Abstract
Background
Up to 25% of stroke survivors develop post-traumatic stress disorder (PTSD) symptoms, yet the predisposing factors remain largely unknown. The C-C-Chemokine receptor-5 gene (CCR5) loss-of-function mutation (LOFM, CCR5-Δ32) has been identified as a protective factor against post-stroke depression. This study investigates whether CCR5-Δ32 also confers protection against post-stroke PTSD, in conjunction with two additional polymorphisms: the 5-HTTLPR in the serotonin transporter gene and the BDNF Val66Met variant.
Methods
We conducted a prospective analysis of 432 survivors of first-ever mild-to-moderate ischemic stroke, assessing PTSD symptomatology at 6, 12, and 24 months post-stroke. Genetic screening for CCR5-Δ32 status and PTSD symptom data were available for these participants.
Results
PTSD was diagnosed in 48 participants (11%) within the first year post-stroke. CCR5-Δ32 carriers exhibited significantly fewer PTSD symptoms at 6, 12, and 24 months compared to non-carriers (P < .001, P < .001, P = .02, respectively), with sustained improvement over time. Multivariate analysis confirmed that CCR5-Δ32 status was independently associated with lower PTSD risk after adjusting for relevant confounders. Furthermore, individuals with a maladaptive coping style who were non-carriers of CCR5-Δ32 exhibited a higher risk of PTSD development (HR = 4.03; 95% CI, 1.95-6.32, P < .001). Carriers of both 5-HTTLPR-L and CCR5-Δ32 had significantly lower PTSD symptoms at 6 and 12 months post-stroke (P = .026, P = .05), as did carriers of both the BDNF Val allele and CCR5-Δ32 at 6 months (P = .022).
Conclusions
Our findings suggest that CCR5-Δ32 carriers are less likely to develop PTSD symptoms following stroke, including individuals with pre-existing maladaptive coping styles. These results highlight a potential genetic target for future intervention strategies, with CCR5 blockade emerging as a promising therapeutic avenue for post-stroke PTSD prevention.
Keywords
Introduction
Neuropsychiatric complications significantly impact stroke recovery, contributing to increased morbidity and mortality. Among these, post-traumatic stress disorder (PTSD) has gained attention as a major concern for stroke survivors, with prevalence rates reaching up to 30%.1,2 PTSD symptoms can manifest even in cases of mild stroke or transient ischemic attack (TIA), exerting profound effects on adherence to treatment, functional recovery, and overall quality of life.3,4 Recently, our group showed that post-stroke PTSD is more common in individuals with certain premorbid coping styles. 5 The etiology of post-stroke PTSD is multifaceted, involving interactions between trauma-related experiences, personality traits, and genetic predisposition. Twin and family studies have indicated a genetic component in PTSD susceptibility, with heritability estimates ranging from 30% to 40%.6,7 Several candidate gene loci associated with PTSD susceptibility have been identified, 8 including a polymorphism in the promoter region of the gene SLC6A4 encoding the serotonin transporter (5-hydroxytryptamine transporter, 5-HTT). SLC6A4 plays a crucial role in regulating serotonergic activity in the synapse and has been reported to modulate sensitivity to stress and susceptibility to psychopathology. The 5-HTT-linked polymorphic region (5-HTTLPR) is characterized by either 14 (short, S) or 16 (long, L) copies of an imperfect 22–23 bp repeat.9,10 Prior studies suggest that individuals carrying the short allele are at a greater risk of developing PTSD following exposure to trauma.11–14 Brain-derived neurotrophic factor (BDNF) has also been implicated in the development of PTSD. BDNF is involved in neurodevelopment, neuronal survival, synaptic plasticity, and protection against stress-induced neuronal damage. Serum levels of BDNF are decreased in PTSD patients compared to healthy controls. A polymorphism in the BDNF gene (Val66Met; rs6265) has been found to affect dendritic trafficking, synaptic localization, and activity-dependent secretion of BDNF. Individuals who carry the Met66 allele have been found to display abnormal hippocampal activation and reduced hippocampal volumes, often observed in patients with PTSD.11,15–20
The C-C chemokine receptor 5 (CCR5) gene has recently emerged as a potential factor in stroke recovery and PTSD susceptibility. CCR5 is a pro-inflammatory receptor involved in immune modulation and neuroplasticity, with its expression increasing in response to ischemic events. 21 It plays a crucial role in mediating leukocyte trafficking to the site of injury, influencing both neuroinflammation and repair processes. Beyond its inflammatory role, CCR5 is known to impact synaptic plasticity and cognitive function, with studies indicating its involvement in memory consolidation and neural network reorganization following brain injury. CCR5 knockdown in rodents was shown to promote substantial motor recovery after stroke. 22 In humans, we have recently reported that carriers of the naturally occurring mutation in the CCR5 gene (CCR5-Δ32), making it partially inactive, had less depressive symptoms post-stroke 23 and a better cognitive outcome two years post-stroke compared with non-carriers. 22 This mutation is assumed to result in diminished inflammatory responses and enhanced neuroplasticity, which may contribute to resilience against PTSD. Despite this, the role of CCR5-Δ32 in post-stroke PTSD remains unexplored, and understanding its potential protective mechanism could offer novel therapeutic insights..6,24,21,25–27,28
This study aims to investigate whether CCR5-Δ32 acts as a protective factor against post-stroke PTSD and to explore potential interactions with other genetic polymorphisms such as 5-HTTLPR and BDNF Val66Met. Additionally, we seek to determine whether pre-existing coping styles modulate the relationship between genetic predisposition and PTSD outcomes, potentially offering insight into personalized therapeutic strategies.
