Abstract
Introduction
The presence of retained intramedullary fibular allograft presents many challenges during reverse shoulder arthroplasty (RSA), which have been discussed in limited fashion. This case series presents a single-surgeon experience with 6 patients treated with RSA following failed osteosynthesis of proximal humerus fractures using intramedullary allografts.
Methods
A retrospective review was conducted of RSA patients with a minimum of 2-year follow-up (exception of 1 deceased patient) for failed plate fixation of proximal humerus fractures. Two cohorts were created based on the presence of an intramedullary allograft during the procedure. Patient-reported outcome measures, active range of motion, time to revision, surgical time, complications, and case descriptions were obtained from chart review and radiographic analysis.
Results
When treating patients with a retained allograft, the surgeon used a guide pin from a cannulated screw set to advance through the graft, and a cannulated drill was used to ream and bypass the allograft, contributing to a 22% increase in surgical time (151 vs 124 min). Intraoperative complications were observed only in the allograft cohort (50%; 3 of 6), including cortical perforation with cement extrusion, humeral loosening, and proximal humerus bone loss. Patients treated with a retained allograft experienced pain relief (P = .001) but did not gain significant functional improvements.
Conclusion
RSA for failed proximal humerus fixation with healed intramedullary allograft is associated with increased operative time and intraoperative complications, and patients can mostly expect pain relief. Techniques for humeral canal preparation and stem placement to bypass the allograft are helpful in managing these revisions.
Keywords
Introduction
The operative treatment of displaced proximal humerus fractures has evolved over time. Although early surgical techniques were associated with high complication rates,1–5 the introduction of modern locked plating 6 , 7 and an emphasis on restoring the medial calcar 8 , 9 has helped to minimize postoperative complications. More recently, the use of intramedullary allograft has been advocated as a method to augment support of the medial calcar and facilitate fracture reduction and stabilization.10–14 This technique has been demonstrated to enhance plate osteosynthesis and fracture site stability, with union rates reported as high as 96%. 15
However, revision surgery following index fixation with intramedullary allograft augmentation may be required in the setting of avascular necrosis of the humeral head, progressive arthritis of the glenohumeral joint, or postoperative infection. Revision surgery can be extremely challenging as the normal intramedullary anatomy is altered by allograft incorporation. Allograft ingrowth, which occurs as early as 3 to 4 months postoperatively, 8 , 9 creates an intramedullary canal that resists reaming and prosthesis placement. The challenges specific to revision reverse shoulder arthroplasty (rRSA) in the presence of retained intramedullary allograft have only been discussed in limited fashion,16–18 with no study describing the complexity of humeral canal preparation and surgical techniques to effectively address the pathology encountered.
The purpose of this case series is to present a single surgeon experience with 6 patients treated with rRSA following failed osteosynthesis of proximal humerus fractures using intramedullary allografts, with a specific focus on the author’s preferred surgical technique used for humeral canal preparation and stem placement. To provide further contextual differences, this group of patients is compared to 8 patients treated with rRSA for failed osteosynthesis of proximal humerus fractures in the absence of intramedullary allografts.
Methods
A retrospective review was performed of our Institutional Shoulder and Elbow Repository to identify all patients treated with rRSA between December 2008 and November 2017 for a failed plate fixation of proximal humerus fractures by a single high-volume fellowship-trained shoulder surgeon. Two cohorts were created based on the presence of an intramedullary allograft used during the surgical fixation of the proximal humerus fracture.
Case descriptions of the patients previously treated with an intramedullary allograft were created from chart review and radiographic analysis. Medical records were studied to identify the time from the index procedure to rRSA and surgical time required for rRSA as well as demographic information. Preoperative and postoperative patient reported outcome measures (PROMs) including Simple Shoulder Test (SST), American Shoulder and Elbow Surgeons total score (ASES), Short Form (SF) 12, and Visual Analog Score (VAS) for pain and function. Active range of motion was also reported through goniometer-based measurements of external rotation and forward elevation. Internal rotation was measured based upon the highest vertebral level reached behind the back. Complications and additional revision procedures were also noted.
Given the small sample size of the cohorts, significant test comparisons were not performed; however, descriptive comparisons, including means and standard deviations, were calculated. Efficiency of treatment was analyzed with nonparametric Wilcoxon signed rank tests to compare preoperative to postoperative changes in variables within the cohorts. These tests were 2-tailed, and P < .05 was used to determine if there was a significance.
Surgical Technique
All operations were performed by the senior author (JCL) using a deltopectoral approach in the beach chair position. Only the proximal locked screws were removed from the humeral head. The remaining distal screws and lateral plate were left in place during glenoid exposure in order to minimize the risk for iatrogenic humeral shaft fracture. The subscapularis was then released off the lesser tuberosity using a subscapularis peel. The glenoid was exposed using extensive pericapsular releases, and the glenoid baseplate and glenosphere (DJO, Austin, Texas) were impacted using standard technique.
