Abstract
The indication for total elbow arthroplasty (TEA) for primary and posttraumatic elbow arthritis has increased, however, its indication after infection remains elusive. Wound closure about the elbow increases the challenge of treating a previously infected elbow, often necessitating soft tissue coverage with local or regional flaps. We present a 75-year-old male patient with an elbow infection following a failed complex intraarticular fracture open reduction and internal fixation of the distal humerus. Initially, he presented with severe functional impairment and pain, also with an active fistula with serous exudate, whose culture was positive for Cutinebacterium acnes. Septic hardware loosening, and septic nonunion with intraarticular involvement of the left elbow was diagnosed. The patient underwent hardware removal, fistulectomy, serial irrigation and debridement and a pedicled antegrade posterior interosseous artery (PIA) flap on staged surgical treatment. Finally, after ruling out infection persistence, a TEA was performed. We aim to report the outcome of a patient treated with a TEA in the context of a previously infected elbow with soft tissue coverage with an antegrade PIA flap. Comprehensive treatment must be done in an appropriate manner, to obtain an expedited and desirable outcome.
Keywords
Introduction
Current total elbow arthroplasty (TEA) indications include inflammatory arthritis mostly due to rheumatoid arthritis, severe acute comminuted distal humerus fractures, 1 some primary osteoarthritis and posttraumatic elbow arthritis. Trauma and posttraumatic conditions have expanded TEA indications, up to 69%. 2 Moreover, treatment of an infected nonunion with bone loss is challenging when the joint is involved and the use of a TEA in these cases has scarce reports in the literature. 3 Several methods have been used to maintain anatomy and function of the elbow after implant removal and debridement before definitive surgery, such as cement spacer with and without antibiotic, Illizarov frame, external fixation, etc. Also, loss of flexor and extensor mechanisms should be avoided, protecting anterior capsule and triceps tendon as possible.
In the case of an active fistula, a fistulectomy should be performed, procedure that usually leads to wound closure problems. 4 Therefore, the soft tissue coverage remains a major aspect during the treatment of an infection about the elbow. The posterior interosseous artery (PIA) flap has been described as an alternative option for soft tissue defects smaller than 40cm2, 5 its advantages include that the PIA is very constant, donor site morbidity is scarce and does not require advanced microsurgical technique. Anconeus, brachioradialis, latissimus dorsi, and flexor carpi ulnaris muscle flaps as well as radial forearm and lateral arm fascio-cutaneous flaps are some of the more commonly reported flaps for reconstruction. However, each of these flaps is associated with potential donor-site morbidity or limited coverage. 6
In this case study we describe the use of antegrade PIA flap and early TEA for treatment of an infected complex articular fracture of the distal humerus, previously treated with an open reduction and internal fixation (ORIF), as a prompt and definitive treatment to improve function of the elbow and quality of life for the patient.
Case
A 73-year-old man with hypertension and non-specified heart arrhythmia sustained, twenty months ago, a fall from a height of approximately eight steps with a direct blow over the left elbow causing severe pain, visual analogue scale 10/10, gross deformity, swelling, and disability of that arm (Figure 1). The patient was diagnosed with an intercondylar and supracondylar fracture, and he was treated with an immediate ORIF with fragment specific medial and lateral plates, the approach was performed via an olecranon osteotomy fixated with a tension-band (Figure 2). Six days after the surgery he developed pain, edema, joint swelling and hyperemia around the elbow, and hematic blisters located on the dorsolateral aspect of the elbow. A second surgery was performed to remove three screws located in the lateral plate and redo the olecranon's tension-band.

Initial AP and lateral x-rays views of the left elbow, showing an intraarticular intercondylar and supracondylar comminuted fracture.

Final AP and lateral postoperative x-rays after the distal humerus open reduction and internal fixation.
At his first visit to the office the patient complained of pain visual analogue scale (VAS) 10/10, and severe functional impairment. The patient also referred paresthesias in the medial side of the forearm and in the ring and small fingers. He also had antalgic attitude and seemed low-spirited.