Understanding the genetic and psychological contributors to post-stroke PTSD could pave the way for novel, mechanism-based interventions targeting CCR5 pathways to improve long-term recovery and mental well-being in stroke survivors.
Methods
Study Population
Included in the present study were survivors of mild-to-moderate first-ever acute ischemic stroke/TIA, who participated in our prospective TABASCO study
29
(ClinicalTrials.gov #NCT01926691), an observational cohort study aimed to identify predictors for post-stroke cognitive decline. Included men and women over 50 years old, admitted within 72 h after a first-ever acute ischemic stroke/TIA, with a total NIH Stroke Scale (NIHSS) < 17. Exclusion criteria: stroke that resulted from trauma/invasive procedures, hemorrhagic stroke, a diagnosis of dementia or cognitive impairment before the stroke (determined by Informant Questionnaire on Cognitive Decline in the Elderly
30
score ≥3.3), severe aphasia or disability after the stroke, as judged by a senior neurologist, which made the possibility of continuous follow-up unlikely, or pre-stroke episodes of major depression or PTSD. The admission neurological assessment included verification of stroke etiology, NIHSS and neuroimaging (3TMRI). The exclusion criteria for the study investigating the effect of
Assessment of Mental Outcomes
PTSD symptoms were assessed at 6, 12, and 24 months after the index event with a modified version of the PTSD Checklist Specific for a stressor (PCL-S), using the stressor “stroke”. 31 The PCL-S is an extensively validated 17-item scale that corresponds to the DSM-IV-TR criteria for PTSD, which was found to be equivalent to the PCL-5, a newer 20-item scale, mapped to DSM-5. 32 Subjects were identified as having clinical PTSD symptomatology according to the following criteria: 1) at least one intrusive symptom; 2) at least three avoidant symptoms; 3) at least two hyper-arousal symptoms. To make the PCL-S more homogenous with the rest of the survey and easier to complete for participants of low health literacy, the instrument was modified from a 5-point (“not at all”, “a little bit”, “moderately”, “quite a bit”, “extremely”) to a 4-point response scale (“not at all”, “a little bit”, “somewhat”, “very much”). To allow our scoring to be comparable to other studies, we re-coded PCL-S scores to reflect the standard 5-point response scale (1 = 1; 2 = 2.33; 3 = 3.67; 4 = 5), so that the range of the total score was the same as the unmodified instrument, as previously reported. 5 Strong diagnostic sensitivity (0.944) and specificity (0.864) of the PCL has been well documented in relation to the Clinician-Administered PTSD Scale (CAPS), and this questionnaire has been used to assess PTSD in stroke/TIA survivors3,4,33 and other populations, including patients with myocardial infarction.34,35 A PCL-S cut-point of 44 and above is generally accepted as indicative of probable PTSD,36,37 and this is what we used in our population to determine the group of patients who have probable PTSD. We also assessed the severity of PTSD symptoms using the PCL-S as a continuous scale.4,38
Depressive symptoms were assessed within 72 h of admission and 6,12 and 24 months later, using the 15-item Geriatric Depression Scale (GDS) that has acceptable internal consistency and reliability (Cronbach's α ≥ 0.70) in older adults. 39
All follow-up assessments were conducted in the clinic through face-to-face interviews.
