Abstract
There has been increase attention to the surgical treatment of the subscapularis (SSC) tendon with risk of postoperative dysfunction and early failures following shoulder arthroplasty. Studies investigating the clinical results after shoulder arthroplasty indicate that SSC tendon detachment or incision techniques and rehabilitation concepts may impair SSC recovery and consequently the postoperative function. In response to these results, technical modifications have been proposed and evaluated anatomically, biomechanically, and clinically. The aim of this article is to give an overview of current SSC take-down approaches, subsequent repair techniques, and postoperative rehabilitation protocols as well as to present the diagnostic algorithm and clinical impacts of the increasingly acknowledged condition of SSC insufficiency following shoulder arthroplasty procedures.
Keywords
Introduction
The subscapularis (SSC) muscle constitutes the largest and most powerful muscle of the rotator cuff and plays a major role in optimal shoulder function. Its insufficiency may therefore lead to vast functional deficits, which needs to be considered during surgical release for shoulder arthroplasty procedures.1–5 Multiple clinical studies investigating the results after shoulder arthroplasty indicate that surgical approaches with partial or complete SSC tendon take-down techniques and certain postoperative rehabilitation concepts may impair SSC recovery and can negatively influence the clinical outcome.6–9 Failure of the tendon repair and/or muscular changes in the form of atrophy and fatty infiltration, resulting in partial or complete SSC insufficiency, has been identified as the cause for loss of SSC function.7,10 Failure rates of SSC repairs following shoulder arthroplasty procedures have been observed as high as 6.7% to 47.2%.10–15 Proposed risk factors for postoperative SSC rupture include multiple surgeries with poor-quality tissue, insufficient repair techniques, compromise of the tendon repair resulting from various SSC lengthening techniques, overstuffing of the joint due to oversized prosthetic components, and trauma or inadequate physical therapy during the early postoperative period.8–10,16,17 In response to these results, different modifications of SSC detachment and repair techniques have been developed, aiming to reduce iatrogenic damage and improve postoperative strength and durability of the SSC tendon.
The aim of this article is to give an overview of the relevant anatomical landmarks for surgical intervention, the different SSC take-down approaches, the subsequent repair techniques, and postoperative rehabilitation protocols as well as to present the diagnostic algorithm and clinical impact of the increasingly acknowledged condition of SSC insufficiency following shoulder arthroplasty procedures.
SSC Anatomy, Function, and the Application for Surgical Intervention
The SSC muscle is a multipennate muscle that originates from the anterior surface of the scapula and inserts at the lesser tuberosity as tendon within the superior 60% and as muscle within the inferior 40% of the insertion.
18
The SSC tendon footprint resembles the shape of a human ear or comma as it is very broad proximally and narrows down distally with a straight medial border that is almost parallel to the longitudinal axis of the humerus.19,20 Cleeman et al. divided the SSC insertion into 3 sections: a thick superior tubular tendon, a flat middle tendon, and an inferior portion with a direct muscular insertion.
16
The anterior humeral circumflex vessels pass laterally and parallel to the tendon, separating the tendinous and muscular portions. The axillary nerve and posterior humeral circumflex vessels mark the inferior portion of the SSC muscle at the anterior aspect of the quadrangular space. The innervation pattern of the SSC muscle may include variations. It is innervated by fibers from SSC nerves, predominantly in the form of a single superior, middle, and inferior branch that arise mostly from the posterior trunk of the brachial plexus or rarely directly from the thoracodorsal or axillary nerve penetrating the muscle belly at its anterior aspect (Figure 1).21–24 Yung et al. observed that the middle SSC nerves were always closely related to the upper nerve and appeared to be a part of the upper nerve group.
24
Furthermore, the lower SSC nerves were found to be in close relationship to the axillary nerve penetrating the muscle posterior or immediately lateral to it. Based on cadaveric, biomechanical, clinical, and electromyographic studies, different authors consider the upper and lower portion of the SSC muscle to be 2 separate units with independent functions and innervations.25–30 The SSC muscle ensures internal rotation, adduction, and abduction movement, acts as a humeral head depressor, and actively stabilizes the glenohumeral joint, depending on the position of the humeral head.1–5,25 As the anterior part of the transverse force couple, it further balances the external rotator muscles and contributes to compression and depression of the humeral into the glenoid concavity. Liu et al. suggested that the superior portions may play an important role in generating abduction torque, and the inferior portions may enhance stability.
