Abstract
Background
We report here on the results, stratified by diagnosis, of a multicenter prospective study by surgeons unaffiliated with the design team of reverse shoulder arthroplasty (RSA) performed using a lateralized glenosphere. We hypothesized that outcomes would be comparable to those reported previously.
Keywords
Introduction
Reverse shoulder arthroplasty (RSA) has revolutionized the treatment of patients with arthritis and rotator cuff insufficiency, and can be performed with implants that either medialize or lateralize the center of rotation, and achieve this on the glenoid side, the humeral side, or both. In particular, studies on the outcomes of RSA employing a lateralized glenosphere have consistently reported improved active external rotation (aER) and low rates of scapular notching, but many of these are the work of the design team.1–8 Furthermore, studies have demonstrated higher complication rates and inferior outcome when RSA is performed for revision of a failed prosthetic arthroplasty. The objective of this study is to report on the 2-year follow-up results of a 9-site multicenter prospective study of RSA using glenospheres with a lateralized center of rotation. The study purpose was to prospectively collect the results of RSA using an implant with a lateralized glenosphere performed by experienced nondesign surgeons and to stratify these results by preoperative diagnosis. The study hypothesis is that the outcomes and complication rates would be comparable to those reported previously by the design team. A secondary hypothesis is that RSA performed for revision of a failed prosthetic arthroplasty would have inferior results to primary RSA.
Methods
Patients older than 60 years with cuff tear arthropathy or glenohumeral arthritis with rotator cuff tear, rotator cuff tear with instability or escape and without glenohumeral arthritis, or a failed prosthetic shoulder arthroplasty who had failed conservative treatment and who were candidates for RSA were enrolled between December 2009 and July 2012 into this prospective clinical study at 1 of 9 sites, with participation at each site capped at 40 RSAs. All patients underwent RSA using a single implant design (DJO RSP, Austin, TX) characterized by a lateralized glenosphere. All prostheses were placed through a deltopectoral approach, and none were implanted for osteoarthritis without rotator cuff tear or for proximal humerus fracture. All humeral components employed in the study were of the original design featuring a modular humeral socket and stem and were inserted with cement. The glenoid baseplate was typically placed at the glenoid center or slightly inferior, but this was not standardized. The subscapularis tendon was taken down by peel, tenotomy, or osteotomy, according to surgeon preference and typically repaired whenever possible, although this too was not standardized. The postoperative rehabilitation protocol was not standardized and was at the discretion of the individual surgeon. Institutional Review Board approval was obtained at all sites.
Patients underwent clinical and radiographic evaluation preoperatively and at 3 months, 6 months, 1 year, and 2 years postoperatively. Clinical examination at each time point included goniometeric measurement of both active and passive range of motion comprising forward flexion (FF), abduction (Ab), external rotation (ER), in degrees, and internal rotation to the back, measured as the highest spinous process attained at midline with the extended thumb. Internal rotation measurements were transformed to a 10-point scale, as described previously 7 (Table 1).
Internal Rotation to the Back Transformed for Constant Score.

aGreater trochanter.
bIschial tuberosity.
cSpinous process.
Radiographic assessment included at each time point a minimum of 2 biplanar radiographs comprising true AP with the arm in ER (Grashey) and axillary-lateral views. Radiographs were reviewed for dislocation, component dissociation, periprosthetic fracture, acromion and scapular spine fracture, as well as for component subsidence and loosening. Advanced imaging such as computed tomography to diagnose these complications was obtained at each surgeon’s discretion. In addition, plain radiographs at 24 months were inspected for scapular notching by an independent reviewer who has not involved in patient care, and scapular notching was subsequently quantified according to the method of Nerot.9,10
Patient recorded outcome measures included Simple Shoulder Test (SST) and American Shoulder and Elbow Surgeons (ASES) scores, obtained at each time point. Pain was assessed at each time point using a 10-point visual analog scale (VAS-pain).
