Abstract
Aim
This study aims to describe the shoulder arthroplasty options for young and active patients (<60 years old) with glenohumeral osteoarthritis.
Methods
A systematic review of the literature was conducted by searching on Pubmed database. Studies that reported outcomes of patients with glenohumeral arthritis, younger than 60 years, that underwent shoulder arthroplasty [(Hemiarthroplasty (HA), Hemiarthroplasty with biological resurfacing (HABR), Total shoulder arthroplasty (TSA), Reversed total shoulder arthroplasty (RSA)] were included. Data include patient characteristics, surgical technique, range of motion, pain relief, outcome scores, functional improvement, complications, need for and time to revision.
Results
A total of 1591 shoulders met the inclusion criteria. Shoulder arthroplasty provided improvements in terms of ROM on the 3 plains, forward flexion (FF), abduction (Abd) and external rotation (ER), in different proportions for each type of implant. Patients submitted to RSA had lower preoperative FF (p = 0.011), and the highest improvement (Δ) in Abd, but the worst in terms of ER (vsTSA, p = 0.05). HA had better ER postoperative values (vsRSA p = 0.049). Pain scores improved in all groups but no difference between them (p = 0.642). TSA and RSA groups had the best CS Δ (p = 0.012). HA group had higher complication rates (21.7%), RSA (19.4%, p = 0.034) and TSA (19.4%, p = 0.629) groups the lowest, and HABR had the highest rate of revisions (34.5%).
Conclusions
HA had the highest rate of complications and HABR unacceptable rates of revision. These implants have been replaced by modern TSAs, with RSA reserved for complex cases. Surgeons should be aware of the common pitfalls of each option.
Introduction
The incidence of glenohumeral osteoarthritis continues to increase as the population ages. Elderly patients reproducibly have success with current shoulder arthroplasty techniques, however, replacement options are less successful in young and active patients (<60 years old).1,2 Even though they represent only approximately 5% to 10% of the shoulder arthroplasty population,3–5 the management of glenohumeral arthritis is particularly challenging in contrast to that in older individuals because: 1) more likely to be in their working prime and higher activity levels further heightening the need for greater durability of the reconstruction; 2) greater functional expectations on the part of the patient; and 3) the greater prevalence of types of arthritis more complex than primary osteoarthritis. 4
Treatment options for this demographic have been pursued with varying outcomes. 2 The best treatment management remains controversial,6,7 and despite the benefits of arthroplasty on pain and functional improvement,8–13 concerns about implant longevity and the need for revision remain a dilemma.3,8,14–20 Numerous surgical options have been proposed including arthroscopic management,2,21–23 hemiarthroplasty (HA),2,3,8,17–19,24–33 hemiarthroplasty with glenoid biological resurfacing (HABR),2,20,31,34–45 anatomical total shoulder arthroplasty (TSA)2–4,14,18,26,27,33,46–50 and reverse total shoulder arthroplasty (RSA).51–56 Generally, TSA consistently improves symptoms and shoulder function,11,57–60 although, glenoid component loosening and need for revisions remain a concern.34,61 HA may be an attractive solution, however, this technique provides significantly less pain relief and functional improvement than does TSA.12,60 HABR was introduced as an alternative 43 and several tissue sources have been used to resurface the glenoid, including autogenous fascia lata (AFL), anterior shoulder joint capsule (ASJC), lateral meniscus allograft (LMA), and Achilles tendon allograft (ATA),34,37,41,62,63 but despite the promising initial results, the high rate of associated complications and revisions identified with longer follow-ups has been discouraging this option.
Although initially implanted in elderly patients with cuff-deficient shoulders, RSA is been used for revision of previously failed shoulder arthroplasty in younger patients, for nonfunctional shoulders after irreparable cuff tears or fracture sequelae, and increasingly in the setting of primary arthritis.51–56 The role of shoulder arthroplasty in young patients with primary glenohumeral osteoarthritis is not clearly defined and most of the literature consists of smaller single-centre studies with heterogeneous patient populations.17–19,27,48,49
This study aimed to gather the available data about the main four shoulder arthroplasty solutions for young patients (<60 years old) with glenohumeral osteoarthritis and to present a descriptive review for each option; secondarily, a comparison between outcomes to address any relevant distinction.
