Abstract
Background
Accurate restoration of anatomy is critical in reestablishing proper glenohumeral joint function in total shoulder arthroplasty (TSA). However, even experienced surgeons inconsistently achieve anatomic restoration. This study evaluates whether a new canal-sparing arthroplasty system, designed using the principles of calibrated bone resection and incorporating a nonspherical humeral head prosthesis, can assist in more accurate and reliable reproduction of proximal humeral anatomy compared to a stemmed arthroplasty system.
Methods
The difference between the anatomic center of rotation (COR) of the humeral head and the postoperative prosthetic COR (defined as ΔCOR) was measured in a consecutive case series of 110 shoulder arthroplasties performed by a single surgeon. The first 55 cases used a stemmed arthroplasty system and the subsequent 55 cases used a new canal-sparing implant system that uses a multiplanar osteotomy (MPO) during humeral head preparation. Cases with ΔCOR ≥3.0 mm were deemed clinically significant outliers.
Results
The average ΔCOR in the MPO group was 1.7 ± 1.2 mm versus 2.8 ± 1.5 mm in the stemmed group (P = .00005). The incidence of outliers was lower (14.5% vs 40.0%, P = .005), and there were more cases with a ΔCOR ≤1.0 mm (32.7% vs 3.6%, P = .0001) in the MPO group compared to the stemmed group.
Conclusion
The MPO TSA system provided improved accuracy and precision in restoring proximal humeral anatomy compared to stemmed arthroplasty systems, even in its initial use. This alternative method of humeral replacement may increase consistency in restoring proper anatomy and kinematics in TSA.
Introduction
Anatomic total shoulder arthroplasty (TSA) is an increasingly prevalent surgical intervention that can provide lasting pain relief and improved range of motion for patients suffering from glenohumeral joint arthritis.1–3 The importance of accurately restoring the anatomy of the glenohumeral joint in shoulder arthroplasty has been recognized at least since the days of Neer’s first prosthesis. 4 Nonanatomic positioning of prosthetic components results in inferior clinical outcomes due to altered kinematics and soft tissue imbalances, and can result in complications such as subscapularis failure, stiffness, glenoid failure, and late supraspinatus tear.5–10 Anatomic reconstruction has been a guiding principle in the evolution of TSA designs. However, despite substantial improvements in implant design, even experienced shoulder surgeons are inconsistently able to accurately restore the native anatomy of the proximal humerus following TSA.7,11
A number of studies have sought to better define the anatomy of the proximal humerus and apply this knowledge to improve shoulder arthroplasty design.12–18 Early TSA systems had fixed angle inclination and offset. Newer third- and fourth-generation prostheses offer multiple offset and inclination options, which have been shown in computer models to better restore the native anatomy.14–16 Unfortunately, even these newer stemmed systems have limitations in their ability to restore proximal humeral anatomy due to variable relationship of the articular surface to the humeral diaphysis. Although humeral head resurfacing and stemless humeral head components theoretically allow for more anatomic reconstruction, clinical evidence has shown that resurfacing implants have been even less accurate than stemmed designs in reconstructing the joint line and humeral head center of rotation (COR). 11 Frequently, there is little or no relationship between the thickness of humeral head bone resected and the height of the final implant placed.
Despite these reported shortcomings of currently available resurfacing and stemless humeral head components, we believe the theoretical benefits of a more anatomic reconstruction with canal-sparing humeral head components are achievable with innovation in implant design and surgical technique. One of the authors (SSG) developed a TSA system that does not require a diaphyseal stem, applies the principles of calibrated bone resection during humeral head preparation, and uses a nonspherical humeral head prosthesis with the goal of helping surgeons more accurately and consistently recreate the native anatomy of the proximal humerus. In order to achieve this, the technique uses cutting guides to create a multiplanar bone preserving humeral head osteotomy, which matches the thickness of the bone resected to the thickness of the prosthetic component (Figure 1). The instruments and technique were previously validated in a cadaver study and have been shown to allow surgeons to reconstruct proximal humeral anatomy with high accuracy and reproducibility. 19

Sequence of steps in the multiplanar osteotomy. A, Placement of guide pin in the center of the articular surface of the humeral head. B, Flat surface reamed with the depth of bone removed calibrated to humeral head size. C, Anterior and posterior chamfer cuts. D, Inferior and superior chamfer cuts with drill holes for humeral head component.
