Abstract
Background
Retrieved clinically failed anatomic total shoulder arthroplasty (TSA) components as well as associated radiographs and medical records were analyzed in order to investigate the origins of clinical failure(s) of TSA components.
Methods
Fifty TSA systems were retrieved from 48 patients including 27 females and 21 males; components were implanted for an average of 68 months (range, 2–267). Data obtained through radiographic analysis, medical records review, and damage mode analysis of each component were performed and statistically analyzed.
Results
Significant correlations were found between glenoid loosening and preoperative pain (r = 1.00, P < .001), humeral stem scratching and humeral loosening (Zone 1: r = .60, P < .01; Zone 2: r = .45, P = .03; Zone 3: r = .45, P = .03) , as well as glenoid damage and rotator cuff tears (r = .36, P = .02).
Discussion
Wear and damage of the glenoid and humeral components, which may result from rotator cuff tearing, may lead to accelerated loosening, clinical failure, and early revision arthroplasty. Improved implant design and/or surgical indications may improve implant performance, thereby reducing the incidence of implant damage and wear, and result in improved clinical outcomes.
Introduction
The number of shoulder arthroplasty procedures, including anatomic total shoulder arthroplasty (TSA) and reverse total shoulder arthroplasty (RTSA), performed per year is rapidly increasing. The Nationwide Inpatient Sample (NIS) database, maintained by the Agency for Healthcare Research and Quality (AHRQ), reported that approximately 47,000 shoulder arthroplasty procedures were performed in the United States in 2008. 1 As the number of shoulder arthroplasty surgeries increases, the demand for revision procedures will also grow and present burdens on patients and the healthcare system. Day et al. 2 reported a 319% increase in TSA and 431% increase in all upper extremity revision procedure rates from 1993 to 2007, with both projection model and Poisson regression analyses indicating increases in primary and revision TSA procedures. 3
Singh et al. 4 retrospectively analyzed TSA rates of revision and described 94.2%, 90.2%, and 81.4% survivorship at 5, 10, and 20 years, respectively. Clinical failure of shoulder arthroplasty is generally categorized as a soft tissue, osseous, and/or implant complication, with reasons for revision including wear, loosening, instability, periprosthetic fracture, rotator cuff tear/insufficiency, infection, or deltoid muscle dysfunction; however, in many cases, the origins of failure are multifactorial.3,5 Recent studies have investigated implant complications through both glenoid survivorship as well as polyethylene damage and/or wear analyses of retrieved components.6–13 Additionally, studies have radiographically examined TSA prostheses in order to correlate radiolucencies with implant performance; however, limited studies have investigated radiographic evaluation, damage mode analysis of both humeral and glenoid TSA components, and clinical data from the medical record.7,14–18
The objective of this study is to explore relationships and document observations of damage modes on retrieved metallic and polyethylene TSA components, radiographic analysis, intraoperative observations, patient factors, and implant design characteristics, in order to derive additional information regarding potential causes of in vivo implant damage and clinical failure of TSA. Through this study, we hope to generate hypotheses regarding implant performance, design, and outcomes as the basis for future studies.
Patients and Methods
Following institutional review board approval, retrieved TSA implants were obtained from the Orthopaedic Implant Retrieval Program at our institution between 2000 and 2012. All implants were retrieved by a single surgeon. Retrieved devices were catalogued within 1 week of surgery and subsequently sonicated in a diluted Micro-90 solution for 5 min, rinsed in ethanol, followed by another sonication in >85% ethanol solution for 5 min. All non-polymer components were also subjected to a 5-min ultrasonic acetone bath. Explants were de-identified through the assignment of an explant identification number, allowed to air dry, vacuum-sealed to prevent oxidation, and stored. Implant manufacturer, model, design, and composition characteristics as well as related surgical information were collected and electronically stored in a database.
