was set at 1.5.
The SBHS content in DWPU was 0, 2, 5, 8, and 10 wt%, which were denoted as DWPU-0, DWPU-2, DWPU-5, DWPU-8, and DWPU-10, respectively. The flowchart for the synthesis of the DWPU is shown in Figure 1.
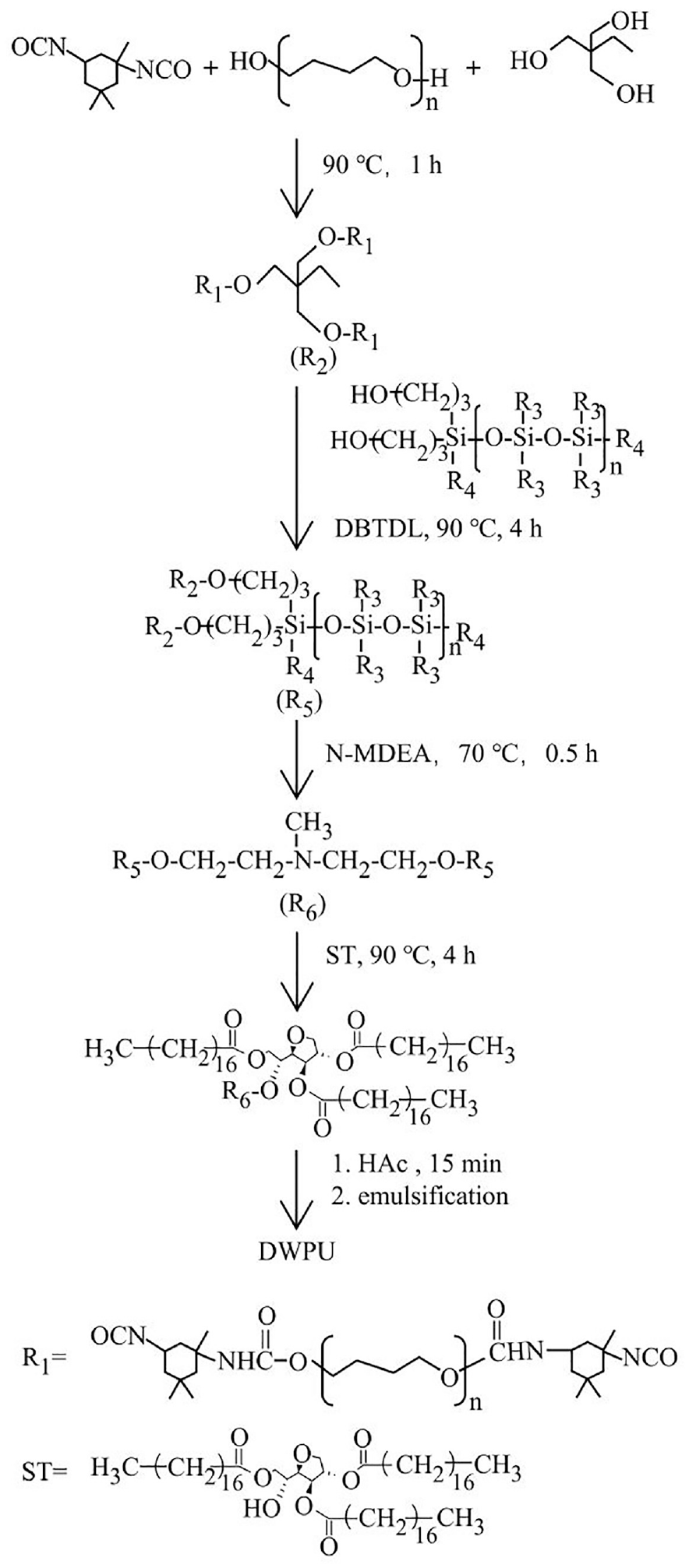
Preparation procedure of the DWPU.
Preparation of DWPU Films
The films were made by casting 1.0 mL DWPU emulsions into Teflon disks at ambient temperature and curing for 7 days. The DWPU films were then tested by drying them in a vacuum oven at 60°C for 24 h.
Water-Repellent Finishing of Cotton Fabrics
DWPU-8 emulsion with deionized water was used to prepare a 100 g/L fabric water-repellent finishing solution. Cotton fabrics were treated by two-dip two-pad method with a water-repellent finishing solution at a roll pressure of 298 kPa and a speed of 5 rpm. Then, cotton fabrics were cured in a heat-setting machine at 170°C for 90 s. The comparison sample X-9 was finished according to the same process.
Characterization and Analysis
FTIR spectroscopy. A Spectrum Two FTIR spectrometer was used for spectral characterization of DWPU films, with 32 scans for each sample, taking air as the background peak, with a scanning resolution of 4/cm, and a spectral acquisition range 3500/cm to 500/cm.
