Abstract
Background
Rotator cuff fatty infiltration and atrophy are risk factors for worse outcomes after total shoulder arthroplasty (TSA). The purpose of this study is to evaluate the prevalence of preoperative fatty infiltration and atrophy in TSA patients compared to control patients without glenohumeral osteoarthritis.
Methods
We retrospectively identified 30 patients undergoing TSA for primary glenohumeral arthritis who had preoperative magnetic resonance imaging. TSA patients were matched by age and sex with control patients without arthritis or full-thickness rotator cuff tears. Blinded observers graded fatty infiltration of the 4 rotator cuff muscles and measured supraspinatus atrophy by the occupation ratio. A multivariate analysis was used to correlate arthritis, age, sex, and body mass index with rotator cuff atrophy and fatty infiltration.
Results
Goutallier grade ≥ 2 fatty infiltration was seen in at least one muscle for 13/30 (43%) TSA patients and 1/30 (3%) control patients (P < .01). The average supraspinatus fossa occupation ratio was 0.72 ± 0.24 in the TSA group compared to 0.92 ± 0.23 for controls (P < .01). Moderate to severe supraspinatus muscle atrophy (occupation ration < 0.6) was observed in 10/30 (33%) TSA patients and 0/30 (0%) controls. Glenohumeral arthritis was an independent predictor of atrophy and fatty infiltration (P < .01). Age, sex, and body mass index were not significantly correlated with atrophy or fatty infiltration.
Conclusion
Glenohumeral arthritis is associated with rotator cuff atrophy and fatty infiltration. In the absence of rotator cuff tendon tearing, glenohumeral arthritis may contribute to the pathophysiology of rotator cuff muscle degeneration.
Keywords
Introduction
With the exponential increase in shoulder arthroplasty procedures over the past decade, there is increasing concern over implant longevity and the rising revision burden.1,2 For anatomic total shoulder arthroplasty (TSA), rotator cuff deficiency remains one of the primary causes of implant failure and need for revision.3,4 Fatty infiltration and muscle atrophy negatively impact the quality of the rotator cuff tissue, decreasing its elasticity and capacity for healing.5,6 More recently, atrophy and fatty infiltration have also been found to predict the development of rotator cuff deficiency and implant failure following TSA.3,4,7
The research surrounding the etiology of rotator cuff fatty infiltration and atrophy has been performed primarily in subjects with large, retracted rotator cuff tears. In the setting of a torn rotator cuff tendon, both mechanical unloading and denervation from tendon retraction may contribute to the pathophysiology of these degenerative changes.5,8–11 However, it remains unknown whether these changes also occur in the setting of primary glenohumeral osteoarthritis without a major rotator cuff tear. The goal of the present study is to evaluate the prevalence of rotator cuff atrophy and fatty infiltration in a patient population undergoing TSA compared to non-arthritic age- and sex-matched controls.
Materials and Methods
The study was approved and monitored by our institutional review board (IRB 10-02764). All subjects received magnetic imaging studies as part of their routine clinical care.
TSA Cohort
All patients undergoing an anatomic TSA at our academic institution were retrospectively identified. Patients were included if they were over 18 years of age, diagnosed with primary glenohumeral arthritis, and had preoperative shoulder magnetic resonance imaging (MRI) available for review. It should be noted that MRI is not routinely used for preoperative assessment prior to shoulder arthroplasty at our institution. Instead, MRIs were usually performed by outside physicians prior to referral to our practice. Patients were excluded for a history of shoulder infection, revision surgery, autoimmune disorders, or fracture. A total of 30 patients met our inclusion/exclusion criteria.
Control Cohort
The control cohort consisted of 30 patients who were selected to match the sex and age of each patient in the TSA cohort. All control patients had plain radiographs of the shoulder that demonstrated no evidence of glenohumeral arthritis. The control patients underwent MRI evaluation of their shoulder for a variety of diagnoses (Figure 1). Patients with full-thickness rotator cuff tears on MRI were excluded as this could impact the muscle quality of the rotator cuff. We included patients with minor rotator cuff pathology including partial thickness rotator cuff tears, tendonitis, and impingement.