Study Population
Fifty TSA implants were obtained from 48 patients. One patient had both primary and revision TSA components included in the study, while another patient had bilateral TSAs, both of which were also included in the study. Thirteen implant models were represented in the analysis, including Bigliani/Flatow (Zimmer, Inc., Warsaw, Indiana, USA; n = 14), Global (DePuy Orthopaedics, Inc., Warsaw, Indiana, USA; n = 9), Bio-Modular (Biomet, Warsaw, Indiana, USA; n = 6), Aequalis (Tornier, Bloomington, Minnesota, USA; n = 3), Cofield II (Richards/Smith & Nephew, Inc., Andover, Massachusetts, USA; n = 3), Comprehensive (Biomet, Warsaw, Indiana, USA; n = 3), Trabecular Metal (Zimmer, Inc., Warsaw, Indiana, USA; n = 3), Aequalis PF (Tornier, Bloomington, Minnesota, USA; n = 2), Global Advantage (DePuy Orthopaedics, Inc., Warsaw, Indiana, USA; n = 2), Solar (Osteonics/Stryker Orthopaedics, Kalamazoo, Michigan, n = 2), Anatomical (Sulzer Orthopedics, Inc., Austin, Texas, USA; n = 1), Select (Sulzer Orthopedics, Inc., Austin, Texas, USA; n = 1), and Cofield I (Richards/Smith & Nephew, Inc., Andover, Massachusetts, USA; n = 1).
Of the 50 retrieved TSA components, 45 included cobalt-chromium-molybdenum (CoCrMo) alloy metallic humeral heads, 35 contained CoCrMo or titanium (Ti) alloy metallic humeral stems, and 46 contained ultra-high-molecular-weight polyethylene (UHMWPE) glenoids (4 cases did not include a glenoid component). Thirty-nine glenoids were composed of conventional UHMWPE, including 1 cross-linked DePuy Global Advantage Anchor Peg Glenoid, and 7 glenoids were composed of Hylamer polyethylene. The retrieved glenoids included pegged (n = 26), keeled (n = 16), and trabecular metal-backed (n = 8) designs. Forty-one of the 46 glenoids were cemented (89%); specifically, 38 components without trabecular metal-back were cemented, 3 trabecular metal-backed components were cemented, and 5 trabecular metal-backed components were uncemented. Eight humeral stems (23%) were cemented.
Damage Mode Analysis
Retrieved TSA components were first visually inspected for the presence or absence of following damage modes: burnishing, dishing (or material removal), embedding, fracture, pitting, and scratching. Intact glenoid components were also analyzed for delamination and edge deformation.6,10,19
Burnishing was defined as discrete regions on the component with a smoother texture and more reflective appearance than the surrounding materials. Dishing/material removal of the component was defined as areas of plastic deformation on the articular surface of the component, while edge deformation was plastic deformation observed at the rim or edge of the polyethylene glenoid component. Delamination, most frequently observed on polyethyelene components, described the separation of layers of the material. Areas of superficial or deep linear scarring were categorized as scratching, while pitting was defined as small voids in the component surface. Third-body particles that had become ensconced into the component surface defined the embedding damage mode. Lastly, fracture was defined as catastrophic failure of components due to overloading (Figure 1).
Representative images of material-level damage on all 3 components—humeral stem, humeral head, and glenoid—of retrieved anatomic shoulder arthroplasty implants. Damage on retrieved humeral stem components, including pitting (a), abrasion (b), and scratching/grooving (c); Damage on retrieved humeral head components, including scratching/grooving (d), pitting (e), and abrasion (f); Damage on retrieved glenoid components, including dishing (g) arrows indicate stepped material removal), burnishing (h) arrows indicate highly-polished surface), abrasion (i), edge deformation (j), delamination (k), scratching/grooving (l), third-body particle embedding (m), and pitting (n).
Components were then evaluated using light microscopy (LM; MZ-16, Leica Instruments) and scanning electron microscopy (SEM; JEOL JSM-6400, JEOL Ltd.), if additional confirmation of damage modes was necessary. Polyethylene components were sputter coated with gold-palladium prior to evaluation under SEM.
