Abstract
Total shoulder arthroplasty is an increasingly popular option for the treatment of glenohumeral arthritis. Historically, the effectiveness of the procedure has largely been determined by the long-term stability of the glenoid component. Glenoid component loosening can lead to clinically concerning complications including pain with movement, loss of function, and accumulation of debris which may require surgery to revise. In response, there has been a push to optimize the design of the glenoid prosthesis. Traditional contemporary glenoid components use pegs for fixation and are made entirely of polyethylene. Variations on the standard implant include keeled, metal-backed, hybrid, augmented, and inlay designs. There is a wealth of biomechanical and clinical studies that report on the effectiveness of these different designs. The purpose of this review is to summarize existing literature regarding glenoid component design and identify key areas for future research. Knowledge of the rationale underlying glenoid design will help surgeons select the best component for their patients and optimize outcomes following TSA.
Introduction
Since the advent of Neer's first glenoid prosthesis in 1974, anatomic total shoulder arthroplasty (TSA) has been established as a reliable procedure that provides pain relief and restores function for patients with osteoarthritis of the glenohumeral joint.1–4 The procedure involves surgical removal of arthritic surfaces around the glenoid cavity and the humeral head, followed by implantation of metal and polyethylene prostheses that articulate with each other (Figure 1). Between 1993 and 2007, the volume of TSA procedures increased at a rate of 6–13% annually, with a further estimated 200–300% increase in procedure rates between 2007 and 2015. 5 In 2017 alone, there were 40 665 recorded cases of anatomic TSA 6 ; according to the American Academy of Orthopaedic Surgeons, about 53 000 TSAs are performed annually in the U.S as of 2021. 7

Shoulder X-rays taken from a patient with severe end-stage arthritis who underwent TSA. During the procedure, a concave glenoid component is fixated within the glenoid fossa and the humeral head is resected and replaced with a semi-spherical prosthesis. The pre-operative X-ray (a) demonstrates the near-total absence of cartilage in the glenohumeral joint, causing the humeral head to grind painfully against the glenoid fossa. The post-operative X-ray (b) shows the anatomic TSA system in place with improved joint spacing. The humeral component is stemless, and the glenoid component is indicated by the radiopaque marker.
The rising popularity of TSA has led to an increased focus on long-term prosthetic implant survival. One major factor in prosthetic survival is the stability of the glenoid component. Loosening and subsequent failure of the glenoid component are common, accounting for as many as 24% of all TSA complications. 8 Radiolucent lines around glenoid implants have long been implicated as a harbinger of glenoid component loosening and possible failure.9,10 However, the association between glenoid radiolucency and clinical outcome remains controversial. The incidence of postoperative glenoid lucent lines has been estimated at anywhere from 30–90%11,12 while glenoid component survival has been estimated at over 97% at 5 years.10,13
Various factors influence glenoid component loosening, including component design, surgical technique, condition of the native glenoid and rotator cuff muscles, and patient characteristics. Modern glenoid components also vary in shape, size, and configuration. Despite the variety of unique designs, there is a paucity of literature on associated long-term outcomes, leaving surgeons to rely on design rationale and existing studies to inform their choice of the component. This review aims to summarize the current literature regarding glenoid component design with respect to optimal peg geometry, non-conventional designs, cementation technique, and radius of curvature. Elucidating these considerations will help inform surgical decision-making and maximize clinical outcomes following TSA.
Peg Design
Current pegged designs use three, four, or five pegs. These vary in geometry; some use three in-line pegs, while others use one central peg in conjunction with three or four peripheral pegs. Evidence suggests that divergent pegs may improve fixation, although 18% of patients displayed radiolucency around two pegs at a mean follow-up of 5.6 years. 14 Glenoid components that utilize pegs for implantation and fixation are widely considered superior to pieces that use keels (Figure 2a). Dillon et al 15 found that cemented keeled glenoids had a 6-year cumulative revision incidence of 2.4% due to glenoid loosening, compared to 2% for all-cemented pegged and 0.8% for central-pegged ingrowth designs. Interestingly, in-line pegged designs have similar long-term radiographic and clinical failure rates compared to keeled designs and have an even higher rate of radiographic loosening. 10 This suggests that peripheral pegs are essential for improving the stability of the glenoid component.