Postoperative complications were recorded and tabulated. These included acromion fracture, scapular spine fracture, prosthetic dislocation, component dissociation, deep infection, nerve injury, and death. In addition, the timing and details of any revision surgery including component revision were carefully recorded.
Statistical Analysis
Data from the 9 individual sites were collected and analyzed independently by a biostatistician. Range of motion, SST, ASES, and VAS-pain scores were compared across the various time points. Statistical analysis was performed on data pooled across study centers using the SPSS® Statistical Software version 25 (Cary, NC). Statistical significance was determined using an alpha of .05; P values are provided in the corresponding tables. Descriptive statistics were used to summarize demographic and baseline data; means with standard deviations and were used for continuous data. Frequency and proportions were reported for categorical data, where appropriate.
Primary end points were stratified across diagnoses, including cuff tear arthropathy, rotator cuff tear with instability or escape, and failed prosthetic shoulder arthroplasty. For each end point and diagnosis, means and standard deviation are presented for baseline and 2-year follow-up scores, along with improvement (change from baseline) scores. Improvement scores were reported with 95% confidence intervals and P values for the resulting paired test statistic.
Results
Two hundred forty-five patients (138 females and 107 males) underwent RSA. All patients were older than 60 years and mean age was 73.1 years (standard deviation 7.6, range 60–92) at the time of surgery. One hundred sixty-three RSAs were performed for cuff tear arthropathy or glenohumeral arthritis with rotator cuff tear, 62 RSAs for rotator cuff tear with instability or escape and without glenohumeral arthritis, and 20 RSAs as a revision. Patient demographics are summarized in Table 2. At 2-year follow-up, 23 patients were deceased and 12 patients had undergone component revision. Thirty-seven patients were unwilling to return or lost to follow-up, despite multiple attempts to contact these patients by both telephone and registered mail. This left 173 patients (71%) available for study at 2-year follow-up.
Patient Demographics.
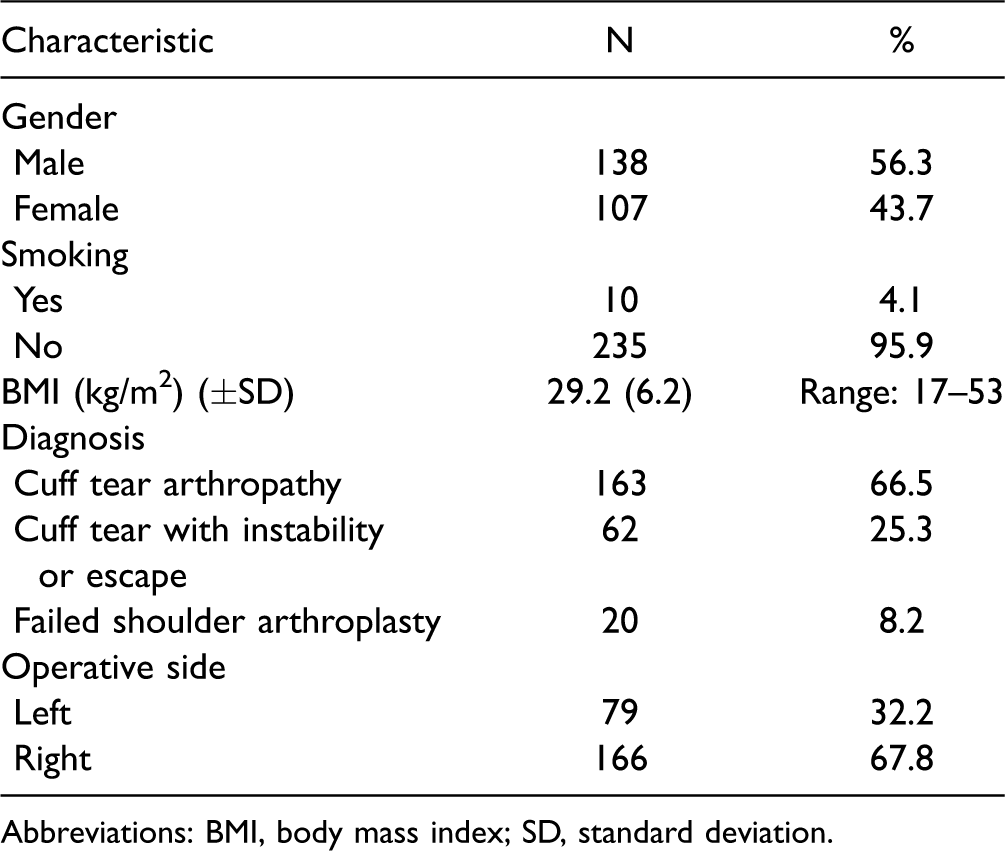
Abbreviations: BMI, body mass index; SD, standard deviation.