Materials and Methods
Literature Search
An electronic search was conducted in January 2021 by searching on Pubmed database the following term: “(shoulder OR glenohumeral) AND (osteoarthritis OR arthritis) AND (arthroplasty OR replacement) AND (young OR younger)”. We analyzed the titles and abstracts and when the abstract indicated a clinical study including patients younger than 60 years who were treated with shoulder arthroplasty, then the study was selected for further analysis.
Eligibility Criteria
A comprehensive systematic review of the literature was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The selected study titles and abstracts were analyzed according to the eligibility criteria. Inclusion criteria were (1) clinical therapeutic studies in the English language; (2) studies reporting outcomes after surgical management of primary or secondary glenohumeral osteoarthritis; (3) mean-age less or equal to 60 years and (4) cases treated with HA, HABR, TSA or RSA. The exclusion criteria were (1) non-pertinent studies as reviews of the literature, technical notes and non-therapeutic studies; (2) case reports and case series with less than 10 patients; (3) studies reporting Arthroscopic Debridement or Ream-to-Run of the glenoid; (4) clinical follow-up of fewer than 18 months; and (5) clinical outcomes not reported at the final of the follow-up. No restrictions were imposed on the publication date or the prosthesis designs. Full articles were reviewed for eligible studies, and their references were screened to identify additional studies that may have been missed. Other three systematic reviews were founded and missed papers were integrated. A PRISMA trial flow shows the study selection algorithm (Figure 1).
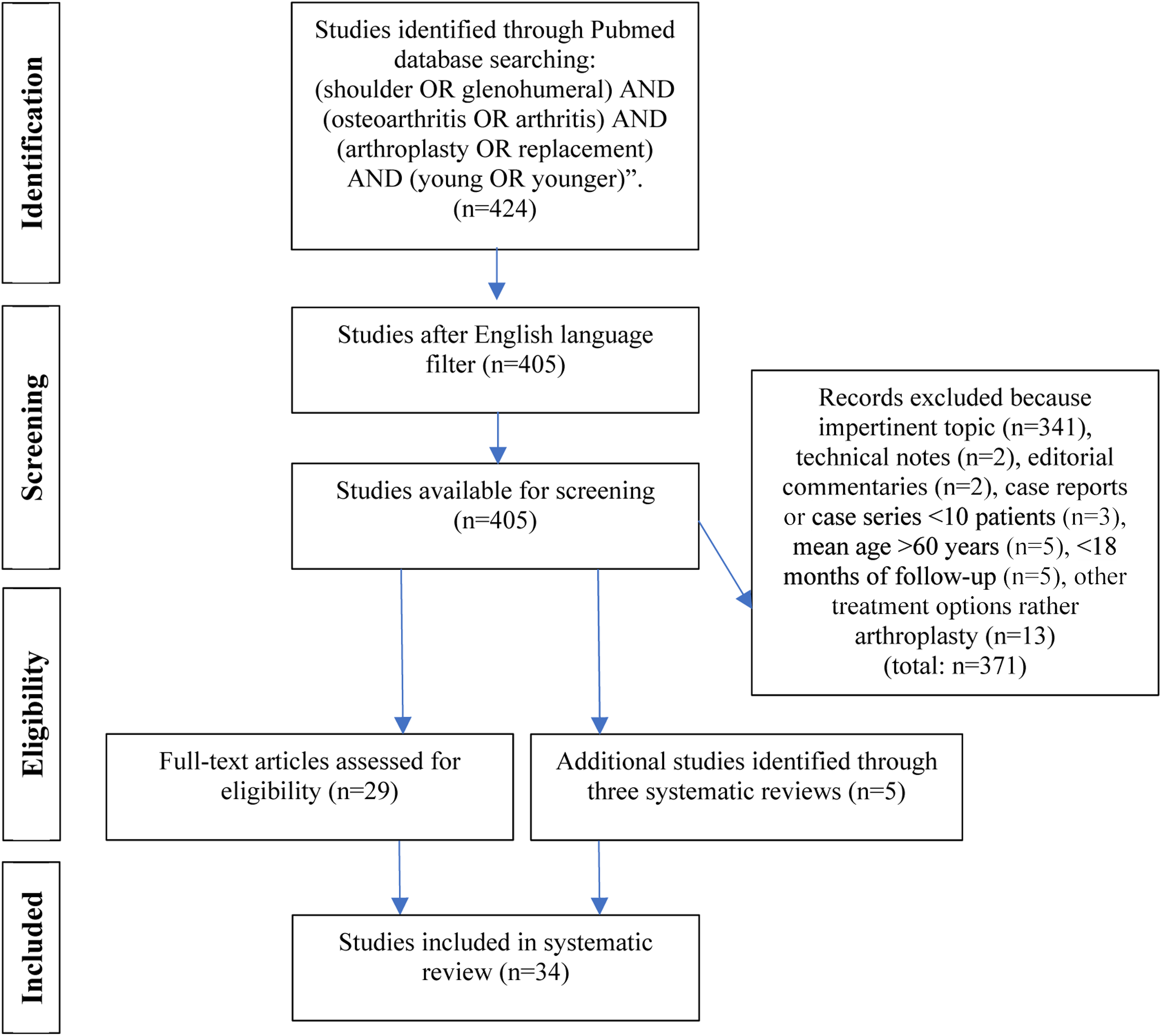
Preferred Reporting Items for Systematic Reviews (PRISMA) flow diagram.