Indications for MRI in Non-Arthritic Control Patients.
MRI Analysis
All patients had MRIs with at least 1.5 Tesla magnets and a T1 sagittal sequence that enabled proper cross-sectional assessment of the rotator cuff musculature. MRIs were read by a musculoskeletal-trained radiologist for the presence of rotator cuff tears. Only two patients in the TSA group had full-thickness rotator cuff tears. In both patients, there was a supraspinatus tear that was less than 1 cm in the anteroposterior dimension without any tendon retraction and did not require repair. The remainder of the MRIs did not have full-thickness rotator cuff tears.
Fatty infiltration for all four rotator cuff muscles (supraspinatus, infraspinatus, teres minor, and subscapularis) was graded from 0 to 4 according to the classification originally described by Goutallier et al.
12
and later modified for MRI analysis by Fuchs et al.
13
Grade 0, normal muscle; 1, some fat streaks; 2, fatty infiltration with more muscle than fat; 3, equal amounts of fat and muscle; and 4, more fat than muscle. This grading scheme was used to analyze the most lateral sagittal-oblique image where the scapular spine was in continuity with the body (Figure 2).
Fatty Infiltration and Muscle Atrophy on MRI. Atrophy and fatty infiltration were evaluated on MRI sagittal T1 sequences. The left image shows normal rotator cuff muscle size and quality. The right image demonstrates grade 2 Goutallier fatty infiltration of the infraspinatus (IS) and teres minor (TM). There is moderate atrophy of the supraspinatus (SS) and subscapularis (Su). Supraspinatous muscle belly outlined in red (A) and Suprasinatus fossa outlined in yellow (B). Occupation ratio is given by A/B.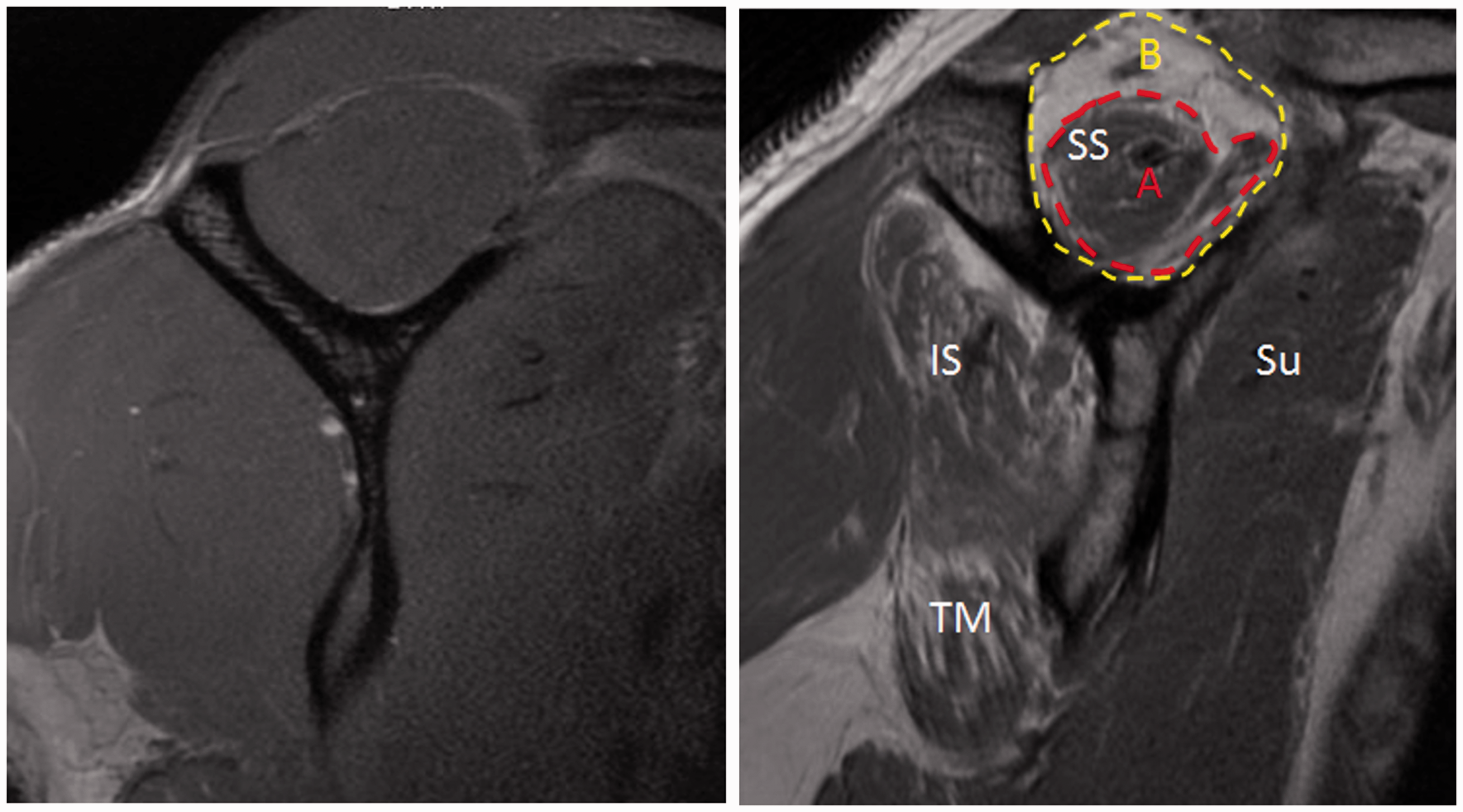
Rotator cuff atrophy was evaluated using the occupation ratio as described by Thomazeau et al. 14 The occupation ratio was defined by the surface area of the supraspinatus muscle divided by the surface area of the supraspinatus fossa (Figure 2). The muscle atrophy was normal/slight if the occupation ratio was 0.6 or greater, moderate if the ratio was 0.4 to 0.6, and severe if the ratio was less than 0.4. Both fatty infiltration and rotator cuff atrophy were graded by observers that were blind to whether the patient was in the TSA or control group.