Particle size and zeta potential test. A drop of DWPU emulsion was diluted to one-thousandth of the mass fraction, and the particle size and zeta potential of DWPU were determined using a Zetasizer Nano zona nanoparticle sizer (Malvern Corp.) at 25° for 120 s.
Thermal analysis test of adhesive film. The DWPU-8 film was thermogravimetrically analyzed by TGA 55 (TA Instruments, USA) with a sample heating rate of 20°/min and a temperature test range of 50–600°C. This test was done under nitrogen atmosphere, and the nitrogen flow rate was controlled at 50 mL/min.
Water resistance test. DWPU films of the known weight (m0) were immersed in water at a given temperature. After 24 h, DWPU films were taken out and reweighed (m). The formula for calculating the water absorption rate is: water absorption rate = (m–m0)/m0× 100%.
Static contact angle and surface free energy of DWPU films. The static contact angles of water and formamide on DWPU films and fabrics were measured using the contact angle meter DSA30. Then 2 μL of test liquid was dropped onto the DWPU film and fabric surfaces. Advance software was used for microscopic measurements of the contact angle and calculation of the surface free energy at the DWPU films by the Owens–Wendt–Rabel–Kaelble model.
Scanning electron microscope (SEM) measurement. The cotton fabric was sprayed with a thin layer of gold before testing. Micrographs of the cotton fabric were obtained using an SEM (Hitachi, TM-1000, Tokyo, Japan) under a voltage of 20 kV.
Spray test. The spray test was conducted using the standard AATCC 22-2014 developed by the American Association of Textile Chemists and Colorists (AATCC), which simulates the spray test of textiles exposed to rain. 19 In this experiment, DWPU-finished cotton fabrics were tested on an AATCC spray tester with a sample size of 180 × 180 mm.
Durable water repellent test. The DWPU-finished fabrics were laundered using test method AATCC 135-2004. Fabrics were tested for WCA after each wash to study the durability of water repellency (DWR) of the fabrics.
The pH resistance test of DWPU-finished fabrics. The WCA of three DWPU-finished cotton fabrics (30 × 30 mm) was tested first. The cotton fabrics were then placed into the same volume of aqueous hydrochloric acid (pH = 2), sodium chloride (pH = 7) and sodium hydroxide solutions (pH = 12). After 12 h, the fabric was removed and washed clean with deionized water. The fabrics were then dried in a 60°C blast oven for 1 h to test the WCA. The final fabric WCA was used as the fabric pH resistance evaluation.
Surface friction coefficient test of DWPU-finished fibers. The fibers were stripped from the cotton fabrics before testing and prepared for use at standard temperature and humidity (25°C, 65% RH). The friction coefficient of the fibers was determined with a fiber friction coefficient meter (XCF-1A) in the friction roll rotation mode. The fibers were tested with a rotational speed of 50 rpm and a pre-tension of 200.0 (10−3 cN). Every sample underwent three tests and the average result was noted. 20