Data Abstraction and Synthesis
Data were extracted to include study and patient characteristics, surgical technique, range of motion, pain relief, outcome scores, complications, need for and time to revision. Patients were stratified into the following treatment groups: HA, HABR, TSA and RSA. After collecting all available data about arthroplasty options for this population, and reviewing descriptively, statistical comparisons between these groups were performed. Continuous data were analyzed through computation of the mean and standard deviation, which were frequency weighted for the sample size. All statistical analyses were performed with SPSS® (v.26, IBM®) and statistical significance was defined by p < 0.05.
Aggregated Demographic and Outcome Statistics.

After removing outliers (TSA with metal-backed glenoid component – MB-TSA); there is only one paper with this type of implant (Gauci et al) reporting complication rates (n = 7; 91.3%).
There are two papers reporting the need for revision that include MB-TSA (Gauci et al; Sperling et al), however the latter didn't stratify the results; for that reason, the outliers were not eliminated for this item.
Results
Literature Search
The search of Pubmed identified 424 studies. After the application of the English language filter, 405 titles and abstracts were assessed. After the application of eligibility criteria, 29 studies published from 2002 to 2019 were included in the systematic review. Five additional references from three systematic reviews were added, encompassing 34 eligible studies [HA (n = 4), HABR (n = 13), TSA (n = 5), RSA (n = 6), more than one type of prosthesis (n = 6)], a total of 1535 shoulders (Figure 1).
Patient Characteristics
Twenty studies presented a mean age under 50 years old, eight from 51 to 55, and six from 56 to 60. The total mean age was 47.61 years, ranging from 44.3 (HABR) to 56.6 years (RSA). The male sex varied from 40.8% (RSA) to 77.5% (HABR). Staging of arthritis was infrequently reported and was conducted using multiple heterogeneous staging systems. There were some differences in patient demographics and preoperative clinical characteristics across treatment groups, including the follow-up interval, age, and sex distribution (Tables 1 and 2).
Included Studies and Characteristics by Year.
