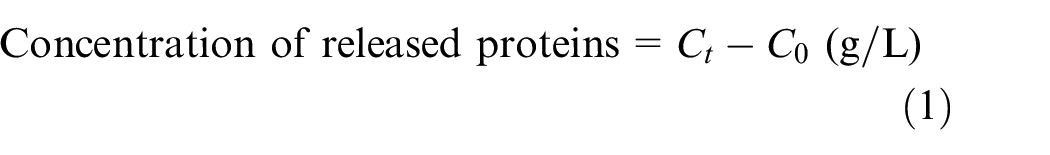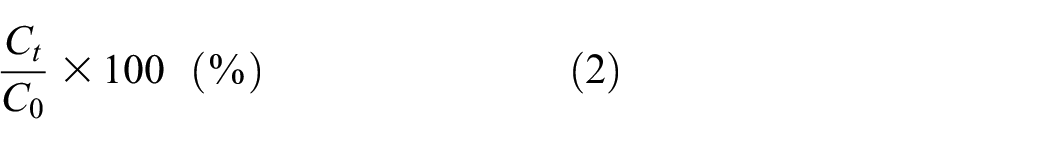Abstract
The low scouring efficiency of mercerized cotton yarns scoured with alkaline and acid pectinases, reported in previously published articles, raised the hypothesis of the lack of pectinase adsorption onto mercerized cotton. In an attempt to identify whether low pectinase adsorption occurs, the adsorption of alkaline and acid pectinases on raw and mercerized cotton yarns is measured using the Lowry protein assay, while laser-scanning confocal fluorescence microscopy is used to visually show the presence/absence of the enzymes on the surface of the cotton yarns. In addition, the surface zeta potential of the raw and mercerized cotton yarns is measured to identify the influence of the surface charge on the adsorption ability of the used enzymes. Data suggest that although used substrates have adsorbed more alkaline than acid pectinase, the adsorption of both enzymes is more intensive onto the mercerized cotton relative to raw cotton. The similar surface zeta potential of the raw and mercerized yarns indicates that higher enzyme adsorption on mercerized cotton is not influenced by the surface charge. Laser-scanning confocal fluorescence microscopy micrographs depict an evident increase of fluorescing signal with scouring time to raw and mercerized cotton yarn. The more intensive signals after 60 min of enzymatic scouring with both enzymes suggest enzyme presence on the yarn surface after a long treatment time. The obtained results confirm the enzyme retention on the yarn surface, thus excluding the hypothesis that the absence of enzyme adsorption is a factor for the low scouring efficiency of mercerized cotton yarns.
Introduction
Raw cotton has about 90% cellulose and surface-located hydrophobic waxes, pectins, and proteins, which limit uniform dyeing. 1 Pretreatment of cotton fabric for dyeing is performed by scouring to remove cuticle components and bleaching to decolorize its natural yellowish color. Traditional scouring is conducted with 1–4% sodium hydroxide solution, in the presence of wetting and washing surfactants at boiling temperature, for 1–2 h, 2 to remove the surface non-cellulosic substances, creating hydrophilic cotton with the excellent ability for uniform dyeing. This treatment is economically unfavorable and produces effluents with high chemical oxygen demand (COD) and biochemical oxygen demand (BOD). 3 Enzymatic scouring is an excellent opportunity to overcome these negative issues by treating the cotton at 40–60°C, for less than 1 h in near-neutral pH conditions. 4 Enzymes are natural biocatalysts that act on a specific substance (or impurity), in some cases hydrolyzing it into water-soluble low-molecular chemicals that can be easily removed from the surface of the substrate. Enzymatic scouring is an energy-saving and environmentally friendly process, and as such its implementation is of great interest to the industry. The enzyme’s effectiveness in a short time is controlled by adjusting the pH, temperature, concentration, treatment time, and mechanical agitation. Besides these, the physical contact between the enzyme and the substrate is of crucial importance for effective enzymatic scouring. Under optimal working conditions, the enzyme possesses an active site that attracts the substrate and develops an enzyme–substrate complex similar to the lock-and-key model. 5 Several enzyme classes such as cellulases, pectinases, lipases, proteases, and their mixtures, are used for the scouring of cotton.6–13 Pectinases alone, or in combination with cellulases, appeared to create the most hydrophilic cotton. Alkaline and acid pectinases were used for enzymatic scouring of raw cotton, alone,6,14–16 or in combination with selective 17 and non-selective oxidants. 18 Pectinases improved the hydrophilicity of cotton, while oxidation of enzymatically scoured cotton imparted aldehyde and carboxyl groups to the cotton without substantial degradation of cotton yarns, confirmed by only a 15% decrease in tensile strength and an 8% decrease in the degree of polymerization.