Attention was then directed to the proximal humerus (Figure 1(A)). The remaining screws and plate were removed. The proximal portion of the intramedullary allograft was grasped in an attempt to remove the graft. Unfortunately, in each case, the graft was incorporated and healed within the humeral canal. In an effort to bypass the intramedullary graft and prepare the humeral canal for the humeral component, a guide wire from a 6.5 mm cannulated screw set was advanced under fluoroscopic guidance through and past the healed intramedullary graft (Figures 1(B) and 2). A 6.0 mm cannulated drill was then passed over this guidewire, reaming the remaining allograft material (Figure 1(C) and (D)). Sequential humeral reamers from the RSA set were then placed into the canal under fluoroscopy to prepare the humerus (Figure 1(E)). Subsequently, humeral broaches and metaphyseal reamers were then used to prepare the humerus. The humeral stem implant is impacted in 30° of retroversion, obtaining an excellent press-fit proximally. If press-fit fixation was not sufficient, the humeral stem was cemented using a previously described technique of distal cementation. 19 Following placement of the final components, the subscapularis was repaired. Patients were placed in a shoulder immobilizer for the first 6 weeks and performed patient-directed pendulum exercises, followed by 6 weeks of active stretching and delayed strengthening for 3 months.

Intraoperative fluoroscopy images of surgical technique used to bypass intramedullary graft and prepare humeral canal for component. A, Anteroposterior fluoroscopy image of patient with humeral head collapse and retained intramedullary allograft. B, Guide pin from a cannulated screw advanced through graft. B and C, 6.0 mm cannulated drill passed over guidewire, reaming the fibula. D, Sequential humeral reamers placed into humeral canal to prepare for component.
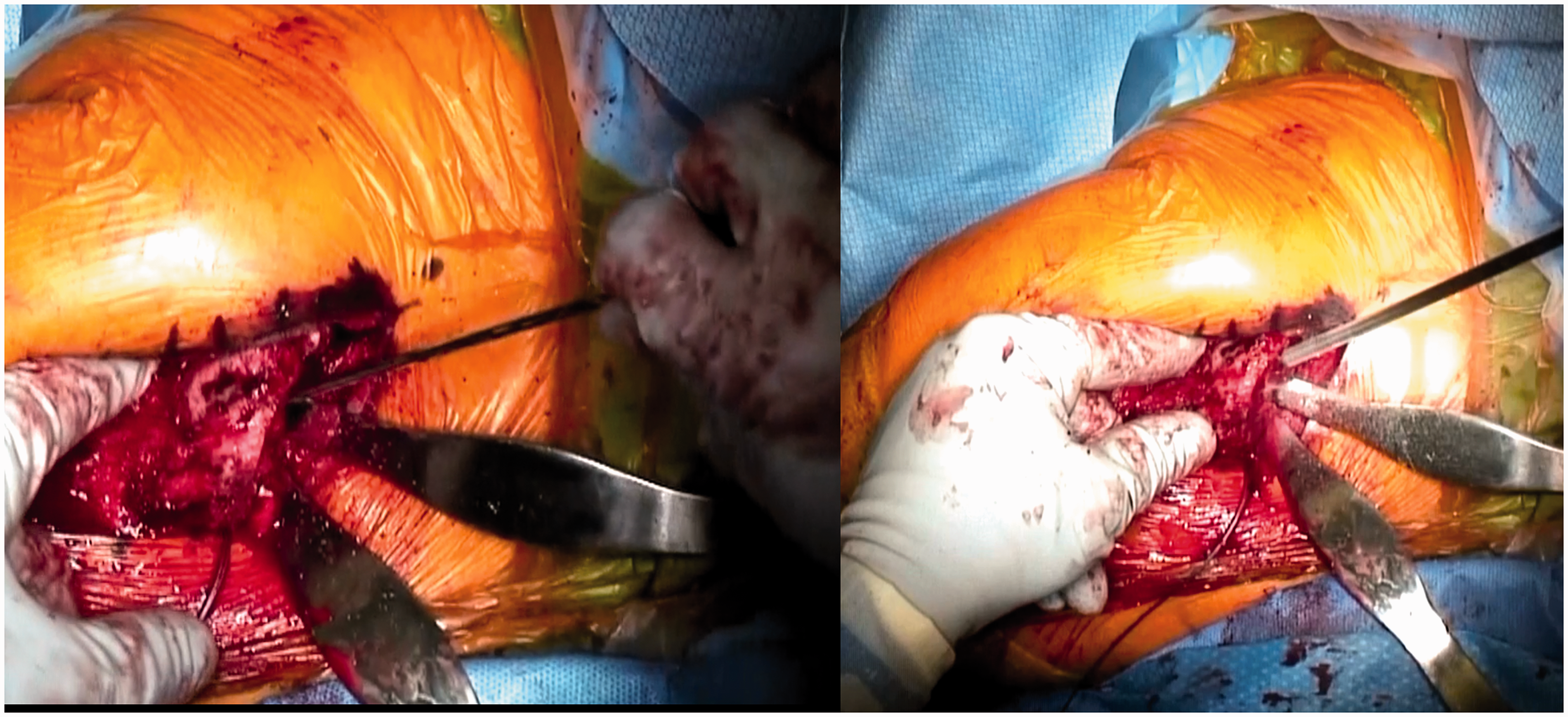
Intraoperative photographs illustrating placement of cannulated guide wire (A) and subsequent intramedullary reamer (B).
