Abstract
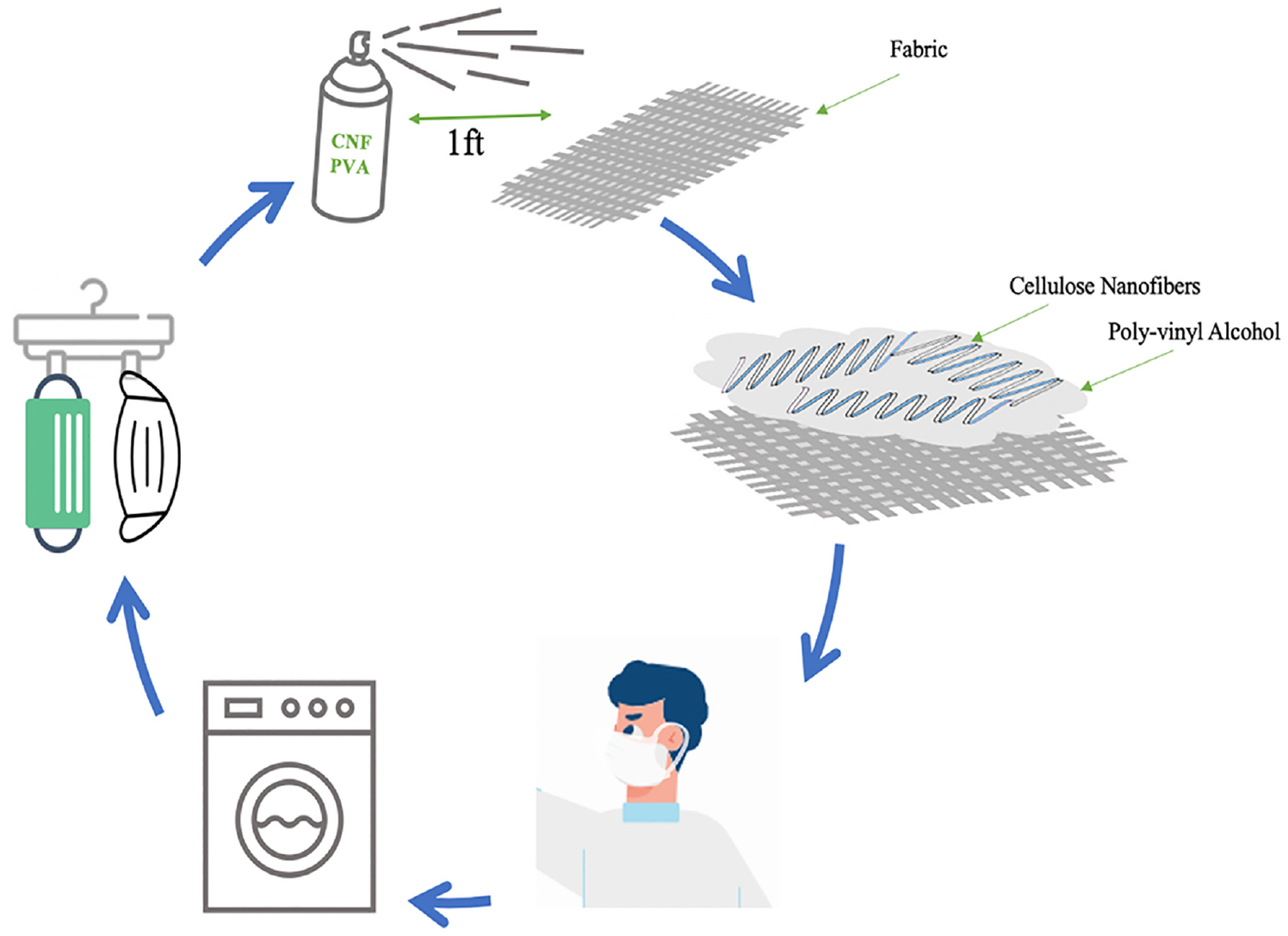
The utilization of face-covering masks as an extended form of personal protective equipment has led to exponential waste measures during the COVID-19 pandemic, with estimations of up to 7200 tons of medical-type waste daily. A primary cause of this waste is surgical layered disposable masks that are constructed by melt-blown nonwovens usually made of non-biodegradable thermoplastic polymers like polypropylene. To increase widespread sustainable options to the public, commercialized or do-it-yourself-based fabric masks serve as a solution, but their resistance to harmful molecules is less than that of the medical-grade masks due to the fabric structure that leaves space for penetration. This project examines a water-soluble dispersion composed of cellulose nanofiber and polyvinyl alcohol, as a spray agent capable of treating the mask fabric surface to promote protection and sustainability against harmful aerosol particles. Cellulose nanofiber spray is also low-cost and biocompatible and could allow multi-use through home laundering. Polyvinyl alcohol was chosen as the water-soluble bonding system and polymer matrix to effectively adhere cellulose nanofiber onto the mask surface. This project follows the biomimic concept of dragonfly wings having uneven nanopillar surfaces to trap and rip bacterial membranes, as the spray decreases the water droplet contact angle on fabric surface, resulting in an increase in adhesion for incident bacteria and/or viruses.
Keywords
Introduction
The onset of the COVID-19 pandemic in December 2019 had a profound effect on individuals around the world. It has disrupted normal routines and led to unprecedented measures such as lockdowns and mask mandates. One of the biggest factors in the pandemic is the usage of face-covering masks as an extended form of personal protective equipment (PPE). 1 This is to protect the wearer and surrounding others because severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is primarily transmitted between people through respiratory droplets and contact routes. 2 Facial masks reign as one of the first lines of defense (as well as vaccination) against easily transmittable COVID-19. Luckily through recent years, various scientific endeavors in the textile industry have enriched textiles with properties (such as improved filtration, antibacterial and antiviral activity, and breathability) crucial for the successful prevention of the spread of infectious diseases. 3 Masks have become the new normal, worn in public places, private places, and other heavily populated areas. One of the most easily attainable types of masks for the common public is the standard surgical mask. These masks are constructed with layered melt-blown nonwovens made of thermoplastic polypropylene, which is non-biodegradable.
Even though masks are efficient, they are made for limited usage, making them nearly non-reusable, causing an exponential amount of disposal waste. New research found that across 11 countries studied, the number of masks that ended up as litter/waste increased 84-fold from pre-pandemic levels. 4 This increase in mask production has led to exponential waste levels, such as up to 7200 tons of medical-type waste daily during the COVID-19 pandemic, and a primary cause of this waste is disposable masks. 5 To promote availability and sustainability during the pandemic, many companies have taken strides in constructing facial masks out of various textiles, inspiring citizens at home to create their own do-it-yourself (DIY) masks. These masks are constructed of natural and synthetic textiles and allow for multi-use wearing through at-home laundering. With the huge demand for facial protection in the present day, DIY masks are the most sustainable application option. One detriment of selecting at-home textiles relates to their pore sizes being too large to stop all aerosol particles. Scientists have determined that the virus can survive on cloth or standard textiles for several days. 3 The demand for sustainable and secure solutions to mitigate aerosol particle transmission has seen a considerable increase in various directions. For instance, a study conducted by Zang et al. 6 in 2020 demonstrated an effective avenue utilizing electrospun cellulose nanofiber/poly-vinyl alcohol (CNF/PVA) biodegradable nanofibrous air filters to combat air particulate matter transmission. In a different approach, Gunathilake et al. 7 in 2022 employed nanocellulose for bioavailable in vitro drug release systems by integrating curcumin-loaded cellulose nanocrystals in a PVA medium.
Cellulose plays a huge role in all types of different media because it is the most abundant, biodegradable, renewable, and recyclable biopolymer in existence. 8 Looking at the demand for cellulose from systemic composition, it has a tight crystal structure, which is enclosed with hemicelluloses and lignin. 8 These endless chains contribute to the tensile strength and durability of the molecule, which further creates plant cell walls. As well as increasing the surface area, nanocellulose combines the key properties of cellulose, such as high specific strength and modulus, hydrophilicity, and extensive ability for chemical modification. 9 The use of CNFs brings numerous benefits including renewability, widespread availability, low-density, excellent mechanical properties, economic value, biocompatibility, and biodegradability. 7 Numerous scientists extract these fibers using chemical processes to further apply them to experiments as nanofibers. Nanocellulose offers a range of benefits across various industries, including food films, wound care, paper, and cotton-based textiles. For example, it can be used as a natural and biodegradable alternative to plastic in food packaging, 10 or as a wound dressing to promote healing and reduce scarring.11–13 In the paper industry, it can improve the strength and stability of paper products, 14 and in the textile industry, they can enhance the durability and performance of cotton-based fabrics. 15 PVA is a polyhydroxy water-soluble polymer, and it is biodegradable and biocompatible in many biomedical applications, which can include surgical sponges, orthopedic stabilization splints, and blood-contacting material. 16 It aids in forming low-cost, hydrophilic, and transparent films with exceptional adhesion properties. 17
This study puts forth the idea of a CNF/PVA water-based dispersion spray applied onto commercial/DIY fabric masks, as a refined approach to foster sustainability and heightened protection against aerosol particles. Despite research by Naik et al., Sing et al., and Meng et al. highlighting cellulose’s antibacterial capabilities, no investigations have been made into the impacts of using a handheld CNF/PVA spray.18–20 To the best of our knowledge, this marks the first time this CNF/PVA deposition technique, employing a handheld spray bottle, has been evaluated. This method is a departure from others, such as a manual coater or airless spray pump.21,22 A handheld spray bottle was chosen for its portability and user-friendliness. CNF/PVA was selected specifically due to its unique properties and potential.