Mercerization is a pretreatment stage that aims to increase the luster, tensile strength, dimensional stability, and dyeability of cotton by treating it with concentrated sodium hydroxide solution (up to 30%), within minutes, at room temperature.19,20 Under the action of a concentrated alkaline solution, physicochemical and structural modifications of cellulose take place. As a result of the penetration of the alkali into the lattice, internal hydrogen bonds are broken, and native cellulose I forms alkali cellulose I. This form, under washing and neutralization, creates cellulose II with a decreased crystallinity and increased number of available hydroxyl groups by around 25%. 20 The major changes during mercerization are divided into fiber and molecular levels. At the fiber level, swelling and cross-sectional morphology changes from beam to round shape occur. At a molecular level, hydrogen bond readjustment, the orientation of molecular chains in the amorphous region, and the orientation of the crystallinity along the direction of fiber length occur. Mercerization is classified according to the form of the treated product (yarn, woven, or knit), mercerizing conditions (water content, tension, alkali concentration, and temperature), stage of treatment (raw, pre-dyeing, or post-dyeing), number of treatments (single or double), and type of alkali used (caustic soda or ammonia). 19 Mercerization of woven fabric is conducted on a raw or bleached fabric after de-sizing/scouring/bleaching. These pretreatments remove the applied size on warp yarn before weaving, enhancing the penetration of caustic soda into the fibers, and giving a more effective mercerization. 19 Yarn mercerization is performed as hank, warp, or cone-to-cone mercerization. Hank mercerization is usually performed on raw cotton, while warp mercerization is a continuous process done after scouring/bleaching. To achieve quick penetration of concentrated sodium hydroxide solution to raw (dry) cotton, the addition of a wetting surfactant, stable in a high alkaline solution, is required. Mercerization after scouring/bleaching, or mercerization on wet cotton yarn imparts good effectiveness to cotton yarn, but the released heat, caused by the reaction between the alkali and water, creates an uneven mercerization. Therefore, the scoured/bleached cotton yarn is dried before mercerization. Even though mercerization on scoured/bleached cotton yarn is more effective, for economic reasons (i.e. to save an additional drying cost), mercerization on raw cotton is practiced. 19
With the arising possibility of applying enzymatic scouring to different substrates, its implementation in a mercerized cotton yarn has enhanced interest. To explore the influences of enzymatic scouring on mercerization and vice versa, 21 the course of scouring-mercerization, 22 and temperature of drying after mercerization, 23 have been altered. Two experiments were conducted on cotton yarn hanks, 22 on an industrial machine, Jaeggli, in a factory that produces mercerized threads, using a procedure comprising mercerization of raw cotton yarns, scouring, bleaching, and dyeing. In the first experiment, enzymatic scouring was conducted before mercerization, while in the second one, enzymatic scouring succeeded mercerization. Scoured-mercerized cotton yarns were obtained by scouring raw cotton yarns with alkaline and acid pectinases, after which they were rinsed, mercerized, rinsed again, and dried. Mercerized-scoured cotton yarns were produced by mercerization of raw cotton yarns. The yarns were then rinsed, dried, enzymatically scoured with alkaline and acid pectinases, rinsed again, and dried.
Enzymatic scoured-mercerized and mercerized-enzymatic scoured cotton yarns were tested in terms of surface characteristics, surface free energy, wettability, absorption capacity, and supramolecular structure. 22 The enzymatic scoured-mercerized cotton yarn has an improved hydrophilicity, higher monolayer capacity, and a higher polar component of the surface free energy than mercerized-enzymatic scoured ones. 22 When applied to the mercerized cotton, the enzymatic scouring has low efficiency and produces yarns with the lowest hydrophilicity. Those results raise the hypothesis that a lack of pectinase adsorption on mercerized cotton yarns occurs.
In this study, the adsorption ability of the alkaline and acid pectinases on raw and mercerized cotton yarns was evaluated in an attempt to examine whether the reason for the low scouring efficiency of mercerized cotton is the absence of pectinases adsorption. For that purpose, adsorption followed by Lowry and laser-scanning confocal fluorescence microscopy (LCFM) to visually show the presence/absence of the enzymes on the surface of the cotton yarns has been made.
Experimental
Materials
Raw (RAW) and mercerized (M80) plied ring-worsted cotton yarn with a linear density of 30×2 tex and spun with 330 twists/m was used. BioPrep 3000L (Novozymes), an alkaline pectate lyase pectinase (EC 4.2.2.2) with enzyme activity 296 U/g enzyme, and NS 29048 (Novozymes), an acidic polygalacturonase pectinase (EC 3.2.1.15) with enzyme activity 378 U/g enzyme, were used for adsorption examination. The enzyme activity was tested according to di-nitro salicylic acid (DNS) method. 24 It is used to determine the amount of reducing sugar as d-galacturonic acid (Sigma Aldrich, St. Louis, Missouri, United States) released from the polygalacturonic acid (Sigma Aldrich, St Louis, Missouri, United States). One unit (U) of enzyme activity indicates 1 µmol of d-galacturonic acid released/min at defined temperature and pH of enzyme activity.
Methods
Mercerization was done on Jaeggli hank mercerization equipment (Vetex AD Veles, North Macedonia) by rolling the hanks in 23.5% NaOH solution and 1 g/L Subitol MEZ-N (CHT-Germany) wetting agent at 18°C for 1 min. Next, the hanks were extended by the application of tension to the original length, rinsed with hot (80°C) and cold (18°C) water for 1 min under tension, neutralized, rinsed, and dried at 80°C.21–23 Optimal scouring conditions for alkaline pectinase were pH 9 (0.15 g/L Na3PO4), 1 mL/L nonionic wetting surfactant Kemonecer NI (KEMO Croatia) at 55°C for 30 min,3,12 and pH 4 (acetate buffer), in the presence of 1 mL/L Kemonecer NI at 45°C for 30 min14,17 for acid pectinase.
The adsorption of alkaline and acid pectinases was tested on raw and mercerized cotton yarn processed under the following steps: hank-mercerization, neutralization, rinsing, and drying at 80°C. Two adsorption experiments, using optimal scouring temperatures of the enzymes (55°C and 45°C) and adsorption at 20°C, were performed. In the first experiment, the adsorption of BioPrep 3000L was performed in a pH 9 bath, with a liquor ratio of 50:1, at 55°C, using 2 mL/L initial concentration of enzyme, in the absence and presence of wetting surfactant, while the adsorption ability of NS 29048 was performed in a pH 4 bath, with a liquor ratio of 50:1, at 45°C, using 2 mL/L initial concentration of enzyme, with and without wetting surfactant. For the second experiment, an adsorption study in a bath with a liquor ratio of 50:1, at 20°C, in the presence of wetting surfactant and 1 mL/L enzyme was conducted (pH 9 for BioPrep 3000L and pH 4 for NS 29048).
Because natural proteins present in cotton could potentially interfere with measurement of enzyme adsorption, the third experiment was conducted in two steps. In the first step, treatment of raw and mercerized cotton was performed with the buffer and wetting agent to remove releasable fiber proteins. Then, the enzyme adsorption on the cleaned cotton (without proteins) was performed in a bath with a liquor ratio of 50:1 at 20°C in the presence of 1 mL/L Kemonecer NI and 1 mL/L initial concentration of enzyme, at pH 4 for acid pectinase and pH 9 for alkaline pectinase.