The purpose of the current study is to expand upon the cadaveric validation with a radiographic study comparing the positioning of the humeral component using this stemless multiplanar osteotomy (MPO) system to a modern stemmed TSA system. Our hypothesis was that the MPO system would result in more accurate and precise reconstruction of proximal humeral anatomy compared to the stemmed system as measured by the difference in COR of the humeral head on plain radiographs. We defined improved accuracy as minimizing the change in the COR of the humeral head between the anatomic COR and the postoperative prosthetic COR (defined as ΔCOR)—with a more accurate reconstruction having a ΔCOR value closer to zero. We defined precision by quantifying the number of clinically significant outliers in each group—with a more precise system resulting in fewer outliers. Clinically significant outliers were defined as cases with a ΔCOR >3.0 mm. The concept of analyzing ΔCOR to assess the accuracy of TSA in restoring native proximal humeral anatomy, and the 3.0 mm cutoff used to define “clinically significant outliers” was previously described by other authors. 11 We chose to use the term “clinically significant outliers” to maintain consistency with this prior work. The 3.0 mm cutoff was chosen as previous biomechanical studies have found that a ΔCOR of this magnitude can adversely affect shoulder biomechanics and kinematics.5,6,8–10
Materials and Methods
Patient Selection
All primary anatomic shoulder replacements (113 consecutive cases) performed by a single fellowship-trained shoulder surgeon over a 2-year period from April 2015 to April 2017 were reviewed. Patients were included in the study if acceptable radiographs, either an intraoperative or postoperative true anteroposterior (AP) radiograph of the shoulder 20 with an unobstructed view of the entire humeral prosthesis in perfect or near-perfect profile, were available. For the stemmed system, this was defined as minimal (<2 mm) overlap of the glenoid and the humeral component. For the MPO system, this was defined as overlap of the 2 central pegs of the humeral component. Patients were excluded from the final analysis if acceptable radiographs were not available. Although the patients were not randomized, consecutive cases were used to minimize selection bias and nearly all of the consecutive cases (110 of 113) were included in the study. The final case distribution included 55 procedures using the Arthrex Univers™ II (Arthrex, Naples, FL, USA) third-generation stemmed total arthroplasty system and the initial 55 procedures using the Catalyst CSR Total Shoulder System (Catalyst OrthoScience, Naples, FL—elsewhere referred to as the MPO or MPO system). Thus, the surgeon used each arthroplasty system exclusively for a period of time to treat the full spectrum of pathology that was a candidate for anatomic TSA. There was no “choosing” of which system to use for a given patient. With 55 patients in each exclusive time period, we assume that both groups had a normalized distribution of severity of arthritis. With large numbers, any significant margin of error with imperfect X-rays would have been normally distributed within each group. In addition, all 55 of the MPO group patients were included in the X-ray analysis (none disqualified or lost to follow-up), thus eliminating the potential to hide or eliminate certain outliers or exclude patients during a “learning curve” period. As this study focuses on the restoration of the anatomy of the proximal humerus following TSA, we did not characterize glenoid morphology. Patient demographic data including age, sex, and laterality of surgery were also collected.
All cases were performed using a standard deltopectoral approach and a subscapularis tenotomy. The subscapularis tenotomy was performed by placing a curved clamp posterior to the subscapularis tendon through the rotator interval and dividing the subscapularis tendon 1 cm medial to its insertion on the lesser tuberosity. This left 1 cm of the tendon on its attachment on the lesser tuberosity for later repair with #5 braided nonabsorbable polyester sutures using figure of eight and Mason-Allen type stitches. Manufacturer’s instructions and instrumentation for humeral head resection were used in all cases. The intramedullary cutting guide was used in all cases in which a stemmed humeral component was implanted. The performing surgeon’s goal was anatomic reconstruction of the proximal humerus in all cases. After final implant placement, the performing surgeon checked for stability by assessing humeral head translation on the glenoid in the AP direction, with greater than 50% translation as a cutoff for instability. The size of the prosthetic humeral head component was not changed in any cases due to findings of instability.