Significant improvements of all measured active motions were observed (Figure 1, Table 3). Mean preoperative active forward flexion (aFF) improved from 73° ± 38° to 102° ± 29° at 3 months, 114° ± 27° at 6 months, 121° ± 27° at 1 year, and 127° ± 29° at 2-year follow-up (P < .0001 for all time points compared with preoperative). Active abduction (aAB) improved from 65° ± 30° to 88° ± 25° at 3 months, 97° ± 24° at 6 months, 102° ± 26° at 1 year, and 109° ± 27° at 2-year follow-up (P < .0001 for all postoperative time points). aER at the side improved from 24° ± 21° to 29° ± 16° at 3 months, 33° ± 15° at 3 months, 37° ± 16° at 1 year, and 37° ± 18° at 2 years (P < .0001 for all time points); and active internal rotation (aIR) from the back improved modestly from the buttock to the L5 spinous process at 2-year follow-up (P < .05). Patients with the greatest preoperative aER deficits demonstrated the greatest gains postoperatively. Fifteen of 17 patients with preoperative aER < 0° were evaluated at 2 years; of these, 14 had aER ≥0° at 2 years with a mean improvement of 42°. Improvements in passive motions were also observed (Figure 1).

Active and passive shoulder range of motion over time. (A) Forward flexion, (B) abduction, (C) external rotation, and (D) internal rotation to the back (2 = thigh, 4 = buttock, 6 = sacrum, 8 = T12 spinous process, 10 = T7 spinous process).
Outcomes at Baseline, 2-Year Postoperative, and Change From Baseline.

Abbreviations: LOS, length of stay; SD, standard deviation; VAS, visual analog scale.
Values listed represent mean ± SD.
Patient reported outcome measures also improved. Mean SST improved from 3.2 ± 2.2 (out of 12), preoperatively, to 5.8 ± 3.1 at 3 months, 7.5 ± 3.0 at 6 months, 8.0 ± 3.1 at 1 years, and 8.5 ± 2.9 at 2 years (P < .0001 for all time points compared with preoperative) (Figure 2). Mean ASES score improved from 45 ± 19, preoperatively, to 73 ± 20 at 3 months, 80 ± 18 at 6 months, 83 ± 17 at 1 year, and 86 ± 16 at 2-year follow-up (P < .0001 for all time points; Figure 3). In addition, mean 10-point VAS pain scores declined from 6.3 ± 2.6, preoperatively, to 1.4 ± 2.0 at 2-year follow-up (Figure 4). These findings are summarized in Table 3.

SST responses over time. SST, Simple Shoulder Test.