Medical Records Review
Medical records were reviewed to determine patient age at implantation and revision, gender, body mass index (BMI), dominant hand status, infection status, and diagnosis/reason for index and revision surgeries, and length of time components were implanted. Operative reports were also reviewed to determine implant fixation methods as well as specific intraoperative observations (eg, instability, presence of wear debris, synovitis). Pathology reports were reviewed for cases of suspected infection to confirm infection status and determine the organism.
Radiographic Analysis
Available immediate pre-revision radiographs (n = 45) were reviewed for the evaluation of device alignment and migration as well as the presence/absence of lucency, cement extrusion/fragmentation, and metallic debris. When present, the location of humeral lucency was defined by zone, using the method described by Sanchez-Sotelo et al.20,21 Glenoid lucency, when present, was graded according to the method described by Lazarus et al. 14
Statistical Analyses
List of Data Elements Captured.

Results
Medical Records Review
The patient population included 27 females and 21 males, with average ages at primary implantation and revision of 59 and 65 years (range, 45–84; range, 48–90), respectively, and average BMI of 30 (range, 21–64). Of the 48 patients, 45 were right-hand dominant and 3 were left-hand dominant, with 31 TSA components implanted on the dominant side. Thirty-two were right shoulder implants, and 18 were left. TSA components were implanted for an average of 68 months (range, 2–267), with 60% of revisions occurring within 5 years of implantation (0 to <1 year [n = 12]; ≥1 to <5 years [n = 18]; ≥5 to <10 years [n = 10]; >10 years [n = 10]). Pain was reported prior to the revision procedure in 48 cases (96%).
Reasons for revision listed on the operative report were multifactorial, including glenoid loosening (n = 31; 62%), rotator cuff tear (n = 20; 40%), infection (n = 9; 18%), humeral loosening (n = 6; 12%), osteolysis (n = 7; 14%), bony fracture (n = 5; 10%), fractured glenoid component (n = 4; 1%), dislocation (n = 3; 1%), and impingement (n = 1; 0.02%). In the 9 shoulders revised due to infection, the known organisms were coagulase-negative Staphylococci (n = 2), methicillin-resistant Staphylococcal aureus (n = 1), staphylococcus (n = 1), and propionibacterium (n = 1), as determined through the medical record review of pathology reports. Four cases were treated as infections despite negative cultures.
Based on surgeon intraoperative observations noted in the operative report, synovitis was documented in 10 shoulders (20%), while glenoid and humeral deficiency were recorded in 19 and 5 cases, respectively. Wear debris (UHMWPE, n = 10; metallic, n = 2) and cement fragmentation were observed in 12 and 2 revision cases, respectively.
Radiographic Analysis
Loosening (humeral and/or glenoid) was observed and graded radiographically in of 35 of 37 aseptic shoulders (95%), with radiographs available for review. Twenty-one pre-revision radiographs exhibited glenoid-only loosening (57%), 13 showed both glenoid and humeral loosening (35%), and 1 demonstrated humeral loosening only (0.03%). Glenoid loosening with Grades 1 (n = 2; 5%), 2 (n = 4; 11%), and 3 (n = 2; 5%) demonstrated the lowest frequency in our series, while 26 pre-revision radiographs (70%) indicated Grade 4 (n = 9; 24%) or Grade 5 (n = 17; 46%). For all cases of radiographically observed glenoid loosening, the average grade was 4 ± 1.3. The highest incidence of humeral loosening occurred in Zone 1 (n = 10; 27%) and Zone 7 (n = 8; 22%), with 10 and 8 cases, respectively (Figure 2).
