Abstract
Background
Total shoulder arthroplasty (TSA) has demonstrated good long-term survivorship but early implant failure can occur. This study identified factors associated with shoulder arthroplasty revision and constructed a risk score for revision surgery following shoulder arthroplasty.
Methods
A validated algorithm was used to identify all patients who underwent anatomic TSA between 2002 and 2012 using population-based data. Demographic variables included shoulder implant type, age and sex, Charlson comorbidity score, income quintile, diagnosis, and surgeon arthroplasty volume. The associations of covariates with time to revision were measured while treating death as a competing risk and were expressed in the Shoulder Arthroplasty Revision Risk Score (SARRS).
Results
During the study period, 4079 patients underwent TSA. Revision risk decreased in a nonlinear fashion as patients aged and in the absence of osteoarthritis with no influence from surgery type or other covariables. The SARRS ranged from −21 points (5-year revision risk 0.75%) to 30 points (risk 11.4%). Score discrimination was relatively weak 0.55 (95% confidence interval: 0.530.61) but calibration was very good with a test statistic of 5.77 (df = 8, P = .762).
Discussion
The SARRS model accurately predicted the 5-year revision risk in patients undergoing TSA. Validation studies are required before this score can be used clinically to predict revision risk. Further study is needed to determine if the addition of detailed clinical data including functional outcome measures and the severity of glenohumeral arthrosis increases the model’s discrimination.
Introduction
Shoulder arthroplasty has become increasingly common over the past several decades in the United States,1–6 Germany, 7 the Nordic Countries, 8 and Australia. 9 Hemiarthroplasty (HA) and total shoulder arthroplasty (TSA) have both been shown to provide good pain relief and improved function for patients with shoulder fracture or arthritis.10–17
Increased arthroplasty utilization means an increased risk of shoulder arthroplasty revision surgery. Various prognostic factors have been identified, which correlate with revision surgery including higher body mass index (BMI), 18 smoking, 19 increased comorbidity, 20 and younger patient age,18–21 as well as increased retroversion,22,23 male sex, 24 the presence of rotator cuff disease, 24 and with sequelae following proximal humeral fractures. 21 A practical scoring system that surgeons can use to accurately predict the risk has, to our knowledge, not been created. Knowing this risk can be helpful to surgeons and patients when weighing treatment options in patients with shoulder arthritis. This risk is of particular concern in the young patient. Younger patients may have a higher risk of periprosthetic infection 25 and higher rates of mechanical failure. 21 Furthermore, revision shoulder arthroplasty carries higher complication rates such as infections 26 and is more complex since bone stock is often compromised, and improvement in functional outcomes is less consistent. 27 Being able to accurately predict revision risk is important when counseling patients regarding treatment options for shoulder problems.
Using population-based data, the purpose of this study was to determine patient and surgical factors that were independently associated with revision risk after anatomic shoulder arthroplasty surgery and to construct a predictive risk score to predict the 5-year risk of revision surgery. Our hypothesis was that younger patient age is associated with higher revision risk.
Methods
Databases Used in the Study
The patients included in this study were part of a previous study that compared HA with TSA using population-based health administrative databases in Ontario, Canada, housed at the Institute for Clinical Evaluative Sciences (ICES). 28 The previous study found no statistically significant difference in survivorship between HA and TSA while controlling for important covariables including arthroplasty type, age, sex, Charlson comorbidity index, income quintile, and presence of rheumatoid or osteoarthritis (OA). Health-care costs for all hospital and physician services are covered by a universal health-care system. Databases used in this study included: the Discharge Abstract Database (DAD), which captures all hospitalizations including all major procedures and diagnoses (recorded using standard codes); the Registered Persons Database (RPDB), which captures each person’s death date and their place of residence over time (along with the median household income of that area); the ICES Physician Database (IPDB) database, which records surgeon factors such as date of medical school graduation; and the Ontario Health Insurance Plan (OHIP) database, which captures all claims submitted by surgeons for remuneration. This study was approved by the research ethics board of the Ottawa Hospital.