Hand of the cotton fabric test. Hand of the cotton fabric testing with reference to the standard AATCC TM 202, PhabrOmeter (Nu Cybertek, USA) used pattern recognition methods to build a mathematical model to extract hand of the fabrics. Cotton fabrics (11 cm diameter) were placed horizontally on the measurement platform of the PhabrOmeter at standard temperature and humidity (20 ± 1°C, 65 ± 2% RH). After entering the fabric’s measurement parameters in the PhES software, the PhabrOmeter completed the testing to obtain an evaluation score of the fabric’s hand with the resilience, softness, and smoothness. 21
XPS characterization. With the use of an X-ray photoelectron spectrometer (Escalab 250Xi, Thermo Fisher Scientific, USA), the elements of the fabrics treated with DWPU were analyzed. By changing the take-off angle, the elemental distribution at different depths was obtained. 22
Results and Discussion
FTIR Analysis
The FTIR spectra of SBHS, ST, and DWPU are depicted in Figure 2(a). For the spectra of SBHS and ST, distinct absorption peaks observed at 2918 and 2852/cm were attributed to the stretching vibrations of –CH3 and –CH2 groups, respectively. These findings indicated the presence of Si–CH3 structures in SBHS and –CH3 and –CH2 structures in ST. Furthermore, intense bands situated at 1233 and 802/cm corroborated the bending and rocking vibrations of –CH3 within the Si–CH3 structure. The strong bands corresponding to Si–O–Si and C–O–C vibrations were evident within the spectral range of 1023–1233/cm. 23
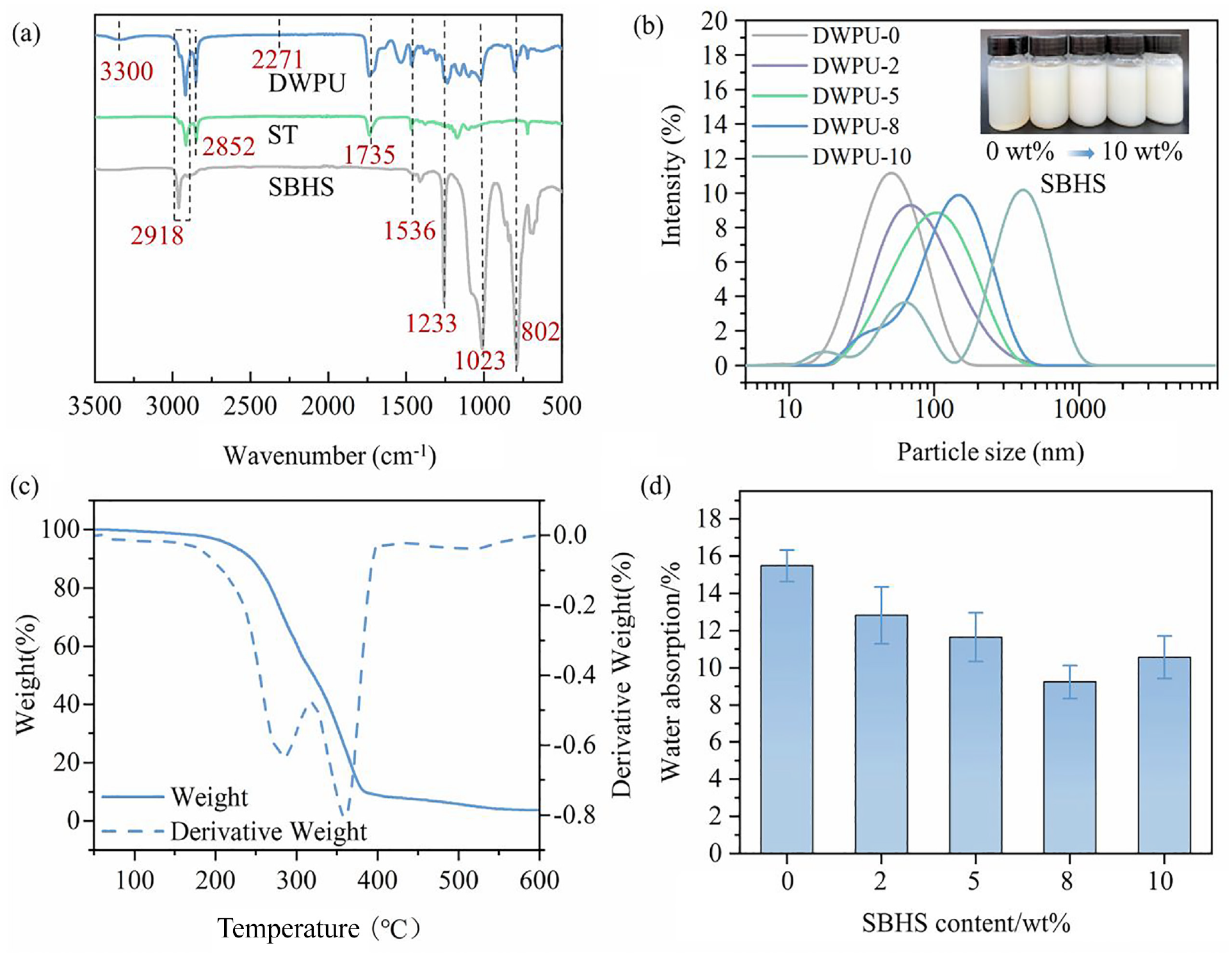
(a) FTIR spectrum of SBHS, ST, and DWPU. (b) Particle size distribution of DWPU with different SBHS content. (c) TG and DTG curves of DWPU-8 adhesive film. (d) Water absorption of DWPU film with different SBHS content.
In the DWPU spectra, the characteristic peaks of SBHS and ST could be observed, which proved that SBHS and ST had been successfully grafted in waterborne polyurethane. Meanwhile, the absorption peaks at 3300, 1735, and 1536/cm corresponded to the formation of urethane (–NH–COO–) by the reaction of–NCO groups and –OH groups, which corresponded to the infrared absorption characteristics expected of waterborne polyurethanes. The disappearance of the characteristic absorption band of –NCO at 2271/cm indicated that the terminal isocyanate reacted with ST to form a DWPU.24,25
Appearance and Stability of the DWPU
The appearance and stability of the DWPU emulsion changed as the SBHS content increased. The color, stability, and zeta potential of the emulsion are shown in Table 1. The light yellow semi-transparent of the DWPU emulsion gradually became milky white and opaque with the increasing SBHS content. The zeta potential can be used as a measure of the surface charge of the DWPU emulsion particles and thus reflects the DWPU emulsion stability. When the SBHS content was 0–8 wt%, a large value of the emulsion zeta potential (42.9−52.5mV) represented a strong charge on the surface of the DWPU emulsion particles. The large repulsive forces between particles gave the emulsion stability. When the SBHS content was 10 wt%, the surface of the emulsion particles was wrapped by more hydrophobic chain segments. The charge was weakened thus leading to a decrease in the zeta potential (39.2 mV) value of the DWPU emulsion. Eventually, the repulsive forces between DWPU particles were weakened and the emulsion agglomerated to form precipitates.