Patients completed a baseline neuropsychological assessment including the Montreal Cognitive Assessment (MoCA) and the NeuroTrax computerized cognitive testing (NeuroTrax Corp., Bellaire, TX). These comprehensive neuropsychological evaluations were repeated 6, 12 and 24 months after the stroke. A Global Cognitive Score was computed as the average of the six index scores (memory, executive functions, visuospatial perception, verbal function, attention and motor skills). Data for each outcome parameter of the NeuroTrax battery were normalized according to stratifications of age (50 to 70 years, > 70 years) and education (≤12 years, > 12 years) to give a distribution with a mean of 100 and a standard deviation of 15 (ie, an IQ-style scale).
Coping Scales
The repressive coping Scale questionnaire 40 was used in our study. This well-established self-report measure 33 consists of 58 items, which combine two well-validated scales: Manifest Anxiety (Taylor Manifest Anxiety Scale) and defensiveness (the Marlow-Crowne Social Desirability Scale). At baseline, participants were asked to indicate whether each item described them on a dichotomous scale – 0(no) 1(yes). The coping style was determined by the median scores on the two scales.34,41 Accordingly, patients were grouped to 4 different coping styles: Participants with anxiety scores below the median and defensiveness scores above the median were classified as repressors. Participants with anxiety and defensiveness scores below the median were classified as low-anxious; high-anxious individuals were those with anxiety scores above the median and defensiveness scores below the median; and defensive individuals had anxiety and defensiveness scores both above the median.
Statistical Analysis
We used the SPSS/WIN (version 29.0, Chicago, IL, USA) software for all statistical analyses.
Comparisons or distributions between categories were assessed using Student's t-test, the Mann–Whitney U, or the chi-square test, as appropriate. The Pearson or Spearman's rank correlation analysis (coefficient estimate r) determined associations between numeric variables.
Descriptive statistics were used to summarize the characteristics of the sample. Missing data were filled in with a multiple imputation method under the “missing at random” assumption. 42 Chi-squared, and t-tests were used to compare the characteristics of participants according to probable post-stroke PTSD status. The Pearson or Spearman's rank correlation analysis (coefficient estimate r) determined associations between numeric variables.
To track differences in PTSD trajectories over time we used the repeated measures approach based on the general linear model (GLM) to compare the PCL-S scores of the three observation points between carriers and non-carriers. Statistical differences between longitudinal PCL-S curves of the different genotypes were analyzed by a two-way ANOVA using the Bonferroni correction. Univariate regression analysis was used to screen for the variables which significantly correlated with severity of PTSD symptoms post-stroke (PCL-S, as a continuous variable). A multiple linear regression model was used to evaluate the relations of the CCR5-Δ32 alleles and PCL-S at 6, 12, and 24 months post-stroke, adjusted for age, gender, education, administration of anti-depressants during the follow-up period, ethnicity, employment before the stroke/TIA and the existence of new infarct on MRI. Next, we conducted univariate correlations of both 5-HTTLPR and CCR5 genotypes and PCL-S scores for each of the four single gene groups separately. For multivariate group comparisons, PCL-S scores were then submitted to a multiple analysis of variance (MANOVA) with two between-subjects factors of CCR5 (w.t., CCR5-Δ32) and 5-HTTLPR serotonin transporter gene (long, short). This MANOVA tested for the main effects of CCR5 and 5-HTTLPR and the interaction of CCR5 × 5-HTTLPR on PCL-S scores. Likewise, we used MANOVA to test the main effects of CCR5 and BDNF Val66Met and the interaction of CCR5 x BDNF Val66Met on PCL-S scores.
Multiple imputations were performed to estimate likely GDS scores for survivors lost to follow-up. The imputation model included selected baseline characteristics as predictor values: sex, age, education, admission NIHSS, smoking, diabetes mellitus, and hypertension. Because the data displayed a nonmonotone pattern of missing values, we imputed using the fully conditional specification method based on a logistic regression model. PCL-S scores were then calculated for each data set and an average was presented.
The prospective contribution of both coping style and CCR5 genotype to the risk for the development of probable PTSD over time was tested using Cox proportional hazards regression, in relation to the duration and timeline until the occurrence of their PTSD. A P-value < .05 was considered statistically significant for all analyses.
Results
A total of 575 consecutive eligible cognitively intact patients at baseline, who were admitted to the Department of Emergency Medicine at the Tel-Aviv Medical Center between April 1, 2008, and December 1, 2014, within 72 h from onset of TIA or ischemic stroke symptoms, were initially evaluated. Of them, the CCR5 genotype was determined in 564 participants. Individuals whose data on PTSD symptoms were missing (N = 129) were excluded, as well as a History of PTSD before the index stroke/TIA (N = 3) and one participant who had an ischemic lesion in the limbic system (left hippocampus).