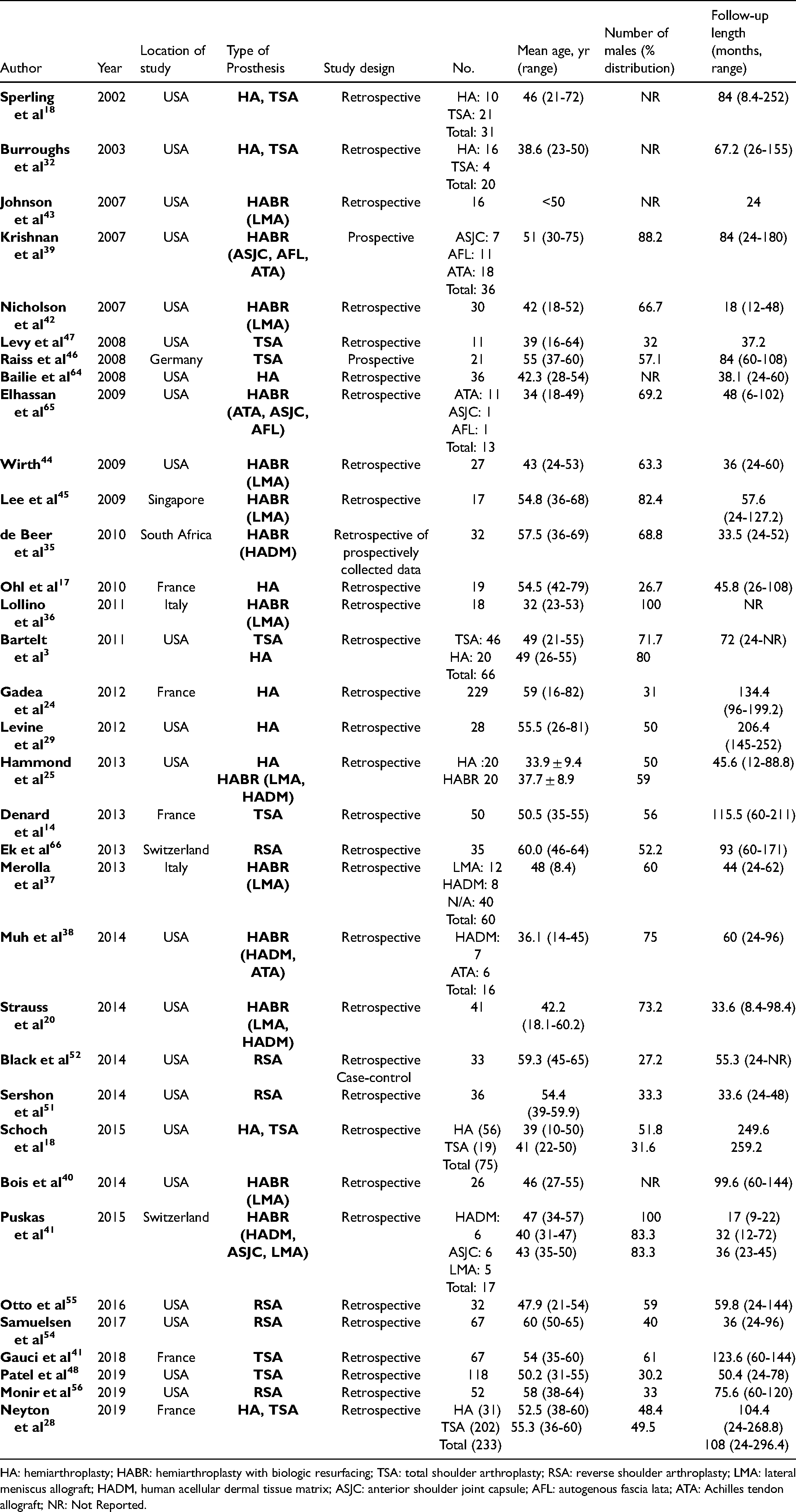
HA: hemiarthroplasty; HABR: hemiarthroplasty with biologic resurfacing; TSA: total shoulder arthroplasty; RSA: reverse shoulder arthroplasty; LMA: lateral meniscus allograft; HADM, human acellular dermal tissue matrix; ASJC: anterior shoulder joint capsule; AFL: autogenous fascia lata; ATA: Achilles tendon allograft; NR: Not Reported.
Surgical Technique
Patients were treated with HA (n = 341), HABR (n = 371), TSA (n = 561) or RSA (n = 262). Concomitant procedures, e.g. biceps tenotomy or tenodesis, repair of rotator cuff tendons, among others, were performed in 16 studies. Studies variably used cemented or uncemented components, stemmed or resurfacing humeral components (HA and TSA), metal-backed versus polyethylene (TSA), standard or lateralized RSA (Tables 2a-d).
Hemiarthroplasty Outcomes.

HA: Hemiarthroplasty; FF: Forward Flexion; Abd: Abduction; ER: External Rotation; IR: Internal Rotation; NR: Not Reported.
Hemiarthroplasty with Biological Resurfacing of the Glenoid Outcomes.