Results
Presence of Intramedullary Allograft
Six patients were revised to an rRSA who had intramedullary allografts. All patients had a minimum of 2-year follow-up with the exception of patient 1, who deceased following a 5-month follow-up. The average age was 66 years (range: 43–89) and 4 were female. The average time to revision was 23 months (range: 6–43 months). Operative time required for revision to RSA was 151 minutes (range: 125–180). Significant improvements were observed in VAS pain and active external rotation (Table 1). However, patients showed no significant improvement in all other PROMs and range of motions, including SST, physical component summary (PCS), mental component summary (MCS), ASES, VAS function, active elevation, and active internal rotation.
Preoperative to Postoperative Improvement in Patient-Reported Outcomes Measurements and Measured Motion of Patients With Fibular Allografts.

Abbreviations: ASES, American Shoulder and Elbow Surgeons; MCS, 12-item Short-Form Health Survey Mental Component Score; PCS, 12-item Short-Form Health Survey Physical Component Score; SST, Simple Shoulder Test; VAS, visual analog scale.
aPatient deceased after 5-month follow-up.
bInternal rotation scale: buttock to greater trochanter (2 points); sacrum to L4 (4 points); L3–L1 (6 points); T12–T8 (8 points); T7–T1 (10 points).
There were 2 intraoperative complications related to bypassing the allograft with subsequent cement extravasation out of the humeral canal. No nerve-related injuries were noted, but there were 2 cases of proximal humeral bone loss of the greater tuberosity after fibula removal. One was managed with a long stem uncemented humeral component, and the other necessitated a larger glenosphere to facilitate soft tissue balancing. In addition, 1 patient developed a postoperative acute infection which was managed with a series of surgical debridements. This patient later developed humeral loosening and is anticipating another revision.
Case 1
Patient 1 was an 89-year-old woman who sustained a left 100% displaced varus-impacted short oblique 2-part proximal humerus fracture after a slip and fall (Figure 3(A)). She was treated using locked plate fixation with an intramedullary fibula allograft using a 3-hole Stryker® (Mahwah, New Jersey) Locked Proximal Humerus plate (Figure 3(B)). Within 1 year, avascular necrosis and collapse of her humeral head was identified with intra-articular penetration of the locked screws as she was experiencing disabling shoulder pain and loss of function (Figure 3(C)).