Flow Chart of Patient Selection.
Baseline and Follow-up Characteristics of post-Stroke Survivors (n = 432).

CRP, C-reactive protein; GDS, geriatric depression scale; GM, gray matter; IQR, interquartile range; MRI, Magnetic resonance imaging; NIHSS, National Institutes of Health Stroke Scale; PCL-S, PTSD Checklist Specific for a stressor; STAI-S, State-Trait Anxiety Inventory-state anxiety subscale; STAI-T, State-Trait Anxiety Inventory-trait anxiety subscale; SD, standard deviation; TIA, transient ischemic attack; WM, white matter. Entries are mean (SD) or n and %, as indicated. Significant results are shown in bold (P < .05).
The mean age was 66.8 ± 9.6 years; 170 (39.4%) were women; 128 (29.6%) were diagnosed as TIA; 360 participants (83.3%) were homozygotes for the CCR5 wild-type allele (non-carriers), while 67 participants (15.5%) were heterozygotes for the CCR5-Δ32 variant allele, and five patients (1.2%) were homozygotes for the variant allele. Allele frequencies were in Hardy–Weinberg equilibrium. As only a small number (N = 5) of participants were CCR5-Δ32 homozygotes, all CCR5-Δ32 variant allele carriers were pooled together in a CCR5-Δ32 carriers’ group (N = 72) and compared to non-carriers homozygotes (N = 360). The frequency of the 5-HTTLPR SS, SL, and LL genotypes was 0.24, 0.50, and 0.26, respectively; The frequency of the BDNF Val/Val, Val/Met, and Met/Met genotypes: 0.57, 0.40, and 0.03, respectively.
Forty-eight participants (11%) developed PTSD symptoms within the first year after the index stroke/TIA (PCL-S ≥ 44) (36 reported symptoms 6 months after the index stroke/TIA, and 32 had probable PTSD 12 months after the index stroke/TIA). Thirty-three participants (7.6%) had probable PTSD two years after the index stroke/TIA. For our analysis, we grouped all participants who had probable PTSD at six and/or 12 months, forming a group of 48 participants (11% of the whole sample) who developed PTSD symptoms within the first year after the index stroke/TIA. Of them, 16 (33.3%) retained their symptoms 24 months after the index event.
A summary of baseline characteristics of participants is presented in Table 1.
Stroke/TIA etiologies (based on TOAST criteria) were as follows: 156 lacunar strokes (51.3%), 41 cardioembolic strokes (13.5%), 34 large-artery atherosclerotic strokes (11.2%), 73 strokes of other or undetermined etiology (24%), 128 patients (29.6%) were diagnosed as suffering from TIA. No relation was observed between etiologies and the development of post-stroke PTSD.
No differences in CCR5-Δ32 distribution were observed across stroke subtypes or between stroke and TIA patients.
CCR5-Δ32 carriers were more educated but did not differ in cardiovascular risk factors or frequency of the ApoE4 allele compared to non-carriers (Table 1); 87.1% of the CCR5-Δ32 carriers were Ashkenazi in their origin, compared with 57% in the non-carriers (the CCR5-Δ32 mutation was previously described as more prevalent among Ashkenazi Jews 22 ). At admission, CCR5-Δ32 carriers had lower C-reactive protein (CRP) (Table 1).
CCR5-Δ32 carriers had lower neurological scores (lower NIHSS) upon admission compared with non-carriers [1.00 (0.00-3.00) versus 2.00 (1.00-4.00), P = .002] and had a lower disability (modified Rankin scale) 3 months after the index event: 1.2 ± 0.9 versus 1.7 ± 1.1, P < .001).
CCR5-Δ32 and post-Stroke PTSD Symptoms
CCR5-Δ32 carriers presented fewer PTSD symptoms (lower PCL-S scores) 6, 12, and 24 months after the index event, compared with non-carriers (P < .001, P < .001, P = .02, respectively, Table 1). After adjustment for age, gender, education, administration of anti-depressants during the follow-up period, ethnicity, employment before the stroke/TIA, and the existence of new infarct on MRI, the association of the CCR5-Δ32 status with PCL-S scores remained significant only at 6- and 12- months post-stroke (Table 2, Figure 2), although the trend was similar at the other time point. CCR5-Δ32 non-carriers tended to remain stable or develop worse PTSD symptoms over time, while CCR5-Δ32 carriers tended to improve (Figure 2).