Study Cohort
We included patients of all ages who had undergone shoulder arthroplasty conducted in Ontario between April 1, 2002 and March 31, 2012 (n = 10 026). We did not use data prior to this study period because different coding systems had been used prior to 2002. At the time the study was started, data were complete to March 2012. Patients coded with anatomic shoulder arthroplasty were included in the current analysis. We excluded patients with reverse shoulder arthroplasty, surgeries that were coded as revisions (n = 763, 7.6%) and whose intervention laterality was unknown (n = 469, 4.7%); subsequent surgeries for patients who had more than 1 surgery on the ipsilateral shoulder during the study period (n = 718, 7.1%); patients who did not survive the index surgery or patients who were not Ontario residents (n = 20, 0. 2%); and patients with shoulder arthroplasty prior to the study period or who were coded with shoulder fracture (ICD10 S422*) at the same time as the arthroplasty (n = 2279, 22.7%). We did not extend the inclusion study period when new data became available because we wanted to ensure a minimum observation of 4 years for each patient.
Study Outcome
The primary outcome of the study was time to revision shoulder arthroplasty. Observation started on the date that the index shoulder arthroplasty had been conducted. Observation continued until patients underwent revision shoulder arthroplasty on the same side as the index shoulder arthroplasty, died, or reached the study observation end date (March 31, 2016). Revision arthroplasty was identified in DAD with the same code criteria used to create the study cohort. Deaths were identified in RPDB.
Covariables Included in the Model
Patient age and sex were determined from the DAD record for the index arthroplasty. Comorbidities were quantified with the Charlson comorbidity index calculated using diagnostic codes recorded in DAD during the index admission. We specifically determined if either rheumatoid arthritis (RA; ICD-10-CA codes M05, M06, M080; ICD-9 code 714) or OA (ICD-10-CA codes M19; ICD9 code 715) was recorded in the DAD. We linked to IPDB to measure surgeon factors including surgeon age and experience (quantified as years since medical school graduation). We used OHIP to calculate surgeon-specific median annual surgery volume performed prior to the index surgery. Finally, we used RPDB to impute patient socioeconomic status using the median household income quintile at the time of the surgery. Specific surgical details regarding the type of anatomic TSA, including the use of cement or glenoid prosthetic type, were not available in the registry.
Statistical Analysis
We used descriptive statistics to define our sample. We followed each eligible patient after the index arthroplasty until revision surgery, end of study, or death. Variables independently accounted for in the model included diagnosis (OA, RA, other [which included patients with avascular necrosis]) and demographic factors including age, sex, rural residence, income quintile, Charlson Index, and surgeon factors. The likelihood of revision was expressed in terms of “revision risk” and was determined using competing risk analysis. A competing risk is an event that occurs (such as death) and that precludes the event of interest (in this case arthroplasty revision). For example, a patient in our study who died of heart disease had a competing event since this would preclude revision arthroplasty surgery from happening. Competing risk analysis is particularly important in older populations who have an increased risk of competing events (such as death). Traditional approaches to statistical analysis of survivorship data, such as the Kaplan–Meier method, would (in our study) result in revision risk estimates that are biased upwards.
The risk of incident revision over time was described using the cumulative incidence function. We used the Fine–Gray subdistribution hazard model 29 to measure the association of arthroplasty type with time to shoulder revision surgery. This model is used to determine an individual’s risk and accounts for the risk of an event (the rate of occurrence of an event in those still at risk of the event) along with competing risks. An interaction term between time and age was included in the model to address the violation of the proportional hazard assumption with age (ie, the ratio of the hazard of revision surgery between age groups was not consistent over time). The best-fitting polynomial transformation that captured the association was determined according to the minimum value of the Akaike information criterion. This criterion estimates the relative quality of statistical models. It was used to determine the polynomial transformation that best fit our data.
We developed a point-based risk score with age, age squared, OA, OA/age interaction, and an age and time interaction, using the methodology defined by Austin (2016). 30 We used calibration plots and observed versus predicted graphs to assess model performance. Calibration is defined as the agreement between observed and predicted risk. This is important because the purpose of the model is to predict future risk in a target population. Calibration was assessed with the Greenwood–Nam–D’Agostino calibration test statistic.
Score discrimination refers to the ability of a model to separate patients with certain factors from those without these factors. In this study, discrimination refers to the model’s ability to determine whether patients with higher Shoulder Arthroplasty Revision Risk Scores (SARRS) have a higher revision risk than those with lower scores. Loss of predictive accuracy of the age-based points scoring system was examined by regressing the incidence of revision on subjects’ score.
Results
During the study period, 4079 TSA cases met the inclusion criteria. These patients had a mean age of 68 years (SD 10.2), were more likely to be female (58%), occasionally lived in rural areas (17.4%), and commonly had OA (74.1%) but rarely had RA (2.7%; Table 1). Patients were followed for a median of 6.0 years (interquartile range: 4.1–8.3).
Demographics.