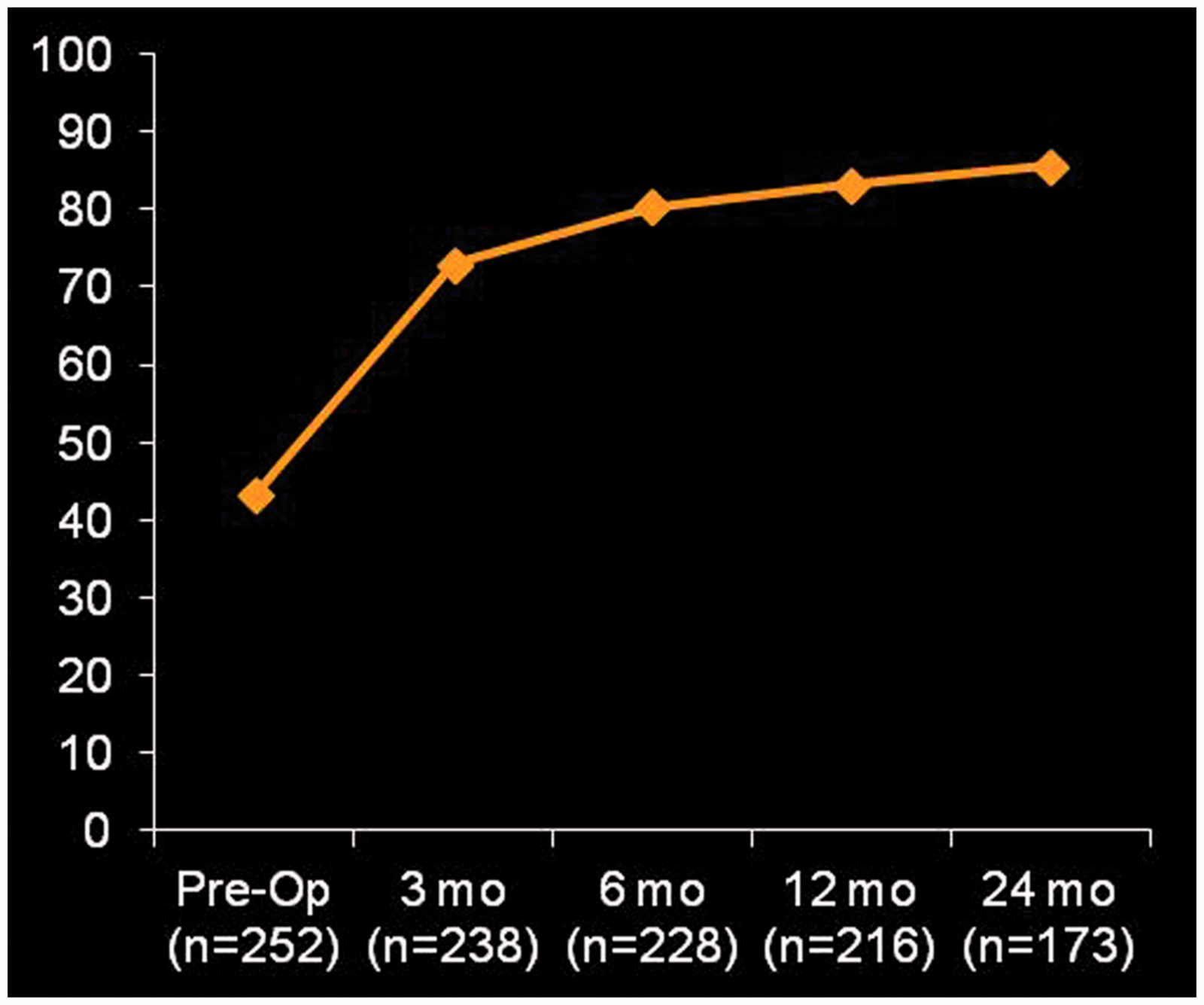
ASES scores over time. ASES, American Shoulder and Elbow Surgeons.
