Abstract
Introduction
Radiocapitellar joint load transfer is poorly understood for both the native elbow and following radial head replacement and lateral collateral ligament (LCL) repair. Radial head arthroplasty is commonly performed in the setting of comminuted radial head fractures. 1 Determining the correct implant length can be challenging when dealing with fracture comminution or delayed reconstruction 2 but is important to restore normal elbow kinematics.3–6 Previous studies have shown that lengthening (“overstuffing”) or shortening (“understuffing”) the radius by 2.5 mm alters elbow kinematics in elbows with the medial collateral ligament intact3,4 or deficient. 5 Overstuffing the radiocapitellar joint has been reported to lead to accelerated cartilage wear and capitellar erosion1,7 as well as decreased elbow flexion. 6 Understuffing could lead to increased load transfer through the ulnohumeral joint, with subsequent degenerative changes. 1
Previous studies have attempted to quantify the change in load through the radiocapitellar joint with lengthening and shortening of the proximal radius. Van Glabbeek et al. quantified native radiocapitellar joint forces using an interpositional load sensor during static loading of the elbow and found a significant increase with 2.5 mm lengthening of the radial neck.3,5 Lanting et al. also reported significant changes in radiocapitellar joint contact pressure with an interpositional load sensor with 4 mm of radial head implant lengthening or shortening but not with 2 mm lengthening or shortening. 8 Cohn et al. reported that as much as 2 mm of lengthening or shortening of the radius could be tolerated without significant changes in radiocapitellar joint contact pressure. 4 To our knowledge, no study has quantified proximal radial loading without disrupting the soft tissues of the radiocapitellar joint.
To date, studies of radiocapitellar joint loading have been limited to static elbow positions and limited orientations, which likely do not accurately reflect loading in the in vivo setting. The effect of arm position on radial head load transfer has important clinical implications following radial head injuries and repair. An improved understanding of these loads will inform changes in rehabilitation protocols, which may lead to better patient outcomes.
Concomitant LCL injury is frequently found with comminuted radial head fractures requiring arthroplasty. 9 LCL injury has been shown to alter elbow kinematics and stability, which are restored following a correctly tensioned anatomic LCL repair. 10 The effects of LCL injury and repair on proximal radius loading have not been established both in the setting of the native elbow and following changes in radial length.
In view of the foregoing, the purpose of this study was to determine the effects of radial length, arm position, and LCL repair on proximal radial loading. Our hypotheses were that increased radial length and the valgus arm position will result in increased load, and that this effect would be the same with the LCL intact and after LCL injury and repair.
Methods
This in vitro study used 8 fresh, previously frozen cadaveric arms (60.9 ± 18.2 years, 8 men) and a previously described elbow motion simulator. 11 A computed tomography (CT) scan (GE Lightspeed VCT 64 slice CT scanner; General Electric, New Berlin, WI) of each arm was performed to screen for deformity, previous trauma, or arthritis.
Specimen Preparation
The arms were mounted on an elbow motion simulator with the humerus rigidly clamped and the elbow and forearm soft tissues unaltered, as previously reported. 11 A posterior longitudinal skin incision with full thickness skin flaps was utilized for specimen preparation. Biceps, brachialis, and triceps were attached to servomotors via 1/8″ braided Dacron® cable, woven into the respective tendons using a locking suture pattern. A second longitudinal incision along the radial aspect of the forearm was utilized to access the brachioradialis tendon. These tendons were attached to pneumatic actuators with number 5 braided polyester suture (Ethibond Excel; Ethicon, Somerville, NJ), routing the sutures through a nylon sleeve attached to the lateral supracondylar ridge. A Steinmann pin was placed retrograde from the long finger metacarpal into the distal radius to stabilize the wrist in neutral position. An electromagnetic tracking system (TrakSTAR; Ascension Technology Corporation, Burlington, VT) was used to quantify elbow motion with sensors attached to the radius and ulna.