CNF and PVA are chosen due to their ability to work together in water to form a gel-like material easily able to be constructed and sprayed in a variety of applications such as coatings, adhesives, and films. This highly water-soluble material is easy to dissolve and remove through a simple washing technique like at-home laundering making it a sustainable multi-use product able to be reapplied after each laundering cycle. Creating a CNF/PVA water-soluble dispersion that can be sprayed on commercial or homemade masks may make them eligible for Food and Drug Administration (FDA) medical mask or NIOSH N95 respirator testing standards. 23 This project aims to compare the CNF/PVA spray deposition on various textiles to NIOSH N95 mask standards by examining the spray’s antibacterial properties following the biomimic concept of dragonfly wings. In previous publications,24,25 it was discovered that nanopillars on the surface of insect wings contribute to the bactericidal properties by individual cell penetration. Individual bacterial cells were observed to be killed within 3 min of contact with insect wings, 26 which are made of chitin with chemical properties similar to those of cellulose. Figure 1 depicts the physical penetration of bacterial particles through the implementation of the biomimic concept. To evaluate the CNF/PVA spray, common synthetic and biodegradable fabrics were tested with instrumental measures such as contact angle testing, optical microscopy, scanning electron microscopy (SEM), machine laundering, textile porosity testing, and bacterial filtration efficiency (BFE) testing. To be comparable to NIOSH N95 mask standards, CNF/PVA sprayed masks must be evaluated according to the US standards for filtration efficiency (ASTM F2100 and ASTM F2101-19). The development of a handheld CNF/PVA spray is novel from prior publications, and it opens up possibilities for future enhancements as a new method for providing biocompatible and portable antibacterial protection.
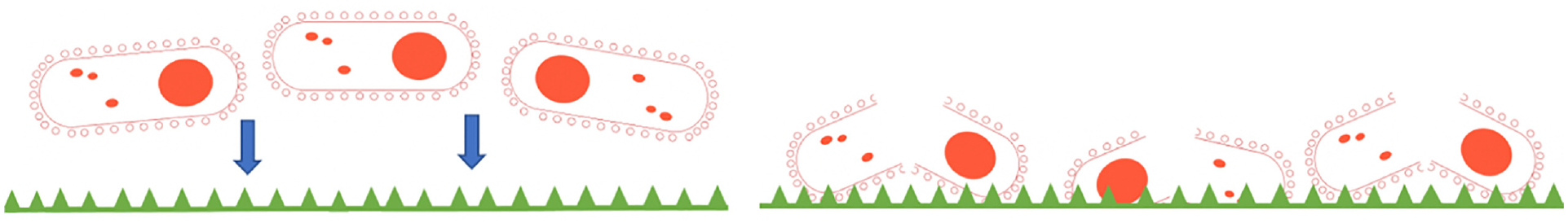
Illustration of the biomimic concept of nanoscale pillars rupturing bacterial cells.
Materials and methods
Materials
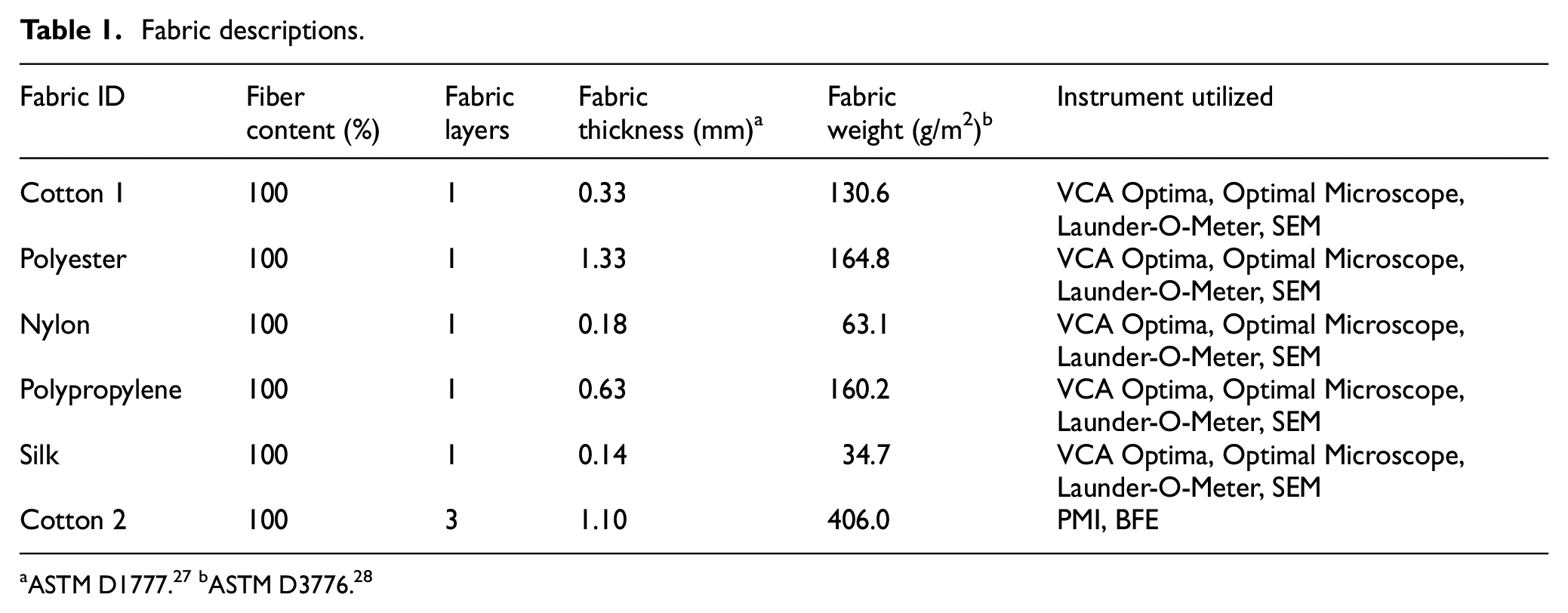
Cellulose in the nanofiber slurry form of a 3.0 wt% aqueous gel, with a lateral dimension of 50 nm wide and lengths of up to several microns, was purchased from Cellulose Laboratory in Canada. PVA with a molecular weight of 85,000–124,000 g/mol and 95.5–96.5% hydrolysis was purchased from Sigma-Aldrich. A cellulose identification strain of Shirlastain D from SDL Atlas was applied to highlight the CNF on the fabric surfaces for microscopic examination. Biodegradable fabric samples of 100% cotton (cotton 1) and 100% silk used for testing were obtained from Testfabrics, Inc. They can be referenced in Table 1. Synthetic textile samples of 100% polypropylene, 100% polyester, and 100% nylon used for testing were also obtained from Testfabrics, Inc., as listed in Table 1. To research a direct comparison with commercial masks, 100% cotton (cotton 2) three-layered masks were purchased from Amazon. These are Hanes Brand masks with the product number MASKBB. According to the packaging information, these masks might contain silver and/or copper. They are not FDA approved; however, they have been authorized by the FDA under an Emergency Use Authorization (EUA) for use by health care professionals (HCP) as PPE to help prevent the spread of infectious illness. It is important to highlight that the masks are machine washable and constructed of 100% cotton. While there is no claim on the mask’s protectiveness against aerosol particles, the inclusion of silver and/or copper could suggest some antimicrobial properties, but there is no direct statement. The container holding the CNF/PVA dispersion was a glass reusable spray bottle (60 mL) from Wedama. Escherichia coli of the BL21 strain was purchased from Bio-Rad Laboratories for bioassay testing and grown in Luria–Bertani (LB) broth (thelabrat.com).
Fabric descriptions.
Preparation of deionized water-soluble dispersion of CNF and PVA
This product was designed to be a handheld bottle with a built-in spray function to coat various fabric surfaces. To create a spray bottle with the most optimizable concentrations of CNF and PVA, a mixing ratio of 0.066% CNF, 0.7% PVA, and 99% deionized (DI) water was used. This pertained to the pure and dry concentrations of CNF and PVA. First, the 3 wt% CNFs were removed from the refrigerator at 1–4°C, and 1.355 g was measured on the scale. The CNFs were placed into a 55-mL beaker of DI water and spread evenly with an IKA RW 20 stirrer at a constant speed of 1000–2400 rpm. Next, to prepare PVA, 5 g was measured on the scale and applied to a beaker with 50 mL of DI water. This created a weighted concentration of 9 wt% PVA, which was later applied to the water-soluble dispersion after heating. The solution was heated in a hot water bath of 175°C for 45–60 min until the bubbles were completely diminished. In total, 5 mL of the PVA solution was measured and distributed to the beaker containing 55 mL of DI water and CNFs. Once distributed into a 60-mL spray bottle, the solution was shaken well and ready to be sprayed in its final homogenized state. The test fabric samples were sprayed from one foot away with five layers and an appropriate minimum drying time of 1 h in ambient conditions. Before proceeding with testing, it was determined that applying a total of five layers of coating is the optimal approach to ensure uniform distribution of water-soluble spray across each textile fabric.