Physical examination revealed an open wound located in the posterolateral aspect of the elbow, of approximately 0.5 cm2 in size, with active non-fetid serous liquid drain (Figure 3), erythema, hyperthermia, swelling, limited range of motion at the elbow joint, along with pain at the superficial and deep palpation in the medial side of the elbow. Also, a positive Tinel sign in the territory of the ulnar nerve about the elbow was present, associated with a diminished strength of the triceps brachii (2/5) and brachialis anterior (3 + /5 in the Daniels Strength Scale). The elbow joint was held in bending position with 90° of flexion, with active range of motion (ROM) of 100° of flexion and −80° of extension. Sequential x-rays revealed loosening of the fixation material, with osteolysis located in the distal humerus (Figure 4). Oral antibiotics were suspended during his initial visit to the office, to prepare the area for wound culture obtention during the first surgery.
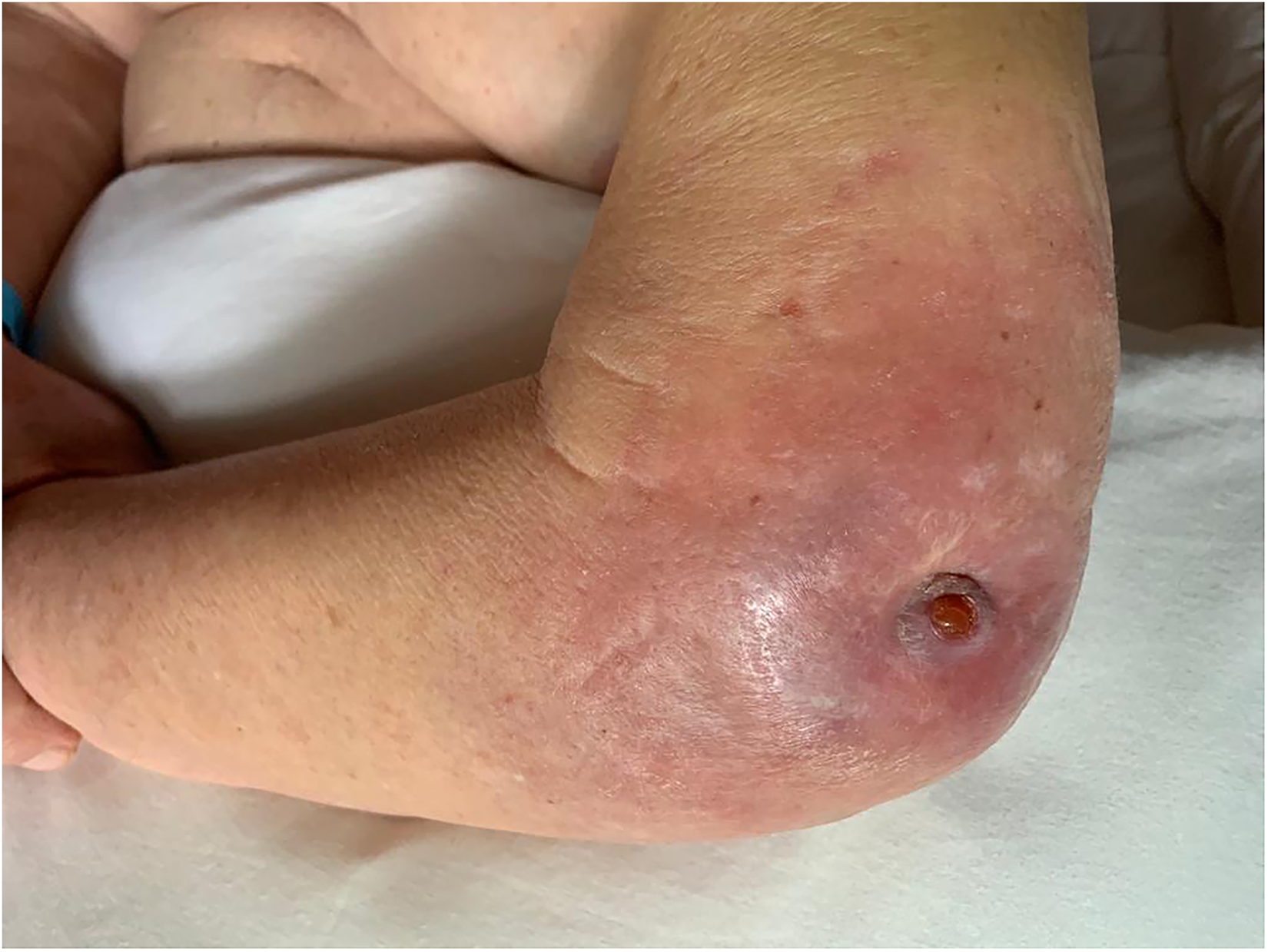
Clinical aspect of the left elbow after the first visit to the office, beneath the active fistula a loosened screw was palpated.