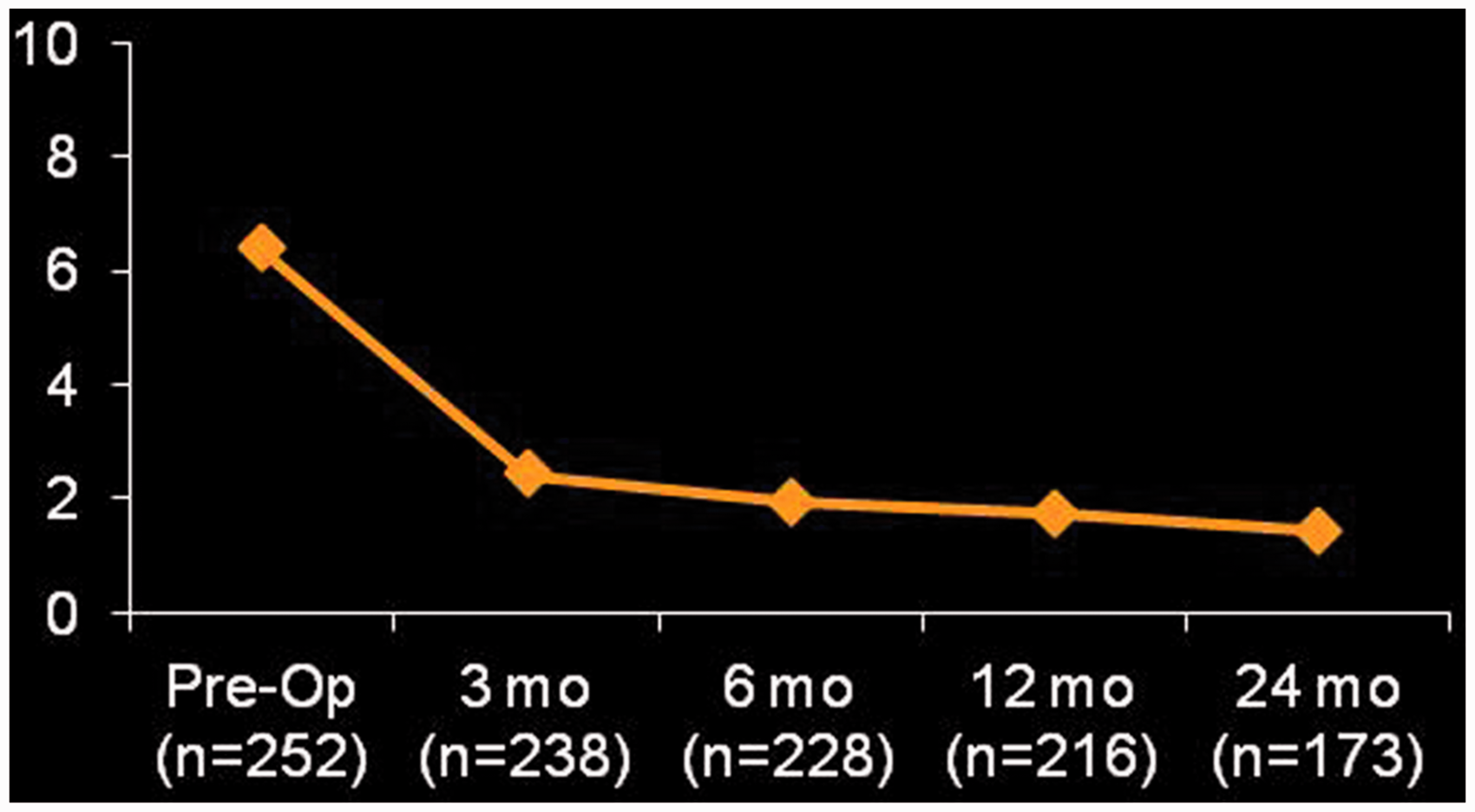
Pain levels (represented as a 10-point VAS) over time. VAS, visual analog scale.
Improvements in active range of motion and patient reported outcome measures (PROMs) between preoperative and 2-year follow-up, stratified by diagnosis, are summarized in Table 4. With the numbers available, active internal rotation did not improve significantly when stratified by diagnosis. In addition, less consistent improvements were observed for patients undergoing RSA for a failed arthroplasty. For example, aER did not improve in these patients. In contrast, the improvement in aFF (63° ± 28°) for patients undergoing RSA for failed arthroplasty was greater than that observed for cuff tear arthropathy (56° ± 43°) or rotator cuff tear with instability or escape (43° ± 49°).
Clinical Outcomes, Stratified by Diagnosis.