Radiographic Analysis
Radiographs were measured using a digital measuring tool, which accounted for the magnification coefficient and allowed for highly accurate measurement to 0.1 mm (IntelePACS InteleViewer™ version 4-12-1-P254, 64-bit-Intelerad Medical Systems Incorporated, Montreal, Quebec, Canada). 21 A previously validated method for determining the difference in the COR (ΔCOR) of the native humeral head and the COR of the proximal humerus following TSA by a best-fit circle technique was used as described by Youderian et al. 22 (Figure 2).
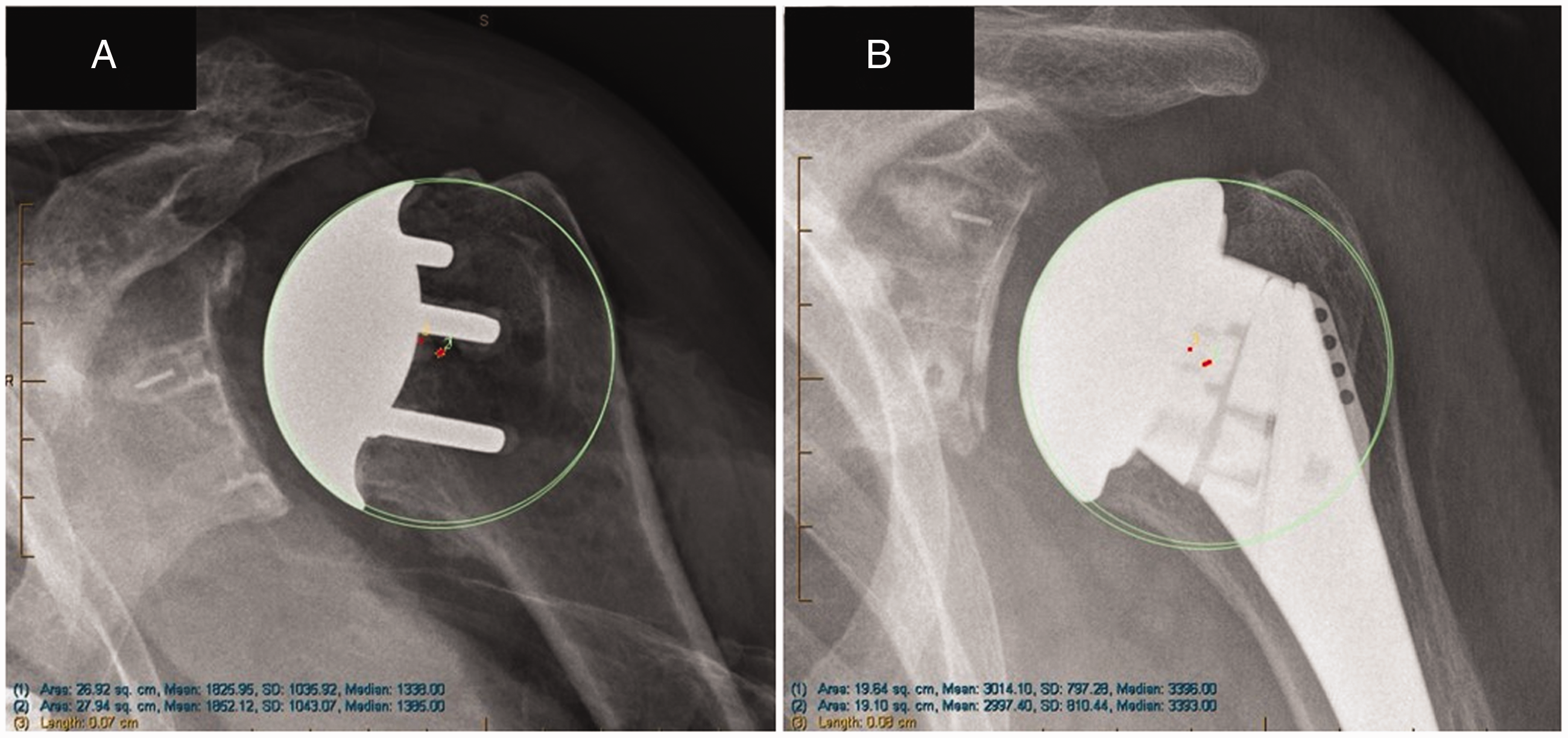
Postoperative radiographs of an MPO arthroplasty (A) and a third-generation stemmed arthroplasty (B) with ΔCOR <1.0 mm.