27
Cadaveric image displaying the SSC muscle, which originates from the deep face of the scapula and inserts at the lesser tuberosity, and its innervation including superior, middle, and inferior SSC nerve branches. In this case, the superior and middle branches arise from the posterior cord of the brachial plexus, while the inferior branch arises from the thoracodorsal nerve (reprinted from Scheibel and Habermeyer,
31
with permission from Elsevier).
Different cadaveric studies have attempted to provide surgical guidelines for safe surgery about the SSC muscle. Yung et al. recommended identification of the axillary nerve at the inferior border of the SSC and protection of the lower SSC nerve. 24 In addition, dissection at the anterior surface should be limited underneath the conjoint tendon or within a margin of approximately 1.5 cm (“safe harbor”). Checcia et al. evaluated the topographic relationship of the penetrating nerves to the anterior rim of the glenoid concavity based on the position of the arm and found that the upper subscapular branch penetrates the muscle as close as 1 cm from the medial border of the glenoid. 21 All the investigated nerve branches were closest to the anterior border of the glenoid when the arm was moved from internal to external rotation. Another study observed an increased risk of denervation, in particular of the upper part of the SSC muscle, when the circumferential release is performed anterior to the SSC muscle beyond the base of the coracoid, especially with a lateral pull on the tendon. 22 Leschinger et al. investigated the location of the SSC nerve branches in relation to the musculotendinous junction, which composes a well-visible landmark during surgical exposure. 23 The nerve entrance was, on average, located 2 cm medial to the musculotendionus junction with minimum distances of 1.1 to 1.3 cm for the different SSC nerve branches. 