The adsorption of alkaline and acid pectinases onto the cotton surface was followed by the Lowry method.25,26 This method allows monitoring of the amount of protein in the bath. Since both pectinases are globular water-soluble proteins, the changes in the protein concentration in the solution were used as a measure of the adsorbed enzymes onto the cotton surface. Two biuret complexes develop the characteristic color in the presence of Folin–Ciocalteu’s reagent using Solution A (20 g/L Na2CO3, 0.2 g/L potassium sodium tartrate, and 0.1 mol/L NaOH), Solution B (5 g/L CuSO4* H2O), Solution C (50 ml solution A and 1 ml solution B mixed before use) and Solution D—Folin-Ciocalteu’s reagent (1 mL reagent dissolved in 2 mL water before use). The constant aliquot of 1 mL of solution with the enzyme was pipetted at 5, 10, 15, 20, 30, 40, 50, 60, 70, 80, and 90 min, mixed with 5 mL of solution C, and left for 10 min at room temperature. Afterward, 0.5 mL Folin–Ciocalteu’s reagent (Solution D) was added, and the mixture was mixed and left for 30 min at room temperature to develop the intense dye. The developed color was measured on an ultraviolet/visible (UV/VIS) spectrophotometer (Model Hitachi-2800, United Kingdom) at λmax=660 nm. The presented results are the mean values of three measurements. A standard calibration curve is constructed with the known concentration of albumin from bovine serum, as a standard protein.
Since Lowry is a nonselective method for protein determination, it should be used for assessing different types of proteins in a solution. In this case, it determines released proteins from the cotton fiber and enzyme proteins. When more proteins are released from the fiber than adsorption of the enzyme, the amount of released proteins from the fiber is calculated according to Equation (1):
where C0 is the concentration of the proteins in the bath at the beginning (concentration of enzyme), and Ct is the sum of the concentrations of the enzymes and released proteins from the cotton fiber at time t.
When the enzyme adsorption occurs over fiber protein release, the percentage of the enzyme (protein) in the bath decreases. The percentage of the adsorbed enzyme on the surface of the cotton fiber is presented as the rate of the concentration of the enzyme in the bath at a given time divided by its initial concentration (Equation (2)):
where C0 is the concentration of the protein (enzyme) in the bath at the beginning, and Ct is the concentration of the protein (enzyme) in the bath at time t. In the beginning, the enzyme concentration in a bath is 100%, and as enzyme adsorption occurs, the adsorption rate decreases.
Untreated (RAW) and mercerized cotton yarns (M80) were used for the analysis of the surface zeta potential, calculated from the measurement of the streaming potential, which occurs when an aqueous solution passes through a capillary channel or a network of capillaries.27,28 For fibrous and granular media, a randomly arranged sample plug is arranged in a measuring cell (cylindrical cell) to provide such a capillary network. For the sake of measurement reproducibility, the sample weight and compression of the sample plug were monitored (the latter in terms of permeability toward water flow). The nature of cotton yarn requires 500 mg of sample for each measurement. An aqueous 0.01 mol/L KCl solution was used as the background electrolyte and the pH dependence of the zeta potential was recorded by an automated pH adjustment with 0.05 mol/L HCl. The initial pH of the aqueous solution was adjusted to pH 10 by adding an appropriate volume of 0.05 mol/L KOH. The results demonstrate the expected linear dependence of the streaming potential on the pressure difference. The slope (i.e. the streaming potential coupling coefficient dUstr/dΔp) is used to calculate the surface zeta potential according to Equation (3): 29
where η and εrel are the viscosity and dielectric coefficient of water, ε0 is the vacuum permittivity, and κB is the electric conductivity of the aqueous solution.
Laser-scanning confocal fluorescent microscopy (LCFM) was used to localize the presence of (auto)fluorescing, non-cellulosic compounds within different cotton yarns as a function of the type of enzyme applied and time of enzymatic treatment (5–60 min). Approximately 1 cm long, cotton yarn pieces were positioned on a transparent glass holder above the 20× dry objective lens of an inverted CFM Leica TCS SP5 II (Leica Microsystems, Wetzlar, Germany), equipped with an LAS AF software program. For better contact with the glass holder, yarn pieces were pressed with a transparent adhesive tape. The excitation was performed with an argon laser with λex = 458 nm, while the obtained signal was collected by a hybrid HyD3 detector, with a wavelength emission range from 520 to 580 nm, which corresponds to selectively collecting the pectin signal as reported in the literature. 30 High-resolution images (1024 × 1024 pixels) were obtained by image-adjusted light gain and 8× line averaging. Yarns were examined at several positions, in the x–y direction, while in the z-direction (yarn thickness), the confocal mode with 20 µm stack thickness was applied. By moving the focal plane in the z-direction, up to 30 optical slices were obtained, and combined further in a three-dimensional (3D) image stack for digital processing. The bright-field images were captured in parallel, to also depict the non-fluorescing areas of the yarn, using a Dodt detector. The images from both the fluorescence and bright-field channels were used in overlaid mode and further processed by the ImageJ program to measure fluorescence intensity. Data were obtained by selecting several regions of interest (ROIs) with an area of 10,000 pixels (100×100 square selections), and software was used to calculate medium, mean, max, and min fluorescence intensity expressed as a gray value. Data were presented as the average of 10 measurements and SD.
Results and Discussion
Adsorption of the Alkaline and Acid Pectinases Followed by Lowry’s Method
Adsorption Kinetics Under Optimal Working Conditions of Enzymes Without Wetting Agent
The adsorption kinetics of alkaline and acid pectinases onto raw and mercerized cotton yarns were followed under their optimal working conditions (pH 9, liquor ratio 50:1 at 55°C for BioPrep 3000L, and pH 4 liquor ratio 50:1 at 45°C for NS 29048) using 2 ml/L enzyme concentration in the absence of a wetting surfactant. Although a decrease in the enzyme concentration in the bath is expected due to their adsorption onto raw or mercerized cotton, the concentration of the proteins in the bath increased or stayed unchanged. The concentration of the proteins increased to about 0.3 g/L after 90 min when raw cotton was treated with both enzymes (see Figures 1(a) and (b)). The increased protein concentration in the bath can be attributed to the release of fiber proteins from the surface of raw cotton.6,7 On the contrary, when mercerized yarns are treated with alkaline and acid pectinases, there are no changes in the concentration of the proteins in the bath (see red line in Figure 1(a) and (b)), indicating either no release or a release of negligible amount of fiber proteins from the surface of the mercerized cotton.