Common variations of glenoid component designs. (a) shows an all-polyethylene (PE) keeled component. (b) shows an all-PE pegged glenoid component with three peripheral pegs and a fluted central peg. The central peg's radial fins allow for bony ingrowth and enhance long-term biologic fixation. (c) shows Zimmer's Trabecular Metal™-backed glenoid component with a PE articulating surface. (d) is a hybrid glenoid with three peripheral PE pegs and a porous central metal peg that allows for bony ingrowth. (e) is an augmented glenoid intended to address posterior glenoid bone loss and retroversion.
Pegged designs continue to evolve. Fluted central pegs allowing bony ingrowth (Figure 2b) are becoming more widespread. Kilian et al 16 compared radiographic lucency rates between a finned, cementless central pegged glenoid and a conventional cemented pegged glenoid component. The authors found no significant difference in the rate of glenoid lucency between the two designs at immediate or 35-month follow-ups. On the other hand, multiple studies report promising outcomes for bony-ingrowth designs. Partially cemented glenoids with radial fins on the central peg allow for long-term biologic fixation, show excellent survival and incorporation, have remarkably low rates of radiolucency and encourage bony ingrowth and osseointegration.17–24 However, the mean follow-up was 3–5 years, and studies of long-term outcomes are lacking.
There is also a lack of literature that focuses explicitly on peg design and configuration. Therefore, further study of the impact of peg length, diameter, number, and geometry on fixation and pull-out strength is warranted to understand better the association between peg design and long-term glenoid stability.
Metal-backed, Hybrid, and Augmented Designs
The search for more stable long-term glenoid fixation has led to several variations on the traditional all-PE component, including metal-backed, hybrid, and augmented designs.
Metal-backed
Metal-backed glenoid components (Figure 2c) made of porous tantalum were designed to mimic bone's mechanical and functional properties and promote bony ingrowth. First introduced by Zimmer in 2003, these Trabecular Metal™-backed glenoids were soon found to have an unacceptably high rate of glenoid failure due to fractures at the keel-glenoid face junction. 25 This forced the manufacturer to revise the design and release a second-generation trabecular metal-backed component in 2009. Merolla et al 26 reported that these second-generation metal-backed glenoids had excellent clinical outcomes at a mean follow-up of 38 months, with radiolucent lines observed around the glenoid component in 5% of patients. In contrast, Watson et al 27 studied the same second-generation metal-backed glenoid component and reported a 25% rate of radiographic metal debris and osteolysis at a minimum 2 years follow-up with one catastrophic failure; the high rate of severe complications led them to stop using the implant in their practice. These results corroborate those of Endrizzi et al, 28 who found that 44% of patients with a trabecular metal porous tantalum glenoid component had metallic tantalum debris formation at a minimum of 2 years follow-up. Fucentese et al 29 also described failure rates (14%) using a pegged, soft-metal-backed glenoid design. Most authors agree that the uncemented, press-fit metal-backed implants offer stable fixation; however, given the troubled history of metal-backed components and conflicting reports in the current literature, longer-term follow-up is necessary to compare the clinical outcomes of metal-backed and polyethylene glenoids.
Hybrid
Hybrid glenoid implants (Figure 2d) were designed to combine the reliable initial fixation characteristic of metal-backed glenoid components with the long-term biologic fixation provided by bony-ingrowth central pegs. These hybrid designs most commonly consist of three peripheral polyethylene pegs that are meant to be cemented during implantation and a central uncemented metal post or cage that encourages bony ingrowth. Grey et al 30 evaluated the outcomes of a hybrid cage glenoid compared to a pegged all-PE glenoid and found the two to be equal in terms of clinical outcome at a minimum follow-up of two years. The hybrid cage glenoid demonstrated radiolucent lines in 13.5% of patients while the all-PE glenoid demonstrated radiolucency in 27.6% of patients; patients who received the hybrid cage glenoid also had significantly less intra-operative blood loss. Gulotta et al 31 also found no differences between a hybrid component with a titanium central post and conventional pegged all-PE components in terms of fixation and clinical outcomes at an average follow-up of 3.2 years. Similarly, Friedman et al 32 reported that hybrid cage glenoids had equally good clinical outcomes to all-PE peg glenoids at mean 50 months follow-up. Taken together, these studies suggest that hybrid glenoid implants are functionally comparable to all-PE pegged glenoids; longer term follow-up is needed to elucidate any differences between the two designs and determine whether one is superior to the other.