A custom, modular load sensing device 12 with an intercalary load cell (Subminiature Model 11; Honeywell, Morristown, NJ) was used to measure axial load through the proximal radius (Figure 1). This consisted of proximal and distal stems, the load cell, and modular spacers, which allowed testing with multiple radius lengths (−4, −2, 0, +2, and +4 mm). The device was implanted in the radial neck through a common extensor tendon splitting approach just distal to the radial head allowing preservation of the annular ligament. A “bone-bridge” technique 13 was used to maintain the anatomic alignment of the proximal radius. A cortical window, to accommodate implant insertion, was opened in the radial neck by removing the lateral half of the bone centered between the radial head and biceps tuberosity. Curettes were used to open the proximal and distal canals. The proximal and distal stems were then cemented into the radius. Prior to the cement hardening, the stems were connected with a temporary spacer block matching the dimensions of the load cell with the 0 mm spacer, ensuring the correct alignment of the stems. Once the cement was fully cured, the temporary spacer block was removed, and the osteotomy of the radial neck was completed by removing a 5 mm section of the medial cortex proximally, preserving the biceps tuberosity distally. The load cell with 0 mm spacer was then connected to the proximal and distal stems, restoring the anatomic alignment of the proximal radius. The skin incisions were closed to restore the normal soft tissue envelope and to ensure the specimen remained hydrated.
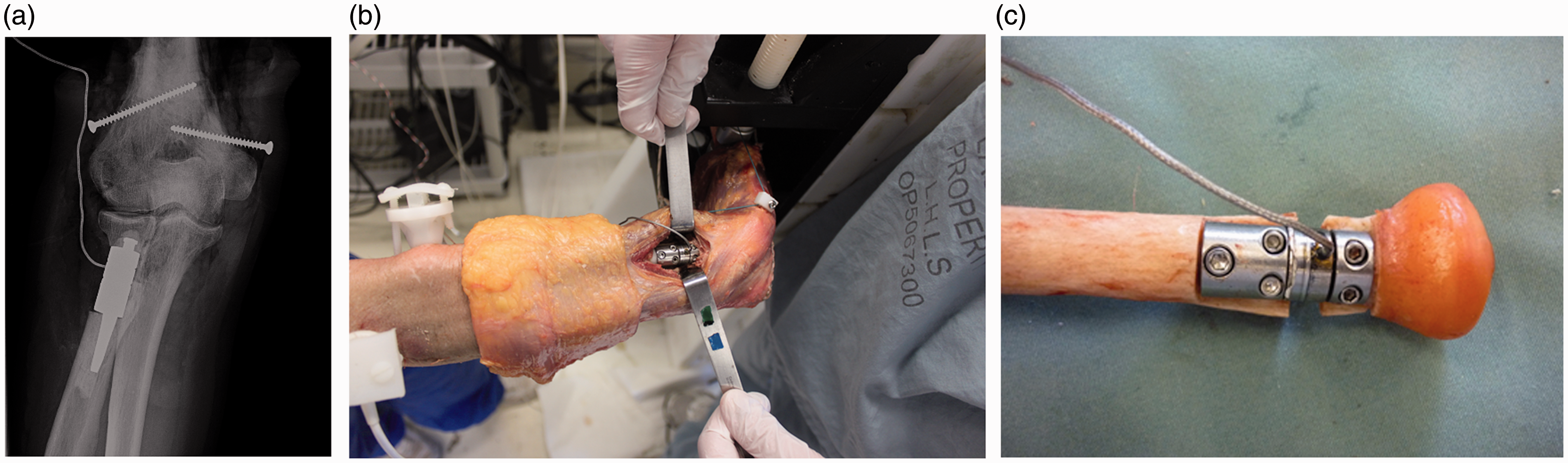
A, A radiograph of the cemented load sensing device implanted in the radial neck between the annular ligament and biceps tuberosity. B, Lateral view of the installed load sensing device in the radial neck. C, Explanted modular load sensing apparatus.
Testing Protocol
Radial neck load was measured during simulated active elbow flexion (at 3 deg/s) with the arm (humerus) in the vertical (gravity dependent), varus, and valgus positions, with the forearm supinated. Active motion was achieved using a custom control system that employs a combination of motors and actuators to produce low-velocity flexion of the elbow, as described in previous studies. 11 The arm was tested first with the 0 mm spacer (native length), then with the −4, −2, +2, and +4 mm spacers in random order. For each testing state, 2 flexion trials were performed, and the load data for analysis were captured from the second trial.
LCL injury was simulated by surgical release from the lateral epicondyle. The ligament was exposed through Kocher’s interval and released sharply off bone, along with a portion of the common extensor origin to allow exposure for repair and to create a more realistic injury pattern. 14 Transosseous ligament repair was performed with a #2 Fiberwire (Arthrex, Naples, Florida) woven through the LCL in locking Krackow fashion. The sutures were passed through a drill hole at the isometric point, exiting through drill holes anterior and posterior to the supracondylar ridge. The sutures were tensioned to 20 N with an actuator and then clamped to maintain that pretensioned length. 10 Kocher’s interval was closed with 0 suture (Ethibond Excel; Ethicon). The same testing protocol was repeated for the LCL repaired state.