Two independent observers, senior-level orthopedic surgery residents, each performed the radiographic measurements for all 110 cases. The measured ΔCOR values for each case were averaged, and this value was used for final data analysis. The radiographic measurements were repeated to allow for calculation of intraobserver reliability. The mean and standard deviation of the ΔCOR for the MPO and stemmed groups were calculated. A ΔCOR ≥3.0 mm for any case was considered a clinically significant outlier (Figure 3).

Postoperative radiographs of an MPO arthroplasty (A) and a third-generation stemmed arthroplasty (B) in cases of clinically significant outliers with ΔCOR >3.0 mm.
Statistical Analysis
Descriptive statistical analysis was performed with categorical and continuous variables analyzed and reported using frequencies and means ± standard deviation, respectively. The Shapiro–Wilk test was used to assess data for normality and confirmed a normal distribution for all continuous variables except the ΔCOR measurements for both groups. The ΔCOR measurements were positively skewed with significantly more data points having smaller ΔCOR measurements than if data were normally distributed. Independent samples t tests were used to analyze normally distributed continuous variables, and the Mann–Whitney U test was used to analyze the nonnormally distributed ΔCOR measurements. Chi-square tests were used to analyze categorical variables. Statistical significance was set at α = 0.05. Sample size calculations and a post hoc power analysis were performed in collaboration with a statistician and determined that the study was adequately powered to detect a difference in all reported values. In regard to the ΔCOR measurements, the study was powered to 0.90 with a minimal detectable difference of 0.5 mm between groups. In regard to the technical outlier measurements, the study was powered to 0.80 to detect a 20% difference in the incidence of surgical outliers between the 2 groups. Lin’s correlation coefficient for continuous variables was used to determine inter- and intraobserver reliability with values above 0.80 representing excellent agreement and significance defined by the 95% confidence interval. 23 All data were analyzed using SPSS Statistics Data Editor version 24 (IBM, Armonk, NY, USA).
Results
In sum, 110 cases were included in the final analysis: 55 stemmed total shoulder arthroplasties and 55 MPO total shoulder arthroplasties. There was no difference in the age, sex, or laterality of surgery between the MPO and stemmed arthroplasty groups (Table 1).
Patient Demographic Data.

Abbreviations: MPO, multiplanar osteotomy, TSA, total shoulder arthroplasty.
The average difference in COR between the anatomic COR and prosthetic COR (ΔCOR) for cases using the MPO TSA system was 1.7 ± 1.2 mm. The average ΔCOR for cases using stemmed TSA systems was 2.8 ± 1.5 mm (P = .00005). Both the inter- and intraobserver reliability of the radiographic measurements for ΔCOR were excellent as measured by Lin’s correlation coefficient (interobserver reliability Rc = 0.91, 95% confidence interval [CI] 0.88–0.94; intraobserver reliability Rc = 0.97, 95% CI 0.95–0.98).
A ΔCOR of <1.0 mm was measured in 18 of the 55 (32.7%) MPO TSAs compared to 2 of the 55 (3.6%) stemmed TSAs (P = .001). A ΔCOR between 1.0 mm and 2.9 mm was measured in 29 of the 55 (52.7%) MPO TSAs compared to 31 of the 55 (56.4%) stemmed TSAs (P = .85). A ΔCOR between 3.0 mm and 5.9 mm was measured in 8 of the 55 (14.5%) MPO TSAs compared to 20 of the 55 (36.4%) stemmed TSAs (P = .015). A ΔCOR ≥6.0 mm was measured in 0 of the 55 (0.0%) MPO TSAs compared to 2 of the 55 (3.6%) stemmed TSAs (P = .50) (Table 2).