23 Release of the anterior aspect of the SSC was therefore recommended no further than 1 cm medial to the musculotendinous junction. 23 Despite the potential risk of denervation of the SSC muscle, Gerber et al. found no sign of direct or indirect neurologic, subclinical damage after total shoulder arthroplasty (TSA) using pre-, intra-, and postoperative electromyographic assessment. 32
SSC Detachment, Repair, and Postoperative Rehabilitation
SSC Detachment Techniques
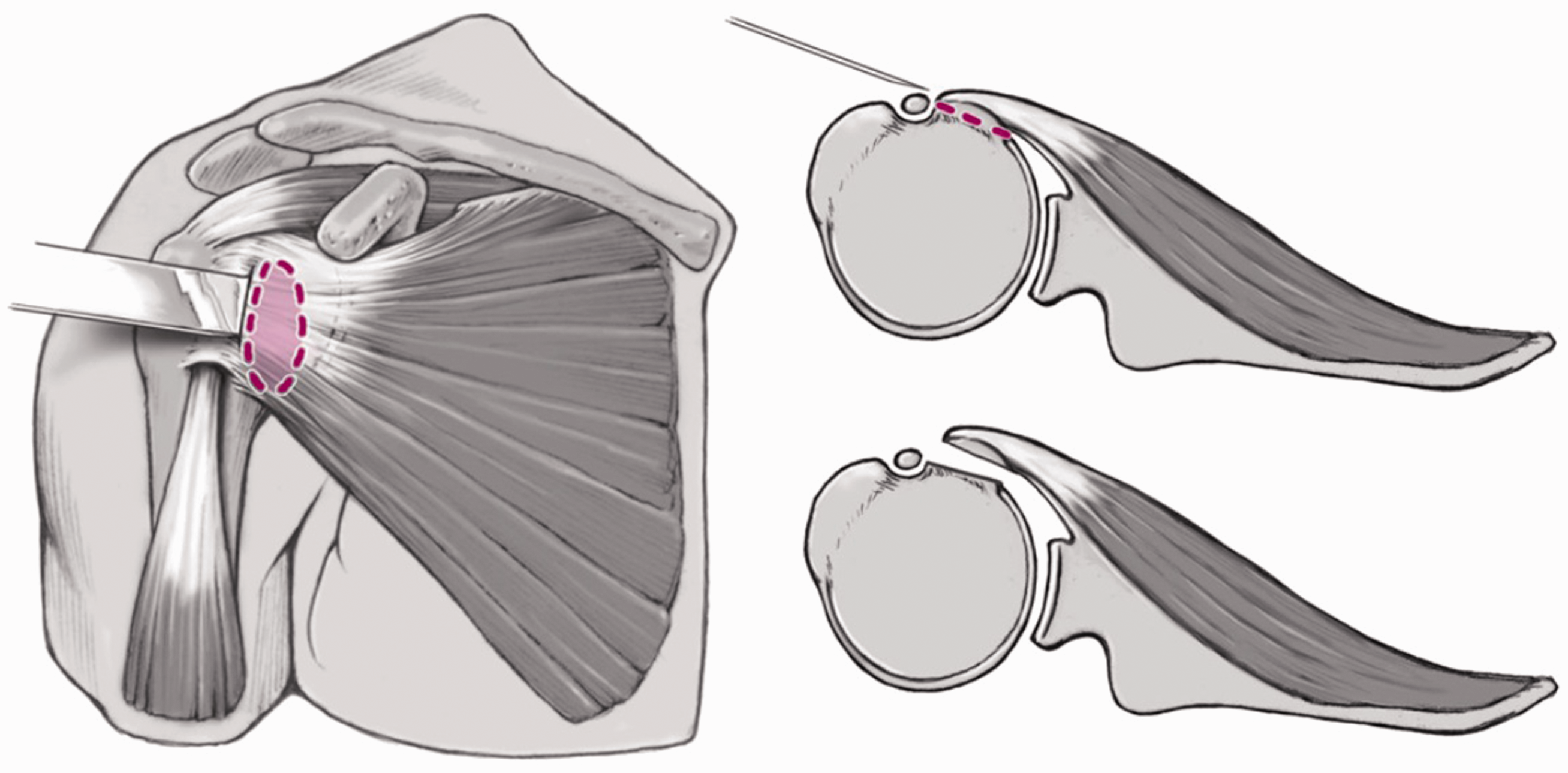
Exposure of the glenohumeral joint can be obtained by performing a SSC tenotomy (Figure 2), SSC peel, or a lesser tuberosity osteotomy (LTO) (Figure 3).7,20,33–38 Rowe et al. initially described complete SSC tenotomy as the standard approach for open shoulder stabilization.
39
The SSC musculotendinous insertion is thereby detached 0.5 to 1 cm medial to the lesser tuberosity. This enables extensive visualization of the glenohumeral joint, mobilization of the detached tendon, and separation of the capsule, followed by an anatomic tendon-to-tendon repair. Potential advantages of the LTO technique are preservation of the SSC tendon integrity, a stronger repair, and bone-to-bone healing that can easily be monitored during the early postoperative period.
Illustration of the complete SSC tenotomy approach with detachment of the musculotendinous junction 0.5 to 1 cm medial from the lesser tuberosity (reprinted from Scheibel and Habermeyer,
31
with permission from Elsevier). Illustration of the lesser osteotomy approach for SSC detachment, preserving the SSC tendon integrity (reprinted from Scheibel and Habermeyer,
31
with permission from Elsevier).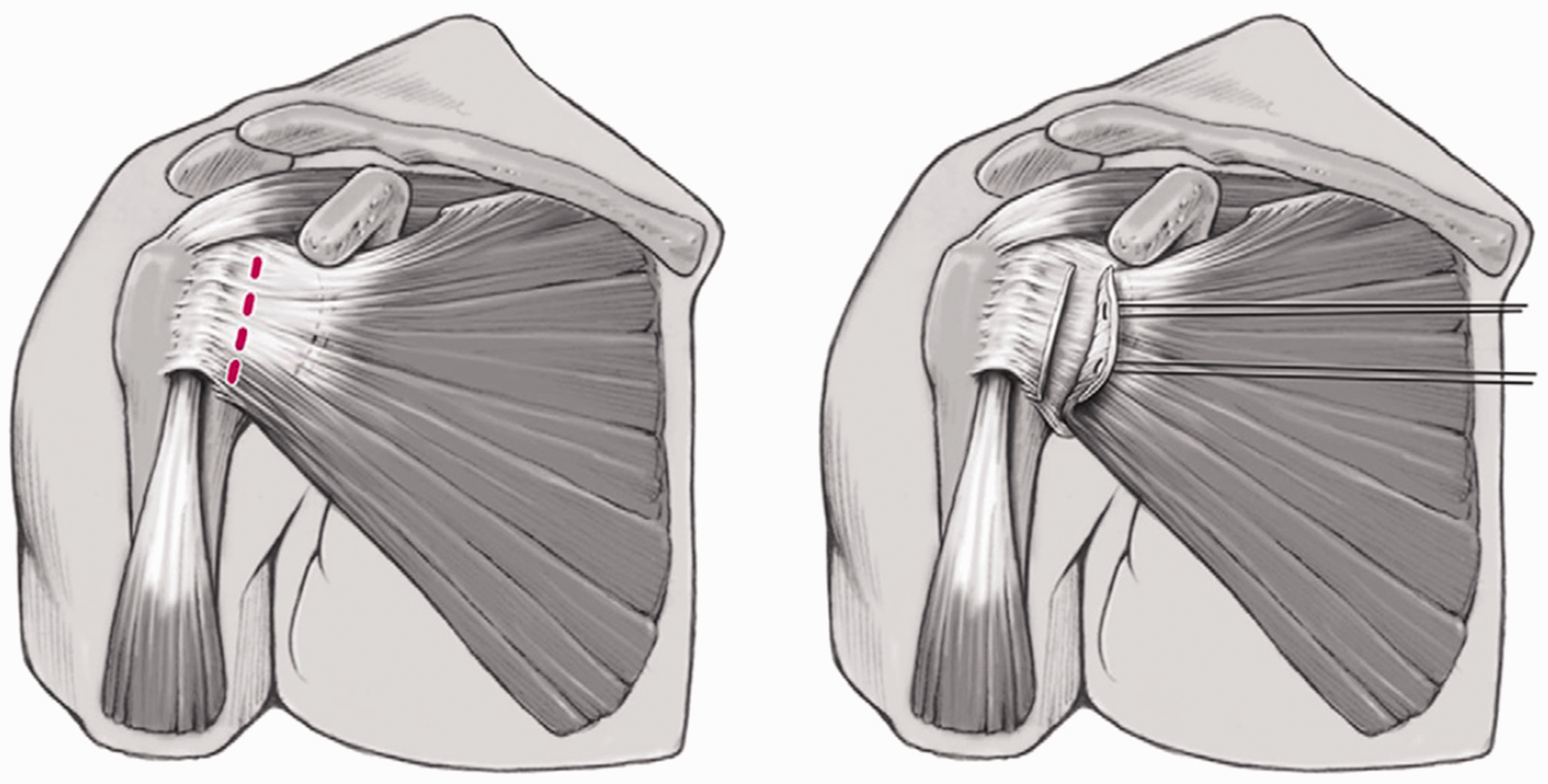