Statistical Analysis
The dependent variable of maximum axial load in the proximal radius throughout flexion was compared with arm position, radial neck length, and LCL state using a 3-way repeated-measures analysis of variance (SPSS Statistics, Version 21.0; IBM, Armonk, NY). Pairwise comparison of means was carried out for any factor found to have a statistically significant effect (P < .05). The maximum axial load is defined as the maximum compressive force through flexion for each trial. One outlier was identified by our statistical software in 1 specimen for 1 trial so this data point was excluded from the analysis.
Results
Effect of Radial Length
In the vertical position, the maximum radial load throughout flexion increased with greater radial length, with the LCL intact and repaired (Table 1; Figure 1). With the LCL intact, increasing radial length from 0 mm to +2 mm increased radial load by 66% (27.6 ± 10.5 N vs 54.8 ± 12.2 N, P < .001), and +4 mm increased load 106% (27.6 ± 10.5 N vs 89.4 ± 17.0 N, P < .001). For the −2 and −4 mm radial lengths, negative loads were seen (−4.8 ± 7.0 N and −20.6 ± 19.1 N, respectively), representing tension on the radial neck. The difference between −2 and −4 mm was not statistically significant (P = .06); however, both lengths produced significantly lower loads compared to 0, +2, and +4 mm (P < .001). Similar results were seen following LCL sectioning and repair (P < .001 for both).
Maximum Compressive Radial Load (N ± SD).
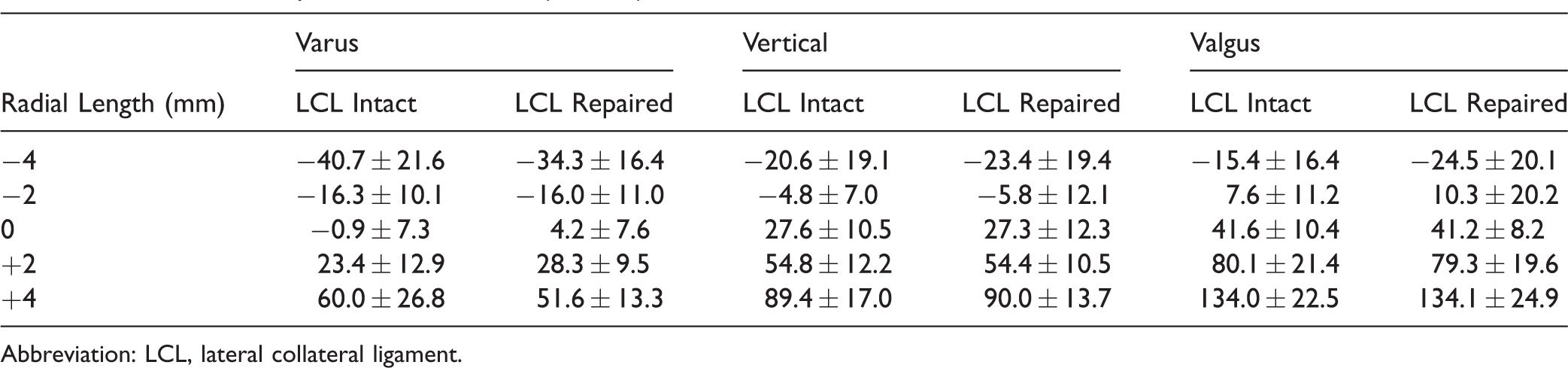
Abbreviation: LCL, lateral collateral ligament.
In the valgus position, there was a comparable pattern of increased radial load with each increase in radial length, for both LCL intact and following repair (P < .001; Figure 2). With LCL intact, increased radial lengths of +2 and +4 mm resulted in 63% and 105% increases in radial load, respectively. Negative loads (tension) were measured with the −2 and −4 mm radial lengths. Radial loads were highest with the arm in the valgus position (P < .001).

Maximum (± 1 SD) radial load throughout flexion with the arm in the vertical position. Radial loads increased with increasing radial neck length (P < .001). Compression forces are positive, and tension forces are negative. LCL, lateral collateral ligament.
In the varus position, there were significant increases in radial load with each increase in radial length, both with LCL intact and repaired (P < .001; Figure 3). In both LCL intact and LCL repaired states, negative loads were found in the 0 mm length, as well as −2 and −4 mm. Radial loads in the varus position were lower than the vertical position (P < .001; Figure 4).