Radiographic Measurement Data.
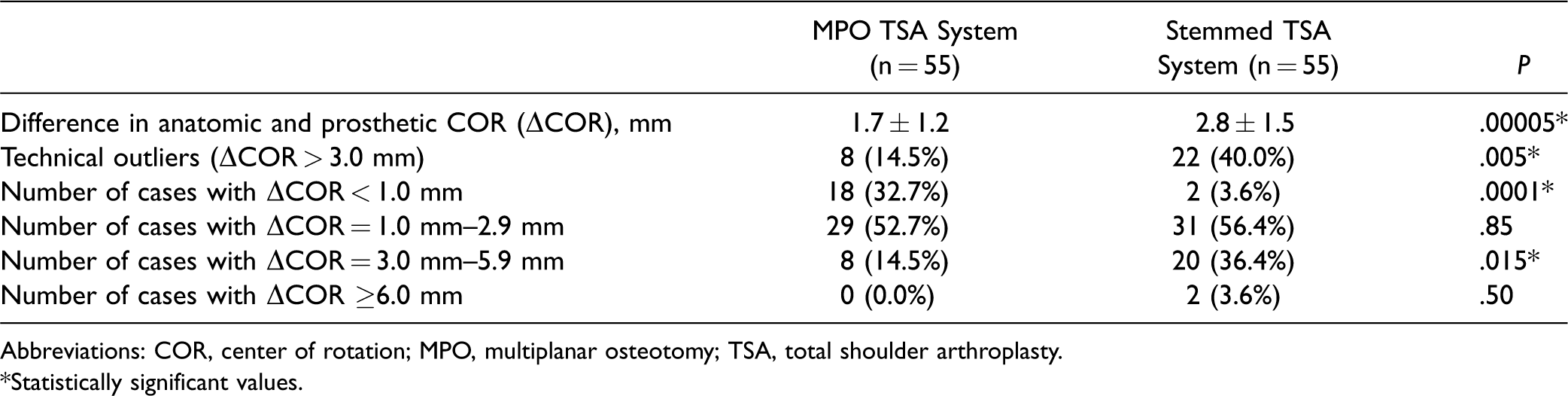
Abbreviations: COR, center of rotation; MPO, multiplanar osteotomy; TSA, total shoulder arthroplasty.*Statistically significant values.
The incidence of clinically significant outliers was lower in the MPO group compared to the stemmed TSA group; 22 of the 55 (40.0%) stemmed TSA cases were outliers, whereas only 8 of the 55 (14.5%) MPO cases were outliers (P = .005) (Figure 3). After allowing for a learning curve of 35 cases using the MPO arthroplasty system, the incidence of clinically significant outliers was 5%, with only 1 clinically significant outlier in the latter 20 cases in this series.
Discussion
Accurate anatomic restoration of the COR of the proximal humerus and maintaining normal glenohumeral relationships is of primary importance in anatomic TSA.24–26 Previous biomechanical studies in both cadaveric and computer models have found that a ΔCOR of as little as 3.0 mm can adversely affect shoulder biomechanics and kinematics.5,6,8–10 The proximal humerus is a complex 3-dimensional structure, and the disease process of glenohumeral joint osteoarthritis further alters this already complex anatomy. Although most shoulder arthroplasty surgeons make every attempt to anatomically reconstruct the humerus, the ability to consistently achieve this goal is limited by the technology currently available.
In an attempt to assist surgeons with anatomic glenohumeral joint reconstruction, third- and fourth-generation stemmed humeral arthroplasty systems provide more options to accommodate greater variability than earlier designs. Unfortunately, these systems are still limited by the relationship of the canal to the articular surface. In a computer model, Pearl et al. demonstrated that most implant systems can restore COR to within 4.0 mm, but extreme head offset can be difficult to reconstruct with currently available prostheses. 15 Alolabi et al. demonstrated that even experienced surgeons were unable to reproduce the COR of the humerus to within 3.0 mm in 31% of cases using modern stemmed implants. 11 In that same study resurfacing systems were found to be even less accurate, with 65% failing to restore the COR within 3.0 mm. 11 The authors found this was mostly due to overstuffing, which may be attributed to an inability to measure the thickness of bone removed during humeral head preparation.