Appearance and stability of the DWPU emulsion with different SBHS content.
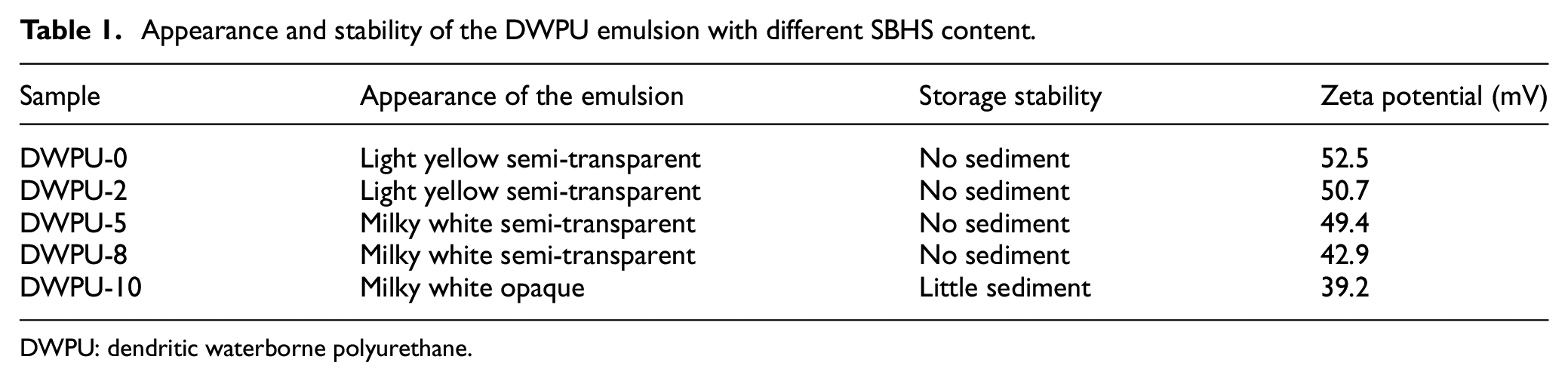
DWPU: dendritic waterborne polyurethane.
Particle Size of DWPU Emulsions
The effect of different SBHS contents on the particle size distribution of DWPU emulsion is shown in Figure 2(b). When the content of SBHS was 0–8 wt%, the particle size of DWPU emulsion was uniformly distributed, and had better stability. When the SBHS content was 10 wt%, most of the DWPU emulsion particle size increased to more than 300 nm and was not uniformly distributed. The reason was that more hydrophobic silicone long chains were grafted into the waterborne polyurethane molecule. Increased molecular weight and entanglement of molecular chains in waterborne polyurethanes resulted in increased particle size and a deterioration in the stability of DWPU emulsions. 26
Thermogravimetric Analysis of DWPU Films
The corresponding thermogravimetric analysis (TGA) curves of the DWPU-8 film is shown in Figure 2(c). Based on the weight change rate curve, the thermal weight loss of the DWPU-8 film is primarily divisible into two distinct stages. First, the thermal decomposition temperature of the polar groups within the molecular chain is approximately 250°. Second, the thermal decomposition temperature of the siloxane chain segment is observed to be approximately 350°. 27 Therefore, DWPU-8 emulsion had good thermal stability and could meet the requirements of heat setting and finishing of cotton fabrics.
Water Resistance Test
The water absorption of the DWPU film with different SBHS content is shown in Figure 2(d). When the SBHS content was 0–8 wt%, the water absorption of the DWPU film decreased from 15.49 to 9.23%. But when the SBHS content was 10 wt%, the water absorption of the DWPU film increased again to 10.56%. This could be explained by the SBHS, which is the DWPU branched chain, having excellent flexibility and hydrophobicity. When the DWPU emulsion was dried, the SBHS was enriched toward the DWPU film surface. This behavior enhanced the hydrophobicity of the DWPU film and prevented water molecules from going into the DWPU film. However, when the SBHS content was increased, the molecular weight of DWPU increased and the intermolecular chains became more easily winding. The migration of SBHS to the film surface became difficult. In addition, the poor compatibility between the silicone chains in the soft segments and the polyurethane in the hard segments resulted in increased phase separation. It is easier for water molecules to enter the DWPU film.