Statistical Analysis
Student’s t test was used to compare categorical means and chi-squared tests were used to compare proportional values between cohorts. A multivariate regression analysis was performed using logistic regression modeling for dependent variables. Sex, age, body mass index (BMI), and the presence of arthritis were each evaluated as potential independent predictors of atrophy or fatty infiltration. Reported values for the independent variables included the coefficient β of the logistic regression fit, the odds ratio (OR) that was calculated from the coefficients as OR = eβ, and the corresponding 95% confidence intervals. P values less than .05 were considered significant.
Results
Cohort Characteristics
Demographics of TSA and Control Cohorts.

Note: BMI, body mass index; TSA, total shoulder arthroplasty.
*Statistically significant values (P < .05).
Average ± standard deviation of demographics for the two cohorts.
Muscle Atrophy
Supraspinatus Atrophy Measured by Occupation Ratio.

Note: TSA, total shoulder arthroplasty.
*Statistically significant values (P < .05).
Average ± standard deviation. Means compared with Student’s t-test, distribution of atrophy groups compared with chi-squared test.
Fatty Infiltration
The Goutallier grade of fatty infiltration for each of the rotator cuff muscles is shown in Figure 3. Across all four muscles, there was a tendency for the TSA group to have higher Goutallier grades, indicating a greater degree of fatty infiltration. Normal muscle (Goutallier grade 0) was seen in over 50% of control patients in each of the muscle groups. Meanwhile, the TSA group had only 1 patient with normal supraspinatus and subscapularis muscles; the remainder of the rotator cuff muscles in the TSA patients all had some degree of fatty infiltration. Goutallier grade ≥ 2 was present in at least muscle in 13/30 (43%) of the TSA group compared to 1/30 (3%) of the control group (P < .01).
Fatty Infiltration of the Rotator Cuff By Goutalier Grade.
Multivariate Analysis
Patient Predictors of Rotator Cuff Fatty Infiltration and Supraspinatus Atrophy.