Abbreviations: IQR: interquartile range; SD: standard deviation; TSA: total shoulder arthroplasty.
Both revisions and deaths were relatively uncommon events with patient observation ending with neither occurrence 76% of the time (Table 2). Death (incidence density 29.5; 95% confidence interval [CI]: 27.8–31.3, events per 1000 patient-years observation [pys]) was more than 3 times more common than shoulder revision (incidence density 8.8 [95% CI: 7.9–9.8] events per 1000 pys). After 5 years, the risk of death was 10.4% (95% CI: 9.6–11.2) and the risk of revision (accounting for the competing risk of death) was 4.9% (95% CI: 4.4–5.5). Kaplan–Meier curves for death are presented in Figure 1.
Outcome Risks.

Abbreviation: CI, confidence interval.
aPresented as number of events per 1000 person-years observation.
bBased on cumulative incidence function (to account for competing risks).

Kaplan–Meier curves for death over (A) 5 years and (B) 30 days.
Of the factors that we offered to the model (Table 1), the only covariables independently associated with time to revision surgery (accounting for the competing risk of death) were patient age and OA status (Table 3). We found that age was best expressed as a combination of a linear and a squared term. In addition, the interaction of age with both OA status and time from surgery also significantly improved model performance. Model discrimination was relatively weak with a c-statistic of 0.55 (95% CI: 0.53–0.61). Calibration was very good with a Greenwood–Nam–D’Agostino’s calibration test statistic of 5.77 (df = 9, P = .76; Figure 2). 31
Competing Risk Survival Model for Time to Arthroplasty Revision Surgery.
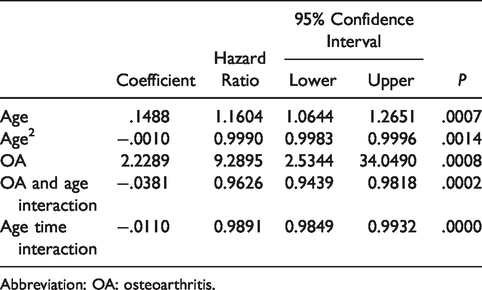
Abbreviation: OA: osteoarthritis.
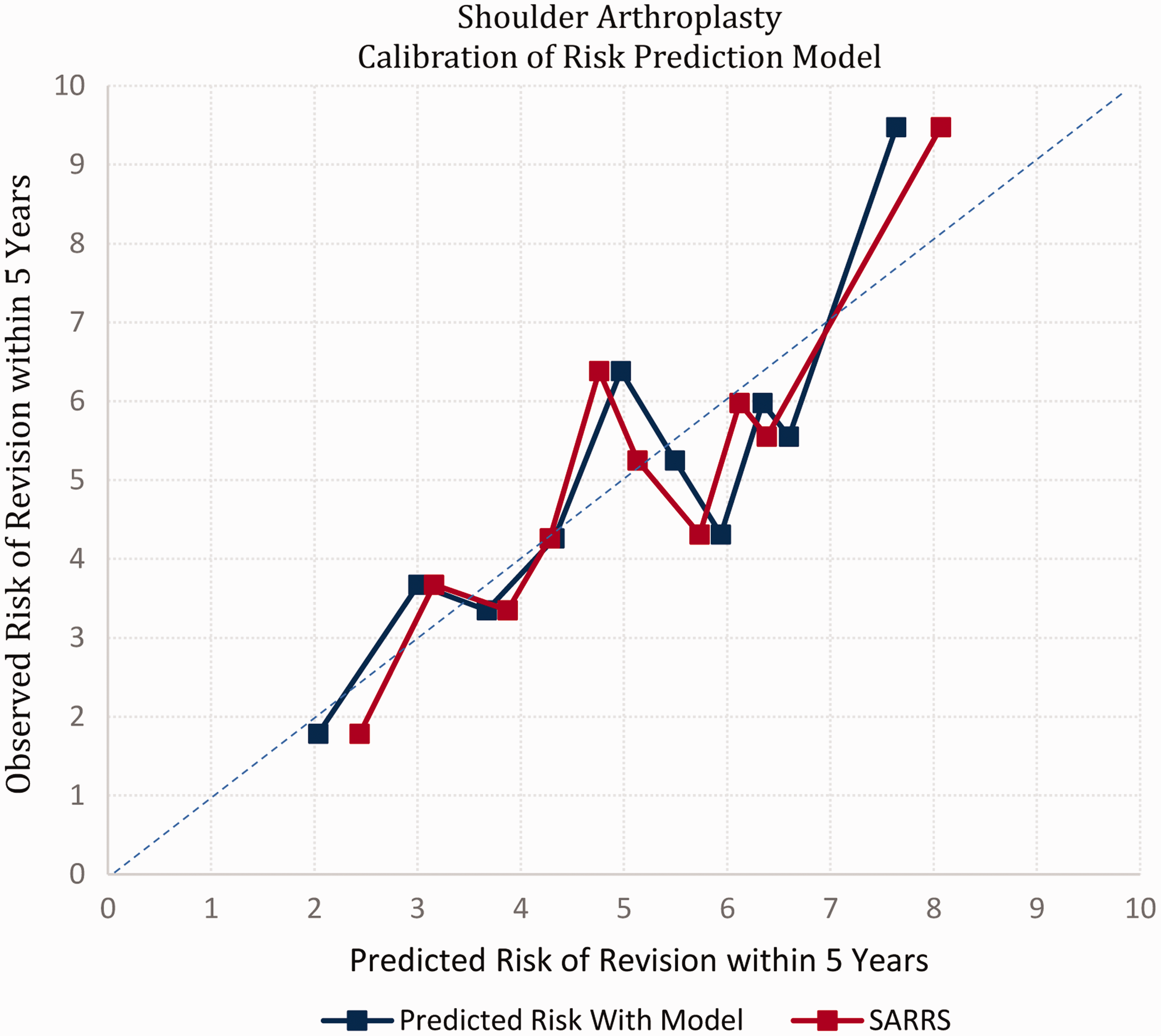