Augmented Glenoid
Augmented glenoids (Figure 2e) were developed for use in patients with significant glenoid wear and deterioration of the native joint line to minimize the amount of bone that must be reamed intra-operatively. These augments help counter glenoid retroversion and posterior subluxation. Wright et al 33 and Priddy et al 34 showed that patients who received posteriorly augmented glenoid components had similar functional and clinical outcomes to patients who received standard glenoid components at minimum 2-year follow-up. Stephens et al 35 and Favorito et al 36 also found significant improvements in pain and range of motion along with high rates of central peg osseous integration in patients with posteriorly augmented, all-PE glenoid components. Augmented glenoids appear to be a reliable method of treating patients with significant posterior glenoid bone loss undergoing TSA.
The Use of Bone Cement
The use of bone cement has traditionally been vital to the glenoid component's initial fixation and long-term stability. Conventional all-polyethylene glenoid implants require cement around each peg for initial and long-term fixation. However, the increasing prevalence of uncemented, bony ingrowth central pegs raises questions concerning optimal cementing practice. Churchill et al found that the exothermic reaction used to set cement reaches temperatures that can cause bone necrosis, and the area of bone at risk correlates with the amount of cement used. 37 Furthermore, cementless implantation can reduce overall operative time. Several studies support the idea that minimal use of cement is desirable. De Wilde et al 38 used a fully uncemented all-PE glenoid implant with autologous bone reamings interposed between the interference-fit central peg's radial fins and found radiologic signs of loosening in only 12% of patients at short-term follow-up. This compares favorably to cemented all-PE glenoids, which can develop radiolucent lines in 60–85% of patients within 3 years. 39 Arnold et al, 40 Churchill et al, 17 and Wirth et al 24 used the same glenoid implant as De Wilde but with minimal cement (<1 cm3 total) on the peripheral pegs. Arnold and Wirth used bone paste between the radial fins of the central peg and each found evidence of glenoid lucency in 7% of patients at a 2-year follow-up. Churchill did not place bone paste between the flanges of the central peg and found evidence of glenoid lucency in 25% of shoulders at a 5-year minimum follow-up. Schoch et al 41 reported radiolucent lines in 24% of shoulders following uncemented fixation of a hybrid ingrowth cage PE glenoid component at minimum 2 years follow-up. On the other hand, Jacxsens et al 42 studied a fully-uncemented central peg bony-ingrowth glenoid component and found progressive radiolucent lines in 74% of shoulders with a revision rate of 12% at minimum 5 years follow-up. These findings suggest that glenoid designs with minimally cemented or completely uncemented peripheral pegs and an uncemented, interference-fit fluted central peg loaded with morselized bone hold great clinical significance and warrant further research.
Interestingly, radiolucent lines are most commonly seen around the inferior pegs of an implanted partially-cemented prosthesis. 23 These clinical and radiographic results support the findings of earlier mechanical tests and finite element analyses, which reported that failure often occurs at the inferior edge of cemented glenoid components. 43 Clavert et al 44 recommended that cement mantles should be as thin as possible between the glenoid implant and the bone, and that cement should be optimized around the keel of the implant. Applying these findings to pegged implants suggests that adjusting cement application to optimize fixation of the inferior pegs may help address a standard mode of failure among pegged glenoid components. However, further research is necessary to test these observations.
Glenohumeral Radial Mismatch
The concept of glenohumeral radial mismatch is essential to the long-term stability of the glenoid component and overall outcomes following TSA. The glenohumeral radial mismatch is defined as the difference in radii of curvature between the articulating surfaces of the concave glenoid component and roughly spherical humeral head (Figure 3). The native glenoid fossa has been described as pear-shaped or irregularly oval-shaped; pear-shaped glenoid components can be used to match native anatomy better and prevent excessive overhang, but these are not superior to standard elliptical implants. This irregular shape reflects that the glenoid cavity usually has a different radius of curvature in the coronal or superior-inferior (SI) plane than in the transverse or anterior-posterior (AP) plane.45,46 The average anatomic glenohumeral radial mismatch is around 2.3 mm in the SI plane. 47

A diagram depicting the concept of glenohumeral mismatch, where R1 represents the radius of curvature of the glenoid fossa in the superior-inferior plane and R2 represent the radius of curvature of the approximately spherical humeral head. Radius of curvature is defined as the radius of the circle circumscribed by the arc. When R1 = R2, the joint is completely captive or fully conforming. Current literature suggests that glenoid component radiolucency is significantly decreased when R1 > R2 by at least 4.5 mm and no more than 10 mm.