Abbreviations: aAB, active abduction; aER, active external rotation; aFF, active forward flexion; aIR, active internal rotation; ASES, American Shoulder and Elbow Surgeons; SST, simple shoulder test; VAS, visual analog scale.
Statistically significant improvements in active range of motion and self-assessed outcome scores were also observed from one time point to the next, through the 1-year follow-up. These improvements are depicted in Table 5 for aFF, aAB, aER, aIR, as well SST, ASES scores, and VAS pain scores.
Level of Significance for Improvements Between Consecutive Time Points in Active Range of Motion and Self-assessed Outcomes Scores.

Abbreviations: aAB, active abduction; aER, active external rotation; aFF, active forward flexion; aIR, active internal rotation; ASES, American Shoulder and Elbow Surgeons; NS, not significant; SST, simple shoulder test; VAS, visual analog scale.
All complications arising following the 245 RSAs were recorded. These included 16 scapular fractures (6.5%), including 9 acromial stress fractures (3.7%) and 7 scapular spine or body stress fractures (2.9%), 7 dislocations (2.9%), 1 component dissociation, and 2 prosthetic joint infections. Twelve complications led to component revision during the first 2 years (4.9%). Two patients required revision surgery for infection at 7 months and 2 years postoperatively. Five patients underwent revision for postoperative instability after RSA including 1 patient requiring 3 revisions. One patient experienced humeral socket dissociation requiring revision to replace the socket, and 2 patients underwent component revision for sequelae of a scapular spine fracture but without internal fixation of the scapula. The distribution of complications is summarized in Table 6.
Complications (and Incidence) Following Reverse Shoulder Arthroplasty.

Standardized radiographs, including Grashey view, were available for 165 of 173 patients at 2-year follow-up; 7 sets were inadequate for evaluation of scapular notching and 1 set could not be located. Scapular notching was observed in 22 of 165 shoulders (13.3%) at 24 months, and of these, 17 shoulders had grade 1 notching, 4 shoulders had grade 2 notching, and 1 shoulder had grade 3 notching.
Discussion
Modern reverse shoulder arthoplasty was pioneered by Grammont,11,12 who designed an implant characterized by a medial center of rotation to minimize shear forces at the interface between baseplate and glenoid, and a 150° humeral angle of inclination that lengthens the deltoid to improve its contribution to active motion. 13 Early results in patients with cuff tear arthropathy demonstrated improvements in active forward elevation following RSA employing a Grammont-type implant.10,14–17 However, concerns were raised regarding the high incidence of scapular notching, exceeding 60% in some early series,15,17 and its implications on long-term functional outcome. In addition, multiple studies have demonstrated that ER does not recover predictably when a Grammont-type implant is used.13,17,18
In 2005, Frankle and colleagues 2 reported on RSA performed using a lateralized glenosphere and a humeral implant with an inclination angle of 135°, with the aim of better replicating more normal anatomy and kinematics and avoiding impingement of the humeral component with the inferior glenoid. Gains in aFF were comparable to those reported for Grammont-type implants but there was as 11% incidence of baseplate failure. 2 Following important changes in implant design (introduction of 5.0 mm peripheral locking screws and HA coating) and surgical technique (avoidance of superior tilt), Frankle and colleagues reported excellent functional gains without baseplate failures. 1 They have documented substantial gains in aER1,2,4,7,8 and have also documented a very low incidence of scapular notching. Several early studies demonstrated a 0% incidence of scapular notching,1,2,4–6 although 2 more recent studies demonstrated scapular notching rates of 9%4–6 and 13.5%. 7 Finally, previous studies have also shown more modest clinical improvements and higher complication rates when reverse RSA is performed for failed arthroplasty.5,6,8
In recent years, there have been several studies by individual nondesign surgeons on the outcomes of RSA using an implant with a lateralized glenosphere. However, these have consisted mostly of retrospective reports, studies comparing RSA and anatomic total shoulder arthroplasty, or studies of surgical technique or complications.