Maximum (± 1 SD) of radial loads with the arm in the valgus position. Radial loads were increased with radial neck length (P < .001). Compression forces are positive, and tension forces are negative. LCL, lateral collateral ligament.

Maximum (± 1 SD) of radial loads with the arm in the varus position. Radial loads increased with greater radial neck length (P < .001). Compression forces are positive and tension forces are negative. LCL, lateral collateral ligament.
LCL Injury and Repair
Overall, there was no significant difference in radial loads between LCL intact and LCL repaired (P = .816). For the −2, 0, and +2 mm radial lengths, the greatest difference between LCL states was 9.1 N (−4 mm, valgus). In varus with the −4 mm length, loads were −40.7 ± 21.6 N and −34.3 ± 16.4 N for LCL intact and repaired, respectively. For +4 mm, loads were 60.0 ± 26.8 N (intact) and 51.6 ± 13.3 N (repaired). In valgus, an opposite pattern was found. For the −4 mm length, loads were −15.4 ± 16.4 N and −24.5 ± 20.1 N for LCL intact and repaired, respectively. For +4 mm, loads were 134.0 ± 22.5 N and 134.1 ± 24.9 N, respectively.
For both the LCL intact and LCL repaired cases, linear relationships between loads and radial length were found using the average force values from Table 1. The coefficients and P values for these relationships can be found in Table 2.
Linear Force Relationships (Force = A[N/mm] × (Radial Length [mm]) + B[N]).

Abbreviation: LCL, lateral collateral ligament.
Arm Position
Significantly greater mean proximal radius loads were seen with the arm in the valgus position compared to vertical and varus (50.8 ± 62.6 N, 28.5 ± 43.1 N, and 5.9 ± 35.7 N, respectively, P < .001; LCL intact and repaired results combined). Distraction forces were common with the arm in the varus position, even with the radius at its native length.
Discussion
This study evaluated the effects of radial length, LCL repair, and arm position on load through the proximal radius. To our knowledge, this is the first study to investigate this with an active motion elbow simulator, which may reflect in vivo conditions more accurately than static testing methods. The results have important implications for radial head arthroplasty, LCL repair, and rehabilitation.
Determining the correct radial length during radial head arthroplasty for fracture can be challenging 2 but is important to reestablish the normal kinematics of the joint. 3 This study demonstrates that the correct radial head length is also important in restoring the normal load through the proximal radius, which should reflect loading through the radiocapitellar joint. Increasing radial length by 2 mm resulted in a 66% increase in load in the vertical position with the LCL intact, while 4 mm lengthening increased load by almost 106%. The same trend was seen in all arm positions and with the LCL intact and repaired. These findings are in keeping with those of 2 previous studies, which both used pressure sensitive film implanted in the radiocapitellar joint under static loading.3,8 Van Glabbeek et al. found radiocapitellar pressure up to nearly 4 times normal with 2.5 mm lengthening in a medial collateral ligament deficient specimen when using the native radial head. Lanting et al. used a radial head implant and found no significant difference in radiocapitellar contact pressure at 2 mm lengthening; however, with a 4 mm lengthening, the forces were nearly doubled. The increased load through the radiocapitellar joint could result in accelerated wear and erosion of capitellum cartilage, with subsequent lateral elbow pain and arthritis.
With radial shortening of 2 mm or greater, tensile loads were found in all arm positions. Presumably, this means load is not being transferred across the radiocapitellar joint, and greater load is being borne by the ulnohumeral joint. One of the goals in performing radial head arthroplasty for nonreconstructible fractures is to restore the normal load distribution between the radiocapitellar and ulnohumeral joints, thus minimizing the ulnohumeral degenerative changes seen with long-term follow-up after radial head excision. 15 By shortening the radius as little as 2 mm, this potential benefit of radial head arthroplasty may be eliminated. 15 These data underscore the importance of accurately restoring radial length when performing a radial head arthroplasty.
It was interesting to observe that there was a rather strong correlation between radial length and load. This suggests that there is a linear response in stiffness of the radius as the height is changed likely due to surrounding soft tissues at the articulations and the intraosseous membrane. In addition, testing was performed in a continuous manner not allowing for the neo-Hookean properties of the cartilage to take effect. Our data suggest that the change in radial load is in the range of 10 to 20 N/mm of length change.