The MPO system was designed to more accurately restore the anatomy of the glenohumeral joint and minimize the variability documented in recent studies of stemmed arthroplasty components. A cadaveric study validating the surgical technique of the MPO arthroplasty system measured numerous points of the humeral head in 3 dimensions before and after the surgical procedure, in an attempt to measure any change in the exact location of the articular surface. 19 The average change in position of each point was below 1.0 mm, and no point deviated from its original location by more than 3.0 mm. The present study was an attempt to demonstrate a similar capacity to improve a surgeon’s ability for precise anatomic reconstruction in clinical practice.
The results of the current analysis show that both the stemmed and MPO arthroplasty systems consistently restored the COR of the humeral head to within 3.0 mm. However, the canal-sparing, MPO system afforded the surgeon a slight increase in accuracy and a considerable decrease in variability in restoring the native anatomy of the proximal humerus. The reduced variability seen in the MPO group, as demonstrated by a significant reduction in the number of clinically significant outliers compared to the stemmed group, is particularly noteworthy. This improved precision was seen early in the use of the MPO arthroplasty system with a 2.5-fold reduction in the number of clinically significant outliers overall (14.5% vs 40.0%, P = 0.005) and a significantly higher percentage of cases with a ΔCOR of ≤1.0 mm (32.7% vs 3.6%, P = .0001). This low rate of clinically significant outliers in the MPO group stands in contrast to the 40% incidence of outliers reported when using a stemmed arthroplasty system, a system that the performing surgeon had multiple years of experience with. This high incidence of outliers is not unique to this series; results from other groups demonstrate a similar incidence of clinically significant outliers when using modern stemmed arthroplasty systems. 11
The design of this system borrows principles from the field of total knee arthroplasty. Most modern total knee arthroplasty systems use instrumentation to direct surgeons in the distal femoral and proximal tibial bone resections. Knee replacement has evolved from freehand estimates of where to cut and where to place implants into higher precision instrument sets, with implant dimensions corresponding to the amount of bone resected by the cutting guides. This has resulted in a more standardized operation, and this concept was influential in the development of the instrumented MPO used in the shoulder arthroplasty system examined in the current study.
We believe that lower rate of clinically significant outliers in the MPO group is due, at least in part, to the application of these principles of calibrated resection and the use of cutting guides to assist the surgeon during the multiplanar humeral head osteotomies. The cutting guides and surgical technique were designed to match the amount of bone resected to the thickness of the humeral head implant and were specifically developed to address the problem of inconsistency in humeral head reconstruction. 27 Minimizing the amount of bony resection during humeral head preparation has a number of benefits including improved implant fixation in the dense subchondral bone of the anatomic neck 28 and preservation of proximal bone stock in the case of revision surgery. 19 Additionally, proper positioning of the implant at the native center of the articular surface is facilitated when there is no stem affecting placement.
Another factor that may have contributed to the lower number of clinically significant outliers in the MPO group is the nonspherical humeral head design. The native humeral articular surface is elliptical rather than spherical. 7 The humeral prosthesis used in the MPO system is designed to replicate this geometry with a radius of curvature in the anterior–posterior axis less than the curvature in the superior–inferior axis. Often with a spherical implant, a surgeon is forced to position the implant to fit the AP dimension of the humeral head, which leaves bone exposed inferior, or conversely position the implant to fit the superoinferior axis, which results in overhang either anterior or posterior. In biomechanical studies, nonspherical humeral prostheses have shown improved kinematics and behave more like native humeral heads.12,29 In addition to the nonspherical humeral head design, the concave undersurface of the implant in the MPO system consists of 4 converging planes that provide a natural resistance to torsion, along with 4 pegs for additional stability. The implant also has a curved border with similar surface area coverage to a traditional hemispherical implant but with reduced implant volume and weight.