Observed and expected risk of shoulder revision surgery within 5 years. SARRS: Shoulder Arthroplasty Revision Risk Score.
The SARRS is presented in Table 4. Based on the patient’s age and OA status, the total number of SARRS points can be determined. For example, a patient with an age of 66 years and a diagnosis of OA has 17 SARRS points (Table 4). Table 5 is used to determine the model-based 5-year revision risk for each SARRS. For example, patients with a SARRS of 17 had an expected 5-year revision risk (accounting for the competing risk of death) of 5.8%. Calibration of the SARRS was statistically indistinguishable from the original model (Figure 1) with a Greenwood–Nam–D’Agostino’s calibration test statistic of 13.9 (df = 9, P = .13). 31
The SARRS.

Abbreviations: OA: osteoarthritis; OR, odds ratio; SARRS: Shoulder Arthroplasty Revision Risk Score.
The SARRS is determined by the number of points based on the patient’s age and osteoarthritis (OA) status (left). The expected 5-year risk of revision surgery (accounting for the competing risk of death) is presented on the right. For example, a patient with an age of 66 years and a diagnosis of OA has a SARRS of 17 (left table). A SARRS of 17 predicts a revision risk of 5.8% over 5 years (right table).
SARRS Score With Corresponding Probabilities of 5-Year Revision.

Abbreviation: SARRS: Shoulder Arthroplasty Revision Risk Score.
The SARRS categorized patients into 3 statistically exclusive risk groups (Figure 3). Based on the patients’ SARRS, patients could be classified into those whose 5-year revision risk was the lowest risk category (SARRS ≤4, Observed revision risk 2.6%, predicted risk 2.5%), medium risk category (SARRS between 6 and 19, observed revision risk 5.0%, predicted 5.2%), and highest risk category (SARRS ≥20, observed revision risk 7.1%, predicted 6.8%).