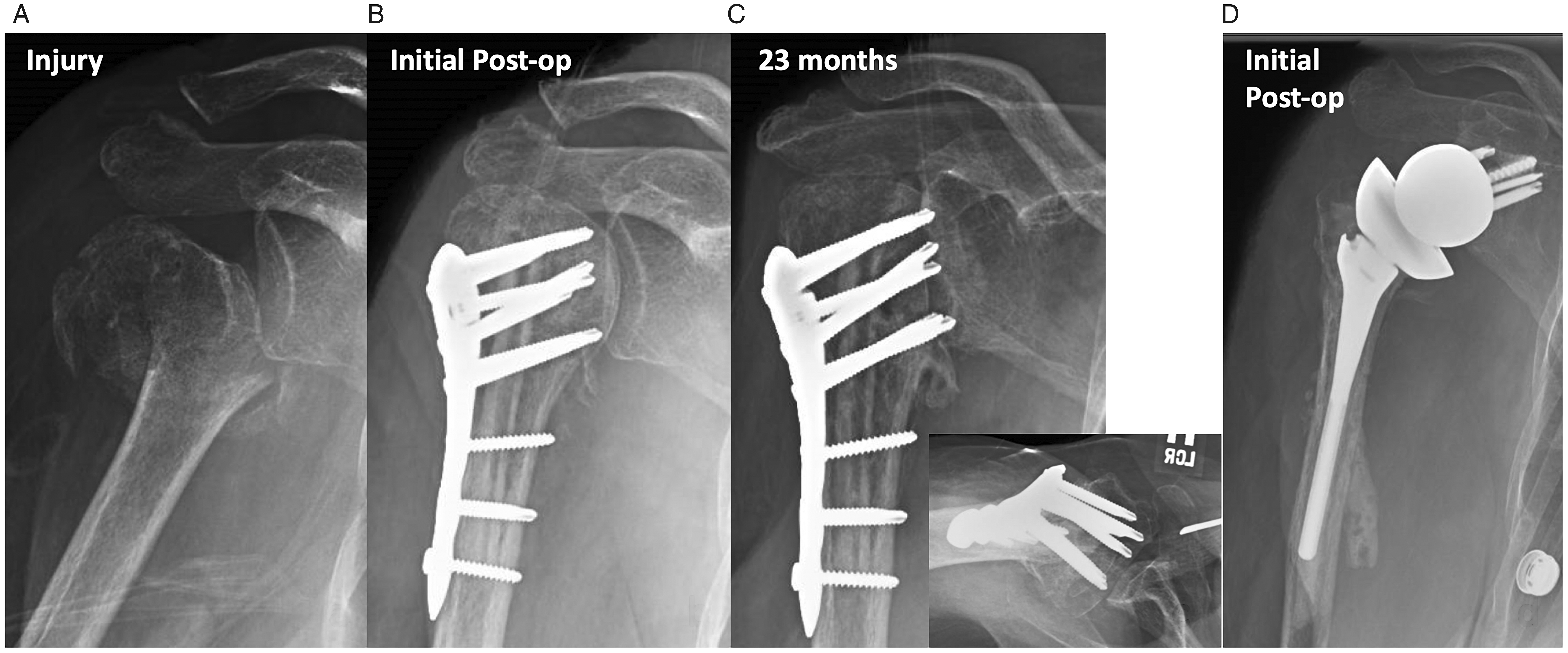
Radiographs of patient 1. A, Injury shows a 100% displaced 2-part proximal humerus fracture, which was (B) treated with an open reduction internal fixation with a locked plate and fibular allograft. C, 23-month postoperative images reveal avascular necrosis, collapse of humeral head, and intra-articular penetration of the locked screws. D, Revision RSA was complicated by cortical perforation of the humerus, and postoperative images show posterior-medial cement extrusion into the triceps.
rRSA (DJO RSP, Austin, Texas) with a cemented humeral component was performed 23 months following the initial surgery (Figure 3(D)). Surgery was complicated by a cortical perforation of the humerus which occurred during humeral canal preparation while attempting to bypass the fibular allograft. Operative time was 172 minutes. Immediate postoperative radiographs revealed posterior-medial cement extrusion into the triceps. However, the patient reported no signs of a radial nerve paresthesia or palsy. Unfortunately, the patient deceased from natural causes after 5-month follow-up. At that early mark, the patient saw mild gains in functional improvement and pain relief, including a decrease in VAS pain from 7 to 5, increase in ASES score from 23.3 to 43.3, and increase in active external rotation from 0° to 45°.
Case 2
Patient 2 was a 61-year-old woman who sustained a left displaced proximal humerus fracture after a slip and fall. She was subsequently treated using locked plate fixation with an allograft strut (Figure 4(A)). She presented 17 months after surgery complaining of constant posterolateral shoulder pain, limited range of motion, and severe pain. Computed tomography (CT) and radiographs revealed avascular necrosis with humeral head collapse and an integrated intramedullary fibular allograft (Figure 4(B)). Prominent screws were noted in the joint, which facilitated the development of severe arthritis and erosive changes of the glenoid. She underwent an rRSA (DJO RSP) with a cemented humeral component 21 months following her initial procedure, with an operative time of 123 minutes. There were no intraoperative complications, and the patient experienced considerable improvements in both function and pain after 2 years, as her VAS pain dropped from 8 to 1 and SST increased from 6 to 12.
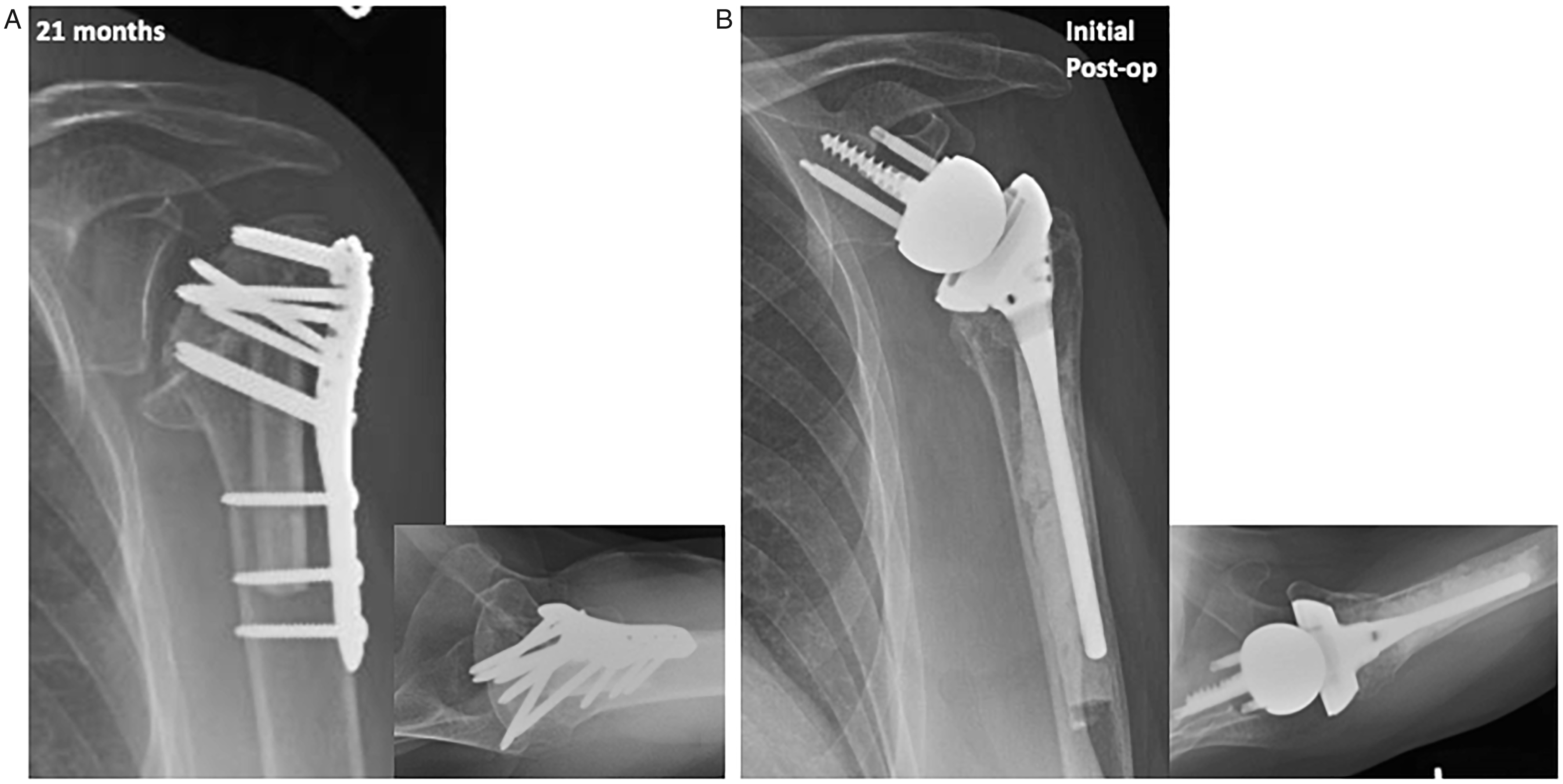