Levy and Blum reported on the first 40 consecutive patients receiving the RSP prosthesis at a mean 18-month follow-up. 19 Complications were identified in 10 patients (25%), but revision surgery was needed in only 2 patients (5%). 19 However, initial and final clinical and self-assessed outcomes were not provided. Levy and colleagues have also published recent studies comparing functional internal rotation and speed of recovery following anatomic and reverse total shoulder arthroplasty using the RSP prosthesis, but long-term outcomes were not a focus of their studies.20,21 In a study of RSA using the RSP prosthesis, Clark and colleagues found that subscapularis repair did not influence postoperative range of motion; 22 at 12-month follow-up, aFF improved 56° in the nonrepair group and 54° in the repair group, 22 which are improvements comparable to those reported here.
Hasan and colleagues reported on the outcomes and effect of experience following the first 60 consecutive reverse shoulder arthroplasties with a lateralized glenosphere. 23 At minimum 2-year follow-up mean improvements in aFF, aAB, and aER were 65°, 54°, and 22°, respectively, and mean aIR improved from the buttock to the L3 spinous process (P < .0001 for all motions). Mean SST improved from 1.8 to 6.9 and mean final ASES score was 72, 23 which represents comparable gains in active range of motion and outcome scores to those reported here and previously by the design team. The gains in range of motion increased and the incidence of complications declined with experience.
In contrast to previous studies, this multi-center study confirms the effectiveness of RSA in improving shoulder mobility and self-assessed shoulder function in a wide range of patients. The ΔFF of 57° and ΔAB of 46° observed in this study between preoperative and 2-year follow-up are comparable to previously reported results for RSA using implants with both medialized and lateralized glenospheres.1,15,17 Improvements in aER were also observed in this study, and this has been related to the lateralized glenosphere design.24,25 The ΔER of 17° observed for patients with cuff tear arthropathy is comparable1,7 to that reported by the design team, but it is greater than that reported in early studies of Grammont-type prostheses employing a medialized glenosphere.14,17 Moreover, the ΔER for patients with less than 0° aER preoperatively was 42°. Tensioning of the residual rotator cuff and posterior deltoid may help explain the improvement in aER.
As demonstrated in Table 5, our study also supports the observation by Levy and colleagues 20 that patients experience false-plateaus in active range of motion following RSA because statistically significant incremental gains in active range of motion were observed for up to 1 year postoperatively.
This study also documented substantial improvements in PROMs, irrespective of preoperative diagnosis. The improvements of 42 points in ASES score for both patients with cuff tear arthropathy and rotator cuff tear with instability or escape are comparable to those reported by the design team, and the mean 2-year ASES score of 88 for patients with cuff tear arthropathy is among the highest reported for RSA. In addition, the ΔSST of 5.7 and 4.8 for patients with cuff tear arthropathy and rotator cuff tear with instability or escape, respectively, are also comparable to previous reports. In contrast, the mean improvements in ASES and SST were 23 points and 3.5 for patients undergoing RSA for a failed shoulder arthroplasty.
Collectively, the mean improvements in aFF, aAB, and aER observed at 2 years exceeded previously reported minimal clinically important differences (MCIDs) of 12° ± 4°, 7° ± 4° and 3° ± 2°, respectively, 26 for all preoperative diagnoses, with the exception of aER for patients undergoing RSA for a failed prosthesis. In addition, the improvements in this study for VAS-pain, ASES, and SST at 2 years also exceed previously published MCIDs26,27 of 1.4–1.6, 14–21, and 1.5–2.4, respectively, for all preoperative diagnoses.
This study also demonstrates that patients undergoing RSA for a failed prosthesis have inferior outcomes compared to the rest of study group. In particular, for RSA performed as a revision, we observed statistically significant improvements in aFF and aAB but no improvement in aER or aIR, with the limited numbers available. This echoes previous reports demonstrating more limited improvements when RSA is performed for revision of a failed total shoulder arthroplasty or hemiarthroplaty.5,6,8 Collectively, this information can help counsel patients preoperatively.