Note: OR, odds ratio.
*Significant P values (<.05).
A multivariate analysis was performed to analyze the correlation of patient characteristics with the presence of grade ≥ 2 fatty infiltration in at least one rotator cuff muscle and moderate to severe supraspinatus atrophy. OR calculated with 95% confidence intervals.
Discussion
We demonstrate that patients with primary glenohumeral arthritis undergoing TSA have worse preoperative rotator cuff atrophy and fatty infiltration compared to non-arthritic controls. Goutallier grade ≥ 2 fatty infiltration and moderate to severe supraspinatus atrophy were found in 43% and 33% of patients in our TSA cohort, respectively. These degenerative changes of the rotator cuff muscles occurred in the absence of large or retracted rotator cuff tendon tears. Age, sex, and BMI were not significant predictors of either fatty infiltration or rotator cuff atrophy in our study.
Other studies have investigated preoperative rotator cuff muscle quality in patients undergoing shoulder arthroplasty using computed tomography (CT) scan. Edwards et al. 15 found Goutallier grade ≥ 2 fatty infiltration in the infraspinatus and subscapularis in 18% and 16% of patients undergoing TSA, respectively. This is lower than our rate of grade ≥ 2 fatty infiltration, which was 30% in infraspinatus and 33% in subscapularis. Lapner et al. reported an average supraspinatus occupation ratio of 77% and Goutallier grade ≥ 2 fatty infiltration in 35% of shoulder arthroplasty patients based on preoperative CT scan. The degree of atrophy and fatty infiltration they observed is similar to our results. Differences in imaging modalities may explain the variable rates of fatty infiltration between studies. Our study employed MRI, which has been shown to have better interrater reliability than CT for Goutallier grading of fatty infiltration. 16
Rotator cuff deficiency is one of the main causes of late failures after TSA.3,4 Traditionally, surgeons utilize preoperative MRI or gross intraoperative assessment to determine cuff integrity, and a large cuff tear is an indication to switch from an anatomic to a reverse TSA. Aside from rotator cuff tendon tears, muscle quality as assessed by atrophy and fatty infiltration may also be an important prognostic indicator for arthroplasty patients. In a study by Edwards et al., 15 patients with preoperative infraspinatus fatty infiltration had worse satisfaction, range of motion, and patient-reported outcomes at an average of 3.5 years after TSA. Similarly, Young et al. 3 showed preoperative fatty infiltration, particularly of the infraspinatus was a predictor of late rotator cuff dysfunction and worse outcomes at 15 years follow-up after TSA.
As part of preoperative planning, many shoulder surgeons will routinely obtain a CT scan to better characterize the osseous anatomy, especially of the glenoid. Prior to this study, we did not routinely obtain any cross-sectional imaging in our practice unless there was concern for a rotator cuff tear on exam or glenoid deficiency on axillary lateral radiographs. Given our findings and the potential importance of muscle quality on TSA longevity and outcomes, we are utilizing more cross-sectional imaging in our practice. We favor using MRI over CT as it provides adequate osseous visualization and is a better tool for evaluating muscle quality. Future studies are still needed to determine the threshold in which muscle quality impacts outcomes and implant longevity in patients undergoing TSA, and if preoperative muscle quality should be used to decide between an anatomic and reverse TSA.
In Lapner et al.’s study, 7 preoperative fatty infiltration of the supraspinatus was associated with worse strength and patient-reported outcomes 1 year after TSA. Additionally, they found small but statistically significant improvements in supraspinatus and infraspinatus fatty infiltration with repeat CT scan at 1 year following TSA. The reversibility of fatty infiltration is a unique finding in this study, as most previous studies in rotator cuff tear patients have demonstrated that fatty infiltration and atrophy fail to improve following surgical repair.17,18 The pathophysiology underlying rotator cuff degeneration in arthritis with an intact cuff may be distinct from chronic cuff tears.