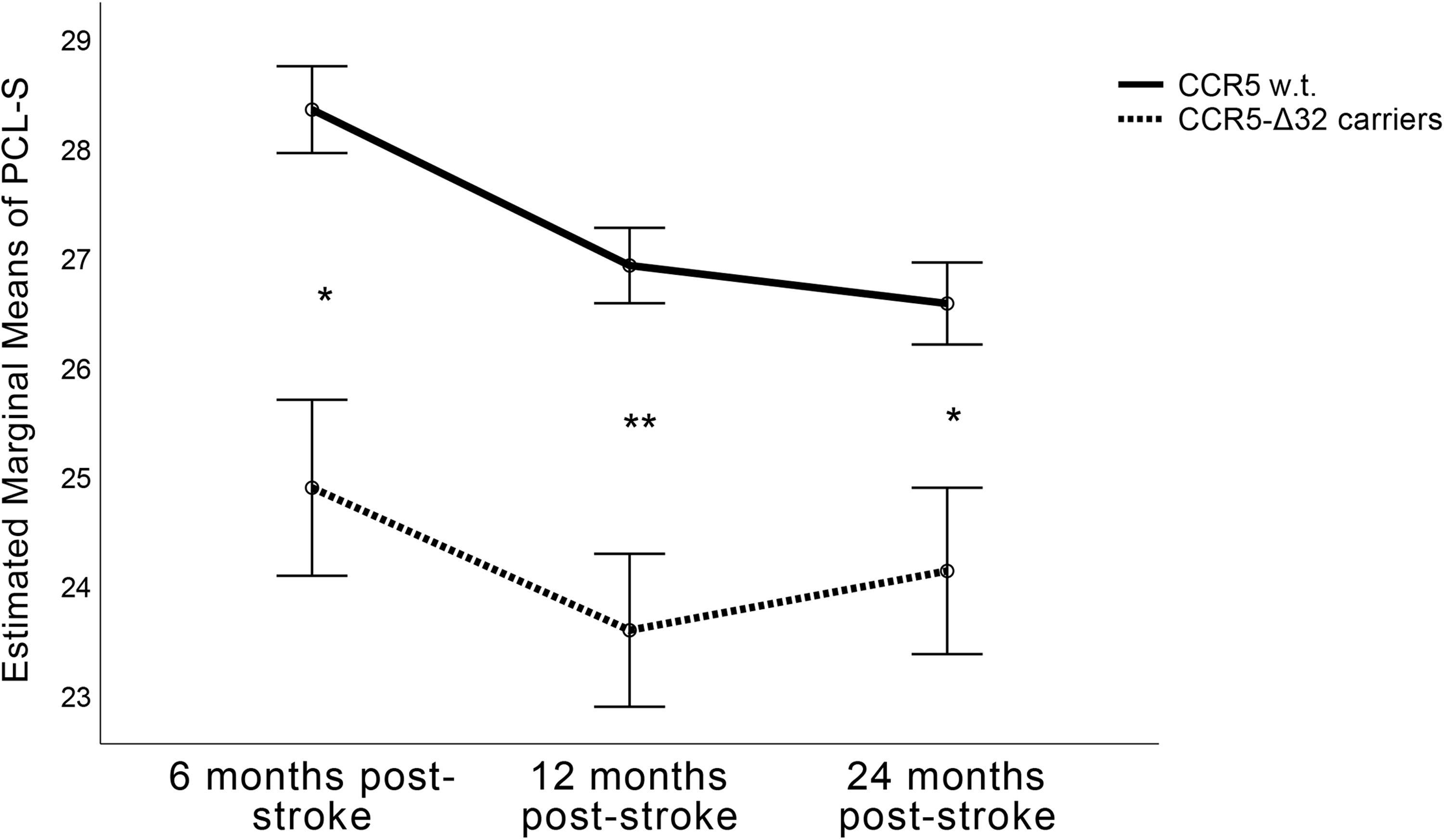
General Linear Model (GLM) Analysis of Repeated Measures of Longitudinal PCL-S Scores Along the Follow-Up, Comparing CCR5-Δ32 Carriers to Non-Carriers. *P < .05, **P < .01.
CCR5-Δ32 Allele in Relation to PTSD Symptoms Severity (PCL-S Score) 6, 12 and 24 Months After the index Event, Unadjusted (Model 1) and Adjusted for Age, Gender, Education, Depression or Administration of Anti-Depressants During the Follow-up Period, Ethnicity, Employment Before the Stroke/TIA and the Existence of New Infarct on MRI (Model 2). Significant Results (P < .05) are Shown in Bold.