For severe internal rotation contracture in advanced osteoarthritis, for example, Habermeyer et al. have proposed detachment of the tendon with the underlying capsule directly from the lesser tuberosity down to the insertion of the latissimus dorsi muscle.34,40 This technique offers excellent exposure of the glenoid, protects the axillary nerve, and allows performance of a bifocal capsulotomy by creating a musculo-tendino-periosteal flap that can be repaired by medialization using transosseous tunnels. In particular, with internal rotation contractures, a 360° circumferential release of the SSC musculotendinous unit is necessary for soft-tissue balancing to gain adequate tendon excursion and restore full glenohumeral motion. This includes the release of adhesions at the upper border to the coracoid at the anterior surface underneath the conjoined tendons, the posterior surface from the capsule along the scapular neck, and the inferior border from the axillary nerve and the circumflex vessels. 36 Circumferential SSC release allows a SSC length increase of about 1 to 1.5 cm with 1 cm corresponding to an external rotation gain of approximately 20°.16,36,41 In addition, Miller et al. reported resection of the anterior capsule located on the undersurface of the SSC to allow further mobilization. 9 In severely contracted cases, release of the intra-articular SSC tendon, including an oblique incision of the superior tubular portion or Z-lengthening of the SSC tendon in the coronal plane, have been described.16,36 Advocating an easier performance and preservation of the SSC strength, Fama et al. further modified SSC contracture management by describing a technique with an initial release of the superior tubular tendon, including a section of the inferior coracohumeral ligament and superior glenohumeral ligament, followed by a deep release of the middle glenohumeral ligament close to the glenoid labrum and the inferior ligament. 42 The SSC tendon length increased by 2.5 cm on average and active external rotation also improved significantly from 8° to 48°.