VCA Optima XE
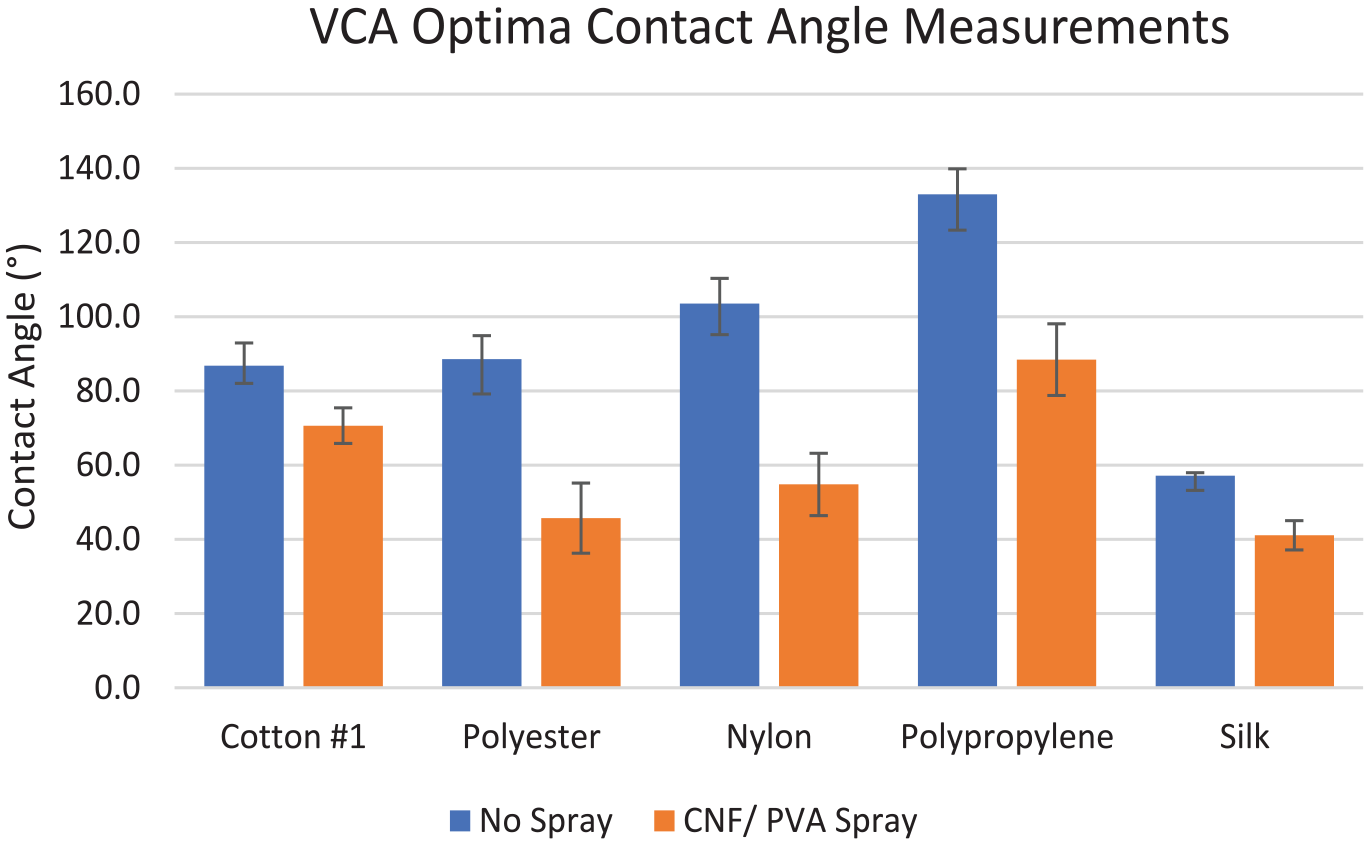
The VCA Optima XE was utilized in the Advanced Textile Testing Laboratory at the University of Texas at Austin (UT) to measure the contact angles of water droplets against various test fabric surfaces (Figure 2). The contact angle of the liquid to the fabric measures the wettability of a solid surface by a liquid. An automated syringe of 100 μL was used, and each measurement was taken by depositing a 2 μL droplet onto the textile surface. These contact angles were calculated automatically with the VCA Optima 2500 software system with a magnification of 35:1 and an accuracy of ±0.5°; each outcome is modified based on the surface tension of the fabric presented. The cotton 1, polyester, nylon, polypropylene, and silk fabrics were measured with this instrument with control samples and CNF/PVA spray-coated samples. Experimental textiles were sprayed with five layers and allowed for a drying time of at least 1 h before testing proceeded.

VCA Optima XE photographs of CNF/PVA water-based dispersion on nylon fabric: (a) control and (b) experimental.
Optical microscope
The Nikon Ni-E Upright Motor optical microscope was utilized in the Center for Biomedical Research Microscopy and Imaging Facility at UT to examine the fabric outer surface with CNF/PVA spray film. The single-layer pure cotton 1, polyester, nylon, polypropylene, and silk fabrics were viewed under the microscope without the CNF/PVA spray and with the CNF/PVA spray. All were chosen to identify whether the CNF/PVA layering structure was similar in distribution on each fabric regardless of its woven structure or fiber chemical makeup.
SEM
The FEI Quanta 650 SEM was utilized in the Texas Materials Institute at the UT to evaluate the surface morphologies of the cotton 1, polyester, nylon, polypropylene, and silk experimental fabrics. The machine contained a voltage of 10.00 kV at a high vacuum and a chamber pressure of 0.60 mbar. The specimens were coated with gold before analysis using the EMS Sputter Coater also located in the Texas Materials Institute.
Launder-O-Meter
The Launder-O-Meter was utilized in the UT Advanced Textile Testing Laboratory to simulate a controlled laundering environment for the CNF/PVA spray washability. Single-layered cotton 1, polyester, nylon, polypropylene, and silk fabrics were laundered for a 45-min cycle with 0.74 g of Seventh Generation Natural Laundry Detergent for 24 total samples; each fabric sample contained three control samples and three experimental samples with five layers of CNF/PVA spray (Figure 3 and Figure 4). Experimental samples were sprayed after measurement and left to dry in ambient conditions before the laundering cycle. Contact angles of the fabrics were measured with the VCA Optima XE to compare surface wettability before and after the Launder-O-Meter laundering. Fabric weight difference was another factor considered to test whether textile weight significantly changes through machine laundering with the CNF/PVA spray applied. Experimental samples were sprayed after measurement and left to dry in ambient conditions before the laundering cycle.

Launder-O-Meter tested samples of fabrics captured pre-laundering: (a) cotton 1, silk, and polyester; (b) nylon and polypropylene.

Launder-O-Meter tested samples of fabrics captured post-laundering: (a) cotton 1, silk, and polyester; (b) nylon and polypropylene.
Advanced capillary porometer (PMI)
The advanced capillary porometer (ACFP-1100AEXLFNBH) was utilized in the UT Advanced Textile Testing Laboratory to characterize pore size and distribution with three control and three experimental measurements (CNF/PVA sprayed) on the cotton 2 fabric. The cotton 2 fabric was selected for its three-layered thickness, aligning with the instrument’s requirement for a more substantial sample. In addition, consistent with the other textiles examined in this study, these masks contain a 100% pure fiber composition. The highly reproducible technique required inputs of fabric diameter and fabric thickness. Out of five measurements of the fabric, the average thickness was calculated to be 0.0169 mm. The advanced capillary porometer followed the dry-up/wet-up testing procedure, which included monitoring the samples in both dry (before placement of liquid) and wet methods (after liquid placement). The test increases the pressure and measures the flow and pressure drop across the dry sample to locate the minimum and maximum detectable pore size on the cotton 2 fabric. The measured pressure curve is inversely proportional to pore diameter, which can be used to calculate pore size distribution. 29 The surface tension of the Silwick fluid used for the samples is 20.1 Dynes/cm, which minimizes likely evaporation during testing. Silwick fluid was chosen as it is the most optimal immiscible and saturated wetting liquid for this research in this field with its contact angle of zero. 30
BFE testing
The BFE testing was performed in Anresco Laboratories in San Francisco, California, to determine protective effectiveness of the commercial cotton masks (cotton 2) with the CNF/PVA spray against exterior bacteria. This is the testing that is most important in relation to COVID-19 because it measures the efficiency of the CNF/PVA sprayed fabric itself comparable to someone wearing a mask. 31 When these results are reported, they are based off percentage, so the higher the percentage, the higher the efficiency. These results are aligned with the applicable standard values for the United States (ASTM F2100 and ASTM F2101-19), which makes them essential to determine whether this is a reliable method to suggest to consumers. While conducting the experimental procedures, the ratio of upstream bacterial challenge to downstream residual concentration determined the filtration efficiency of medical face mask materials. This method was specifically designed using Staphylococcus aureus as a challenge organism. Materials to be tested were conditioned at 21 ± 5°C and 85 ± 5% relative humidity (RH) using a humidity chamber for 4 h. The number of specimens was five in total with two control samples and three experimental (CNF/PVA spray) samples. Cotton 2 fabric was selected for its resemblance to standard surgical masks commonly available to consumers. This choice ensured a more direct comparison with regulated benchmarks. Furthermore, like the other textiles examined in this study, these masks contain a 100% pure fiber composition. Each experimental sample was sprayed 5 times and an ample amount of dry time was allowed in ambient air. The area of specimens tested was ∼40 cm2, and the temperature during testing was 26°C. The mean particle size was 3.0 ± 0.3 μm, and the flow rate was 28.3 L/min utilizing the GB-XF100 instrument.