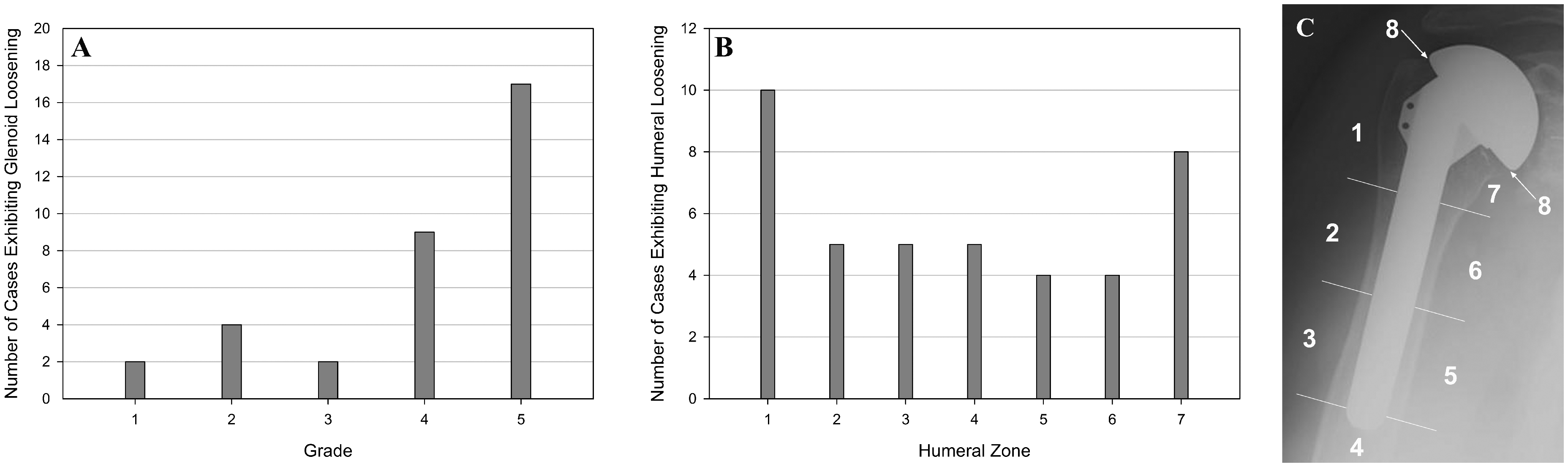
Damage Mode Analysis
Of the described damage modes, only scratching, burnishing, and pitting were observed on the metallic components (Figure 3). Thirty-six humeral heads (80%) and 24 humeral stems (69%) demonstrated at least 1 damage mode. On the humeral stem, pitting was the most common damage (49%), while scratching (60%) and pitting (47%) were the most common modes on the humeral head. Three of 7 (43%) Hylamer glenoid components were fractured, which prohibited edge deformation and delamination damage mode analyses. Of the 46 glenoids, scratching, dishing, and/or pitting were observed on more than 70% of components. Edge deformation and delamination were present on 58% and 14% of the analyzed components (n = 43), respectively (Figure 4).
Representative SEM micrographs showing pitting (a), abrasion/plowing (b), and scratching (c) of the humeral head. Percentage of observed damage modes on retrieved TSA components, including humeral head, humeral stem, and glenoid components. *Three glenoids were fractured and unable to be analyzed for delamination and edge deformation damage modes.

Statistical Analyses
Statistical analyses provided insight into factors potentially contributing to in vivo implant damage. Several statistically significant relationships were found between damage modes, implant characteristics, patient factors, and radiographic analysis data.
A very strong, positive correlation was found between pre-revision, patient-reported pain, and glenoid loosening (r = 1.00, P < .001) and a strong, positive correlation between presence of synovitis and wear debris was also demonstrated (r = .76, P = .02).