SSC Repair Techniques
Different techniques have been described to repair the detached SSC tendon following shoulder arthroplasty, including tendon-to-tendon, tendon-to-bone, and bone-to-bone approaches. During SSC tenotomy, the detached tendon is armed using a modified Mason-Allen suture, 43 the mechanical properties of which have been shown to be superior to single and mattress stitches.43,44 The required number of sutures thereby depends on the amount of tendon detached from the lesser tuberosity. For the LTO approach, dual-row fixation with four sutures seems to achieve the strongest repair strengths, also permitting a qualitatively more secure fixation with regard to rotational motion.45,46
Biomechanical investigations of the different SSC repair techniques have been conducted with respect to the reconstructed SSC length and repair strength. However, biomechanical properties reported in the current literature are somewhat controversial and, at present, no repair technique can be identified as clearly superior. Furthermore, investigations of cadaveric shoulders only permit evaluation at time zero, without consideration of potential differences among the biologic healing rates. Tendon-to-tendon repair is susceptible for tendon shorting with a resultant deficit in external rotation, while the tendon-to-bone SSC refixation allows for tendon lengthening and therefore constitutes a viable option in cases of SSC contracture. 47 Also, the SSC peel technique enables a gain of tendon length and allows for adjustment of the point of tendon reinsertion. Comparing the strength of tenotomy and LTO approaches, the LTO is predominantly observed to enable a significantly stronger repair.45,46,48 The tendon-to-tendon repair technique has been further criticized as not allowing adequate balancing of the anterior soft tissue. With medialization of the SSC insertion, an increase in SSC excursion can be achieved. 49 However, Ahmad et al. found that transosseous tunnel repair alters SSC insertional anatomy, resulting in weaker strength of fixation and less contact area when compared with a combined transosseous tunnel and direct tendon-to-tendon repair. 50
Contrarily, other biomechanical investigations found no significant difference in tendon elongation or failure loading among the repair techniques and suggested in vivo healing rates as the durability-determining factor.51–53 Gap formation was, however, tested to be significantly greater after tenotomy versus LTO techniques and the LTO approach yielded a better SSC repair integrity. 51 The mode of failure, whether due to tendon, muscular, or osseous insufficiency did not show a statistically significant trend among the repair techniques.51,53 Tendon and bone failure occurred with approximately equal frequencies after SSC tenotomy, whereas osseous failure was the predominant cause after LTO. 53 Based on these results with similar or slightly better biomechanical properties, and bearing in mind potentially superior healing of the bone-to-bone repair in vivo, the LTO approach may be favorable. The osseous quality is thereby important to consider also with respect to the associated weakening of the metaphyseal part of the proximal humerus.
Postoperative Rehabilitation
Early range of motion has been a major tenet of rehabilitation following TSA for many years, with most protocols emphasizing immediate passive mobilization. 54 Denard and Lädermann prospectively compared immediate versus delayed mobilization following anatomic TSA. 6 Although patients who immediately began to mobilize showed a higher primary functional outcome, these initial differences ceased after 3 months postoperatively. Moreover, the rate of osteotomy healing was 81% in the immediate mobilization group compared to 96% in the delayed group (P = .101). Therefore, it appears that immediate mobilization does not affect the final outcome of TSA. These early gains may further come at the cost of an impaired SSC tendon healing and at present, a more restricted protocol is consequently recommended.
SSC Sparing Approaches
With increasing awareness of the risk of postoperative SSC dysfunction, modified surgical approaches have been developed, aiming to reduce muscular damage. In 2009, Lafosse et al. proposed a novel technique, performing primary anatomic TSA entirely through the rotator interval. 55 However, this procedure allowed only for a restricted visualization of the humeral head, especially of the inferior aspect. Consequently, concerning complication rates were observed, including residual osteophytes in 47%, malposition of the humeral component in 35%, and a mismatching humeral head in 29% of the patients.55,56 Malpositioning of the humeral head potentially exerts asymmetric stress on the glenoid, resulting in glenoid erosion and loosening over time. 57 Simovitch et al. attempted to improve the rotator interval technique in 2013 by introducing the use of modified instruments and an adaptable prosthesis with dual eccentricity but did not report any clinical or radiologic outcomes. 58 Other authors evaluated incomplete SSC tenotomy as a less invasive alternative. Savoie et al. described the release of only the inferior 30% to 50% of the SSC tendon to preserve the crucial superior aspect and observed recovery of full SSC strength in all patients and absence of tendon ruptures during ultrasound and magnetic resonance imaging (MRI) evaluation. 59 Alternative techniques have also been developed to reduce incision of the SSC tendon during reverse shoulder arthroplasty using either superior or SSC and deltoid sparing approaches.60–62
A recent study proposed a posterior glenohumeral joint access through the internervous interval between the infraspinatus and teres minor muscles. 63 This novel technique was observed to allow a significantly greater joint exposure in comparison to the rotator interval approach and the SSC splitting approach, often used for instability procedures, while reducing the rotator cuff damage in cadaveric shoulders. It offers the potential advantage of an improved access to the posterior glenoid, which is commonly subject to greater abrasion, and in cases of humeral head retroversion.63,64 However, good anatomical knowledge and careful instrumentation is required due to the proximity of the axillar nerve and the potential advantages need to be validated in clinical setting.