Although the present study involved a single surgeon without randomization, we chose to use all eligible 110 consecutive operations over a 2-year period as we felt this would be the most objective method of directly comparing the 2 groups. The patients in the stemmed group represented the most recent 55 consecutive procedures using an arthroplasty system that the performing surgeon had multiple years of experience. A single type of implant system was chosen in order to create a homogenous study group and eliminate any variability in radiographic measurements that would be introduced by examining multiple stemmed arthroplasty systems. The patients included in the MPO group were the first 55 cases performed by the same surgeon after immediately transitioning to the MPO system. All 55 of the MPO group patients were included in the X-ray analysis (none were excluded or lost to follow-up), thus eliminating the potential to hide or eliminate certain outliers or exclude patients during a “learning curve” period.
Instability following implantation of the final humeral head prosthesis, defined as greater than 50% translation of the humeral head on the glenoid, was not encountered in any of the cases in this series. A commonly utilized technique to increase stability in this setting is to increase the size of the prosthetic humeral head. Increasing the size of the humeral head prosthesis would undoubtedly result in a change in the ΔCOR measurement. Theoretically, if the ΔCOR were zero and the shoulder were unstable, upsizing the humeral head prosthesis would increase the ΔCOR, resulting in a less anatomic reconstruction but a more stable shoulder. Conversely, if a shoulder were unstable because of an undersized prosthetic humeral head component, increasing the humeral head size could result in increased stability and a more anatomic reconstruction with a ΔCOR closer to zero. As there is currently not enough evidence on the long-term functional outcomes of TSA based on intraoperative stability assessment findings or radiographic parameters of anatomic reconstruction, surgeons should continue to use whichever method they are comfortable with to assess and address intraoperative shoulder instability regardless of the type of prosthesis used.
Additional limitations of this study include the inability to blind the independent observers performing the COR measurements to the type of prosthesis being measured, a single surgeon series with the surgeon who performed the procedures involved in the design of the prosthesis used in the study, the lack of randomization of prosthesis types, and reliance on 2-dimensional radiographs to determine 3-dimensional geometric parameters. Although the design surgeon was performing the surgeries in this study, he was not involved in the radiographic measurements or statistical analysis in an attempt to eliminate bias. With regard to the radiographic assessment, an even more accurate method of measuring COR could have used computed tomography scanning but would subject patients to additional radiation exposure, as this is not currently part of our treatment protocol. Furthermore, this study does not attempt to characterize glenoid morphology, analyze the position of the glenoid component, or determine an association between improved implant positioning and clinical outcome following TSA. Larger prospective series correlating the radiographic accuracy of component positioning with long-term radiographic, functional, and patient-reported outcome measures are needed to determine whether radiographic parameters can predict clinical outcome following TSA.
Conclusion
Using measurements of the anatomic COR of the proximal humerus as a reference, the MPO canal-sparing TSA system helped improve a surgeon’s ability to more accurately and reliably restore the anatomy of the proximal humerus compared to stemmed total arthroplasty, considerably reducing the number of clinically significant outliers. These results may be attributable to the MPO technique of humeral head preparation and the nonspherical humeral head design of this prosthesis.
Footnotes
Authors’ Note
This study was performed at Physicians Regional Medical Center – Pine Ridge, Naples, FL and at the NewYork-Presbyterian/Columbia University Medical Center, New York, NY.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Baranek, Dr. Trofa, and Dr. Levine declare no conflicts of interest. Dr. Goldberg reports personal fees and other from Catalyst OrthoScience, during the conduct of the study; personal fees and other from Catalyst OrthoScience, outside the submitted work. In addition, Dr. Goldberg has a patent US9814587 licensed to Catalyst OrthoScience, a patent US9289306 licensed to Catalyst OrthoScience, a patent USD735338 licensed to Catalyst OrthoScience, and a patent USD746989 licensed to Catalyst OrthoScience.
Ethical Approval
This study was approved by the performing hospitals institutional review board (1174942).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