Kinetic of released proteins from RAW and M80 cotton during treatment with (a) 2mL/L alkaline pectinase BioPrep 3000L (pH 9, liquor ratio 50:1 at 55°C in the absence of a wetting agent), (b) 2mL/L acid pectinase NS 29048 (pH 4, liquor ratio 50:1 at 45°C in the absence of a wetting agent), and (c) kinetic of released proteins from RAW cotton during treatment with BP and NS at their working conditions in the presence of 2 mL/L initial concentration of enzymes and 1 mL/L Kemonecer NI (wetting agent).
Adsorption Kinetics Under Optimal Working Conditions of Enzymes With Wetting Agent
In the next experiment, adsorption under optimal working conditions of both enzymes in the presence of a wetting agent was tested. Wetting surfactant dramatically affects the outcome of the enzyme treatment or more precisely the concentration of released proteins (see Figures 1(c) and 2). It plays a crucial role in enzymatic scouring either by enhancing the penetration of the solution inside the fiber or by improving the pectinase adsorption on the fiber surface. 7 Wetting surfactant also acts as a scouring stimulator, enhancing the release of non-cellulosic components from the cotton surface. 7 Treating raw cotton with alkaline and acid pectinases in the presence of a wetting agent (see Figure 1(c)) shows that, instead of stimulating the adsorption of enzymes, the nonionic wetting agent Kemonecer NI intensifies the release of proteins from the raw cotton when both enzymes are applied. After 90 min, 0.942 g/L of proteins are released in the presence of alkaline pectinase BioPrep 3000L and 0.452 g/L in the presence of acid pectinase NS 29048. Twice as much protein is released when alkaline rather than acid pectinase is applied, which corresponds to the finding that solubilization of proteins in an alkaline medium (pH 9) is more intensive than in acid conditions.2,31
(a) Kinetic of released proteins from the mercerized (M80) cotton yarns during treatment with alkaline pectinase (pH 9 at 55°C) and (b) acid pectinase adsorption (pH 4 at 45°C) presented as the rate of adsorbed proteins (enzymes) on the mercerized cotton. Both experiments were performed in a bath with a liquor ratio of 50:1 in the presence of 1 mL/L Kemonecer NI and 2 mL/L initial concentration of enzyme.
When mercerized cotton is treated with alkaline pectinase under optimal working conditions in the presence of a wetting agent (see Figure 2(a)), increased protein concentration in the solution indicates that natural proteins are released from M80 cotton. In an alkaline medium, the thin waxy film is easily removed, making the mercerized and scoured cotton with alkaline pectinase more hydrophilic than the same one scoured with acid pectinase. 