When comparing damage modes with implant characteristics, clinical data from the medical record and radiographic data, moderate, positive correlations were found between scratching damage on the humeral stem (at/near the head-stem interface) and radiographically observed humeral lucency in Zone 1 (r = .60, P < .01), Zone 2 (r = .45, P = .03), and Zone 3 (r = .45, P = .03). A weak, positive correlation was found between length of time components were implanted and burnishing of the glenoid (r = .33, P = .03). Similarly, a weak, positive correlation between keeled glenoid components and burnishing of the glenoid was established (r = .32, P = .04). In keeled components, 69% exhibited burnishing of the glenoid, while 38% of pegged components were burnished. When comparing edge deformation of the glenoid component and presence/absence of rotator cuff tear, a weak, positive correlation was found (r = .36, P = .02). In the patient population with rotator cuff tears (n = 20; 40% of total series), 65% of glenoid components exhibited edge deformation, compared to 39% of glenoid components in patients without rotator cuff tear. When each muscle (subscapularis, supraspinatus, infraspinatus, teres minor) was evaluated separately, there was also a weak, positive correlation between subscapularis tears and edge deformation of glenoid components (r = .32, P = .04).
Discussion
Analyses showed statistically significant correlations, both for the glenoid components and humeral components. On the glenoid side, loosening was associated with increased patient-reported pain. Regarding damage observed on retrieved glenoid components, burnishing damage was associated with longer implantation time as well as keeled implants; edge deformation was associated with presence of rotator cuff tear, specifically subscapularis tears; and Hylamer polyethylene implants had increased rates of fracture compared to other polyethylene implants. On the humeral side, scratching damage at/near the head-stem interface on the humeral stem was associated with humeral lucencies in Zones 1, 2, and 3. Globally, concurrent intraoperative glenohumeral synovitis and presence of wear debris were strongly associated.
Despite improvements in implant design and surgical techniques, glenoid component loosening remains a dominant mode of failure of TSA.2,23,24 The influence of wear debris generation on aseptic loosening of total hip and knee arthroplasty implants has been well established25,26; similarly, results from several TSA retrieval studies have reported significant wear and damage to the polyethylene glenoid components leading to wear debris-induced osteolysis, subsequent component loosening, poor patient outcomes (eg, limited range of motion, pain), and ultimately revision.4,27 Nho et al. evaluated 78 retrieved TSA glenoid components (n = 57 revised due to glenoid loosening; 73%), assessing numerous damage modes, including burnishing, abrasion, scratching, pitting, delamination, focal wear, surface deformation, embedded third-body debris, and fracture. They found the inferior region of the glenoid exhibited the greatest wear scores and all metal-backed glenoid components generated metallic third-body wear debris, which was found embedded in the polyethylene surface. Nho et al. 6 reported scratching (100%) and pitting (94%) were the most common damage modes observed. Burnishing (69%) and abrasion (68%; 37% occurring at the edge of components) were also common, followed by surface deformation (62%) and edge deformation (23%). In a separate study, Nho et al. 9 showed that articular conformity of retrieved glenoid components was significantly associated with differential wear scores; specifically, non-conforming surfaces showed greater burnishing and conforming surfaces exhibited greater abrasion and delamination. Gunther et al. 10 evaluated glenoid components retrieved from 10 consecutive revision surgeries and indicated that the surface and subsurface damage modes observed, including scratching (90%), abrasion (68%), pitting (60%), delamination (58%), deformation (40%), embedded wear debris (28%), and burnishing (8%), played a role in glenoid failure. Scarlat and Matsen 11 described rim erosion (68%), surface irregularity (68%; includes “polishing, scratches, and pits”), fracture (29%), and central wear (22%) damage in a study of 41 retrieved glenoids (not including implants retrieved at autopsy), implanted for an average of 1.04 years.