HABR: hemiarthroplasty with biologic resurfacing; LMA: lateral meniscus allograft; HADM, human acellular dermal tissue matrix; ASJC: anterior shoulder joint capsule; AFL: autogenous fascia lata; ATA: Achilles’ tendon allograft ; . FF: Forward Flexion; Abd: Abduction; ER: External Rotation; IR: Internal Rotation; NR: Not Reported.
Total Shoulder Arthroplasty Outcomes.

TSA: Total Shoulder Arthroplasty; PE: polyethylene; FF: Forward Flexion; Abd: Abduction; ER: External Rotation; IR: Internal Rotation; NR: Not Reported.
Reverse Total Shoulder Arthroplasty Outcomes.

RSA: Reverse Shoulder Arthroplasty; FF: Forward Flexion; Abd: Abduction; ER: External Rotation; IR: Internal Rotation; NR: Not Reported.
Range of Motion (ROM)
Shoulder arthroplasty provided improvements in terms of ROM on the 3 plains - forward flexion (FF), abduction (Abd) and external rotation (ER), in different proportions for each type of prosthesis.
Twenty-two studies reported pre and postoperative FF angle values3,14,17,20,31,34–37,42,44,45,50–53,55,56,64 (Tables 2a-d). Patients submitted to RSA had lower preoperative FF (71.2°, p = 0.011). There were no postoperative variances or improvement (Δ) differences between groups (Table 4). Fifteen studies reported pre and postoperative range of active Abd18,26,29,36,42,48,50,51,53–56 and twenty of ER3,14,17,18,20,26,29,31,34–37,39,42,44,45,48,50–52,54–56,64,65 (Tables 2a-d). RSA group had the highest Δ in terms of Abd (51°) but the worst in terms of ER (11.4°), particularly when compared to TSA (29.8°, p = 0.05). When we compare exclusively postoperative ER values, there is a difference between HA and RSA groups (46.3° vs. 31.7°, p = 0.049). Moreover, there were no other statistically differences at the end of the follow-up (Table 4). Internal rotation (IR) values were listed whenever present, but a comparison was not made due to the high heterogeneity on the way they were presented in each study (Tables 2a-d).
Outcome Scores
Improvement in pain status, using an aggregate of standardized pain scores, was reported in twenty-two studies,,3,18,20,28,30,31,34–36,41,42,44,45,50,52,56,64,66 Constant Score in twelve,14,29,30,36,41,42,44,48,50,51,56,64 Subjective Shoulder Value (SSV) in three,36,41,51 American Shoulder and Elbow Surgeons Shoulder Score (ASES) in twelve,20,28,31,34,35,37,45,50,52,53,55,56 and Simples Shoulder Test (SST) in eleven studies.20,31,35,37,44,50,52–56 (Tables 2a-d)
Pain scores improved in all groups but no difference between them (p = 0.642). TSA and RSA groups had the best CS improvement (33.8 and 34.6) and the HABR group had the poorer, with a statistical difference between them all (p = 0.012).
Patients with HA and TSA had the highest postoperative SSV values (73.2 and 72.3) and the HABR the lowest (41; p = 0.003), however, the Δ was not possible to quantify due to the lack of preoperative data. There were no differences regarding other functional outcomes (ASES and SST). (Table 4)
Complications
Twenty-three studies reported the complication rates associated to the surgical treatment3,14,17,27,29,30,32–35,37,38,41,45,48,50–52,54,55,64–66 (Tables 2a-d).
HA group had a complication rate of 21.7%, HABR of 19.7% and RSA of 19.4%. After eliminating the outliers of TSA metal-back glenoid prosthesis (complication rate of 91% in the only study reporting this outcome) the complication rate was 19.4%, the same as the RSA group. There was a statistical difference in the multivariable analysis between HA and RSA groups (p = 0.031) (Tables 3 and 4).
Complications.