23 On the contrary, when mercerized cotton is treated with acid pectinase, the percentage of the protein in the bath decreases because enzyme adsorption occurs over fiber protein release. Therefore, the adsorption of acid pectinase on mercerized cotton, shown in Figure 2(b), is presented as the rate of the concentration of the enzyme in the bath at a given time divided by its initial concentration. As the enzyme adsorption occurs, the adsorption rate decreases. Since the adsorption rate does not significantly change after 30 min, those adsorption data are presented in Figure 2(b). The results show that the rate of the adsorption of acid pectinase has the highest intensity in the first 10 min, where about 12% of NS is adsorbed on the surface of mercerized cotton yarn.
Two-Step Adsorption Kinetics at Ambient (20°C) Temperature in the Presence of Wetting Agent
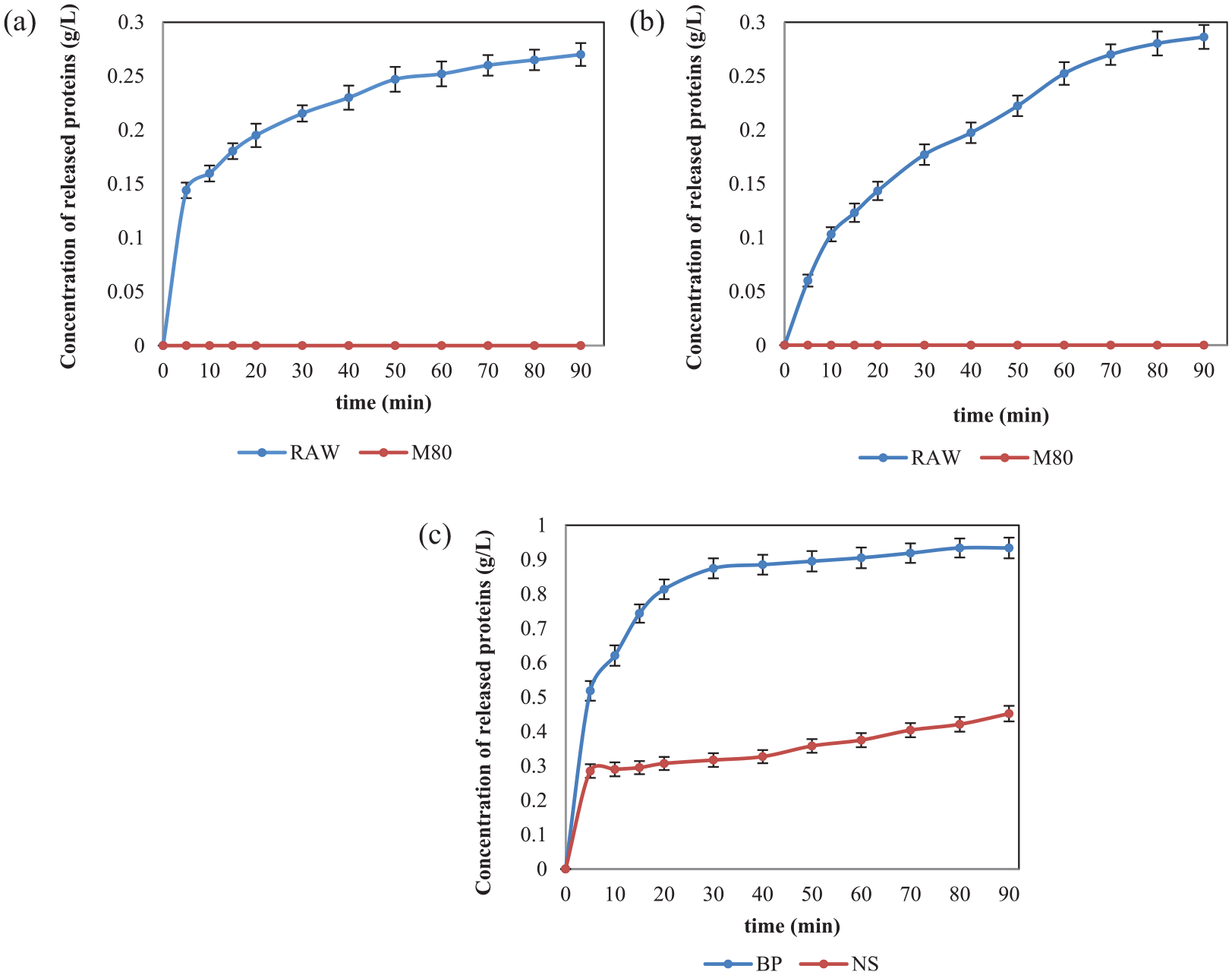
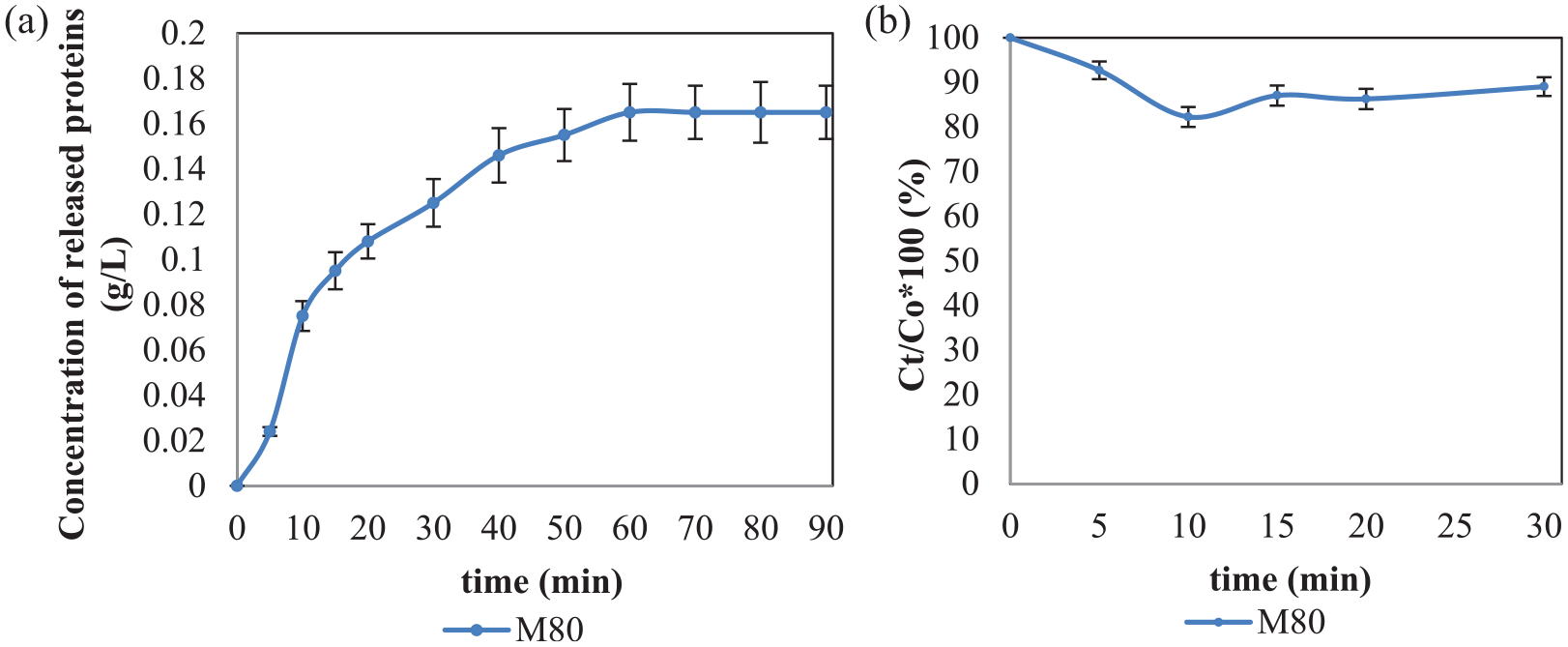
In Figures 1 and 2, the adsorption kinetics of alkaline and acid pectinases onto raw and mercerized cotton yarns were followed under their optimal working conditions using 2 mL/L enzyme concentration in the absence and presence of wetting surfactant, respectively. These experiments were conducted by adding an enzyme at the beginning, and then the concentration of the protein (enzyme) during the time was tested. Unfortunately, instead of decreasing the protein concentration (decreasing the enzyme concentration as a result of its adsorption onto the cotton surface), the concentration of proteins increase as a result of the release of fiber proteins. These two overlapped processes occurred at the same time disabling real examination of enzyme adsorption. A two-step experiment was conducted to split these two processes (enzyme adsorption and the release of the fiber proteins). In the first step, a 15 min treatment of raw and mercerized cotton was performed with the buffer and the wetting agent, and in the second step, the cleaned cotton (without proteins on its surface) was treated with 1 mL/L enzyme at 20°C in the presence of wetting surfactant.
During the first step of this experiment, the fiber proteins are released from the surface of the raw, but not from that of the mercerized cotton yarns (Table 1). Next, the enzyme is added, and the change in enzyme concentration in a bath is measured. Even though the adsorption rate is followed for 90 min, only the adsorption in the first 30 min has changed substantially, so only the results for this time interval are presented in Figures 3(a) and (b).
Concentration (g/L) of released protein in the presence of 1 mL/L Kemonecer NI and alkaline (pH 9) and acid (pH 4) buffers in a bath with liquor ratio 50:1 at 20°C.