From left to right, sequential x-rays showing the progressive hardware loosening, the previous surgeon decided to remove some of the screws of the lateral plate as shown in the AP and lateral of the far right. Above, AP views; below, lateral views.
A diagnosis of septic hardware loosening, and septic nonunion with intraarticular involvement of the left elbow was established. Our work-up after diagnosing a bone and/or joint infection with an active fistula includes discontinuation of antimicrobial therapy, plain orthogonal x-rays, blood tests including serum markers for active infection. The measurement of these serum markers will be useful during all treatment stages to evaluate the response to the antibiotic therapy and the clinical response to treatment. A CT scan of the elbow was also obtained. It demonstrated changes due to the previous fracture and hardware material in the olecranon and distal humerus with loss of contact in some segments associated with peripheral inflammatory changes. We do not routinely perform scintigram nor the obtention of cultures in the office.
We performed a surgical debridement and thorough irrigation with sterile saline solution. During the procedure the first step is always obtention of cultures using swabs from wound exudate along with tissue. We furthermore performed a fistulography using methylrosanilinium chloride (crystal violet), to identify the fistula path, the involved tissues, and the scarring around the infection. Once the affected tissues were identified, a broad fistulectomy and debridement was performed, until tissue with adequate bleeding and coloration was obtained. Antibiotic-impregnated cement pearls were placed in the area formed after the joint and subchondral bone loss (Figure 5). Subsequently, decompression neurolysis and anterior subcutaneous transposition of the ulnar nerve plus hardware material removal was performed. During this surgery, the evaluation of the triceps status is mandatory. In this patient the extensor mechanism was in continuity, still attached to the olecranon process, with good muscle vitality. A negative pressure assisted wound closure system was placed in the soft tissue defect after the fistulectomy in left elbow. The obtained cultures were positive for Cutinebacterium acnes therefore the patient received parenteral specific antibiotic therapy. Serial surgical debridements were performed until we decided to remove the negative pressure assisted wound closure system and perform a pedicled antegrade PIA flap, to close the skin defect (Figure 6, Video 1).

Free osteochondral fragment obtained during the second irrigation and debridement. The fragment is partially stained with methylrosanilinium chloride, used during the fistulography.

Above, left; defect remaining on the fistula site after the culture obtention and fistulography with methylrosanilinium chloride. Above, right; following the crystal violet staining a thoroughly fistulectomy was performed leaving a 3 × 4cm defect. Below, aspect of the elbow and forearm after the closure with an antegrade posterior interosseous artery flap.

Trans operative video after the antegrade posterior interosseous artery flap placement, good coloration and capillary refill are observed.