Radiographs of patient 2. Displaced proximal humerus fracture treated with an open reduction internal fixation with a locked plate and fibular allograft. A, 21-month postoperative images reveal avascular necrosis with collapse of humeral head, locked screw penetration into the joint, causing glenoid erosion and arthritic changes. B, Revision RSA performed with cemented humeral component without complications.
Case 3
Patient 3 was a 67-year-old woman who sustained a displaced 4-part proximal humerus fracture following a slip and fall. She was treated with locked plate fixation using an intramedullary fibular allograft strut and a Zimmer® (Warsaw, Indiana) 10-hole Proximal Humerus Locking Plate (Figure 5(A)). She presented 3 years later with severe glenohumeral arthritis with humeral head collapse and functionally disabling shoulder. Radiographic analysis demonstrated penetration of locked screws which were eroding into the glenoid (Figure 5(B)). She underwent an rRSA (DJO Altivate Reverse, Austin, Texas) 34 months following initial surgery, and an uncemented long stem humeral component was used due to loss of proximal humeral bone support (Figure 5(C)). Total surgical time was 125 minutes. There were no complications. At follow-up 25 months after surgery, the patient experienced restoration in functional mobility with an SST increase from 2 to 7 and no pain with a VAS pain score of 0.

Radiographs of patient 3. A, Proximal humeral fracture treated was an open reduction internal fixation with a locked plate and fibular allograft. B, 34-month postoperative images reveal collapse of humeral head and locked screw penetration into the joint. C, Revision RSA performed with uncemented long stem due to loss of proximal humeral bone support.
Case 4
Patient 4 was a 43-year-old man who sustained a right comminuted proximal humeral fracture secondary to a fall. He was treated with a locked plate fixation augmented with an intramedullary fibular allograft strut. One year following surgery, he developed avascular necrosis with humeral head collapse with associated severe pain and loss of function (Figure 6(A)). A CT scan revealed prominent screws that protruded into the joint causing erosion of the glenoid. In addition, 11 months following the initial surgery, he underwent an rRSA (DJO Monoblock, Austin, Texas), which amounted to 148 minutes of operative time, using an uncemented humeral component (Figure 6(B)). No complications were noted, and she achieved significant improvements in all PROMs and active range of motions.