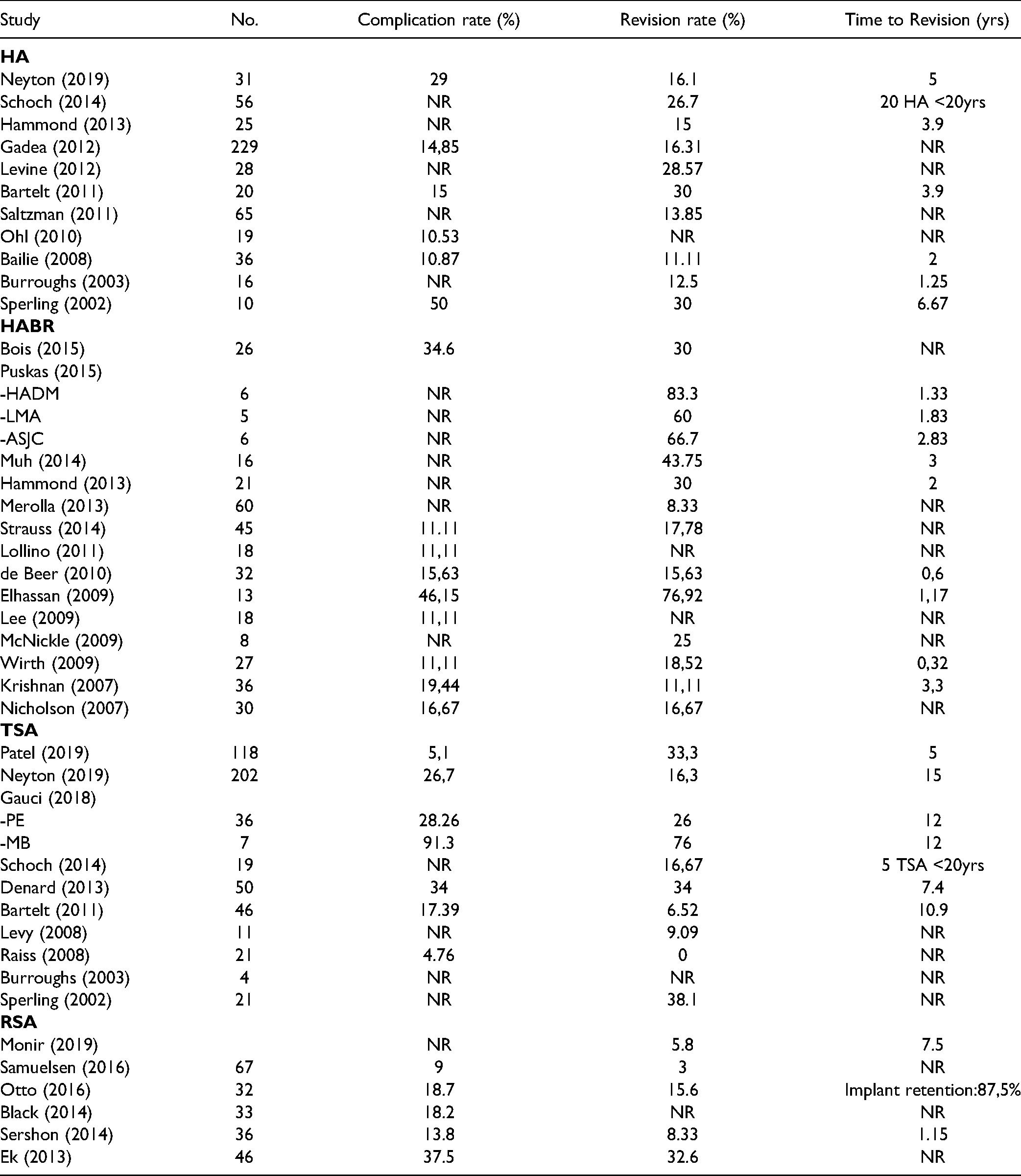
Thirty-one studies reported Revision rates.3,14,17,18,20,26–28,30–36,38,39,42–45,48,50,51,53–56,64,66 HABR group had the highest with 34.5%, HA 25.7%, TSA 25.6%, and RSA 13,1% (Table 3). Two papers reported the need for revision that includes MB-TSA (Gauci et al; Sperling et al), however, the latter didn't stratify the results for each implant. For that reason, the outliers were not eliminated in this section. Despite the range of values between groups (34.5-13.1%, p = 0.170), the modest number or contributing papers didn't allow a reliable statistical analysis (Table 4).