During the PIA procedure, a bedside doppler was realized to corroborate PIA presence. Once confirmed the artery presence, the patient was placed on supine position. Analgesia was acquired with the use of a supraclavicular peripheral nerve block. A tourniquet was applied to the proximal third of the arm. Then, the limb was exsanguinated using only the aid of gravity and the ischemia cuff was insufflated up to 220mm Hg. The defect at the elbow was measured, with dimensions of 3 × 4cm, then a piece of Esmarch bandage was used to draw the template of the defect size. The template was made after the debridement, because most of the times debridement results in a larger defect. A straight line was then marked in the dorsal aspect of the forearm from the lateral epicondyle to the distal radioulnar joint (DRUJ), this line corresponds to the course of the artery in the posterior compartment of the forearm, the artery runs in the septum between the ECU and the EDM. The fasciocutaneous branches perforate the deep fascia of the posterior compartment of the forearm along the septum between the fifth and sixth extensor compartments and perfuse the skin of the posterior aspect of the forearm. The drawn line was used to divide the forearm in three equal thirds. The donor site for soft tissue coverage was planned in the distal third of the dorsal forearm previously marked. The flap design was drawn using the Esmarch template with the measurements obtained from the defect to cover the elbow. The pivot point was planned at the union of the proximal and middle thirds of the forearm (approximately 8- 11 cm distal to the lateral epicondyle). At or just above this level, the PIA crosses the posterior interosseous nerve (PIN) which is already in the compartment.
Then the fasciocutaneous flap was elevated in the distal third, the flap has an average of three cutaneous perforators in the distal third of the forearm dorsum arising from the PIA between the EDM and ECU. We recommend elevating first the ulnar aspect of the flap with the fascia over the ECU to reach the ECU-EDM interval, in this way the risk of raising a flap between the EDM and EDC is avoided. Once the PIA perforators were observed between the ECU and EDM at the base of the interval, elevation of the remainder of the flap could be completed. Stitches between the skin and flap fascia were temporary placed to avoid shearing forces over the perforators. Then, the PIA was carefully dissected proximally and raised always between the ECU and EDM. Good hemostasis of the muscular branches of the PIA was done using a bipolar cautery avoiding proximity to the PIA. Skeletonizing the artery should be avoided as well, maintaining the soft tissue around the artery will prevent vasospasm. At the junction of the proximal and middle thirds of the forearm (8-11 cm from the lateral epicondyle) the pivot point of the flap was reached. In our experience we have found this pivot point is sufficient to reach the posterior aspect of the elbow for coverage.
The tourniquet was deflated; vascularity of the flap was confirmed. The flap can be transferred to the elbow defect making a subcutaneous tunnel or by incising the skin to the defect avoiding any pressure over the pedicle. In this case the flap was tunneled. Careful attention should be placed to avoid twisting or kinking the flap pedicle. The flap was sutured to the defect edges using Nylon simple stitches avoiding tension. Donor site defect was closed with a full thickness skin graft (FTSG) obtained from the patient's groin.
Two months after, and once clinical and laboratory parameters were normal, the elbow joint replacement was planned. A PET-CT scan with fluorodeoxyglucose (18F-FDG) was performed to rule out an acute bone infection. To solve the elbow joint destruction once an ongoing infectious process was ruled out, we decided to perform a TEA with a cemented Discovery Elbow System (Biomet, Warsaw, IN, USA) prosthesis (Figure 7). We used this elbow system because at the current moment it is the only elbow replacement system available in our country. The triceps tendon was reflected from its olecranon insertion for ulnar reaming and steam placement. The reinsertion was performed with standard osseous tunnels and steel core sutures. The patient was placed on a soft dressing and immobilized with an extension blocking splint for two weeks, after that period an elbow hinged orthosis was placed and the ROM were gradually increased until we allowed for full range of motion four weeks after surgery. The orthosis was discontinued at six weeks.

Final AP and lateral postoperative x-rays after the cemented total elbow arthroplasty.
At six-months postoperative evaluation DASH score was 21 and Mayo Elbow Score 80. Elbow ROM showed 45° of flexion and 45° of extension. Supination was measured on 45° and pronation in 75°, pain VAS was 0/10. Triceps muscle strength was 4/5, biceps 5/5, hand intrinsic muscles 5/5, in the Daniels Strength Scale. Skin scars were eutrophic, and the flap area showed euthermic, with capillary refill of two seconds, with no signs suggestive of infection (Figure 8, Videos 2 & 3).