Antimicrobial activity measurement
The method of cell growth quantifier (CGQ) was used to test CNF antimicrobial activity (Scientific Bioprocessing, Baesweiler, Germany). The GCQ implements a responsive approach of backscattered light measurement with sensor-based technology, allowing for efficient and precise growth biomass monitoring. This continuous data stream permits close observation of microbial or cell culture growth phases, thereby gaining insights into growth kinetics. Three 250-mL flasks were prepared for experimental measures, each sterilized through autoclaving prior to addition of ingredients. To observe growth and suppression, backscattered light measurements of E. coli were employed as the primary indicators in all flasks. The assembly included a control flask with a single E. coli colony in LB broth, and two experimental flasks consisting of one E. coli colony, LB broth, and CNF. E. coli colonies were introduced in each flask using aseptic technique; the inoculating loop was twisted 5 times to uniformly dispense cells. The CNF concentration was standardized at 1.355 g and the LB broth at 28 mL. Experimental testing with the CGQ was conducted at 37°C at 150 r for 27 h. Two main measurements were recorded: backscatter data and optical density at 600 nm (OD600).
Results and discussion
Testing the contact angle with the VCA Optima XE
The contact angles are measured using the VCA Optima XE instrument. The average of six individual values recorded on each fabric was taken to congregate a mean and standard deviation value of both the right and the left angle measurements. Each fabric was sprayed 5 times with a drying time of 1 h. Cotton 1 measured an increase in wettability by 16.15° with the CNF/PVA spray application, and polyester measured an increase in wettability by 42.9° largely greater than the cotton 1 textile. Nylon demonstrates the highest increase in wettability by a decrease in 48.70° out of the measured fabrics, indicating that there is an increased hydrophilicity to the surface with the spray applied. The polypropylene fabric decreased its contact angle by 45.55°, and the silk fabric decreased its contact angle by 16.10°. The increase in surface roughness and −OH presence of the CNF/PVA layering can significantly impact the morphological changes of the fabric surface and, therefore, enhance the fabric samples’ hydrophilicity, similar to the effect of the Chen et al. 32 approach. There is maximum adhesion on the nylon masks by the greatest decrease in contact angle, which could indicate that it contains the highest wettability property. The contact angle decrease can be described by an increase in ions at the surface and less compact structure with increased water absorption, which is comparable with previously published data.33,34 The results represented in Table 2 indicate that this development extends to most textiles regardless of their biodegradability.
Contact angle p values from analysis of variance analysis (probability of obtaining an F statistic greater than the computed F); filled cells.

Very significant.
Figure 5 addresses the significance of the CNF/PVA addition by conducting a two-way analysis of variance (ANOVA) statistical analysis measuring variance between the four different textiles. Different textiles were utilized to measure the increased hydrophilic effects across various diameters of porous areas and various fiber makeups, which addresses CNF/PVA adaptability. Before statistical analysis, the data were checked and evaluated for normal distribution. Overall, the various fabrics exhibited a high statistical significance regarding the CNF/PVA addition of two p < 0.01. Regarding spraying, one p < 0.01 (Table 2) examined the effect of the CNF/PVA spray between the control and experimental samples, with the significance confirming the change of ion structure at the surface of the textiles. Regarding fabric comparisons, p < 0.01 (Table 2) addresses the statistical effect of each fabric on one another, with significance confirming the differences in chemical makeup of the fiber formation. Polyester, a synthetic fabric usually derived from petroleum, is normally characterized by its large surface area per unit mass and small pore size.35,36 Nylon, a synthetic fabric produced from coal, exhibits hydrophilicity due to the presence of amide bonds. These linkages form hydrogen bonds with water molecules in the amorphous regions. 37 Cotton 1, one of the most abundantly used biodegradable fabrics, exhibits high moisture-wicking and breathability. 38 Polypropylene, a synthetic fiber derived from a thermoplastic polymer of the polyolefin group, is used in many medical textiles (including standard surgical masks) with its microporous membrane and extreme hydrophobicity.39,40 Silks are classified as high molecular weight organic polymers, exhibiting characteristic repetitive peptide sequences with hydrophilic properties. 41 The uncertainties in the standard error bars represent individual standard deviation.
Two-way ANOVA results of VCA contact angle measurements.
Examining surface morphologies with a Nikon Ni-E Upright motor
As shown in Figure 6, the Nikon Ni-E Upright motor was used to capture images of the single-layer surface morphologies of the cotton 1, polyester, nylon, polypropylene, and silk fabrics, both without and coated with a CNF/PVA spray. The cotton 1, polyester, and polypropylene images were taken with a magnification of 10 × using three-layering high dynamic range (HDR) and differential image contrast. The nylon images were taken with a magnification of 40 × using three-layering HDR and differential image contrast. The CNF/PVA coating is clearly visible in the porous areas of the fabric and creates a physical barrier that helps to reduce the transmission of aerosols and changes the surface roughness of the exposed outer area of the fibers present. The silk images were taken with a magnification of 20 × using three-layering HDR and differential image contrast. Regardless of the fabric, the CNF/PVA coating is similarly present on the porous areas, reducing the size of the pores across the entire fabric. However, the experimental images show a non-uniform layer of CNF/PVA. It is suggested that the uneven distribution of the CNF/PVA on the fabrics may be due to factors such as the size, shape, and velocity of the droplets produced when the water-soluble dispersion is sprayed. Although this does not necessarily affect the overall amount of CNF/PVA applied to the textiles, there is still similar film deposition, which is evident by other experimental procedures utilized in this study.
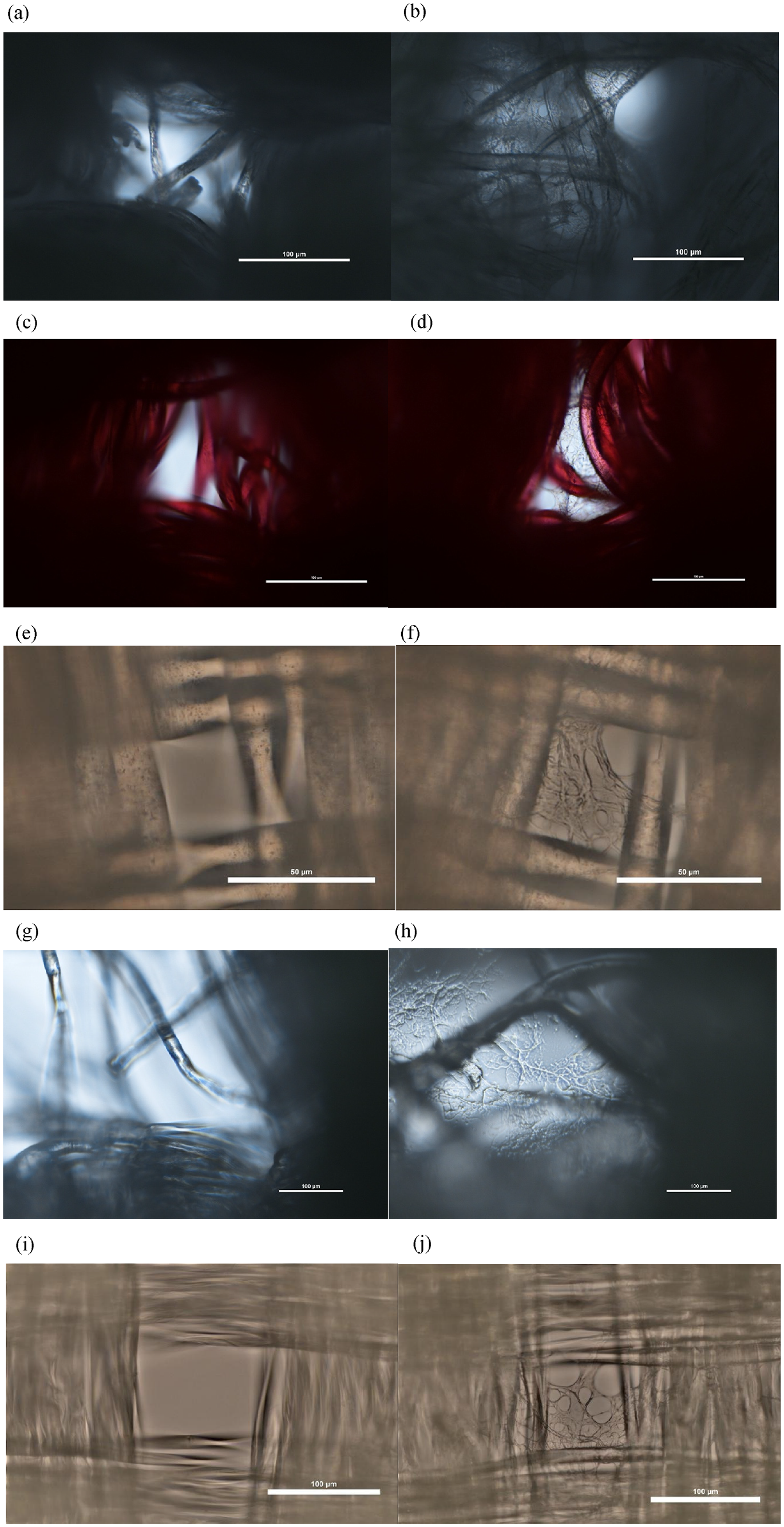
Control and experimental photographs of CNF/PVA water-based dispersion on textiles: (a) cotton 1 control at 10×; (b) cotton 1 experimental at 10×; (c) polyester control at 10×; (d) polyester experimental at 10×; (e) nylon control at 40×; (f) nylon experimental at 40×; (g) polypropylene control at 10×; (h) polypropylene experimental at 10×; (i) silk control at 20×; and (j) silk experimental at 20×.