PCL-S, PTSD Checklist Specific for a stressor; SE, standard error.
In the probable PTSD group (PCL-S ≥ 44) 6 and 12 months after the index stroke/TIA, 46 patients (97.9%) were CCR5-Δ32 non-carriers, and only 1 (2.1%) was CCR5-Δ32 carrier (χ2 = 8.03, P = .005). This trend also appeared at 24 months (in the probable PTSD group, 30 patients, 90.9%, were CCR5-Δ32 non-carriers, and only three patients, 9.1%, were CCR5-Δ32 carriers), but the results at this time point were non-significant (χ2 = 1.15, P = .284).
On average, the non-carriers` group (both probable PTSD and non-PTSD) presented higher PCL-S scores than carriers at all-time points, and post hoc tests revealed that their scores differed significantly (P = .006, for the linear difference between curves, Table 1 and Figure 2).
The subjects who were CCR5-Δ32 homozygotes (N = 5) presented even lower PCL-S scores compared with heterozygotes CCR5-Δ32 and non-carriers at six months: 22.9 ± 3.8 versus 24.7 ± 8 and 28.5 ± 10.8, respectively; at 12 months: 24 ± 5.7 versus 23.9 ± 5.7 and 28.2 ± 11.2, respectively; at 24 months: 24 ± 4.7 versus 24.1 ± 8.1 and 27.3 ± 10.7, respectively. Thus, there was a trend toward dose-response of the number of CCR5-Δ32 alleles, but the differences were non-significant, probably due to the small sample.
5-HTTLPR Polymorphism, Val66Met SNP, and post-Stroke PTSD Symptoms
Univariate analyses demonstrated that participants with either one or two copies of the 5-HTTLPR-L allele presented fewer PTSD symptoms only 12 months after the index event, compared with non-carriers (P = .013). In addition, univariate analyses demonstrated that participants with either one or two copies of the BDNF-Val variant presented fewer PTSD symptoms 6 and 24 months after stroke, compared with non-carriers (P = .007, P = .039, respectively).
Figure 3 plots PCL-S scores in GLM analysis for the four allelic groups (available for 404 participants). As shown in Figure 3(a), carriers of both 5-HTTLPR-L and CCR5-Δ32 had the lowest level of PTSD symptoms at all-time points, followed by the 5-HTTLPR-S and CCR5-Δ32 carriers, and then by the 5-HTTLPR-L, CCR5 non-carriers (wild-type, w.t), with 5-HTTLPR-S, CCR5 w.t, endorsing highest PTSD symptoms 6 and 12 months after the index event (P = .026, P = .05, for PCL-S scores at 6 and 12 months post-stroke, respectively). Submitting these PCL-S scores to a multivariate analysis of variance (MANOVA) with two between-subjects factors of 5- HTTLPR (L, S) and CCR5 (w.t., CCR5-Δ32) revealed no significant 5-HTTLPR x CCR5 interaction (P = .946).

(a) General Linear Model (GLM) Analysis of Repeated Measures of Longitudinal PCL-S Scores Along the Follow-up, Comparing the Four Allelic Groups of 5-HTTLPR and CCR5. *P < .05, for Differences Between 5-HTTLPR S & CCR5 w.t. to 5-HTTLPR L & CCR5-Δ32. (b) General Linear Model (GLM) Analysis of Repeated Measures of Longitudinal PCL-S Scores Along the Follow-up, Comparing the Four Allelic Groups of BDNF Val66Met and CCR5. *P < .05, for Differences Between BDNFMet S & CCR5 w.t. to BDNFVal & CCR5-Δ32.
As shown in Figure 3(b), carriers of both the BDNFVal allele and CCR5-Δ32 had the lowest level of PTSD symptoms six months after the index event (P = .022), followed by the BDNFMet and CCR5-Δ32 carriers, and then by both the BDNFVal allele and CCR5, CCR5 non-carriers (wild-type, w.t), and BDNFMet allele and CCR5 w.t, endorsing highest PTSD symptoms 6 months after the index event. Submitting these PCL-S scores to a MANOVA with two between-subjects factors of BDNF (Val, Met) and CCR5 (w.t., CCR5-Δ32), revealed no significant BDNF Val66Met x CCR5 interaction (P = .951).