Static Contact Angle and Surface Free Energy of DWPU Films
The contact angles of water and formamide on DWPU films with varying SBHS content were precisely measured, and the corresponding surface energies were subsequently calculated using Advance software. The surface energy values of the DWPU adhesive films with differing silicone concentrations are tabulated in Table 2. With the grafting of the hydrophobic chains SBHS to the polyurethane side chains, the WCA of the DWPU membrane increased from 104.28° to 108.40°, and the surface free energy decreased from 22.54 to 19.90 mN/m. The results showed that the surface energy of the DWPU films decreased with increasing SBHS content. In addition, DWPU-0 also demonstrated that the dendritic polyurethane formed by ST-termination had excellent water repellency.
Surface energy of the DWPU films with different silicone content.
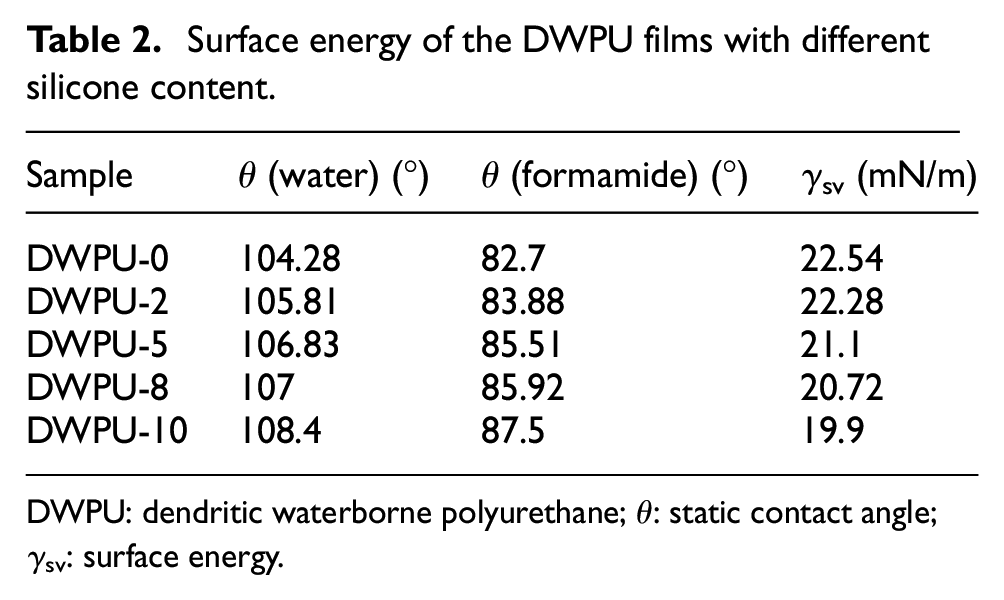
DWPU: dendritic waterborne polyurethane; θ: static contact angle; γsv: surface energy.
Water Repellency of the Finished Fabrics
The evaluation rating with the water repellency of the different DWPU-finished cotton fabrics at the standard spray test is shown in Table 3. The water repellency of cotton fabrics finished with ST-terminated waterborne polyurethane (DWPU-0) could reach 70 points. This demonstrated the feasibility of capping polyurethanes with multibranched long alkyl chains. The water repellency of the DWPU-finished cotton fabrics was improved from 70 to 80 points after SBHS grafting to polyurethane side chains. As the SBHS content increased, the water repellency of cotton fabrics could reach 95 points. The water repellency of cotton fabrics finished with DWPU-10 decreased slightly because ST and SBHS-grafted form a dendritic structure in the polyurethane molecular chain. During the pad–dry–cure process of DWPU-finished fabrics, long-chain alkyl and SBHS chain segments underwent migration to the fabric surface, ultimately resulting in the formation of a dendritic polyurethane film exhibiting low surface energy. However, with the further increase of hydrophobic chain segments, the intertwining of molecular chains prevented the migration of hydrophobic chain segments to the fabric’s surface. The fabric surface could not form an effective dendritic polyurethane protective layer, resulting in poor water-repellency.
Standard spray test ratings of DWPU-finished fabrics with different SBHS content.

DWPU: dendritic waterborne polyurethane.
Durable Water Repellency of DWPU-Finished Fabrics
The DWPU-8-finished cotton fabrics were tested for washing resistance and the specific test data are shown in Figure 3(a). After five standard washes, the WCA of the cotton fabrics changed from 148.6° to 143.4°. Because the cationic groups in the DWPU molecule formed a large number of hydrogen bonds with the oxygen-containing groups in the cotton fibers, the DWPU-finished cotton fabrics had good durability of water repellency.