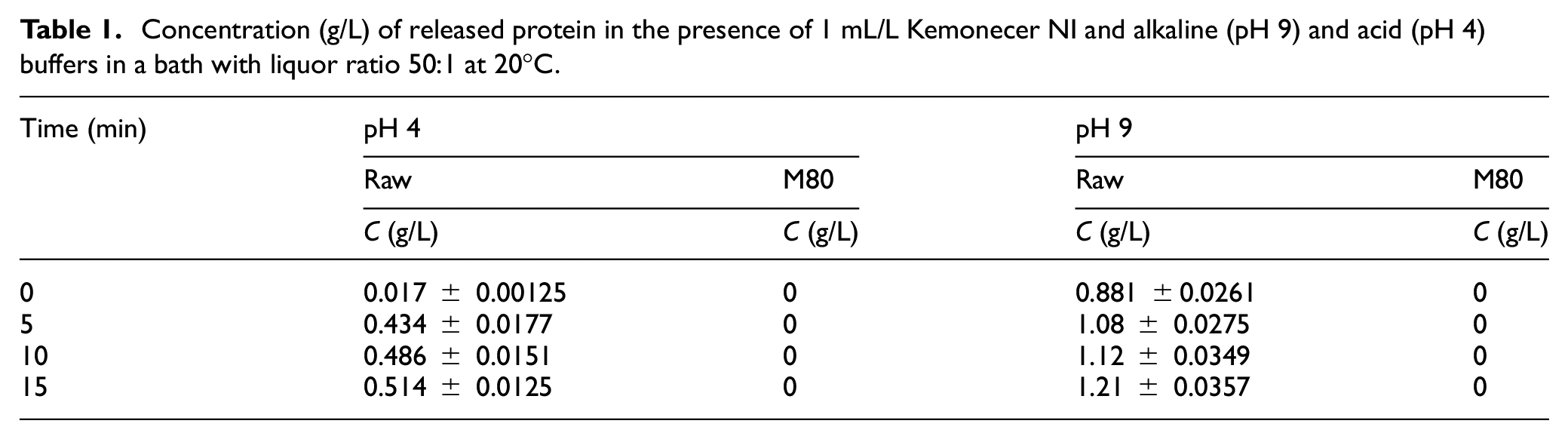
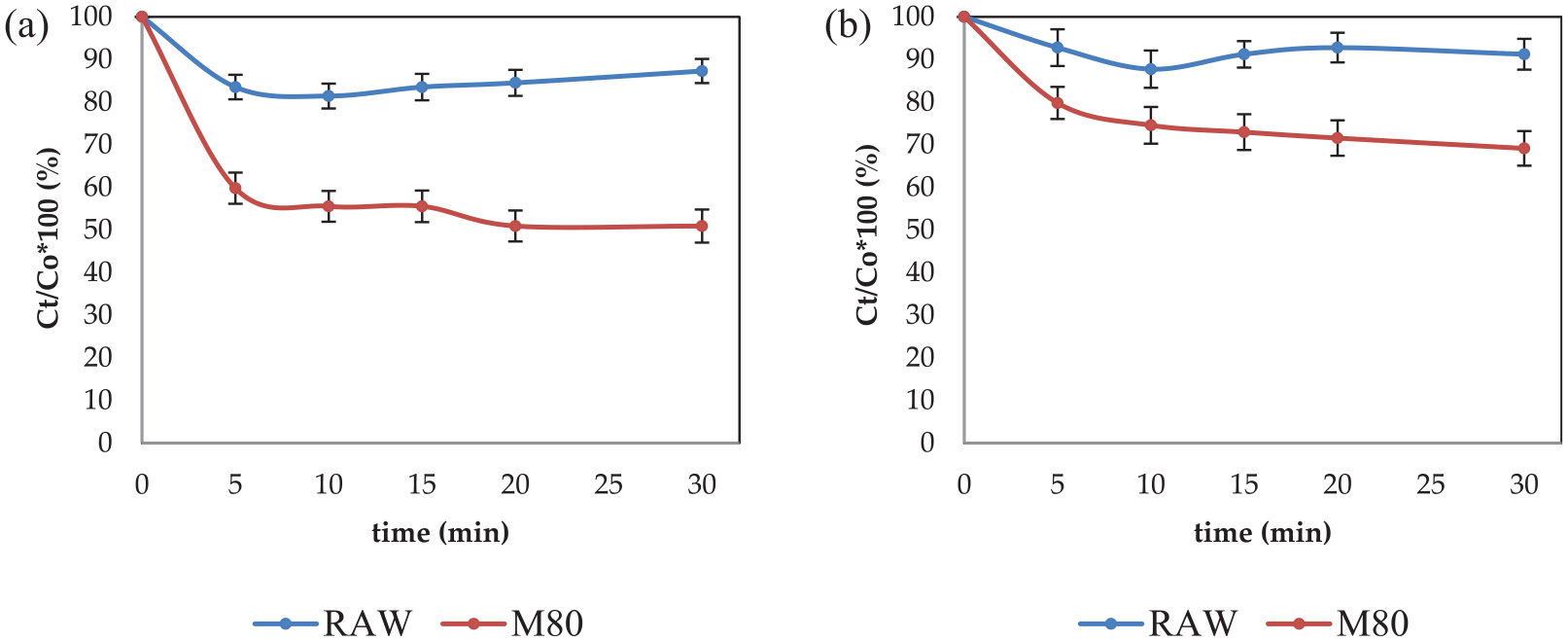
Rate of (a) alkaline and (b) acid pectinase adsorption on RAW and M80 cotton yarns prewashed in the buffer to remove native proteins, before adding enzymes at time 0. The experiment was performed in a bath with a liquor ratio 50:1 at 20°C in the presence of 1 mL/L Kemonecer NI and 1 mL/L initial concentration of enzyme.
The adsorption of alkaline pectinase (see Figure 3(a)) is more intensive than the adsorption of acid pectinase (see Figure 3(b)) to all tested substrates and is substantially higher to mercerized than raw cotton yarn. Eighteen percent of alkaline pectinase BioPrep 3000L is adsorbed on RAW cotton at 10 min and almost 50% on the mercerized cotton yarn after 30 min. Acid pectinase is adsorbed 12% on RAW and 30% on mercerized cotton, at 10 and 30 min, respectively. The adsorption of both pectinases onto mercerized cotton excludes the hypothesis that the reason for the low scouring efficiency of mercerized cotton is the absence of enzyme adsorption. Since enzymatic scouring of mercerized cotton yarn was less efficient than that of raw cotton using the same concentration of enzyme, to achieve desired hydrophilicity of mercerized cotton, a greater concentration of the enzyme is necessary.