Our results also support the effectiveness of the LCL repair technique to restore the normal load transfer through the proximal radius. A previous study has shown that this repair technique recreates the normal kinematics of the elbow 10 ; however, no study has looked at restoration of load transfer. In our study, there was no significant difference in proximal radius load between the LCL intact and repaired states overall for any of the positions. No significant difference between LCL intact and repaired states indicates restoration of normal soft tissue tension across the lateral side of the elbow. With the arm in varus, sectioning and repairing the LCL had a small mitigating effect of radial length changes on proximal radius loading: Lower tensile loads were found with −4 mm, and lower compressive loads were found with +4 mm. This was an expected finding, as presumably the LCL was repaired in a lengthened state as the joint may not have been as well reduced with the varus load while the ligament was being tensioned. Conversely, with the arm in valgus, LCL repair seemed to accentuate the effects of increased radial length, likely due to the gravity compressing the radiocapitellar articulation during ligament tensioning. Overall though, these effects are not statistically significant and likely not clinically important (P = .816).
Another important outcome of this study is the finding of tensile loads through the proximal radius with the arm in varus with the native radial length. This is important with respect to radial head arthroplasty design and fixation. With contemporary modular designs, tensile loads on the radial head, in the setting of a fixed stem, could conceivably cause stem loosening or even dissociation of head–stem junctions. For press fit stems relying on bony ingrowth for definitive fixation, tensile forces in the early postoperative period could increase micromotion of the stem enough to cause fibrous fixation rather than bony ingrowth. Thus, for ingrowth implants, avoidance of varus positioning in the early rehabilitation period should be a consideration for all patients following uncemented ingrowth radial head arthroplasty not just those with concomitant LCL injuries. Tensile forces acting at the radial neck may explain the frequent disassociation of a bipolar radial head implant reported in patients with a total elbow arthroplasty. 16
Strengths of this study include the use of an active motion simulator, which likely more closely replicates in vivo conditions more accurately than static simulators. Additionally, we quantified radial loading throughout elbow flexion with mean values reported herein. Similarly, our test arms recreated the in vivo environment as closely as possible: muscles and ligaments were left intact, with all surgical approaches utilizing muscle-splitting approaches. A noninvasive load measurement technique was utilized avoiding issues with interpositional force sensors in the radiocapitellar joint altering load measurements by virtue of their thickness as well as the need to section soft tissues to insert them. Furthermore, the native radial head position was maintained by using a bone-bridge technique during insertion of the radial neck implant.
Using the native radial head instead of an arthroplasty, implant creates both strengths and weaknesses. Issues surrounding variation in component positioning, design and fixation were avoided by using the native radial head. In using any given implant design, results of a study may not be generalizable to other implant designs. With the native radial head, we could reliably reproduce the normal position and therefore accurately induce states of overlengthening and underlengthening. However, by using the native radial head, our results may not accurately reflect the forces acting on radial head implants when used clinically.
Another weakness of the study is that we measured force through the radial neck instead of the radiocapitellar joint. It is reasonable to expect that compressive load through the radial neck is very closely related to, if not the same as, compressive load through the radiocapitellar joint, since the surrounding structures (the proximal radioulnar joint and annular ligament) are unlikely to affect compressive loads. The tensile forces measured at the radial neck were likely induced by the annular ligament restraining the radial head and/or shear resistance at the proximal radioulnar joint articulation. These tensile forces are likely different than those seen acting at the radiocapitellar articulation.
This study further demonstrates the deleterious effects of overlengthening the radius, with significant increases in load with 2 mm or more of overlengthening, potentially leading to capitellar cartilage wear and erosions. It also shows that underlengthening the radius has repercussions: significant increases in tensile loads could affect implant fixation and failure, and failure to recreate normal load transfer across the radiocapitellar joint may lead to an increase in ulnohumeral arthritis. These adverse effects persist despite LCL injury and repair. Accurate restoration of radial length, with or without LCL injury requiring repair, can successfully restore normal load transfer through the radius and should be the goal in patients undergoing radial head arthroplasty.
Conclusions
The results of this study have shown that restoration of correct radial length during radial head arthroplasty is likely important to restore physiologic loading. Overlengthening or underlengthening a radial head implant can cause increased wear of the capitellum cartilage and tensile forces in the radial neck potentially affecting fixation of radial implants. This is in agreement with previous findings.2,3,5,8 Distraction forces occur in the radial neck with the arm in varus, suggesting that this position should be avoided postoperatively when using uncemented press fit implants as it may cause loosening and disassociation of fixed stem bipolar implants.
Footnotes
Acknowledgment
The authors thank Mark Welsh for his technical lab support.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: One of the authors receives royalties and is a consultant for Wright Medical.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Sciences and Engineering Research Council of Canada and Canadian Institutes of Health Research.