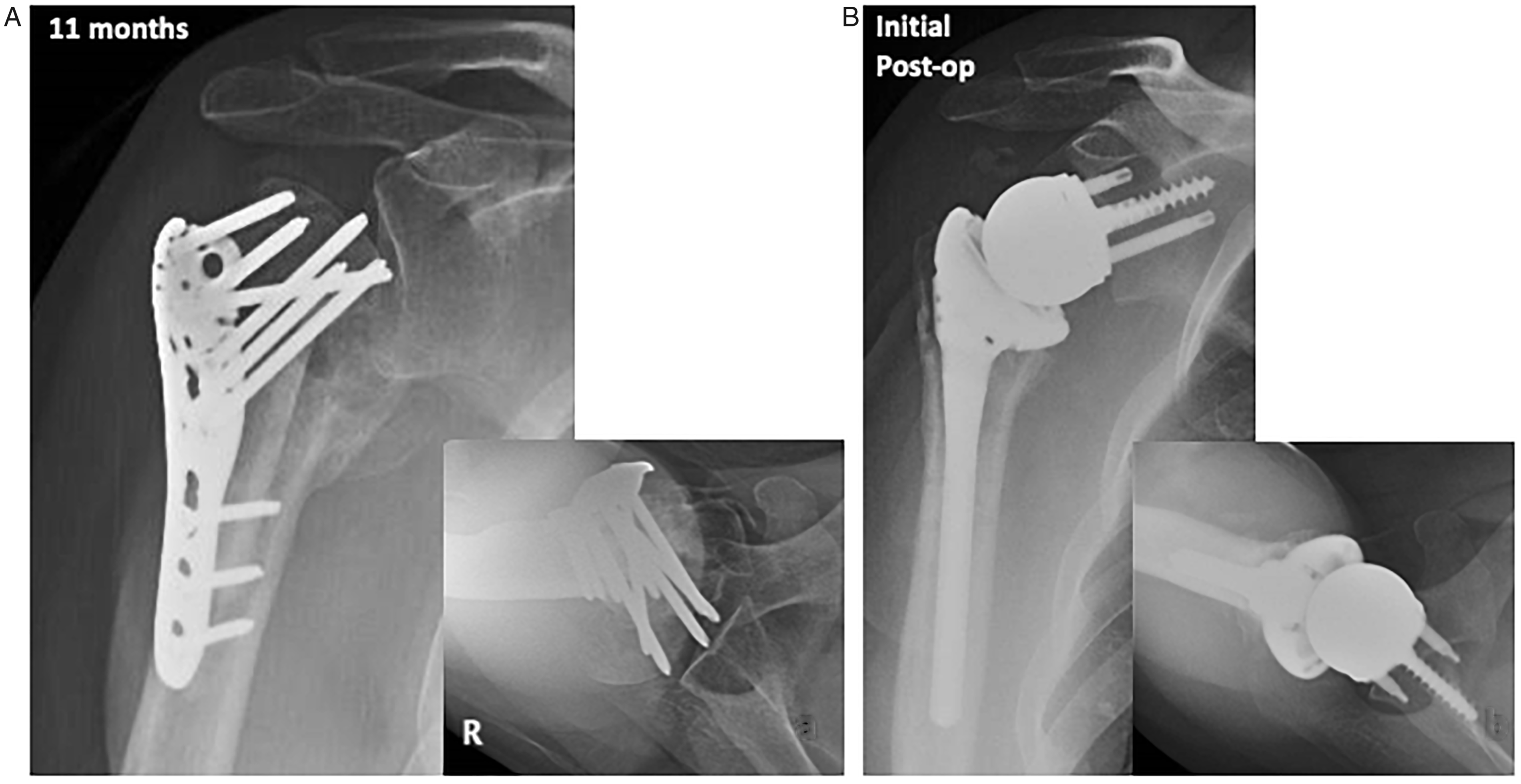
Radiographs of patient 4. Right proximal humerus fracture treated with an open reduction internal fixation with a locked plate and fibular allograft. A, 11-month postoperative images reveal avascular necrosis, collapse of humeral head, and locked screws penetration into the joint, causing glenoid erosion. B, Revision RSA performed with uncemented humeral component.
Case 5
Patient 5 was a 71-year-old woman who underwent locked plate fixation of a displaced proximal humerus fracture augmented with an intramedullary allograft. One year later, she underwent an arthroscopic capsular release with little improvement in her pain and shoulder function. Two years following, she presented complaining of pain at rest and associated limitation in shoulder function. Radiographs demonstrated avascular necrosis with collapse of the humeral head with a lesser tuberosity malunion (Figure 7(A)). She underwent an rRSA (DJO Monoblock, Austin, Texas) using a cemented humeral component, which took 147 minutes. An unrecognized intraoperative penetration of the humeral cortex occurred during humeral canal preparation while attempting to bypass the fibular allograft. Cement was extruded through this cortical breach as noted on the postoperative radiographs (Figure 7(B)). Fortunately, there were no signs of postoperative nerve deficits, but the patient did develop significant proximal humeral bone loss over time. At the 28-month follow-up, there were no changes in function or pain when compared to baseline measures other than a reduction in active motion for elevation.

Radiographs of patient 5. Proximal humerus fracture was treated with an open reduction internal fixation with a locked plate and fibular allograft. A, 43-month postoperative images reveal avascular necrosis and collapse of humeral head. B, Revision RSA performed with unrecognized intra-operative penetration of humeral cortex while attempting to bypass allograft and cement extrusion occurred through breech.