Zeta Potential of Raw and Mercerized Cotton Yarns
The electrokinetic properties describe the electrical potential adjacent to a solid surface when it is surrounded by a liquid phase. It is described by the zeta potential, which is the potential at the borderline between the stationary and mobile liquid phases. 32 The potential of the fiber is crucial to its adsorption ability. It depends on the surface composition, the liquid’s chemical properties, the chemical nature of dissociable surface groups, the specific adsorption of ions, surfactants, and poly-electrolytes at the solid surface, and the ionic strength and the pH value of the liquid phase. 31 In a range of pH 2–9, cotton has negative zeta potential due to the presence of carboxyl and hydroxyl groups. 33 Different processing steps of cotton, such as scouring, bleaching, and mercerization, change its zeta potential, and therefore, change its adsorption properties too.34,35
Since the changes in the cotton during mercerization could influence the zeta potential 35 and hence the adsorption ability of the used enzymes, the surface zeta potential of the raw and mercerized cotton was measured. The correlation pH versus zeta potential for untreated and mercerized cotton yarns presented in Figure 4 shows that above pH 5, the surface charge of either untreated or mercerized cotton yarn is indistinguishable. Below pH 4, a distinct evolution of the zeta potential with pH for untreated and mercerized cotton yarn is seen. Even though the surface zeta potential of the tested samples is similar, the adsorption of the tested enzymes is more prominent in the mercerized relative to raw cotton. This result excludes the hypothesis that the surface charge (zeta potential) of mercerized cotton influences the different adsorption between raw and mercerized cotton as explained in Figure 3.
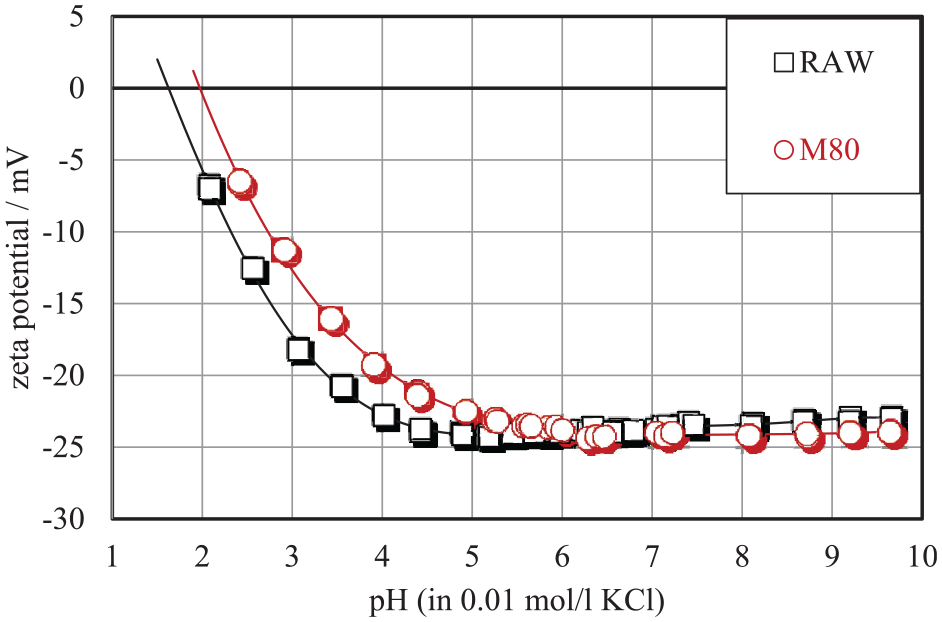
Zeta potential versus pH for raw and mercerized cotton yarn in 0.01 mol/L KCl.
LCFM Imaging of Raw and Mercerized Cotton Yarns Before and After Alkaline and Acid Pectinases Adsorption
LCFM imaging (see Figure 5) is a non-destructive method for microimaging of samples, without demanding preparation steps. This technique permits the localization and depiction of the (auto)fluorescing compounds at light microscopy resolution, allowing us to use it for the examination of cotton yarns containing (auto)fluorescing, non-cellulosic matter, and adsorbed enzymes. Figure 5(a) presents micrographs obtained from the maximum intensity of the consecutive images, comprising the z-stack. The micrographs reveal obvious differences in fluorescent signal intensity between raw and mercerized cotton yarns, implying that all compounds which are released from the raw cotton at the mercerization step may contribute to the signal, including waxes (presenting a mixture of alkanes, fatty acids, fatty alcohols, and plant steroids), proteins, pectin compounds (mainly in the form of pectic acid and rhamnose), and even foreign matters.