Two-month follow-up of the antegrade posterior interosseous artery flap placement, good coloration and capillary refill are observed.
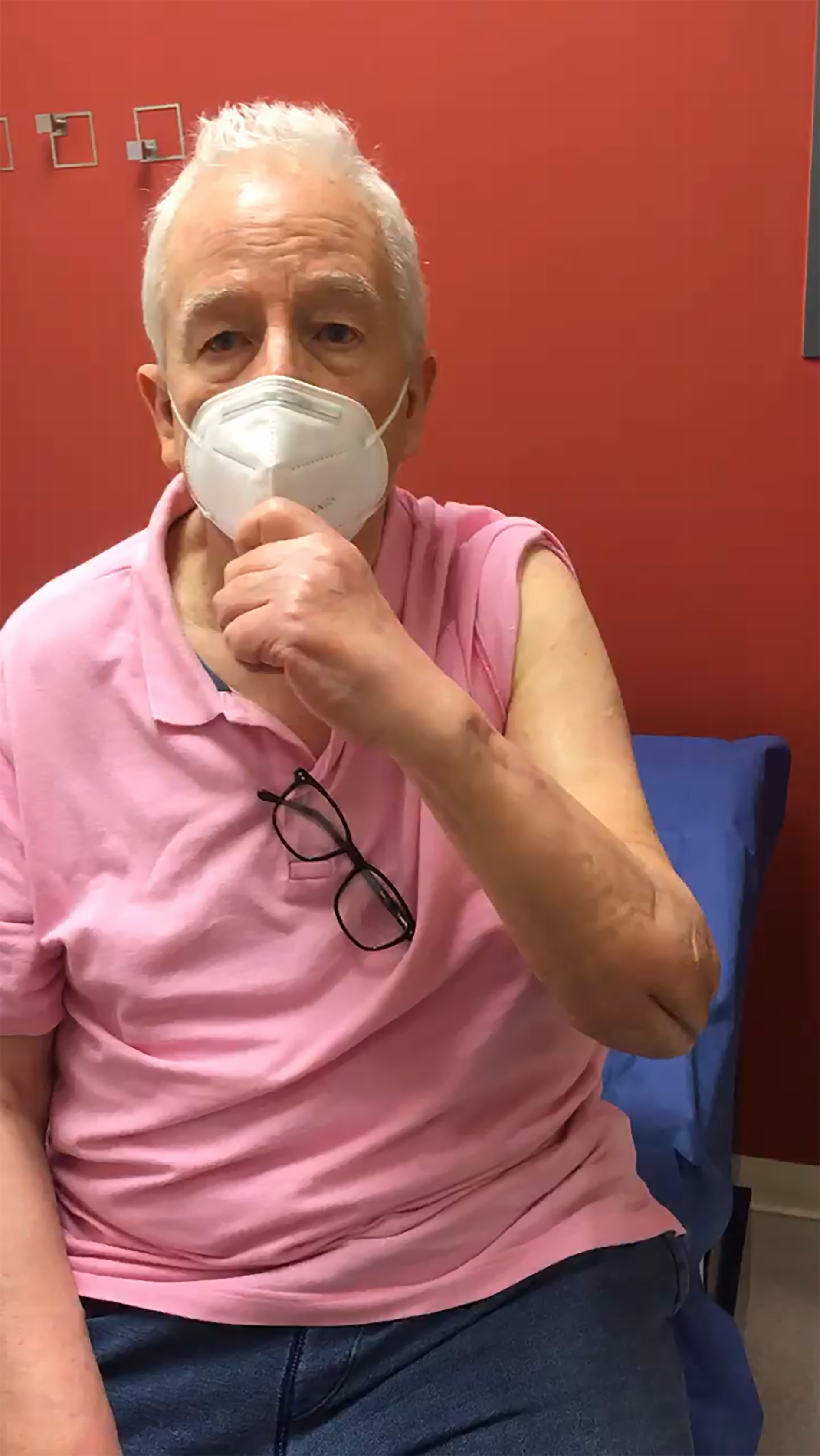
Ten-week follow-up clinical video showing elbow and forearm range of motion.