Walch et al 48 initially found that a radial mismatch between 5.5 mm and 10 mm was optimal for reducing glenoid component radiolucency in anatomic TSA systems. However, the upper limit was not definitively established. For example, one recent study indicated that there is no significant difference in rates of glenoid component lucencies in shoulders with a radial mismatch between 3.4 and 7.7 mm. 49 Another recent study found that a mismatch of greater than 4.5 mm was significantly associated with improved clinical outcomes and protection against radiolucencies. 50 Biomechanical and finite-element analyses suggest that high-conformity designs may protect against abrasive wear while less-conforming designs reduce the potential for mechanical loosening of the glenoid component.51–53 Overall, the literature indicates that less-conforming designs confer protective benefits up to a radial mismatch of +10 mm, after which the risk of glenoid component micromotion increases. 54 In practice, the optimal radial mismatch is multifactorial and likely depends on the patient's native anatomy, on their movement patterns, and on the particular system selected by the surgeon. Table 1 shows recommended mismatch ranges for common glenoid systems according to manufacturer guidelines.
This Table Contains a Sample of the Most Common Glenoid Components Available on the Market. It Lists Key Design Features and Recommended Mismatch Ranges as Obtained from the Manufacturers' Surgical Technique Guides.

Inlay Glenoids
Inlay glenoid components (sometimes referred to as mini-glenoids) are smaller than traditional onlay glenoids. Inlay glenoids are designed to sit within the glenoid fossa surrounded by a rim of native bone (Figure 4). This is thought to reduce eccentric loading and physiologic rocking-horse forces during humeral head translation, thereby improving the implant's stability. Cadaveric and finite element studies have demonstrated that inlay glenoids possess superior biomechanical properties and resist loosening better than pegged and keeled onlay designs.55,56 Inlay TSA has successfully treated primary glenohumeral arthritis, resulting in significant functional improvement and pain relief for varying glenoid morphologies. 57 Davis et al 58 studied inset mini-glenoids in nine shoulders with severe glenoid dysplasia and medial glenoid bone loss and found that the component led to satisfactory clinical outcomes. Gunther and Tran 59 reported significant long-term clinical improvements with no loosening or revision in a study of 21 patients with severely deficient bone treated using inset glenoids. Rondon et al 60 also studied inlay glenoids in the setting of glenoid deficiency and found improved outcomes with low revision rates. Studies have shown that inlay glenoids are also effective in active populations, showing no signs of loosening at short-to-midterm follow-up and allowing a high rate of return to sport and weightlifting.61,62 While few glenoid inlay components are currently available on the market, studies to date are encouraging, and further testing is warranted to evaluate potential benefits over traditional onlay designs.