Diagnosis of SSC Dysfunction
Diagnosing a failed tendon repair requires a high degree of suspicion, as the condition can be asymptomatic. A potential SSC rupture should be considered especially in patients who underwent any kind of anterior approach with detachment of the SSC and who present with pain, anterior instability, weakness in internal rotation, and increased passive external rotation in the early postoperative period.8,22 An inciting event, with the patient noticing a pop-like feeling during external rotation or extension of the upper arm, may have occurred. The clinical evaluation of the SSC function includes assessment of the range of motion, strength measurement and specific SSC tests with the lift-off test, 65 internal rotation lag sign, 66 as well as the belly-press test67,68 and belly-off sign.68,69 In order to avoid false-positive results, a reliable clinical evaluation using these specific SSC tests is only possible in cases of a free range of internal rotation. 10 However, results published by Jackson et al. demonstrate that postoperative SSC ruptures are often unable to be diagnosed by physical examination. 14 In a collective of 15 patients, no correlations between tendon integrity and subjective patient outcome, pain levels, active internal and external rotation, or clinical SSC tests (lift-off, belly press, and shirt tucking) were thereby detected. The lift-off test was false negative in 4 of the 7 patients with a complete SSC tear and false positive in 3 cases. Only bear hug dynamometry and internal rotation strength correlated with SSC integrity.
For radiologic assessment of the SSC musculotendinous unit, ultrasound evaluation is especially useful in the postoperative period, where a detailed clinical evaluation cannot be performed with regard to tendon healing.
10
Postoperative radiographs with standard axillary views are performed in cases of LTO approaches to evaluate the integrity of the repair (Figure 4(a) and (b)).
7
In cases of long-standing SSC tendon defects, anterior subluxation of the humeral head can be observed on the axillary views.
70
Different authors used (Arthro-) computed tomography to detect tendon tears and grade the fatty infiltration as per Goutallier et al. (Figure 4(c)).7,71,72 MRI has also been described to evaluate atrophy and fatty infiltration of the SSC muscle;68,73 however, after TSA, MRI is much less reliable due to metal artifacts.
Preoperative (a) and postoperative (b) radiograph of an anatomic total shoulder arthroplasty with failed lesser tuberosity osteotomy repair of the SSC tendon (red arrow). Postoperative computed tomography image (c) showing fatty infiltration of the SSC muscle (green arrow).
Clinical Results
Miller et al. were one of the first to specifically report on clinical postoperative SSC dysfunction following shoulder replacement surgery. 9 Forty-one patients were included in this study and had been operated on using a complete SSC tenotomy with circumferential release of the SSC tendon. The SSC tendon was repaired using either isolated soft-tissue anatomic side-to-side repairs or with additional bone tunnels from the anatomic neck to the lesser tuberosity to reinforce the repair. After a mean follow-up of 1.9 years, the authors found abnormal results for the lift-off test in 67.5% and 66.6% for the belly-press examination. In addition, 68.2% of patients reported having difficulties tucking their shirt in the back of their pants. However, no imaging studies had been performed, and conclusions concerning the structural integrity of the SSC could not be drawn.
Ponce et al. published one of the initial investigations of clinical SSC function following LTO in 76 patients after TSA. 37 Postoperative SSC function, as evaluated by belly-press, lift-off, or shirt-tuck tests, was normal in 62 patients, abnormal in 5, and not documented in 9. Radiographic evaluation demonstrated a single rupture of the LTO repair. These results of the clinical examination appeared to be much better than those of Miller et al. 9 Gerber et al. published on their technique of LTO in 36 patients who underwent TSA. 7 After a mean follow-up of 39 months, the tuberosity osteotomy fragment healed in an anatomical position in all cases. The clinical examination of the SSC muscle revealed negative lift-off and belly-press test results in 75% and 89% of patients, respectively. However, despite radiological healing of the bone in all cases and absence of musculotendinous tears, 40% of patients had a postoperative increase in fatty infiltration by at least 1 stage and 15% of patients by at least 2 stages.