Examining surface morphologies with SEM
As shown in Figure 7, the FEI Quanta 650 SEM collected various images of cotton 1, polyester, nylon, polypropylene, and silk fabrics after they were sprayed 5 times with CNF/PVA spray with optimal drying time. Within each captured photograph, the fiber surface area increases as it is layered with the CNF/PVA spray film. Individual CNFs are clearly identified through the PVA film among each textile fiber and porous area similarly regardless of if they are synthetic or biodegradable fibers. All textiles were examined at two different magnifications of 500 × and 3000 ×, which present a textile cross-section and fabric porous area. CNFs are dispersed from fiber to fiber while also encompassing individual fibers, and this generates an increased physical defense against aerosol particles. CNF/PVA spray film visually adheres within and on the surface of the textile fibers as a rugged-like texture. These surface morphologies are like the study by Tyagi et al. 42 and contain uneven, sharp edges on the substrate surface that could support the biomimic concept that physical cellulosic barriers increase antibacterial resistance.

FEI Quanta 650 SEM of textile swatches layered with CNF/PVA water-based dispersion spray: (a) cotton 1 at 500×; (b) cotton 1 at 3000×; (c) polyester at 500×; (d) polyester at 3000×; (e) nylon at 500×; (f) nylon at 3000×; (g) polypropylene at 500×; (h) polypropylene at 3000×; (i) silk at 500×; and (j) silk at 3000×.
Machine-laundering capabilities with the Launder-O-Meter
Table 3 lists the contact angle measurements using the VCA Optima XE instrument, and the average of 10 individual values was taken to congregate a mean value for both pre- and post-laundered samples on cotton 1, polyester, nylon, polypropylene, and silk. CNF/PVA experimental samples were sprayed 5 times after the contact angle measurements were recorded to test whether CNF/PVA leaves residue post-machine laundering. Machine laundering was conducted with the Launder-O-Meter with simulated abrasion to replicate an at-home washing cycle. To further evaluate CNF/PVA spray adhesion, a two-way ANOVA statistical analysis was conducted for each textile, and results are presented in Table 5. Before statistical analysis, the data were checked and evaluated for normal distribution.
Surface contact angles for cotton 1, polyester, nylon, polypropylene, and silk fabric samples, both pre- and post-laundering.
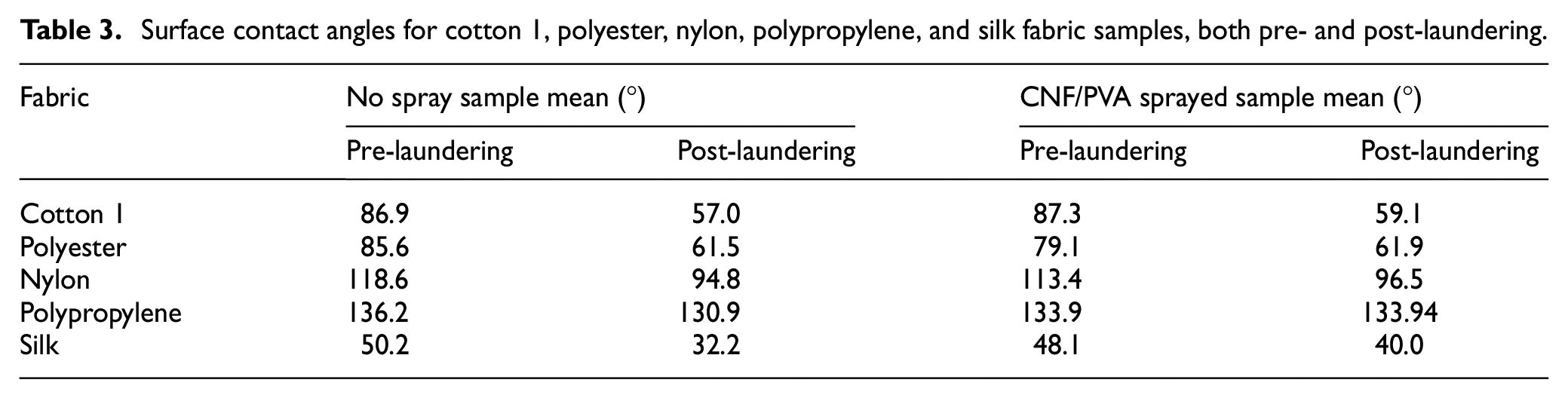
Table 4 addresses weight as variable that can affect the overall textile property based on CNF/PVA adhesion through machine laundering. Ten samples of cotton 1, polyester, nylon, polypropylene, and silk 2 × 2 inch squares were measured on the precision scale, and after measurements, five layers of CNF/PVA spray were applied to five of each textile sample. Optimal drying time of at least 1 h was allowed for the experimental samples in ambient air. Machine laundering was conducted with the Launder-O-Meter with simulated abrasion to replicate an at-home washing cycle, and after a 45-min cycle, samples were air-exposed for at least 24 h for complete dehydration. To further evaluate CNF/PVA spray adhesion regarding weight, a two-way ANOVA statistical analysis was conducted, and results are presented in Table 5. Before statistical analysis, the data were checked and evaluated for normal distribution.
Weight measurements for cotton 1, polyester, nylon, polypropylene, and silk fabric samples, both pre- and post-laundering.
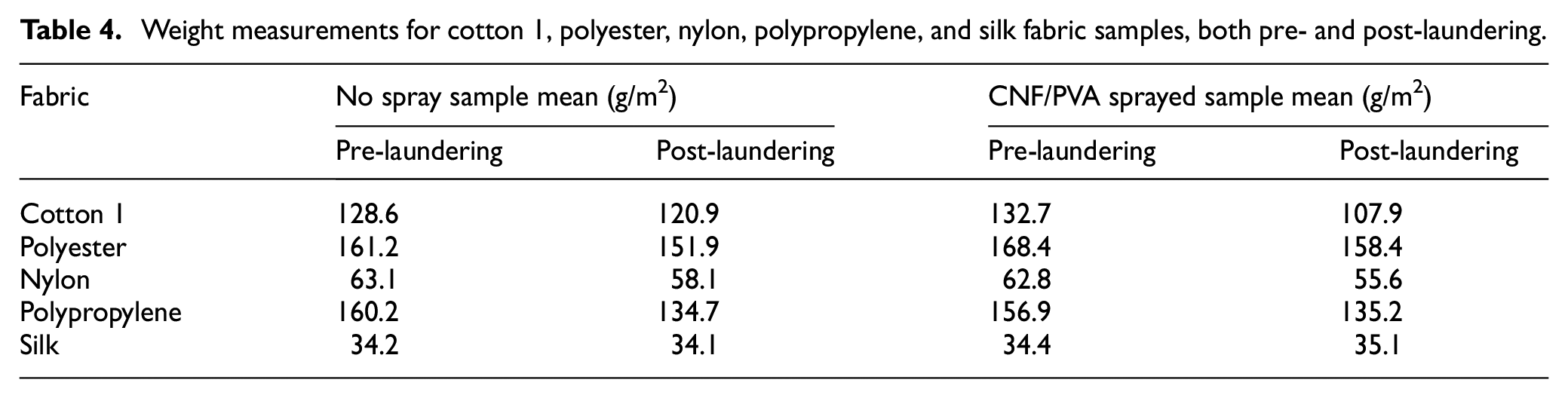
Machine laundering p values from ANOVA analysis (probability of obtaining an F statistic greater than the computed F); filled cells.
Very significant.
Contact angle (CA); weight (W); spraying (S); washing (Wa); interaction (I).
Cotton 1: CA: S (df: 1.00, F: 0.27), Wa (df: 1.00, F: 145.98), and I (df: 36, F: 0.13); W: S (df: 1.00, F: 7.99), Wa (df: 1.00, F: 0.56), and I (df: 1.00, F: 2.23).
Polyester: CA: S (df: 1.00, F: 0.89), Wa (df: 1.00, F: 41.28), and I (df: 36, F: 1.16); W: S (df: 1.00, F: 3.18), Wa (df: 1.00, F: 1.61), and I (df: 1.00, F: 0.00).
Nylon: CA: S (df: 1.00, F: 0.65), Wa (df: 1.00, F: 87.5), and I (df: 1.00, F: 119.70); W: S (df: 1.00, F: 4.12), Wa (df: 1.00, F: 26.13), and I (df: 1.00, F: 3.15).
Polypropylene: CA: S (df: 1.00, F: 0.05), Wa (df: 1.00, F: 2.31), and I (df: 1.00, F: 2.38); W: S (df: 1.00, F: 0.35), Wa (df: 1.00, F: 88.3), and I (df: 1.00, F: 0.49).
Silk: CA: S (df: 1.00, F: 0.70),Wa (df: 1.00, F: 23.52), and I (df: 1.00, F: 0.12); W: S (df: 1.00, F: 0.28), Wa (df: 1.00, F: 1.03), and I (df: 1.00, F: 0.46).