Case 6
Patient 6 was a 67-year-old dominant man who tripped and fell over in his driveway, sustaining a left 4-part proximal humeral fracture with significant varus angulation and anterior subluxation (Figure 8(A)). He was subsequently treated with a locked plate fixation augmented by an intramedullary fibular allograft strut (Figure 8(B)). Seven months postoperatively, he returned with severe functionally disabling shoulder pain. Imaging revealed avascular necrosis and complete collapse of the humeral head with arthritic changes of the glenoid related to locked screw erosion on the glenoid (Figure 8(C)).

Radiographs of patient 6. A, Injury shows a left 4-part proximal humeral fracture, which was (B) treated with an open reduction internal fixation with a locked plate and fibular allograft. C, 23-month postoperative images reveal avascular necrosis, collapse of humeral head, and locked screw erosion in the glenoid from arthritic changes. D, Revision RSA performed to restore humeral component height and balance soft tissue from the significant humeral bone loss. Patient developed an oxacillin-sensitive Staphylococcus aureus infection 25-week postoperative. E, Recent follow-up shows humeral component loosening.
Patient 6 underwent an rRSA (DJO Altivate, Austin, Texas) using a cemented humeral component. The procedure took 180 minutes due to the challenges of bypassing the fibula allograft including loss of a significant amount of proximal humeral bone. Given the significant proximal humeral bone loss, restoration of appropriate humeral component height was difficult, and soft tissue balancing was best achieved using a 44 mm glenosphere (Figure 8(D)). He subsequently developed a postoperative infection with Oxacillin-Susceptible Staph Aureus identified 25 weeks’ postsurgery. The infection was treated by a series of surgical debridements with exchange of modular components and 6 weeks of intravenous antibiotics. Radiographs taken 37 months’ postsurgery demonstrated humeral component loosening and proximal humeral bone loss. The patient demonstrated worse functional mobility compared to his preoperative level, shown by the decrease in SST from 5 to 2 (Figure 7(E)). However, the patient did have slightly better active range of motions and a VAS pain score of 0.
Absence of Intramedullary Allograft
There were 8 patients revised to an rRSA without intramedullary allografts. These patients also had a minimum 2-year follow-up. The average age of this cohort was 74 years (range: 63–82) and 6 were females. The average time to revision was 26 months (range: 6–55 months). Operative time required for rRSA was 124 minutes (range: 94–168). Improvements were observed in all PROMs and motion for patients, with only 1 exception being active internal rotation (Table 2). There were no intraoperative complications, but 1 patient developed a postoperative infection requiring surgical debridement and placement of antibiotic spacer for an oxacillin-sensitive staph aureus infection.
Preoperative to Postoperative Improvement in Patient-Reported Outcomes Measurements and Measured Motion of Patients Without a Fibular Allograft.

Abbreviations: ASES, American Shoulder and Elbow Surgeons; MCS, 12-item Short-Form Health Survey Mental Component Score; PCS, 12-item Short-Form Health Survey Physical Component Score; SST, Simple Shoulder Test; VAS, visual analog scale.
aInternal rotation conversion scale: buttock to greater trochanter (2 points); sacrum to L4 (4 points); L3–L1 (6 points); T12–T8 (8 points); T7–T1 (10 points).
Cohort Comparison
The operative time on patients with intramedullary allografts was 22% higher (151 vs 124 min). Intraoperative complications were observed only in the allograft cohort, and both cohorts experienced a single postoperative infection. Improvements in PROMs and ROMs were more significant in the cohort without retained allografts. The allograft cohort showed no improvements in SST, PCS, MCS, ASES, and VAS function as well as active elevation and internal rotation, emphasizing the challenge of effectively operating and obtaining positive outcomes on patients with a retained allograft.
Discussion
To our knowledge, this is the first study to report and compare the mid-term outcomes following conversion of failed open reduction and internal fixation to an rRSA. As this case series highlights, a healed intramedullary allograft creates difficulties during revision surgery. While attempting to bypass the allograft, humeral cortical perforation and loss of proximal humeral bone can occur (as demonstrated in 2 of the 6 patients), and surgical time required for the revision increases (22% longer operative time).
When first introduced, locked plating was thought to represent a superior treatment option both biomechanically and clinically, given the inherent principle of angular stability in the setting of poor quality bone. 20 , 21 Yet, this biomechanical advantage did not appear to fully negate adverse outcomes, with complication rates reported to be as high as 49%, 2 , 22 , 23 with continued concern for varus displacement, fracture settling, and screw penetration. 4 , 24
Surgical techniques have evolved to integrate intramedullary allografts and locking plate fixation to provide additional biomechanical support and healing potential. 9 , 25 , 26 Cadaveric studies on fibular strut graft augmentation in combination with a locking plate have shown great promise in minimizing complications. 11 , 27 Bone graft augmentation constructs stiffness, enhances volumetric fracture reduction, and increases resistance to locking plate screw-pullout, malunion, and humeral head varus collapse. 28 Studies also indicate significantly less displacement, 27 cyclical motion, and reduced plastic deformation 29 compared to locking plate fixation alone.