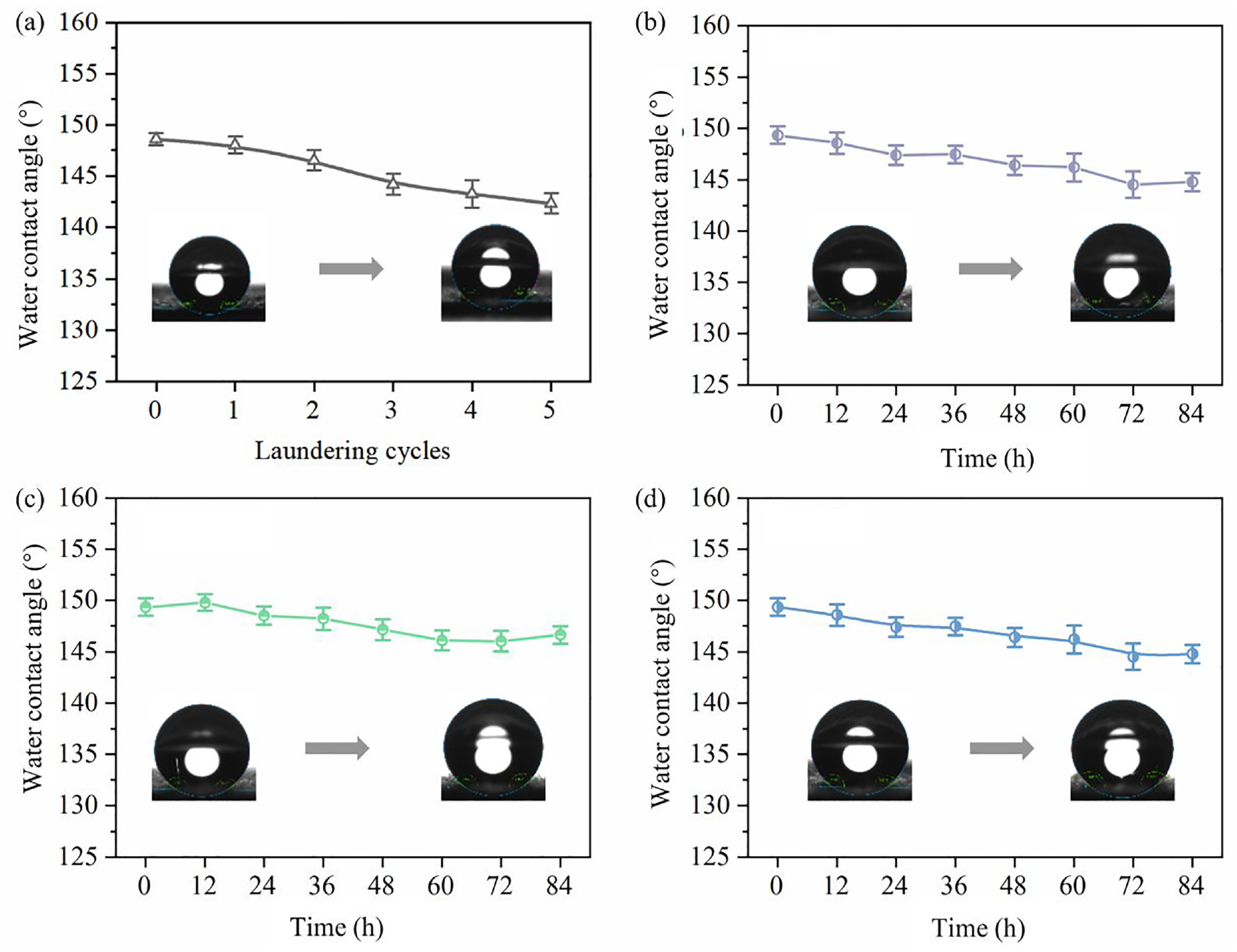
WCA of DWPU-8-finished fabrics (a) after washing, and at (b) pH = 2, (c) pH = 7, (d) pH=12.
Resistant to Acid, Alkali, and Saline Solution
The WCAs of DWPU-8-finished cotton fabrics immersed in different pH aqueous solutions are shown in Figure 3(b, c, d). The WCA of cotton fabrics decreased to varying degrees as the soaking time increased. In the pH = 12 solution, the WCA of the fabrics decreased from 147.6° to 142.3° (see Figure 3(d)). This was due to the accelerated hydrolysis of the urethane bonds in the waterborne polyurethane structure in alkaline solutions. The DWPU film on the fabric surface was disrupted, which led to a greater degree of WCA reduction. In summary, upon immersion of the fabrics in various pH solutions, a marginal reduction in the WCA was observed. Nonetheless, the WCAs remained consistently above 140°, indicative of the fabrics’ sustained hydrophobicity.
Fabric Surface Morphology
SEM was used to compare the surfaces of unfinished, DWPU-8-finished and washed cotton fabrics as shown in Figure 4. The surface of the original cotton fibers was relatively smooth as shown in Figure 4(a). Cellulose fibers are known to contain hydrophilic groups that do not prevent water molecules entering. However, a DWPU film was formed on the fibers’ surface after finishing as shown in Figure 4(b). The DWPU membrane with low surface free energy isolated the hydrophilic groups in the fiber from outside water molecules, effectively preventing water molecules entering the cotton fiber. In addition, the DWPU membrane was still tightly bound to the cotton fiber after five washes, as shown in Figure 4(c). The excellent fastness of DWPU on cotton fabrics could be attributed to the hydrogen bonding between the cationic groups in DWPU and the oxygenated groups on the fiber surface. 28

SEM micrographs of (a) unfinished cotton fabrics, (b) DWPU-8-finished cotton fabrics, and (c) DWPU-8-finished cotton fabric after five washes.