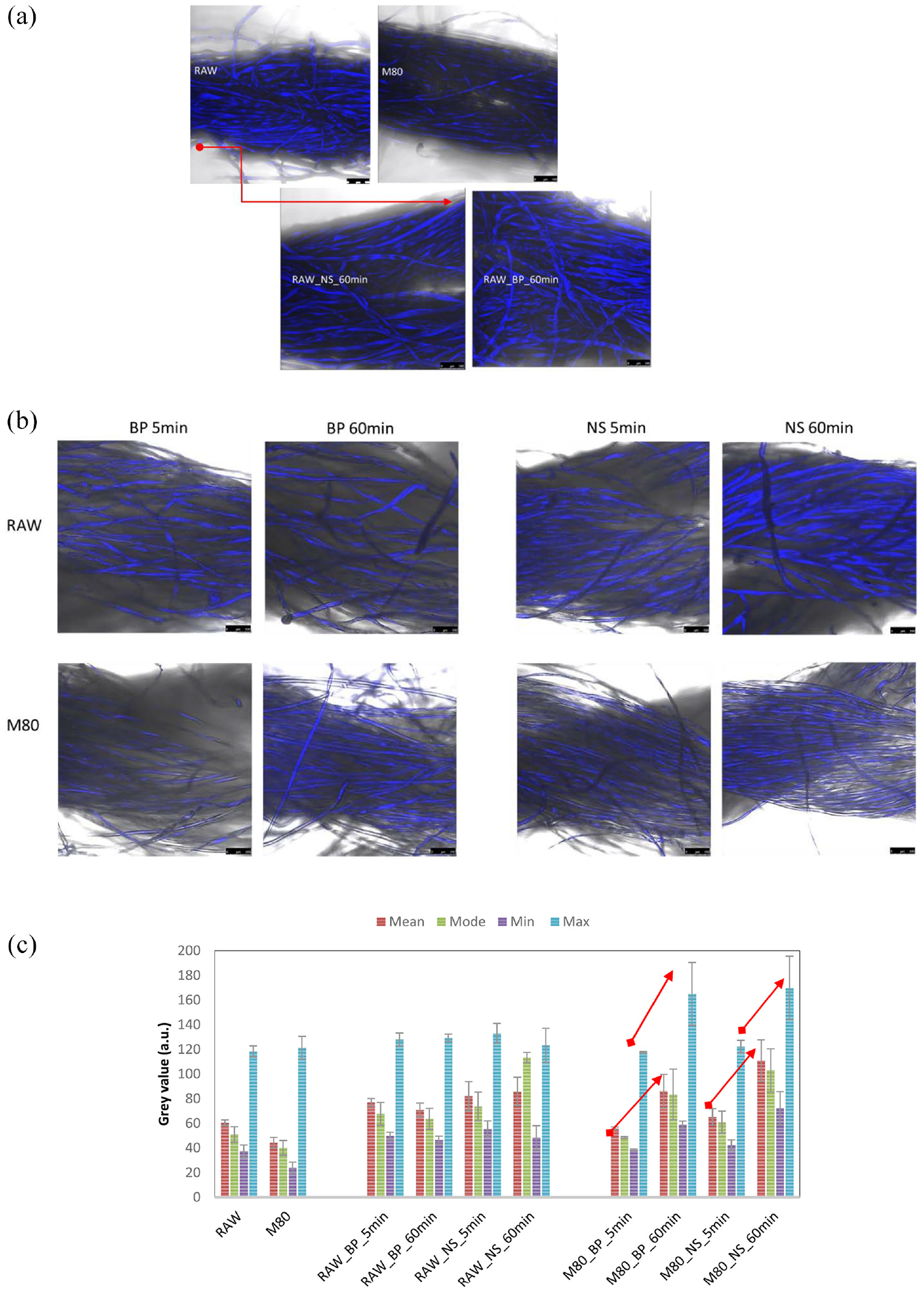
Laser-scanning confocal microscopy micrographs from the different cotton yarns (raw and M80), before and after 5 and 60 min exposure to alkaline (BP) and acidic (NS) pectinase. Images are obtained from the z-stack, as maximum z-projections (a) or from single, x-y focal plains (b). Scale bar in all micrographs corresponds to 100 µm. Overlaid signals of fluorescent and bright field channels are presented. Fluorescence intensity expressed as gray values (arbitrary units (a.u.)) presented under (c) is extracted and calculated by Image J program.
Even when the selected range for collection of fluorescent signal (i.e. 520–580 nm) is per the setup for pectin detection from a previous study, 30 the contribution of another non-cellulosic compound to this signal cannot be completely excluded. Indeed, the identification of a single source of fluorescence is a complex task and it requires comprehensive analytical tools. Furthermore, the pectin-specific labeling with the fluorescent probe may upgrade this finding; however, this requires an additional analytical toolbox, which is beyond the scope in this study.
To track the change in fluorescent signal after different times of exposure of RAW and M80 yarns with both enzymes, the respective micrographs are presented in Figure 5(b). All micrographs capture the presence of fluorescent compounds on the surface of RAW and M80 cotton yarns. As the time of enzymatic treatment lapses, the fluorescence intensifies in M80, while remaining similar in the case of RAW cotton yarn. The differences in calculated gray values related to fluorescence intensity (see Figure 5(c)) are most pronounced within the max and mode data, where both imply significant change (increase) in the case of M80 cotton, for both enzymes. The calculated gray values also increase with the duration of treatment, that is, for ~70% (mode value) and ~40% (max value) in the case of both. This suggests that enzymes contribute to the signal, implying the adsorption of enzymes onto the cotton yarn surface. The fluorescence of pectinases is already reported for pectinases with exo-polygalacturonase and pectin lyase activities 36 because of the presence of fluorescing amino acids, such as tryptophan. The micrograph in Figure 5 and the fluorescence intensity data certainly supplement the findings that adsorption of both enzymes occurs, and the amount of the adsorbed enzyme increases with time.
Conclusion
The adsorption ability of alkaline and acid pectinases on raw and mercerized cotton yarns is examined using the Lowry’s method for determining protein concentration in the bath. Alkaline pectinase adsorbs to a greater extent than acid pectinase. The adsorption of both enzymes is more intensive onto the mercerized than raw cotton. Eighteen percent of alkaline pectinase BioPrep 3000L and 12% of acid pectinase NS 29048 are adsorbed on raw cotton for 10 min, while 50% of BioPrep 3000L and 30% of NS 29048 are adsorbed on mercerized cotton after 30 min. The surface zeta potential, which is similar to that of raw and mercerized cotton, does not significantly influence the adsorption ability of the used enzymes. Raw and mercerized cotton have indistinguishable zeta potentials with values of pH above 5, while differences between them become obvious at a pH below 4. The adsorption of enzymes is also suggested by the LCFM study, where an evident increase in fluorescing signal with scouring time was detected. The adsorption of alkaline and acid pectinase onto mercerized cotton excludes the hypothesis that the low scouring efficiency of mercerized cotton is due to the absence of pectinase adsorption.
Footnotes
Acknowledgements
The authors acknowledge Azra Osmić for the preparation of samples for confocal analysis and Prof. Dr Andraž Stožer (Institute of Physiology, Faculty of Medicine, University of Maribor, Slovenia) allowing access to the LCFM unit. The author would like to acknowledge the Research Core Program Group for Textile Chemistry P2-0118, financed by the Slovenian Research and Innovation Agency (ARIS).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.