Discussion
The correct management of young and active patients with glenohumeral arthritis continues to be debated in the literature. Although TSA is more common and has been reported as a reliable treatment for pain secondary to glenohumeral degenerative disease, historically the results in younger patients have not been as favorable as in older patients and concerns remain regarding the early failure of the glenoid component.20,26 HA avoids complications related to prosthetic loosening of the TSA glenoid component,67,68 thus the optimal candidate would be the young patient with unipolar involvement of the humeral head and a relatively preserved glenoid articular surface.2,68,69 HA alone has been reported to provide short-term pain relief and improved function, but studies with longer follow-up have demonstrated progressive joint space narrowing, glenoid erosion, and diminishing outcomes.32,69–71 Levine et al 17 reported that 74% of shoulders achieved satisfactory results, with outcomes correlated most significantly with the status of posterior glenoid wear, thus suggesting that HA be reserved for patients with a concentric glenoid. These patients were reevaluated at an average follow-up period of 17.2 years and 25% were satisfied. In a review of 78 hemiarthroplasties, Sperling et al 19 reported that at 15 years of follow-up, unsatisfactory results in 45% of their patients. According to the literature, this option is not recommended due to early failure rate, poor pain and functional outcomes. In our analysis, HA had the highest rate of complications (21.7%), which is statistically different from RSA (19.4%, p = 0.031). The same lower rate was reported in the TSA group when we don't consider patients with the metal-backed glenoid components.
To improve the results seen after HA and to avoid the complications associated with the glenoid component of TSAs, biological resurfacing of the glenoid was reassessed in 1988 by Burkhead and Hutton., 34 and since then variable results have been seen. Options include Achilles’ tendon, lateral meniscus, and fascia lata autografts, and acellular dermal matrix-based scaffold grafts. Besides, conflicting reports exist in the literature. 2 Encouraging results were reported by Krishnan et al 34 in their 2 to 15-year follow-up of 36 patients. Other studies, however, have reported contrasting outcomes, with rapid deterioration, return of pain, and a high rate of conversion to TSA.20,41 Significantly worse outcomes were reported by Elhassan et al 41 in their retrospective review of 13 patients aged younger than 50 years treated with HABR with a 92.3% failure rate. The authors concluded that this treatment is unreliable. More recently, Lee et al 65 reported their experience and among their 19 treated patients monitored for a mean of 4.25 years, poor clinical outcomes, and a complication rate of 32%, all requiring revision surgery, led the authors to conclude that glenoid resurfacing produced inconsistent results with a high incidence of complications. Strauss et al 20 reported an unacceptable failure rate of 51.2%, alongside persistent pain, poor function, and poor outcome scores postoperatively, leading to a conversion to a TSA or RSA. In our review, patients that underwent HABR experienced less improvement in terms of outcome scores as CS and SSV, with a high rate revisions (34%).
Although originally thought to be more suited for lower-demand patients, TSA outcomes have been improved through time, and there is increasing evidence supporting this option for the treatment of this population. The American Academy of Orthopaedic Surgeons (AAOS) clinical practice guidelines support its use. 72 In comparison with HA, TSA leads to a significantly better pain score and range of motion improvements but with similar satisfaction and revision needs. 2 Some authors have noted unsatisfactory results despite improvements in pain and motion for this patient demographic, others have considered it a viable treatment option with low complication rates and excellent intermediate to long term results. Bartelt et al 3 reported an implant survival rate of 92% at 10 years for TSA and significantly less pain, greater active FF, and higher satisfaction than their counterparts who underwent HA. Raiss et al 48 prospectively evaluated 21 patients with a mean age of 55 years, and at a mean follow-up of 7 years, there were no revision requirements, and 95% of patients were either “very satisfied” or “satisfied”. The subjective outcome scores increased significantly and no clinical or radiologic signs of periprosthetic loosening were reported. Denard et al 14 examined 52 TSAs, and the survivorship of the glenoid component was 98% at 5-year follow-up and 62.5% at 10-year follow-up. On the largest meta-analysis to date, Radnay et al 12 reported that TSA resulted in significantly better pain relief, postoperative range of motion, and patient satisfaction, with a lower revision rate. In our review TSA group had the highest ER improvement, with the lowest improvement in terms of Abd; better CS and SSV when compared to HABR. After eliminating the TSA metal-back glenoid prosthesis outliers with a complication rate of 91.3%, 64 the rate was 19.4%. This is the lowest value along with the RSA group as stated before. Although the worries over glenoid component loosening with TSA over time have been legitimized in some recent follow-up studies, some authors refer that these potential complications seem to occur over the long-term, providing the patient with years of symptom-free improved function. For appropriately selected patients, TSA decreases pain and improves shoulder function.11,73