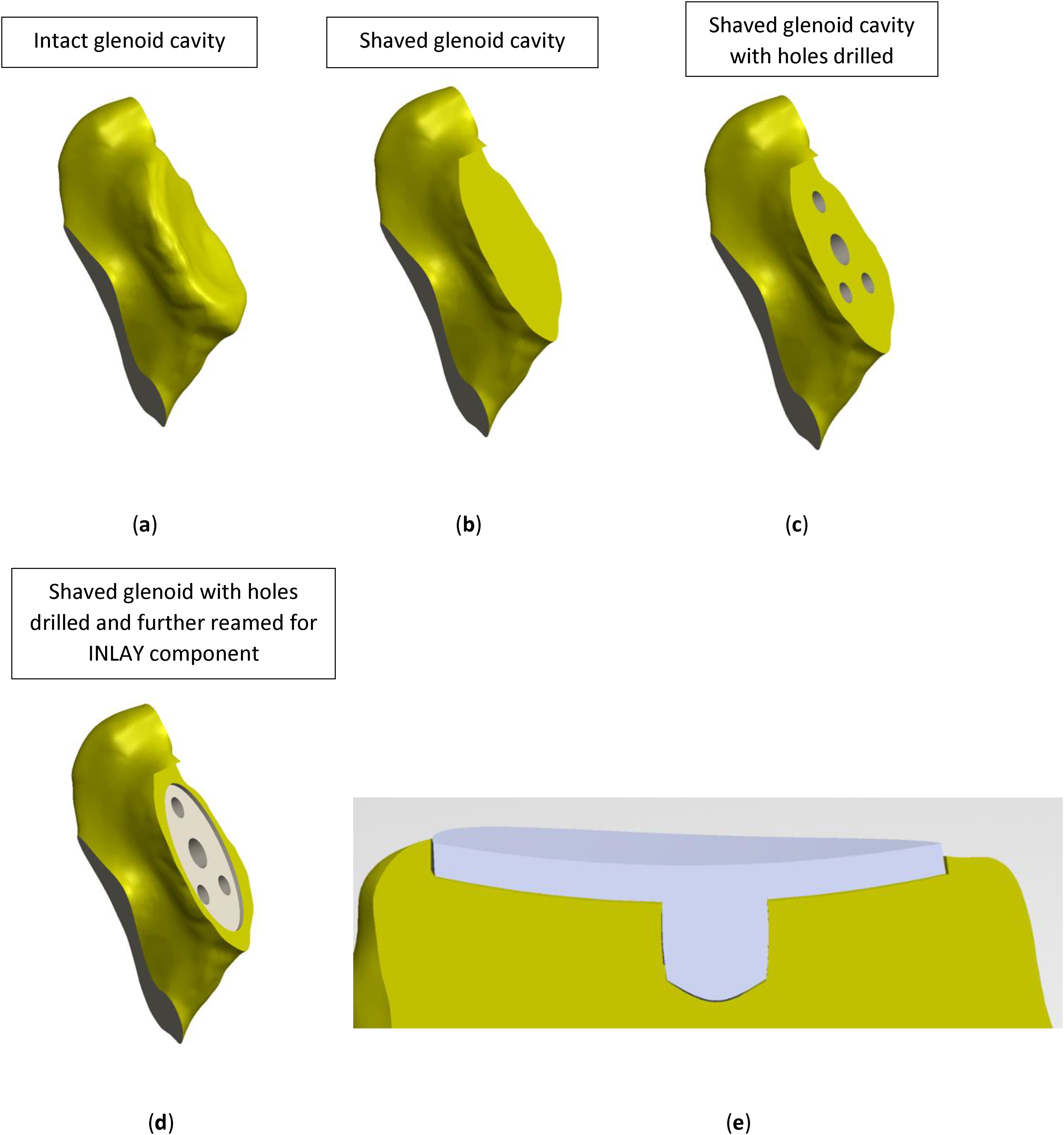
A diagram showing the differences in surgical preparation for inlay and onlay glenoid components. (a) represents an elliptical native glenoid prior to reaming. (b) shows the glenoid face after reaming. This exposes the subchondral layer of bone and leaves an even surface for implantation of an onlay glenoid component. (c) shows the peg holes. (d) also shows the glenoid after reaming and drilling. Here, the glenoid is further reamed and a thin rim of superficial chondral bone is preserved so the inlay glenoid component can be implanted within the deeper subchondral bone. (e) depicts a cross-section of an inlay glenoid implant fixated within the glenoid. The surface of the implant may lie flush with the peripheral rim of bone or protrude slightly.
Conclusion
The glenoid component has undergone many design iterations since TSA first became widely accepted in the 1970s. The glenoid component will continue to evolve as surgeons and manufacturers seek to improve the procedure's reliability, most prominently by ensuring stable long-term fixation. There is currently a lack of research on specific glenoid design considerations such as peg geometry and dimensions. There are also promising avenues for future research on optimal cementing practice and unconventional (such as hybrid) designs. Additionally, while the field has a solid approximation of the ideal radial mismatch, further research is warranted to narrow the range and provide a firm rationale for the accepted values. Other notable areas of interest for glenoid design include inlay components, particularly for bone loss, and the emerging field of patient-specific 3D printing in arthroplasty. While TSA has traditionally been more challenging than total hip or knee arthroplasty due to the shoulder's anatomy, advances in glenoid component design hold great promise for streamlining the procedure and improving patient outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