Focusing on spraying through contact angle measurement, all textiles indicate p > 0.05 (see Table 5) regarding their pre-laundering contact angles compared with post-laundering contact angles. This result is in agreement with our prediction, if the textile fabric follows a controlled machine-washing cycle, the surface wettability and weight of fabric treated with CNF/PVA spray will not exhibit significant differences compared with untreated fabric post-laundering due to the hydrophilic properties of CNF and PVA. Following our hypothesis, post-laundering shows that the contact angles of the nylon and polypropylene experimental samples did not have a significant reduction, indicating that washing may not cause a dramatic surface tension change for those applied with the CNF/PVA spray. This supports the assumption for complete removal of the CNF/PVA adhesion to textiles within one machine-laundering cycle comparable to samples with no spray. Focusing on washing through contact angle measurement, polypropylene contains a p > 0.05, and cotton 1, polyester, nylon, and silk contain a p < 0.05 (see Table 5). This indicates that polypropylene contact angle remains similar before and after laundering, and it did not have a significant effect on hydrophobicity after one cycle of laundering. On the contrary, the contact angles of pre-laundering cotton 1, polyester, nylon, and silk are significantly higher than post-laundering contact angles, meaning machine laundering is a statistical factor affecting hydrophobicity. Increasing mechanical abrasion of nylon during laundering was a probable cause of significant differences in wettability before and after a cycle, which most likely causes fibers to shed from the fabric surface. 43 This physical structure change can be referenced in Figures 3 and 4. To know the impact of machine laundering of the two independent variables, the interaction between spraying versus washing was recorded in relation to the contact angle measurement. In a two-way ANOVA analysis, there is no notable interaction effect between spraying and washing on the contact angles for each fabric type. The variables of spraying and washing are independent, exhibiting no interactive effects, and show consistent differences attributed only to the spraying or washing processes. This outcome aligns perfectly with our initial prediction, as the intended factor to change is the complete removal of the CNF/PVA spray through a single laundering cycle.
Focusing on spraying through weight measurement, all textiles contain p > 0.05 (see Table 5) regarding their pre-laundering weight compared with post-laundering weight. Following our hypothesis, post-laundering shows that the measured weight of the cotton 1, polyester, nylon, polypropylene, and silk experimental samples did not have a significant change, indicating that washing does not cause a dramatic weight change for those with the CNF/PVA spray applied compared with those with no spray applied. This supports the assumption of complete removal of the CNF/PVA adhesion from textiles within one machine-laundering cycle. In another section of the two-way ANOVA analysis, the weighted effect was tested between the pre- and post-laundering samples aiming to measure whether weight significantly changed before and after one cycle including spray presence. Focusing on washing through weight measurement, cotton 1, polyester, and silk showed a p-value of p > 0.05, which confirms an insignificant difference in weight for both no spray and CNF/PVA samples. Nylon and polypropylene showed a p-value of p < 0.05 (see Table 5), which confirms a significant difference in weight for both no spray and CNF/PVA samples. This follows the published work by Tapia-Picazo et al. 44 which reported that the washing process used in their study resulted in a decrease in the average molecular weight and, subsequently, a reduction in the elongation at breakage. To determing the impact of machine laundering of the two independent variables, the interaction between spraying versus washing was recorded in relation to the weight measurement. In a two-way ANOVA analysis, there is no notable interaction effect between spraying and washing on the weight for each fabric type. The variables of spraying and washing are independent, exhibiting no interactive effects, and show consistent differences attributed only to the spraying or washing processes. This outcome continues to follow our initial prediction, as the intended factor to change is the complete removal of the CNF/PVA spray through a single laundering cycle.
Through both experiments evaluating differences in contact angles and weight, it was especially important to analyze each textile for the spraying and washing effect. Each one of the two factors plays a role in determining the longevity of the textile in two separate ways. Spraying evaluated the CNF/PVA film adhesion through machine laundering and increased abrasion. The hydrophilic nature of the CNF/PVA film contributed to the complete removal through one laundering cycle, which makes it more attractive to restart the cycle of daily mask usage and continue the reapplication of CNF/PVA film.
Textile porosity testing with the advanced capillary porometer (PMI)
The dry-up/wet-up extraction method conducted with the advanced capillary porometer was utilized to compare porous differences between cotton 2 fabric and cotton 2 CNF/PVA layered fabric as shown in Figure 8. Cotton 2 fabric was used to simulate CNF/PVA spray application to a commercialized mask, which contained three-layered cotton fabric. Particle filtration measured with dry-up/wet-up testing examined three control and three CNF/PVA textiles to limit testing discrepancies.
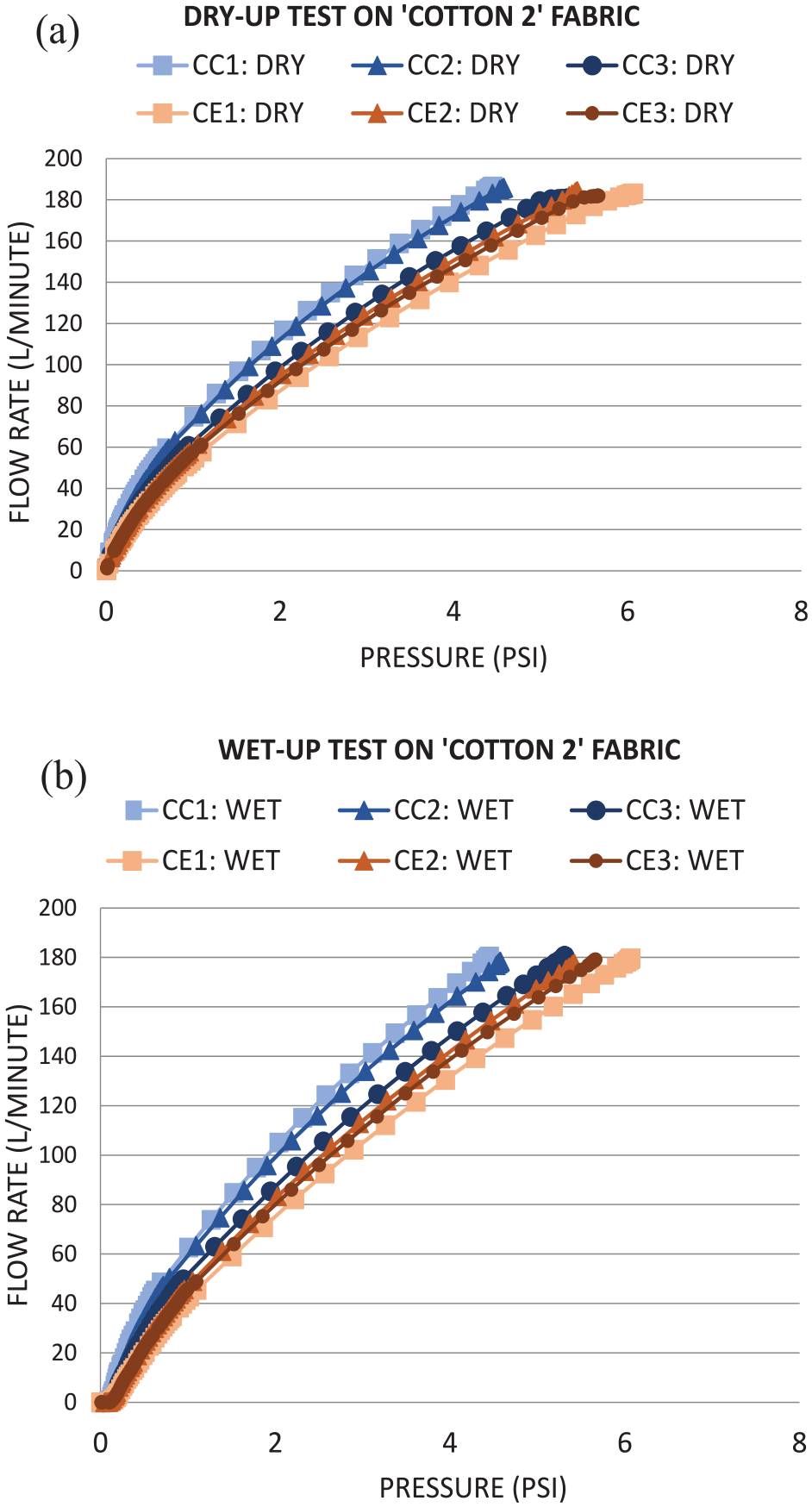
Dry-up/wet-up curves on cotton 2 fabric between control and CNF/PVA spray: (a) dry-up and (b) wet-up.
Figure 8 represents both parameters of the dry-up and wet-up testing method with increasing pressure (PSI) values correlating with increased flow rate (L/min). The initial intersection of all the linear models corresponds to the maximum pore size, also known as the bubble point. The ending points of all linear models on the dry-up (8a) and wet-up (8b) graphs correspond to the minimum pore size that includes all the liquid resulting in the porous areas being emptied. 45 In both dry and wet parameters, there are visual discrepancies between the experimental and control fabrics, indicating that there could be a measured difference in porous areas with application of the CNF/PVA spray.
As shown in Table 6, the control cotton contained a mean flow pore diameter of the three measurements at 31.37 µm and a mean bubble point pore diameter of 650.16 µm. The experimental CNF/PVA cotton contained a mean flow pore diameter of the three measurements at 22.74 µm and a mean bubble point pore diameter at 1489.78 µm. There is a clear correlation between the CNF/PVA application and decrease in porous diameter values and an increase in bubble point pore diameter values. Bubble point pore diameter demonstrates a great fluctuation in values due to change in textile surface patterns, and this contrasts with the mean flow pore diameter in the inverse direction as comparable to the publication. 45 In addition, maximum pore size distribution is recorded for the cotton 2 fabric samples. To find the numerical value, pore distribution must be measured for its change of filter flow between wet and dry samples, and it can be calculated from the equations below.