The complication rate at 2-year follow-up is substantially lower than those published in early reports on RSA, 28 but is higher than the 6.4% reported by the design team 1 for patients with cuff tear arthropathy. In addition, the 4.9% revision rate at 2 years is only slightly lower than the 6% reported by the design team at up to 12-year follow-up. 29 The rates of specific complications reported here reconcile with other previous studies: for example, the dislocation rate is comparable to that reported recently23,30,31 and the 1.6% rate of deep infection at 2 years is comparable to the 4% and 0.9% reported for 2 consecutive large patient series by Walch and colleagues 31 and the 0% and 3% reported by Nowinski and colleagues 32 for implants inserted with and without antibiotics-impregnated cement.
We observed a 6.5% incidence of scapular fracture, including both acromion and scapular spine fractures. This is lower than the 10.2% reported by Levy and colleagues 33 using the same implant system and higher than the 3.1% incidence reported recently by Frankle and colleagues. 34 Our observed incidence is comparable to the 5.5% incidence previously reported by Crosby and colleagues 35 and the 4.9% incidence reported by Hamid and colleagues 36 for Grammont-style prostheses. More recently, Neyton and colleagues reported a 1.3% incidence of scapular fracture a mean 97 months following RSA, also using a Grammont-style prosthesis. 37 However, 46% of patients were unavailable for follow-up, so the actual incidence may be higher. A recent report has demonstrated that scapular fractures can occur well beyond 2 years postoperatively, so our observed incidence may increase over time. 34 Recent studies33,38,39 have explored various patient, surgery, and implant factors to determine predictors of postoperative scapular fracture, but only osteoporosis has emerged as a risk factor. 33 Still, the nearly 10-fold variability in the published incidence of scapular fractures suggests that factors such as implant design, implant positioning, and soft tissue management play a role.
Finally, our 13.3% incidence of scapular notching is comparable to the 0% to 13.5% incidence observed in multiple reports by the design team and similar to the 11% observed by Hasan and colleagues. 23 A recent study has suggested that larger more lateralized glenospheres and placement of the humeral component to replicate native retroversion may help minimize scapular notching. 40
Limitations
This study has several limitations that warrant mention. Only 71% of the 245 patients enrolled in this study were available for 2-year follow-up. This percentage is lower than that for similar RSA studies that report the number of eligible or enrolled patients. Cuff and colleagues 1 noted that 96 of 114 RSAs (84.2%) performed by a single surgeon for rotator cuff deficiency were available for study at 2-year follow-up. Wall and colleagues 41 reported on 240 RSAs performed by 2 surgeons in patients with a mean age of 72.7 years. At mean follow-up of 39 months, 191 (79.6%) were available for study. In our study, nearly one half of the 29% (72 of 245) lost to follow-up were either deceased from causes unrelated to the surgery (23 of 245) or had undergone component revision (12 of 245) at 2-year follow-up. This left 37 of 245 (15.1%) patients who could not be reached despite multiple attempts, chose not to follow-up, or elected to withdraw from the study.
In addition, all participating surgeons are fellowship-trained and experienced in RSA using an implant with a lateralized glenosphere. As such, the results may not generalize to low volume RSA surgeons who are unfamiliar with this prosthesis. In addition, we report here on minimum 2-year follow-up, but as with all arthroplasty studies, longer follow-up will be essential to establishing long-term durability of this procedure and its outcomes. Study strengths include prospective and standardized data collection and its balanced multi-center nature, with 6 sites contributing greater than 30 patients.
Conclusion
RSA with a lateralized glenosphere improves active shoulder motion and self-assessed function in carefully selected patients with cuff tear arthropathy and/or pseudoparalysis, or a failed prosthetic shoulder arthroplasty. The overall complication rate is not dramatically different from that reported by the design team, although acromion and scapular spine fractures warrant additional analysis to determine causative factors and preventive strategies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Several of the authors are consultants for DJO Global.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by DJO Global, Inc.