Clinical aspect of the patient's left upper limb after 6 months. Top, dorsolateral aspect of the forearm in resting position. Second from top to bottom, a slight extension lag of the small finger, asymptomatic. Third form top to bottom, donor site completely healed, with a barely noticing scar. Bottom, flap and posterior scar showing appropriate healing and no signs of infection.
Case Discussion
Elbow arthroplasty was originally developed as a treatment for end-stage inflammatory arthropathy of the humeroradial compartment.7,8 Promising results derived in more indications for elbow replacement in recent years. Prior to the widespread use of disease modifying anti-rheumatic drugs (DMARD's), 43% of all elbow replacements were done in patients with rheumatoid arthritis. 9 It is estimated that currently, only 19% of patients with an elbow arthroplasty suffer from RA.
Predictable results can be obtained in elbow arthroplasty with carefully selected patients. 10 Nonetheless, these surgeries are not as widely performed as knee of hip replacement. This is due to the more recent development of the implants and a lack of research in the field. 11 Also, it is important to notice that series of patients have reported complication rates ranging from 15% to 80%. 11 Being the most frequent complications periprosthetic infection (between 2 & 4%), implant loosening (2%), periprosthetic fracture, ulnar neuropathy (5%), triceps insufficiency (3%), and implant failure.12,13
The ulnar nerve was anteriorly transposed looking for appropriate soft tissue coverage to the nerve. In this case the nerve was not covered with nerve wrapping. However, if substantial scarring is noted, we cover the nerve with vein graft wrapping as described by Sotereanos et al 14 since in our practice we do not have access to porcine small intestine submucosa (SIS, Axoguard Nerve Connector) nor cross-linked bovine collagen type I (CLC, Integra NeuraGen®). However, these biomaterials could be used for wrapping as well. Submuscular anterior transposition of the ulnar nerve could be another option when the nerve has a poor vascular wound. 15
The triceps tendon was reflected from its olecranon insertion following Brian and Morrey, 16 because it offers an extensive posterior exposure to the elbow joint. At the end of the procedure the extensor apparatus was reattached using heavy nonabsorbable braided sutures through olecranon tunnels. After the triceps repair, elbow extension was protected with an elbow hinged orthosis and extension against resistance was forbidden, to prevent triceps insufficiency as recommended. 17 In this case, since the patient had preoperative severe pain and ROM limitation, we were aiming to a functional painless ROM, not expecting full extension recovery.
In recent decades, research has focused on elbow arthroplasty and hemiarthroplasty as treatment for complex and non-fixable fractures of the distal humerus. 18 Currently, open reduction and bicolumnar plating is the gold standard for treatment of these fractures, although failure in fixation, postoperative stiffness and other complications can result with this treatment method. These findings have fueled interest on elbow replacement as treatment for complex fractures. 19
The authors acknowledge that there is contradictory evidence regarding the use of elbow arthroplasties in patients with previous local infection in the elbow.3,18 However, recent research, mainly consisting in case series reports, have demonstrated high success rates in reconstruction of the elbow in patients with treated local infection. 20 Yamaguchi et al, 3 reported on ten consecutive elbow arthroplasties, implanted in patients with the background of local infection, which was treated with newer antibiotics. These patients were given a minimum of two-year follow-up, with implant survival rates of 80% without new signs of infection. The patient reported on this study has a follow up of twenty months to date.
Our patient presented severe elbow post-traumatic arthritis and infection, markedly limiting his activities of daily living. After the removal of the loose hardware and provided adequate debridement and skin coverage, multidisciplinary care including a coadjutant antibiotic therapy dictated by an infectious disease specialist, the local infection was eradicated. Being a low demand patient, with severe limiting arthritis and pain with desire to regain mobility in his elbow, we offered a comprehensive treatment including debridement, soft tissue coverage and a TEA. We believe that this case summarizes a perfect indication for both a primary TEA and a PIA antegrade flap.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