Hand of DWPU-Finished Fabrics
The surface friction properties of fibers changed after cotton fabrics were finished with water repellents. The highest friction coefficient was found in fibers finished with X-9 water repellent agent. The lowest friction coefficient was found in the unfinished fibers. The surface friction properties of DWPU-finished fibers were changed due to the low surface free energy of SBHS. The static and dynamic friction coefficients of the fibers were reduced by the addition of SBHS (see Figure 5(a)). The dynamic coefficient friction of DWPU-10 finished fibers was almost the same as that of the original fibers. Furthermore, an inverse correlation was observed between the friction reduction value and the increasing SBHS content, potentially stemming from the saturation of the fiber surface with SBHS, thereby limiting its further friction-reducing effect. The reduction of the fibers’ friction coefficient improved the softness and smoothness of fabrics. As shown in Figure 5(b), the hand of the fabrics finished with water repellents decreased compared to the original textiles. However, the softness and smoothness scores of DWPU-finished fabrics gradually increased with the increase of SBHS content. The hand of DWPU-10-finished fabrics was comparable to that of the original fabrics. The results showed that the addition of SBHS contributed to the improvement of the hand of fabrics finished with DWPU.
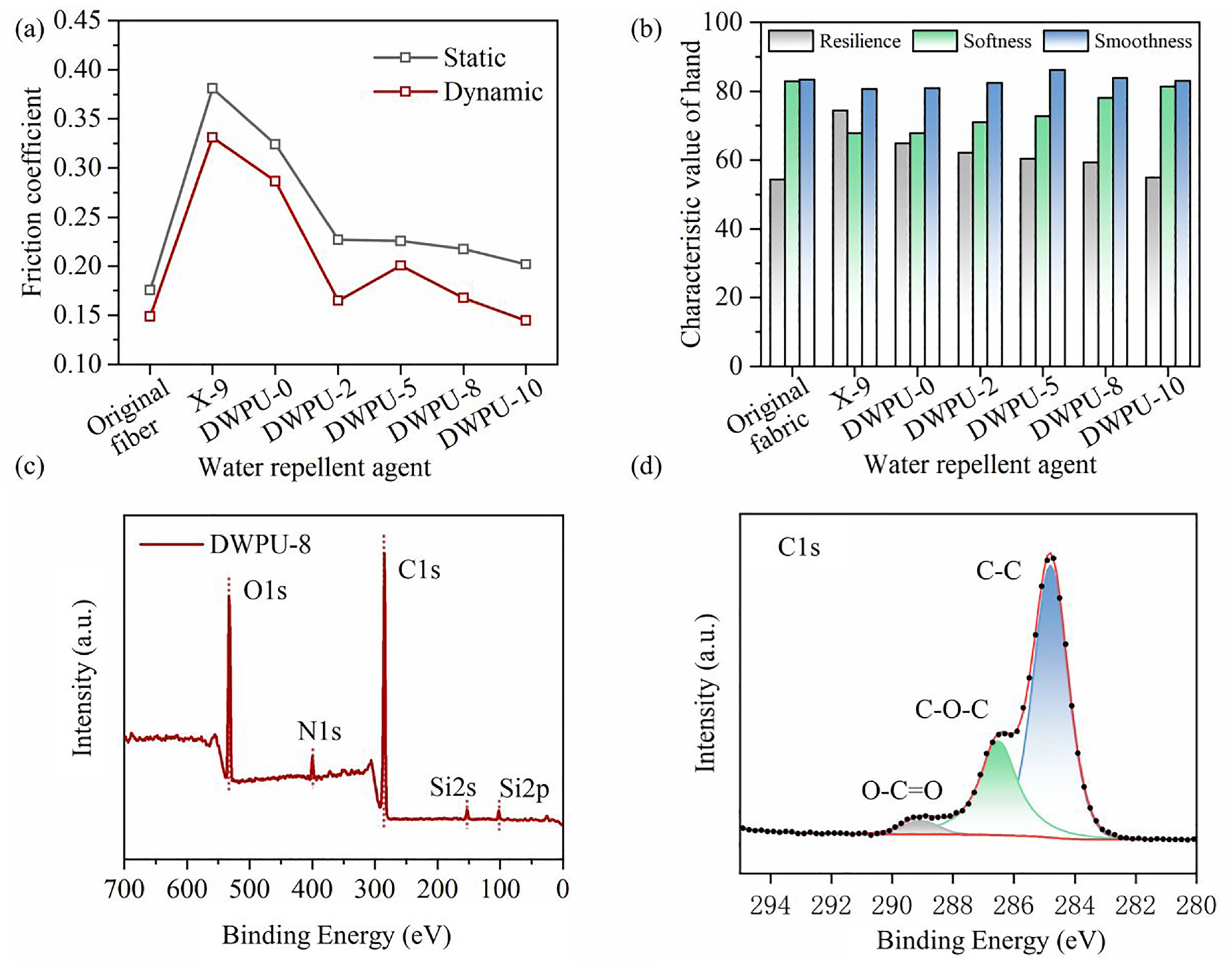
(a) Surface friction coefficient of fibers with different water-repellent agents. (b) Hand of fabrics with different water-repellent agents finished. (c) XPS full spectra of DWPU-finished fabric. (d) C1s peak fitting of the DWPU-finished fabric.