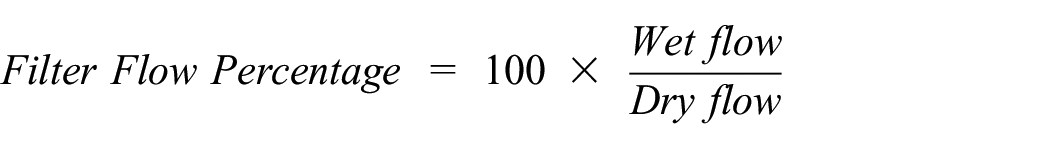


The mean of the cotton control values of maximum size pore distribution is 64.82, and the mean of the cotton experimental values of maximum size pore distribution is 40.61. This is representative of a differential change of filter flow between the control and experimental samples with the porous distribution being more miniscule. Maintaining a continuous flow through the advanced capillary porometer further emphasized the importance of physical closure of fabric porous areas without diminishing total air permeability but decreasing aerosol leakage. Air permeability is important as masks are used as a breathing apparatus to help the wearer maintain normal respiration levels, while continuously being protected from outer infectious inhalants.
Control and experimental (CNF/PVA spray) cotton fabric porosity on cotton fabric.
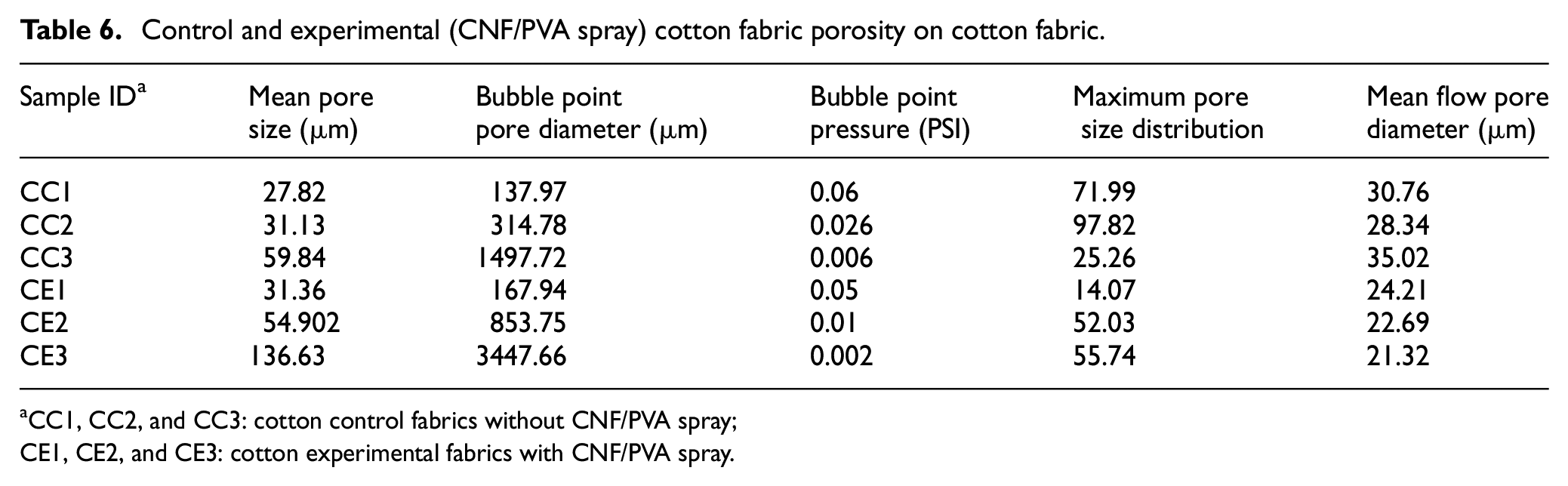
CC1, CC2, and CC3: cotton control fabrics without CNF/PVA spray;
CE1, CE2, and CE3: cotton experimental fabrics with CNF/PVA spray.
In assessing air permeability and breathability, it is pivotal to draw insights from comparative studies. Akduman 46 created cellulose acetate nanofiber mats for N-95 respirators and reported a mean flow pore size ranging from 2.38 to 5.71 μm. Although cellulose acetate is different from CNFs, their compositional studies provide a relevant comparison. Akduman’s findings suggest with even smaller pore sizes than our study with a range of 2.38−5.71 μm, and adequate breathability can be accomplished.
Evaluating BFE
As shown in Figure 9, the BFE of five cloth masks sprayed 5 times each was measured using the ratio of S. aureus bacterial challenge from upstream to downstream. Cotton 2 fabric was used to simulate CNF/PVA spray application to a commercialized mask, which contained three-layered 100% cotton fabric in the form of a cloth face covering. Recipe concentrations through the samples consisted of three levels (0.602, 0.903, and 1.355 g) of 3 wt% CNF, and an identical concentration of PVA (5 g of PVA along with additional 55 mL H2O). To objectively evaluate the relationship between increasing CNF concentrations and associated BFE values, we employed Spearman’s rank correlation analysis. The resulting coefficient of approximately
Spearman’s Rank Correlation Coefficient

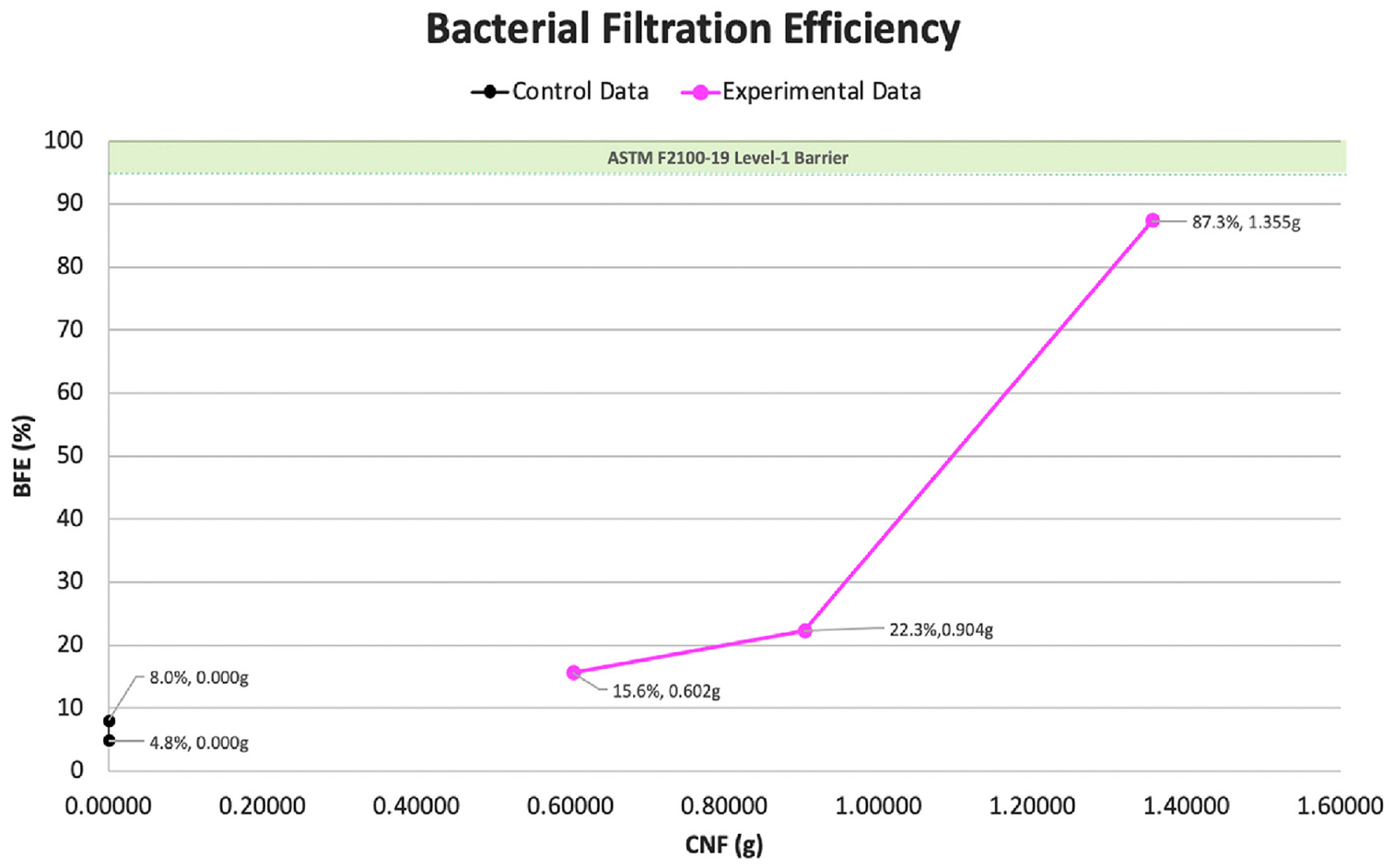
Bacterial filtration efficiency regarding ASTM F2100-19 specification requirements.
CNF antimicrobial property
Figure 10 illustrates the growth curves obtained by the CGQ, utilizing E. coli as the model organism. Escherichia coli is a Gram-negative, rod-shaped bacterium, which serves as a fundamental tool in both teaching and research laboratories due to its rapid growth and maintenance characteristics. 51 A visual analysis of E. coli’s growth patterns identifies four phases in the following order: lag phase, log phase, stationary phase, and death phase. The CGQ enhances the analysis by quantifying viable cell numbers to identify growth phases. The apparatus provides two main measurement methods: backscatter data, which reflects the light intensity after its interaction with the cells, and the traditional OD600 (optical density at 600 nm), which gauges cell density in a liquid culture based on light absorbance at the 600 nm wavelength. Both these techniques are geared toward shedding light on the growth dynamics of microbial or cell cultures, albeit through differing light interaction methods: scattering and absorbance.