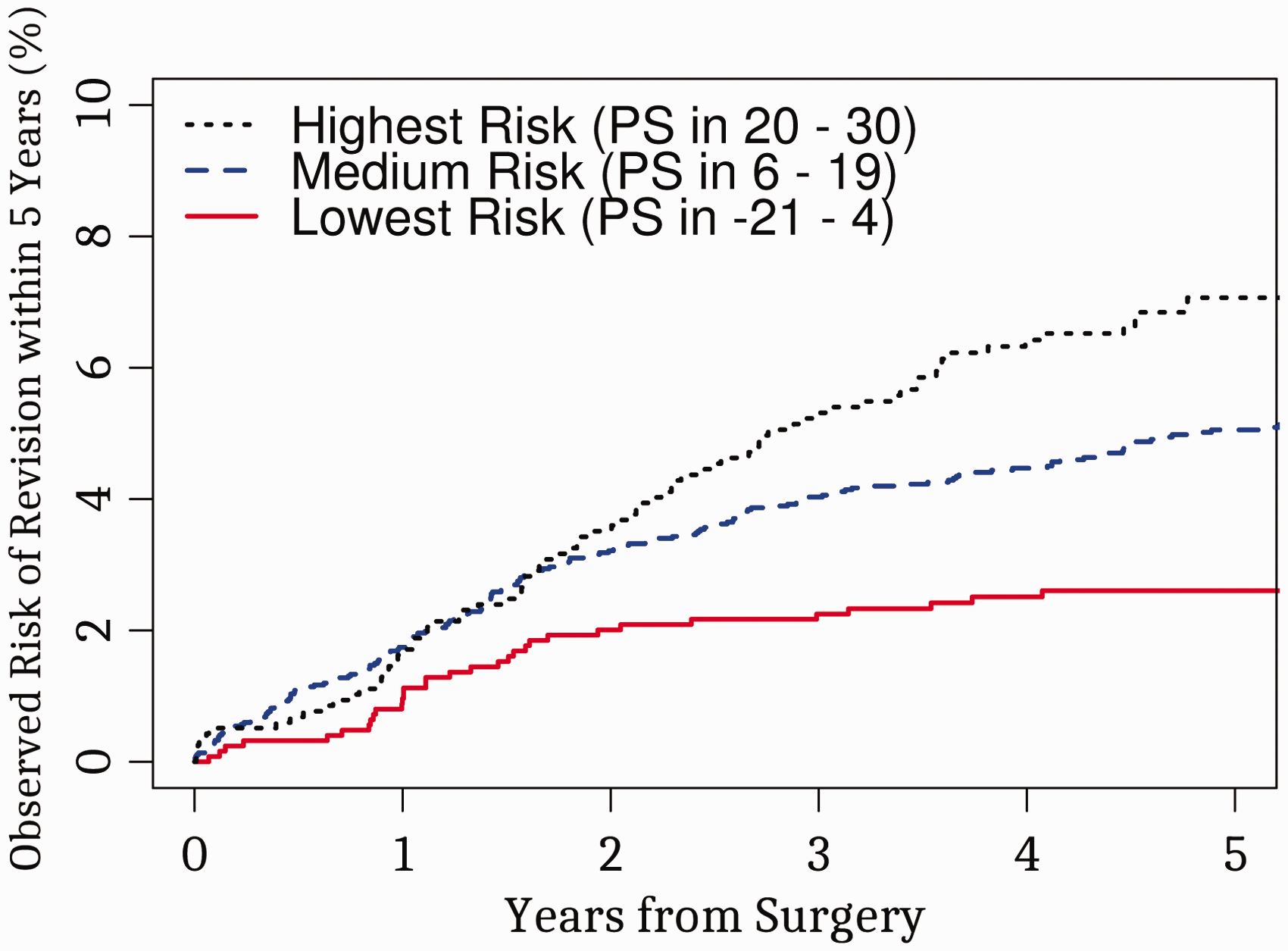
Risk of 5-year shoulder arthroplasty revision surgery by Shoulder Arthroplasty Revision Risk Score (SARRS) group. We divided the subjects in the study into 3 risk groups where first and last groups have 20% of the subjects with highest point score and lowest point score, respectively. Cumulative incidence functions for shoulder revision were estimated within each of the 3 groups.Abbreviation: PS: point score.The observed risk curves in this group are statistically distinct (Gray’s test score: 20.24, P < .001; c-stat: 0.5762; 95% CI: 0.5354–0.617).
Discussion
The purpose of this study was to determine patient and surgeon factors associated with revision risk after anatomic shoulder arthroplasty surgery and to construct a predictive risk score to predict the 5-year risk of revision surgery. To our knowledge, this is the first study that uses population-based data to derive a score that predicts the risk of revision surgery in patients undergoing anatomic shoulder arthroplasty. We found that patient age had a nonlinear effect on revision risk and interacted significantly with the presence of OA. In patients without OA, the revision risk increased with age while patients are <35 years, plateaued until an age of 55, and then decreased afterwards. In patients with OA, revision risk progressively decreased as patients aged. Our point-based risk score accurately predicted risk of revision surgery. This score was used to categorize patients into significantly distinct revision surgery risk groups. The group with the lowest SARRS scores (with SARRS ≤4) had a revision risk of 2.3%. The middle SARRS group had more than double the revision risk at 4.8%. The highest SARRS group had a revision risk of more than triple that of the lowest risk group at 7.1%. The SARRS point system summarized in Table 4 illustrates the association between age, OA status, and revision risk: the higher the points, the greater the risk.