XPS of DWPU-Finished Fabrics
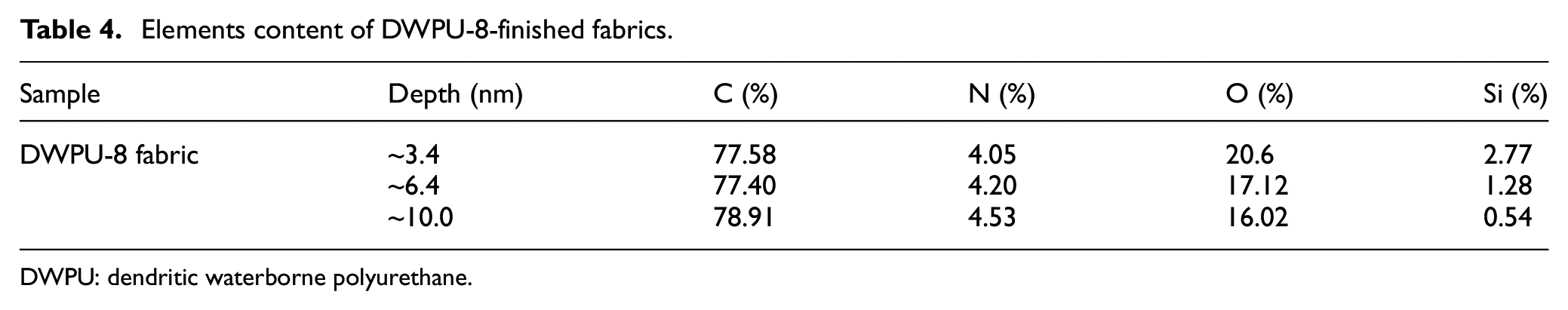
The full spectra of GWPU-8-finished cotton fabrics are shown in Figure 5(c), and the peaks in the full spectral maps of DWPU-8-finished fabrics are C1s, N1s, O1s, Si2s, and Si2p. This indicated that the SBHS had been successfully accessed into the DWPU. For more detailed information on the distribution of elements on the DWPU-8-finished fabric surface, the peak fitting process was performed on the C element (see Figure 5(d)). The binding energy of elemental C in polyurethanes was categorized into three main types: C–C/C–H, C–O, and O–C=O/–NCOO–. This was further evidence of the success of DWPU synthesis. The atomic mass concentrations are shown in Table 4. As the take-off angle decreases, the elemental content of C and N is higher and the elemental content of O and Si is lower. This indicates that the hard segments in the DWPU film on the fabric surface were concentrated on the inside and the soft segments on the outside. The results further confirmed that ST and SBHS were enriched on the surface of DWPU, reducing the surface free energy and conferring excellent water repellency to the fabric.
Elements content of DWPU-8-finished fabrics.
DWPU: dendritic waterborne polyurethane.
Conclusion
In summary, DWPU fluorine-free water-repellent capped with multibranched alkyl groups (ST) and introduced with long-chain silicones (SBHS) in the side chains were successfully prepared in this study. The data show that DWPU has better performance when the SBHS content is 8 wt%. The thermal decomposition temperature of polar groups in the molecular chain, such as carbamate bonds, was about 250°C and the thermal decomposition temperature of siloxane chain segments was about 350°C. The emulsion of DWPU-8 had an average particle size of 178.1 nm, a zeta potential of 42.9 mV, a surface energy of 20.72 mN/m, and a water absorption of 8.2%. The ST-terminated DWPU-treated fabric had a large WCA, but only 70 points on the spray test. With the addition of SBHS, the fabric rated 90–95 on the spray test. DWPU-finished fabrics had excellent resistance to wetting, with a static WCA of 148.6°. Compared with the water repellents in the market, DWPU had the advantage of making the fabrics closer to the original hand. In conclusion, DWPU fluorine-free water repellent capped with multibranched alkyl groups (ST) and introduced with long-chain silicones (SBHS) in the side chains has the zeta potential to be applied in the textile industry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