In patients mainly with secondary and complex forms of osteoarthritis, as severe rotator cuff deficiency, which is uncommon in young patients (<60 years old), TSA may not be a viable treatment option. These patients represent a rare and special population that needs to be prudently addressed. Reports of primary repair of the rotator cuff at the time of arthroplasty have had good results with function and pain, though these patients must be carefully selected. RSA has been used in elderly patients with biconcave or severe glenoid bone loss. 74 In 2013, several authors started to report their results of RSA in younger and active patients with more severe forms of arthritis, with a growing body of literature since 2017. In a recent systematic review, Chelli et al 75 reported a rate of 17% of postoperative complications, leading to a new surgical procedure in 10% of cases at a mean follow-up of 4.2 years. FF and ER were restored in most patients, although the functional results were modest. The results of these authors tend to support the idea that younger patients expect higher functional levels and experience less satisfaction. The range of motion obtained with RSA seems lower than what is reported with anatomic TSA. Thus, RSA can be a potential option in young patients with a critical cuff-deficient shoulder, with a glenoid with severe bone erosion, or a failed previous arthroplasty, when nonoperative treatment has failed, with reliable clinical improvements and midterm complication rates comparable to those of older patients. In our study, we found that this group of patients experienced the highest improvement in terms of Abd but the worst in terms of ER, particularly when compared to TSA (p = 0.05). When we compare exclusively postoperative ER, the RSA group experienced lower improvements, especially when we compared with HA (p = 0.049). We need to keep in mind that patients receiving RSA probably present distinctive and more severe forms of the disease. Moreover, this solution interestingly presented a low associated rate of complications (19,4%) and a low rate of revisions (13%), which might be considered a promising solution for selected patients. However, studies with longer follow-ups are needed for reliable conclusions.
This review has several limitations. The data were obtained from non-randomized trials, but to date, no high level prospective randomized trials have been published. Twelve papers included some patients who were older than 60 years. As the data were not stratified for each patient in each study, a subgroup analysis of those exclusively under this age wasn't possible. With 82.4% of the studies presenting a mean age lower than 55, we consider that this aspect hasn't a major impact on this review. Underlying diagnoses in cases of secondary osteoarthritis aren't presented consistently, and not including them weakens further comparisons. Regarding the concomitant procedures and implant variability, the goal is to be mainly descriptive, and all this data is presented in the tables. A variety of techniques and graft choices in the HABR group were also used. Although a difference in the type of soft tissue covering is a confounding variable, we believe including a comparison of all is needed as one is not definitively clinically superior. There isn't enough data available regarding the wear pattern or the Walch classification of glenoid morphologies, and the eventual influence of this feature on the treatment modality choice and respective outcomes. Despite the descriptive nature of this review, heterogeneous results were reported in each paper, which limited a comprehensive statistical comparison between groups. There is a relevant variance in the clinical and outcomes scores chosen through the different studies, and we opted to use those that were more reliable. Though, we believe relevant conclusions can still be drawn from the comparisons.
Conclusion
The management of young, active patients with symptomatic glenohumeral arthritis continues to be debated in the orthopaedic surgery literature. Alternative treatments to total shoulder arthroplasty have been investigated in this patient population to improve postoperative outcomes and avoid the likely need for revision surgery secondary to failure of the glenoid component over time. Hemiarthroplasty has the highest rate of complications in this population and hemiarthroplasty with glenoid resurfacing has been abandoned gradually due to the unacceptable rate of revisions. These implants have largely been replaced by modern TSAs with cemented polyethylene glenoid components, with reverse shoulder arthroplasty as an increasingly utilized treatment of severe cases. Optimal management of young patients with end-stage disease remains an important topic of investigation. Surgeons should be aware of the common complications and pitfalls of each option.
Footnotes
Author Contributions
The authors declare the contribution of Carolina Lemos PhD, from the School of Medicine and Biomedical Sciences, University of Porto, Portugal, in the preparation of the statistical analysis of the results of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