Different retrieval studies demonstrate a broad range of observations due to differential patient, surgical, and outcomes factors in each series. However, our observations are similar to reports by Nho et al., 6 Gunther et al., 10 and Scarlat and Matsen. 11 In our study population, with 21 cases revised due to glenoid loosening (57%), scratching (98%), dishing/material removal of the surface (74%), and pitting (72%) damage were the most common observations. Burnishing damage was observed in half of the retrieved glenoid components. These findings are consistent with observations of specific damage modes by Nho et al. 6 and Gunther et al. 10 as well as surface irregularities identified by Scarlat and Matsen. 11 However, our series showed a higher rate of edge deformation (54%) than Nho et al. 6 (23%), but a lower rate than Scarlat and Matsen 11 (68%). Nho et al. 6 reported 52% of revisions demonstrated rotator cuff tendinopathy, while 40% of our series had a rotator cuff tear; however, no additional details were available to compare each muscle. Also, our series had lower rates of embedded wear debris (13%) and delamination (13%) than the Gunther series (28% and 58%, respectively). 10 Keeled glenoid implants were associated with an increased incidence of burnishing in our series, which is described as a “less severe” damage mode generally indicative of the time-dependent relationship of joint contact stresses, as reported in total knee arthroplasty retrieval literature,28,29 which may correspond with keeled glenoids being implanted for an average of 25 months longer than pegged glenoids in our study population (keeled glenoids: implanted on average 89 months and pegged glenoids on average 64 months, prior to revision). However, a review by Matsen et al.14,27 described poorer “seating” and overall “less secure fixation” with keeled glenoid components compared to pegged glenoid components, which may also affect joint kinematics and lead to in vivo component damage.
In addition to design characteristics of the different retrieved TSA implants, some patient factors were also found to correlate with the presence of specific component damage modes. Notably, edge deformation, or rim loading, on the glenoid components was shown to be correlated to the presence of a postoperative rotator cuff tear in our series, while Nho et al. 6 concluded that rim loading of retrieved glenoid components was associated with increased rates of glenoid loosening. A recent multi-center review by Young et al. 30 of 518 primary shoulder arthroplasties with at least 5-year follow-up found that the incidence of secondary rotator cuff dysfunction was 16.8%, while our study population showed an increased incidence of 40%. Matsen et al. 27 discussed patient factors related to glenoid component failure, which included patients with glenohumeral instability and eccentric loading profiles of the shoulder due to rotator cuff deficiency; specifically, superior subluxation following massive rotator cuff deficiency was defined as a cause of glenoid component loosening. In addition to identifying pre-operative risk factors, Young et al. 30 also found an association of secondary rotator cuff dysfunction with the development of radiolucent lines, glenoid component migration and radiographic evidence of loosening, ultimately leading to decreased implant survivorship. While the magnitude of glenohumeral contact forces has been shown to decrease following rotator cuff tear, Yu et al. 31 confirmed the migration of the humeral head as a result of an imbalance of muscle forces across the joint. These previously described deviations in glenohumeral kinematics may contribute to the edge deformation observed primarily on glenoid components retrieved from patients with post-arthroplasty rotator cuff tears.27,31
Hylamer glenoid components are associated with high rates of fracture, delamination, and early failure, compared to survivorship of conventional polyethylene components. Hertel and Ballmer 12 analyzed 7 retrieved Hylamer and conventional polyethylene glenoid components, which were implanted 3 to 12 years, and observed rim deformation and/or erosion, abrasion, and scratching damage modes, as well as central wear. Rockwood and Wirth presented observations of 7 Hylamer polyethylene glenoids, which had been implanted 32–99 months. They found erosion, cracking, fragmentation (catastrophic failure), pitting, and/or delamination in 86% of the components. 13 Our series also showed greater rates of catastrophic failure (fragmentation) of Hylamer polyethylene components (43% within the Hylamer polyethylene cohort; 6% of total series) compared to no fragmentation of conventional and cross-linked glenoid components.