Despite the fact that the majority of our cohort of patients with primary glenohumeral arthritis undergoing TSA did not have rotator cuff tears, we found a high prevalence of both rotator cuff atrophy and fatty infiltration in the absence of tendon tearing. Only 2 patients in our TSA cohort had full-thickness rotator cuff tears that consisted of small, less than 1 cm supraspinatus tears without retraction. The TSA group had at least one muscle grade ≥ 2 fatty infiltration in 43% of patients, compared to 3% in the non-arthritic control patients. Similarly, moderate to severe supraspinatus fatty infiltration occurred in 33% of TSA patients but was not seen in any of the control patients.
The etiology of muscle atrophy across injury and disuse models is relatively well understood. In chronic rotator cuff tears as well as denervation injuries, hind limb suspension models, muscle atrophy occurs due to mechanical or neurologic unloading that causes a decrease in muscle protein synthesis and an increase in pathways of muscle degradation.19–22 In the setting of chronic rotator cuff tears, atrophy occurs predictably in patients with larger tears of increased chronicity, suggesting that mechanical unloading and disuse results in muscle atrophy. Therefore, the fact that many patients with poor range of motion and shoulder osteoarthritis have muscle atrophy of the rotator cuff muscles is not surprising. As patients do not use their arm as much with shoulder osteoarthritis, decreased use and function of the rotator cuff likely leads to the chronic atrophy changes seen in this study. Future studies are needed to determine if improvements in function after TSA result in improvements in muscle size after surgery, and whether these muscle changes affect patient outcomes.
The etiology of rotator cuff fatty infiltration, however, is not fully understood.20,23 In the setting of chronic rotator cuff tearing with retraction, both mechanical unloading and denervation appear to play a role in degenerative muscular changes. Meyer et al. 6 described rotator cuff tendon tearing with retraction changes the pennation angle of the muscle fibers, creating gaps that are infiltrated by fat and fibrous tissue. Using a sheep model, Gerber et al. 5 sectioned rotator cuff tendons and found that over the course of 40 weeks, the muscle developed progressive intrafasicular fat content without a change in vascularity. 5 Other research suggests that retracted rotator cuff tears may increase tension on the suprascapular nerve as it passes through the suprascapular notch, leading to denervation, which may contribute to atrophy and fatty infiltration.5,8,11,24
These changes are probably not relevant in the scenario of osteoarthritis when the rotator cuff is still intact, as there is less mechanical unloading and denervation is not likely. Therefore, one must consider other potential causes of fatty infiltration. Recent basic science studies suggest that a newly described stem cell population in muscles can contribute to the development of fatty infiltration and fibrosis after rotator cuff injury, and these cells are regulated by changes in transforming growth factor-β (TGF-β) activity. 25 TGF-β is upregulated in chronic inflammatory conditions and has been shown to be present in both chronic large rotator cuff tears and the synovial fluid of patients with osteoarthritis. This may provide a common pathway to explain why glenohumeral OA and rotator cuff tearing can both independently lead to changes in rotator cuff muscle quality. 26 Further, chronic inflammation, as seen in osteoarthritis, may cause distinct local muscle changes including fibrosis and atrophy which may be an important underlying cause of muscle degeneration in this subset of patients.27,28 Therefore, the development of rotator cuff fatty infiltration may be multifactorial and be related to the underlying disease process occurring in the shoulder.
The study had a few limitations. The MRI scans were obtained with variable Tesla strengths and techniques based on the technology that was available over the study period, which could affect radiographic muscle assessments. With a retrospective study, we can only identify correlations between a subset of available variables and rotator cuff degeneration. While we accounted for sex, age, and BMI in our analysis, we did not assess other comorbidities such as diabetes that might serve as confounding variables affecting rotator cuff muscle quality. Additionally, we can only establish correlations and not causation between glenohumeral arthritis and rotator cuff degeneration. Finally, the current study did not have post-operative outcomes after TSA. Further follow-up research is needed to correlate atrophy and fatty infiltration with outcomes after TSA and to identify patients at risk for unsatisfactory outcomes after anatomic TSA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Level of Evidence
Level III, retrospective case–control study