CCR5-Δ32 and post-Stroke PTSD in Susceptible Individuals
We have recently reported that premorbid coping styles, high-anxious and defensive, were associated with an increased risk of developing PTSD after stroke. 5
In our cohort, 191 patients (44.2%) were classified as repressors, 83 (19.2%) as low-anxious, 75 (17.4%) as high-anxious, and 83 (19.2%) as defensive. Here, we examined the unique contribution of both premorbid coping styles and the CCR5 allele genotype to the risk of developing PTSD after stroke.
Cox models for the prediction of the development of post-stroke PTSD during the follow-up period according to both coping style and CCR5 allele are shown in Table 3 and Figure 4. Compared with the highly anxious and defensive patients (Maladaptive coping style) carriers of CCR5-Δ32 (none of this group developed probable PTSD), the Maladaptive coping style carriers of CCR5 w.t. had a higher risk of developing probable PTSD (HR = 4.03;95% CI,1.95-6.32, P< .001) after adjusting for age, years of education, lesion existence in MRI, stroke severity (NIHSS) and baseline depression. Compared with the low anxious and repressors patients (=Adaptive coping style) carriers of CCR5-Δ32 (none of this group developed probable PTSD), the Adaptive coping style carriers of CCR5 w.t. had a higher risk of developing probable PTSD (HR = 3.36;95% CI,1.46-7.77, P< .01) after same adjustments (Table 3).

Survival Curves Showing Time to Post-Stroke PTSD Symptomatology by Both Personality Trait and Coping Manner and CCR5 Genotype.
Adjusted Hazard Ratios (HR) for the effect of coping style and CCR5 genotype on development of probable post-stroke PTSD.

*P < 0.05, **P < 0.01, ***P < 0.001.
CI confidence interval. The hazard ratio (HR) was calculated by using the stratified Cox proportional regression method during the 2-year follow-up period. Adjustments were made for the following variables: age, years of education, lesion existence in MRI, stroke severity (NIHSS), and baseline depression.
Discussion
In this study, we demonstrated that a loss-of-function mutation (LOFM) in the CCR5 gene is associated with a reduced likelihood of developing PTSD symptoms after a stroke or TIA. This effect is more pronounced in individuals with a maladaptive coping style, who are known to be more susceptible to PTSD than those with an adaptive coping style.
When analyzing known genetic variations linked to PTSD, both the BDNF and 5-HTTLPR polymorphisms, examined separately, had a minor effect on the development of post-stroke PTSD symptoms compared to the impact of the CCR5-Δ32 allele. Our findings suggest that this allele may play a role in mitigating, if not reversing, the risk effects of the 5-HTTLPR-S or BDNF-Met66 alleles, both of which have been well-documented as risk factors for developing PTSD following trauma exposure. 43 Notably, no interaction was found between CCR5-Δ32 and either BDNF or 5-HTTLPR.
Both PTSD and ischemic insults are associated with synaptic loss and inflammatory processes.6,24 Brain connectivity deficits observed in PTSD may interact synergistically with stroke-induced brain injury, exacerbating cortical network dysregulation, cognitive dysfunction, and functional impairment, ultimately leading to worsened PTSD symptoms.44,45
Considering the established neural substrates of PTSD development, CCR5 depletion (resulting from the CCR5-Δ32 mutation) may help prevent the pathological changes that contribute to PTSD development after stroke. Our findings align with previous evidence demonstrating that the CCR5-Δ32 mutation is associated with a reduced likelihood and severity of post-stroke cognitive impairment and depression.22,23 These conditions are closely linked with PTSD, suggesting that the CCR5-Δ32 mutation may influence a common neurobiological pathway across these disorders.
In our cohort, genetic variations previously linked to PTSD had only a minor effect compared to the CCR5-Δ32 mutation. This may indicate that BDNF and 5-HTTLPR play a lesser role in the specific context of post-stroke PTSD compared to PTSD from other causes.
Personality traits play a significant role in determining PTSD risk after stroke, influencing an individual's ability to cope with the distress associated with stroke/TIA. Our findings also support this observation. 5 Since certain personality traits render individuals less likely to develop PTSD, it is not surprising that the impact of the CCR5-Δ32 mutation was less pronounced in these individuals. However, in the maladaptive coping style group, the presence of the CCR5-Δ32 mutation significantly reduced PTSD risk, underscoring the complex interplay between genetics and personality traits in stroke recovery.