Likewise, clinical studies have demonstrated good results with locked plating with adjunct intramedullary allograft, with Gardner et al. 8 reporting no loss of reduction in a series of 7 patients. Neviaser et al. 14 also reported that the use of plate and fibular allograft appears to enhance stability, resulting in greater patient functional scores by indirectly reducing complications (lower rates of reduction loss, screw cutout, and osteonecrosis).
Although it has been suspected that failure of locked plate with intramedullary graft fixation would be associated with challenging revision procedures, there remains a paucity of discussions regarding these failures. Robinson et al. 17 reported 2 cases (9.5%) of fixation failure and recurrent varus deformity 2 weeks following locking plate and fibular graft utilization. Failure secondary to plate malposition was attributed to poor technique in placement of the posteroinferior locking plate screws. One patient underwent revision fixation with maintenance of the index hardware, while the other went onto revision replacement arthroplasty. Unfortunately, the applied surgical technique was not discussed. Neviaser et al. 14 reported a single case (2.6%) of reduction loss and varus collapse that did not require further intervention. Chen et al. 10 reported 2 complications (13.3%) following fibular graft and plate fixation. Satisfactory results were obtained after plate removal in a patient with avascular necrosis, while another patient with varus displacement achieved good function without revision.
Our case series describes a 50% complication rate (3 of 6 patients) in patients with intramedullary allografts during rRSA. One patient developed an acute postoperative infection at 25 weeks requiring surgical debridement, exchange of the modular components, and antibiotic bead placement. Two intraoperative complications were observed directly attributed to the healed intramedullary allograft, both resulting in cement extrusion. Fortunately, both patients were safe from nerve related injury, as previous reports have described significant neurovascular compromise from similar cement extrusion.30–33 It is also possible that cortical perforations may have occurred where uncemented humeral component fixation was utilized. Although the surgical technique described using fluoroscopic guidance for guide pin placement and cannulated reamers can help to bypass and clear away the intramedullary allograft, it is not free from the potential complication of cortical breakthrough.
The described surgical technique for bypassing the healed intramedullary allograft is similar to an intramedullary cement removal technique previously described. 34 The incorporated intramedullary allograft is typically quite dense, preventing the routine passage of humeral canal reamers. By using a guide pin under fluoroscopic guidance, it is possible the drill though the healed graft in line with the intramedullary canal. Once the intramedullary graft is bypassed, standard reamers can then be used. This technique requires careful attention to the trajectory of the guide pin, as the intramedullary graft may have been placed at an angle which can cause the guide pin to migrate out of the humeral cortex. This may have occurred in the 2 cases where cement extravasation occurred. When possible, avoidance of cement in these cases can help mitigate the risk of cement extrusion and nerve related injury.
Despite the increased complication rate and longer operative time for patients with retained intramedullary allografts, the clinical outcomes following rRSA demonstrate significant improvements in pain and modest improvements in function from the preoperative status. These findings are consistent with previous reports of managing failed fixation of proximal humerus fractures with RSA. Hussey et al. 35 reported improvements in pain and functional scores among 19 patients treated with RSA for failed internal fixation of proximal humerus fractures, citing a 25% major complication rate. In the largest reported series of patients treated with RSA for similar indications, Grubhofer et al. 36 reported improvements in pain and functional outcome scores with a revision rate of 7% (4 patients).
This case series is not without limitations. As a small series of patients, the ability to detect significant differences among the 2 cohorts is difficult. Increased operative time and intraoperative complications were seen in patients treated with intramedullary allografts; nonetheless, meaningful statistical conclusions were not possible. Selection bias was mitigated by including all patients treated with rRSA for failed proximal humerus locked plate fixation at this institution. As a single surgeon case series, the results may not be extrapolated to the experience of all surgeons. Revision of failed fixation of proximal humerus fractures to RSA represent challenging procedures with high levels of complexity related to dense surgical scarring and altered anatomy from prior surgery and fracture healing.
Conclusion
RSA is a reasonable option for patients with failed proximal humerus fracture fixation, even in cases with healed intramedullary allografts. However, healed intramedullary allografts are associated with increased operative time and a higher rate of intraoperative complications. Surgical techniques to bypass the healed allograft are helpful in managing these challenging cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Levy is a paid consultant for DJO Orthopaedics and Globus Medical. He receives royalties from DJO Orthopaedics and Innomed. No Federal funds or outside sponsorship helped fund this study. All remaining authors, their immediate families, and any research foundation which they are affiliated have not received any financial payments or other benefits from any commercial entity related to the subject of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
IRB Approval
This study received Category 4 Exemption status per CFR 46.101(b)(4) on 06/17/2019.