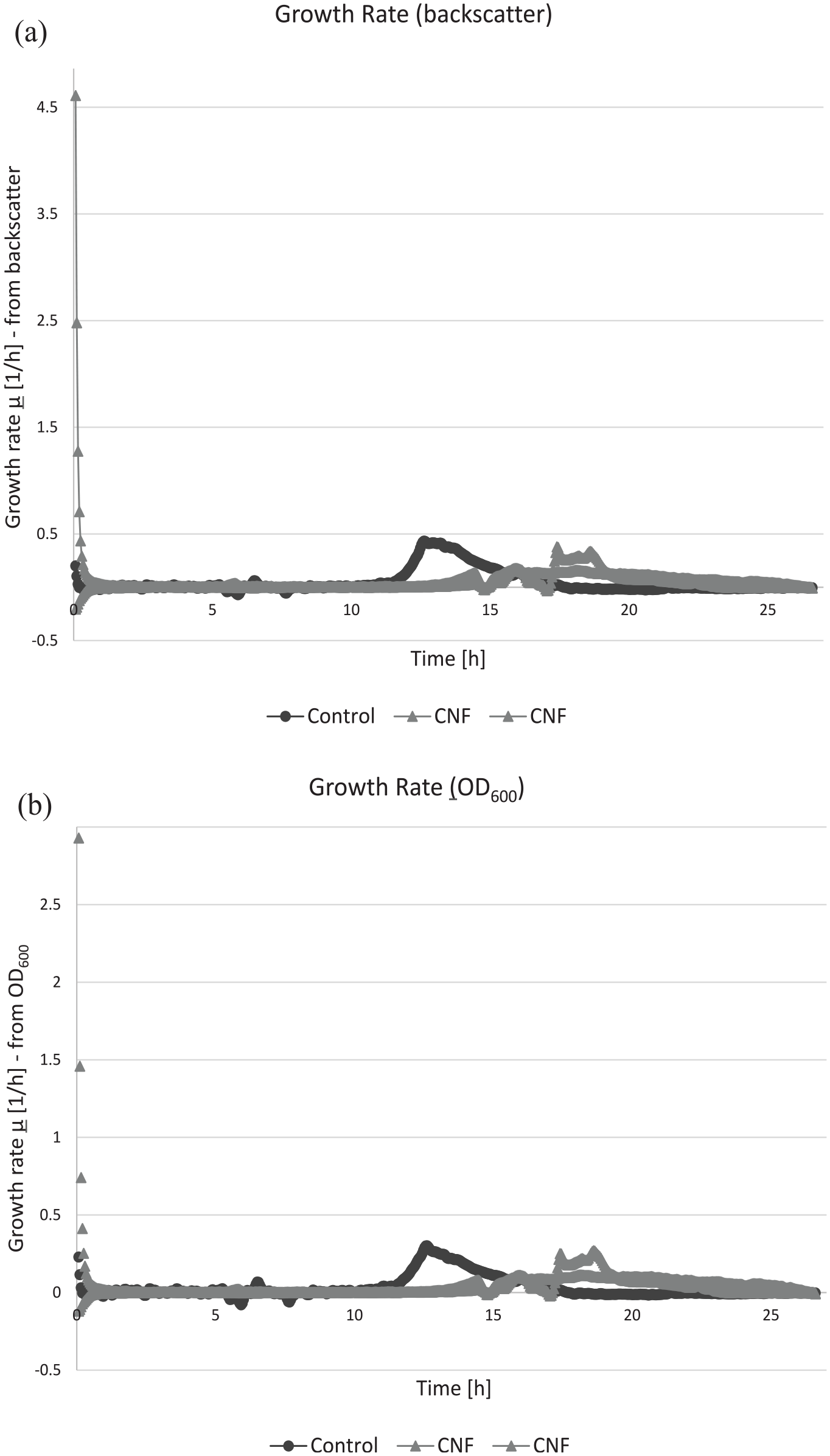
CGQ growth curves between control and CNF containing flasks: (a) backscatter and (b) OD600.
Figure 10(a) portrays the progression of time against the growth rate µ(1/h) in the context of backscatter intensity. These measurements offer insights into the dynamics of culture expansion, where the higher values on the y-axis denote increased growth rates, suggesting minimal antibacterial interference, while lower values suggest increased antibacterial presence. The control flask exhibited a growth rate peaking at 0.54 per hour, establishing a baseline. Both experimental flasks infused with CNF contained reduced growth rates, peaking at 0.48 per hour and 0.30 per hour, respectively. This decline in growth rates in the presence of CNF suggests cellulose’s efficacy in suppressing E. coli proliferation. The discrepancy in growth rates between the control and the CNF-infused flasks underscores the potential antibacterial properties of CNF. Given the reduced rates overall in the experimental flasks, it suggests an immediate influence on of CNF on the initial stages of E. coli binary fission. Figure 10(b) plots the duration of culture growth against growth rate µ(1/h), derived from the OD600 measurements. In the CGQ growth curves, a spike in OD600 values implies heightened cell density, accredited to increased light scattering and absorption by cellular division. The control flask showcased a growth rate that peaked at 0.33 per hour, which provides a point of reference of the E. coli under normal conditions. Contrarily, the experimental flasks infused with CNF recorded suppressed growth rates, reaching peaks at 0.29 per hour and 0.12 per hour, respectively. The considerable dip in growth rates outlines the inhibitory action of CNF on E. coli binary fission.
The consistently reduced E. coli growth rates in the subsequent experiments, especially in flasks containing the antibacterial spray ingredients, indicated noticeable effect of antimicrobial properties. The control CGQ growth curves follow the traditional growth mechanisms of E. coli with each of the four phases observed and a peak occurring at 12.53 h. 52 The inclusion of CNF appeared to not only suppress the overall growth rate but also delay time to reach maximum growth, underlining CNF’s effectiveness as an antibacterial agent.
Conclusion
In summary, various tests of biodegradable and synthetic fabrics were conducted to test for a positive correlation of decreased aerosol transmission with various instruments: VCA Optima, optical microscope, SEM, machine laundering, PMI, and BFE. Through testing the CNF/PVA water-soluble dispersion applied onto the mask fabrics, it is found that there is a significant correlation between the CNF/PVA loading and the hydrophilicity increase. The CNF/PVA spray changed the surface texture, decreased the porous area, and improved the barrier property against aerosol bacteria. The lower contact angles indicated there was significant stronger adhesion in the CNF samples due to the hydrophilic nature of cellulose and PVA. Post-laundering showed that the contact angles did not have a significant reduction in comparison with the pre-laundering contact angles, indicating that washing does not cause a dramatic surface tension change for fabrics applied with the CNF/PVA coating spray, which was the target outcome. Regarding the fabric weight reduction through washing, machine laundering was not affected by the addition of CNF/PVA spray on each of the textile samples, but differences in weight were from mechanical abrasion. Regarding the optical microscope and SEM images, the CNF/PVA solution visually demonstrates increased adhesion.
The PMI porosity testing revealed smaller mean flow pore diameters when the test fabrics were applied with the CNF/PVA spray to create an increased physical barrier against foreign particles. The test of BFE in accordance with ASTM F2100-19 revealed that the application of our CNF/PVA spray could increase the BFE of the mask fabrics to 87.3%, close to the 95% needed for professional masks with level 1 barrier performance. The CGQ growth curve assessments emphasized CNF and its ability to suppress E. coli proliferation, indicating antibacterial activity. Overall, the positive results from this study effectively responded to our research question, showcasing the various capabilities of the CNF/PVA spray and substantiating the biomimicry concept. A significant strength of the study includes antibacterial clearance against bacterial pathogens with an increase in mass percentage, which is parallel to the findings of Naik et al., 18 in which both studies use S. aureus at a test organism.
Moreover, a potential limitation could involve the versatility of the spray’s deposition on different textiles. Our study tested five distinct textiles, both synthetic and biodegradable, to evaluate this, but deeper exploration is still required. Further testing of CNF/PVA antimicrobial spray applications can be expanded to other forms of PPE, such as surgical gowns, caps, and air filters to limit aerosol particles. Additional research is recommended to expand on the spray’s effectiveness against other bacterial species, such as mold, pollen grains, foreign bodies, and viruses to further understand how the CNF/PVA spray can combat these microorganisms.
Supplemental Material
sj-docx-1-aat-10.1177_24723444241237302 – for Cellulose nanofiber as mask/personal protective equipment surface agent for enhanced anti-bacterial performance
sj-docx-1-aat-10.1177_24723444241237302 for Cellulose nanofiber as mask/personal protective equipment surface agent for enhanced anti-bacterial performance by Sophia Jackson and Jonathan Chen in AATCC Journal of Research
Footnotes
Acknowledgements
We would like to thank Amy Leksana and the Austin Community College, Biotechnology Department for their helpful instruction with the CGQ experimentation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research grants utilized on this work were awarded by American Association of Textile Chemists and Colorists, and The University of Texas at Austin Texas Innovation Center, the College of Natural Sciences, and the Office of Undergraduate Research.
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article or its supplemental materials. Most data and calculations were conducted at the UT by SJ. Raw data for BFE were generated at a commercial facility. Raw data for antimicrobial activity measurement were generated at Austin Community College, Biotechnology Department. Policy: Basic, Share upon Request
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