Stroke recovery is a complex, multifactorial process involving both brain and non-brain factors. Brain-related factors include stroke location, size, and specific vulnerabilities such as personality traits and genetic predispositions like the CCR5-Δ32 mutation. Non-brain factors encompass environmental and social factors, rehabilitation resources, and support systems. Our findings suggest that the CCR5-Δ32 mutation may play a crucial role in recovery. However, given its relatively low prevalence in the general population, its effects could potentially be replicated using CCR5 blockade therapy. Indeed, the CCR5-blocking agent Maraviroc is currently being tested in a clinical trial for post-stroke cognitive decline, 46 highlighting its potential as a therapeutic target to enhance stroke rehabilitation.
This study has several strengths, including systematic prospective follow-up and comprehensive clinical data collection in a cohort with a relatively high prevalence of the CCR5-Δ32 mutation. The longitudinal administration of the PTSD questionnaire by trained personnel, alongside depression and cognitive assessments, genetic data, and inflammatory markers, adds robustness to our findings. However, there are limitations to consider. First, while the PCL-S provides valuable insights into PTSD symptomatology, it does not replace a formal psychiatric diagnosis. Although we used a conservative cut-off to identify probable PTSD cases, it is possible that the actual prevalence is underestimated. Second, our study cannot establish causality between the CCR5-Δ32 mutation and PTSD development. Third, the exclusion of patients with severe or hemorrhagic strokes limits the generalizability of our findings to this subgroup. Further research is warranted to explore these aspects. Additionally, the demographic characteristics of our cohort, where 62.1% of participants were of Ashkenazi origin, may limit the broader applicability of our findings. However, the association between CCR5-Δ32 and PTSD scores remained significant even after adjusting for ethnicity. While the loss to follow-up was relatively modest, selection bias cannot be entirely ruled out. We applied multiple imputation techniques to address this and estimate probable PTSD scores for participants lost to follow-up (because we used a large number of predictor variables, and since no differences were observed in baseline characteristics between participants attended at follow-up and those who were missing or lost to follow-up, it seemed reasonable to assume that data were missing at random). Furthermore, more patients with probable PTSD received antidepressant treatment 12 months post-stroke/TIA, though the precise indications for these prescriptions remain unclear. Lastly, the lack of a non-stroke control group is a limitation that should be addressed in future studies.
In conclusion, the CCR5-Δ32 allele is associated with a lower likelihood of developing PTSD after stroke in the TABASCO cohort. This protective effect is particularly pronounced in individuals with a maladaptive coping style. CCR5 blockade may represent a promising, mechanism-based therapeutic target to improve stroke recovery and manage neuropsychiatric complications. Future clinical trials should aim to validate our findings and refine patient selection for personalized interventions based on genetic and psychological profiles.
Supplemental Material
sj-docx-1-css-10.1177_24705470251345245 - Supplemental material for Post-Stroke PTSD: The Protective Role of CCR5-Δ32 Polymorphism
Supplemental material, sj-docx-1-css-10.1177_24705470251345245 for Post-Stroke PTSD: The Protective Role of CCR5-Δ32 Polymorphism by Hen Hallevi, Oren Tene, Jeremy Molad, Aviva Alpernas, Dana Niry, Saly Usher, Lital Feldinger, Estelle Seyman and Einor Ben Assayag in Chronic Stress
Footnotes
Author Contributions
Hen Hallevi: Writing – original draft, Writing – review & editing, Investigation, Conceptualization. Oren Tene: Writing – original draft, Writing – review & editing, Investigation, Conceptualization, Methodology. Jeremy Molad: Writing – review & editing, Investigation. Aviva Alpernas: review & editing, Investigation. Dana Niry: Writing – review & editing, Investigation. Saly Usher: Writing – review & editing, Investigation. Lital Feldinger: Writing – review & editing, Investigation. Estelle Seyman: Writing – review & editing, Investigation. Einor Ben Assayag: Supervision, Methodology, Writing – original draft, Writing – review & editing, Investigation, Conceptualization, Funding acquisition, Formal analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by grants RAG11482 from the American Federation for Aging Research (to EBA), 2011344 From the U.S.-Israel Bi-national Science Foundation (to EBA), 3-0000-17856 from the Israeli Ministry of Health (to EBA, HH, OT, JM), AARG-16-442861 from the Alzheimer's Association and grant from Rekanati Foundation (to EBA).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EBA, HH, OT, and the Tel-Aviv Sourasky Medical Center have a pending patent application filed #62/978,324 (U.S. Patent Application No. 17/799,920 and European Patent Application No. 21757047.2). The specific aspect of the manuscript covered in the patent application: using CCR5 antagonists for treating post-stroke depression.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