Our data indicated that revision surgery was very uncommon with a 5-year risk of 4.9%. The incidence of revision was much less common than death (5-year risk 10.4%). Therefore, a competing risk model to control for the competing risk of death (as used in our study) was highly important to attain unbiased parameter estimates and risk estimates. The 5-year incidence of death of 10.4% observed in this study was slightly higher than the probability of death cited in provincial life tables from 2006 to 2008, which indicate the probability of a 68-year-old person dying in 5 years was 8.5%. In the first 30 days following surgery, the Kaplan–Meier curve for death (Figure 1) did not appear to demonstrate an increase over baseline. In this cohort, 71.6% of patient had a Charlson score of 0, and 87.3% had a maximum of 1, which was not worse than population norms. 32
Previous studies have also identified an association between younger patient age and poorer survivorship outcomes in shoulder arthroplasty.8,18,20,21,33–36 It is possible that these poorer outcomes may be attributed to the higher incidence of mechanical failure and aseptic loosening 21 associated with more complex and multifactorial pathology that occurs in younger populations including congenital deformity, posttraumatic arthropathy, and RA. In addition, younger patients may place a higher demand on their implants postoperatively and have greater expectations.37,38
In a multivariate analysis, Singh et al. found that underlying diagnosis was not associated with revision risk. 18 This contrasts with a study by Farng et al. 20 in which implant failure rates were lower in patients with RA, a finding consistent with this study in which both age and an underlying diagnosis of OA were highly associated with revision risk. It is possible that the higher risk of revision in OA patients may be related to greater demand and corresponding stresses on the glenohumeral joint as well as higher expectations compared with RA patients but further study is required for this to be fully elucidated.
In addition to age, other factors have been identified to be associated with increased revision risk.39–41 Patient with higher BMI have been shown to have higher revision risk, 18 and superobese patients have higher incidences of infection, dislocation, component loosening, venous thromboembolism, and medical complications than nonobese controls. 41 Smoking was also identified as an independent risk factor for revision in shoulder arthroplasty. 19 Charlson index was not associated with revision risk in this study, although our data did not specifically include smoking status or BMI. In a study by Werner et al., 19 male sex was associated with early revision following reverse shoulder arthroplasty but this variable was not significantly associated with other arthroplasty types. Male sex was not associated with revision risk in this study.
Surgeon experience has been shown to be associated with implant survivorship in other joints. In knee arthroplasty, revision rates were lower in hospitals with higher surgical volumes. 42 In upper extremity arthroplasty, Schairer et al. 43 found that hospitals with a lower volume of HHR or RSA had higher 90-day readmission rates. Higher volume providers have been shown to have better outcomes44,45 and were more likely to perform TSA versus HHR for OA. In this study, surgeons with more experience performed TSA more frequently, although surgeon experience was not associated with revision risk. It is possible that the relatively low surgeon volumes in this cohort limited the ability of this variable to influence revision risk; nevertheless, this variable appeared to have less influence on revision risk than other factors in the model.
Several issues should be considered when interpreting our results. First, patient-reported functional outcomes were not available, and therefore, the current model reflects revision risk only. It is possible that the addition of these variables might improve the accuracy of the SARRS model. Second, it is not possible to obtain individual health behaviors and sociodemographic components from administrative health data. The database had limited ability to differentiate between different diagnostic categories, and patients were coded with a diagnosis of OA, RA, or other. Although patients with rotator cuff tears were analyzed in the “other” category, more granularity in terms of diagnosis would have been beneficial and may form the basis of future research in this area. Third, the SARRS model distinguished risk into 3 distinct groups. It was not possible to include other factors that may affect revision risk such as the severity of the arthritis, level of complexity associated with revising certain types of implants, or other surgical factors. Future work should focus on adding surgical and clinical data to the skeletal model presented in this study to improve the model’s discrimination so that medium risk groups can also be distinguished. Finally, our study captured all outcomes (both revision surgeries and deaths) since we used population-based datasets. These factors ensure accurate risk estimates and generalizability of the results.
Summary
Two factors were found to significantly influence revision risk in anatomic TSA: patient age and diagnosis. We developed a score based on the patient’s age and OA status that can be used by surgeons to estimate the risk of revision surgery (accounting for death). Future efforts should focus on validating this model’s performance in other cohorts and on building upon the current model by adding functional and surgical data.
Footnotes
Authors’ Note
This review was performed at the Ottawa Hospital, Ottawa, Ontario, Canada.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CvW is supported by a University of Ottawa Department of Medicine Clinician Scientist Chair.