Comparative studies of tendon-to-tendon versus bone-to-bone repair techniques tend to favor the LTO approach.38,74,75 Jandhyala et al. described overall better clinical outcomes in LTO patients with a significantly better belly-press test and better, but not statistically significant, range of motion results. 74 Scalise et al. did not observe a difference in internal rotational strength and also in overall higher clinical outcomes and better SSC healing after LTO with abnormal SSC tendons seen in 10% of the LTO patients compared to 47% of the tenotomy patients. 75 However, a systematic review with evaluation of 20 studies and a total of 1420 shoulder observed similar complication rates, but a greater range of motion and fewer revisions (10.0% vs 16.2%) after SSC tenotomy versus LTO. 76 Comparing the SSC peel to the LTO repair, Lapner et al. and Buckley et al. observed no significant difference in strength or clinical scores during their investigations, whereas Shafritz et al. described a significantly worse SSC function following the SSC peel and thus recommend the LTO procedure.33,77,78 On ultrasound evaluation, Buckley et al. identified abnormal SSC tendons in 4 of the 32 patients in the SSC peel group, which correlated with significantly worse functional outcomes, whereas all tendons were intact in the LTO group. 33
Anterior shoulder instability following TSA was described by Moeckel et al. in 7 of 236 patients. 17 These patients were all found to have SSC tears on operative exploration. Despite tendon repair, 3 of 7 patients continued to have instability, thus requiring further surgery. Terrier et al. confirmed the instability-inducing effect of SCC deficiency after shoulder arthroplasty using a computer-based electromyography-driven musculoskeletal model of the glenohumeral joint to measure muscle forces, contact patterns, and humeral head translation. 79 A deficient SSC initiated a decrease of the infraspinatus muscle in order to reestablish a mechanical equilibrium, which consequently required the force exerted by the supraspinatus and middle deltoid to increase. These changes in muscular forces lead to an upward migration of the humeral head, eccentric contact patterns, and a decrease in the contact pressures by 2%.
Few studies investigated the clinical impact of postoperative SSC dysfunction after TSA. Denard and Lädermann prospectively compared healed versus nonhealed osteotomies. 6 The belly-press test was more likely to be negative and improvement of the American Shoulder and Elbow Surgeons (ASES) score was significantly greater when the osteotomy was successfully healed. In addition, there were trends toward a greater improvement of the Simple Shoulder Test and Single Assessment Numeric Evaluation, which, however, did not reach significance. 6 In a multicenter study, Edwards et al. found that postoperative fatty infiltration of the SSC in patients after TSA negatively influenced mobility, strength, and overall Constant scores. 71 However, in a study by Armstrong et al., in 3 of 4 patients with documented SSC tears by ultrasound examination, the postoperative ASES scores for shoulders with an intact SSC were equivalent to those on the side with the torn SSC tendon. 10
Conclusion
Exposure of the glenohumeral joint is an inevitable surgical step of shoulder arthroplasty procedures, which is associated with a risk of causing iatrogenic damage to the SSC musculotendinous unit and may consequently lead to vast functional implications. Variable innervation patterns of the SSC muscle constitute one of the surgical challenges, requiring thorough anatomical attentiveness during SSC detachment and mobilization. Specific landmarks have been described to provide surgical guidance. Sufficient access to the glenohumeral joint can be obtained by different SSC tendon take-down techniques, including SSC tenotomy, SSC peel, and LTO approaches. A strong and, if possible, anatomic SSC tendon repair should be performed. Although at present, no SSC repair technique has been identified as clearly superior, slight trends toward better functional outcomes using LTO techniques have been observed. Weakening of the metaphyseal part of the proximal humerus, however, represents a potential drawback of this technique. For the postoperative treatment, restricted rehabilitation concepts are recommended. Aiming to protect the integrity of the musculotendinous unit, anatomically feasible SSC sparing approaches have been described, but clinical investigations of these are still awaited.
Footnotes
Authors’ Note
The presented article constitutes an update of “Subscapularis dysfunction following anterior surgical approaches to the shoulder” by Scheibel M and Habermeyer P, published in the Journal of Shoulder and Elbow Surgery, 2008;17(4):671–683, and contains contents of that article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EB declares that no conflicts of interest exist. MS receives consultant fees from Wright, Arthrex, Exactech, Storz, and Medacta. AL receives consultant fees from Wright, Arthrex, and Medacta.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