While other retrieval studies have previously reported wear and mechanical failure of polyethylene glenoid components, no shoulder arthroplasty retrieval studies have extensively characterized damage to the humeral head or humeral stem. In this study, scratching was the most commonly observed damage mode on the CoCrMo humeral heads, generally located on the articulating surface, but did not correlate to any radiographic parameters or clinical data from the medical records. Mechanical scratching damage on the humeral stem, which was generally located at/near the head-stem interface, was observed in 29% of retrieved components in our series, which correlated with several of the humeral zones of loosening. Although wear-induced osteolysis is most often associated with polyethylene debris, metallic particulate and ionic debris generated at the femoral head-trunnion interface has been associated with component loosening and reduced survivorship of total hip arthroplasty. 32 It is hypothesized that metallic debris generated at the head-stem interface may contribute to the presence or progression of osteolysis and component loosening, through wear-debris induced and/or metal hypersensitivity immune processes. 33 Teeter et al. 34 also recently documented corrosion and fretting damage on the lower zone of the humeral stem taper, which may lead to trunnionosis, as described in the total hip arthroplasty literature. 35
This study had several limitations. Due to the retrieval-based nature of this study, a control was not established to validate if revision rates of retrieved, “clinically failed” TSA components are increased due to damage. Glenoid components were not included with every retrieved set of implants, and the ability to grade glenoid components through surface mapping, as previously described in literature, was complicated by symmetric glenoid designs and the inability to accurately determine anatomic orientation in vivo. Also, the medical records were incomplete, with only 44 post-index and revision to post-index sets available for review. Although this data set included a heterogeneous population of implants, comparisons were drawn to understand implant performance based on implant characteristics. Due to many factors, including the heterogeneity of the retrieved implant population and retrospective review of records, the statistical analysis was not inclusive of all possible data elements and Type II error is therefore possible.
The multi-modal analysis of this study indicated relationships between implant damage modes (on polyethylene and metallic components), radiographic, and clinical data, including radiolucencies and reasons for revision. Damage was most prominently and frequently observed on polyethylene glenoid components; however, metallic humeral heads and humeral stems of retrieved TSA components also demonstrated several damage modes. Specific implant design features may be associated with increased rates of damage, as a greater incidence of burnishing was observed in keeled glenoid components compared to pegged components. Biomechanical considerations, such as post-implantation rotator cuff tears, may alter the joint kinematics, thereby leading to component damage, such as edge deformation.
Anatomic TSA remains an effective treatment option for patients with arthrosis of the glenohumeral joint and an intact rotator cuff. However, with rates of revision of a 3-decade cohort reported as 5.8%, 9.8%, and 18.6% after 5, 10, and 20 years, respectively, there remains a need for further clinical, translational and basic science research to improve TSA component survivorship. 4 Accelerated prosthetic wear or damage may lead to early device failure and/or need for revision. This study explored trends and observations related to clinical failure of TSA through analyses of retrieved implants, radiographs, and clinical records and showed associations between glenoid loosening and patient-reported pain, burnishing damage on keeled glenoid components, edge deformation damage and rotator cuff tears, humeral stem damage and humeral stem loosening, as well as synovitis and presence of wear debris. These findings may provide a platform for future multi-center and prospective studies to elucidate significant factors leading to early revision, develop algorithms and/or techniques for determining patients and prostheses at risk for clinical failure, and recommend novel prosthesis designs.
Footnotes
Authors' Note
This study was approved by the Human Investigation Committee at Beaumont Hospitals Research Institute under the study ID #2011-247.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AAOS Disclosure of J Michael Wiater: Board or committee member American Shoulder and Elbow Surgeons: Board or committee member Biomet: Paid consultant Clinical Orthopaedics and Related Research: Editorial or governing board DePuy, A Johnson & Johnson Company: Paid presenter or speaker Eleven Blade Solutions, Inc: Stock or stock Options Journal of Bone and Joint Surgery—American: Editorial or governing board Journal of Shoulder and Elbow Surgery: Editorial or governing board Journal of the American Academy of Orthopaedic Surgeons: Editorial or governing board Sanders/Mosby-Elsevier: Editorial or governing board Synthes: Research support Tornier: Research support Zimmer: Paid consultant; Paid presenter or speaker; Research support *Please note that upon IRB protocol approval, a management plan was formalized and executed defining Dr. Wiater's role in the study. Kevin Baker, PhD, (as research PI) and Erin Baker, PhD, (as family member) have received research support from Zimmer (per AAOS disclosure); however, this funding occurred after this study was completed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Orthopaedic Implant Retrieval Program at Beaumont is underwritten by generous donations to the Dorothy and Byron Gerson Implant Analysis Fund.
